Current issue
- Page Path
- HOME > Articles and issues > Current issue
Review Articles
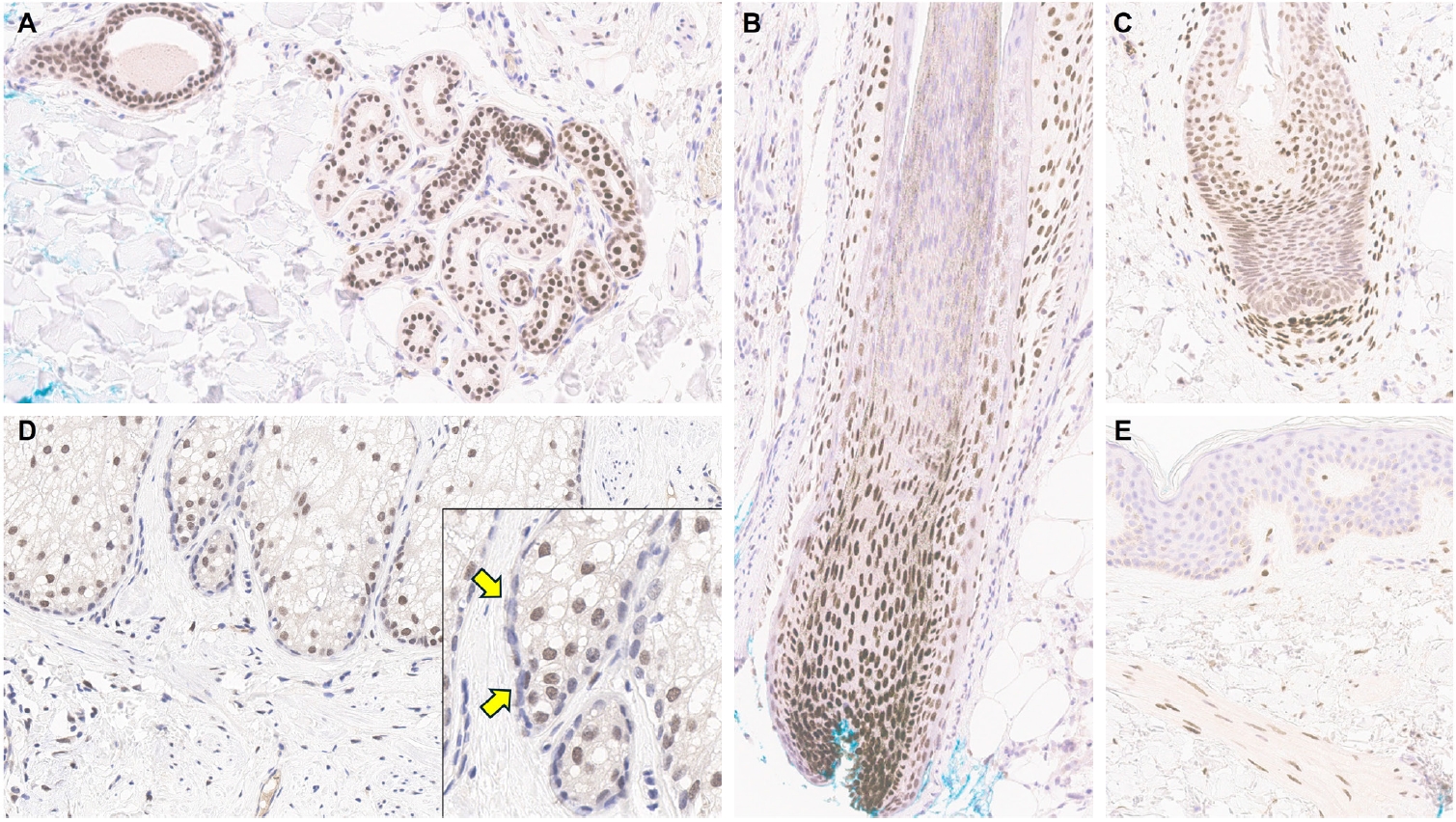
- The evolving role of TRPS1 in dermatopathology: insights from the past 4 years
- Mokhtar H. Abdelhammed, Woo Cheal Cho
- J Pathol Transl Med. 2026;60(2):129-143. Published online January 29, 2026
- DOI: https://doi.org/10.4132/jptm.2025.11.25
- 1,582 View
- 112 Download
-
 Abstract
Abstract
 PDF
PDF - Over the past 4 years, trichorhinophalangeal syndrome type 1 (TRPS1) has rapidly gained attention among practicing pathologists, with numerous studies emerging that both support and question its diagnostic utility. Initially regarded as a highly specific marker for tumors of mammary origin, TRPS1 is now recognized to have broader expression patterns, including in a variety of cutaneous neoplasms. This is likely due to embryologic parallels between breast tissue and skin adnexal structures, an overlap that was underappreciated in early investigations. Although TRPS1 lacks absolute specificity—even among cutaneous neoplasms—it can still offer meaningful diagnostic value when interpreted alongside conventional immunohistochemical markers and within the appropriate morphologic context. Noteworthy diagnostic applications include mammary Paget disease, primary extramammary Paget disease, rare adnexal neoplasms such as endocrine mucin-producing sweat gland carcinoma and primary cutaneous NUT adnexal carcinoma, and cutaneous metastases from breast carcinoma. In this review, we present the most comprehensive and up-to-date evaluation of the utility and limitations of TRPS1 immunohistochemistry in dermatopathology. Our aim is to deepen understanding of this emerging marker and provide practical guidance on its optimal integration with established immunohistochemical panels to enhance diagnostic accuracy in routine practice.
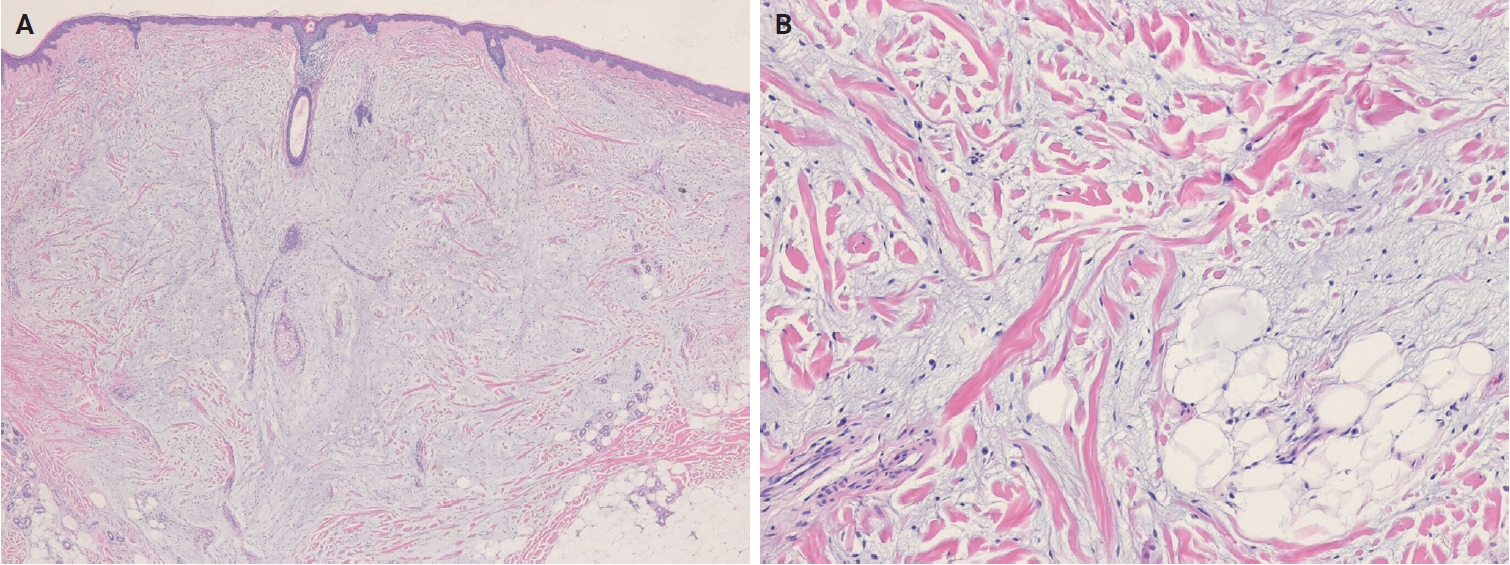
- Cutaneous soft tissue tumors in the 5th edition of the World Health Organization classification of skin tumors: key updates and new entities
- Joon Hyuk Choi
- J Pathol Transl Med. 2026;60(2):144-183. Published online March 13, 2026
- DOI: https://doi.org/10.4132/jptm.2026.01.09
- 353 View
- 38 Download
-
 Abstract
Abstract
 PDF
PDF - The 5th edition of the World Health Organization (WHO) classification of skin tumors introduces a dedicated chapter on cutaneous soft tissue tumors, providing a comprehensive, standardized reference with updated diagnostic criteria that directly inform routine dermatopathology practice and molecular diagnostics. This edition incorporates several key changes, including newly recognized entities such as EWSR1::SMAD3-rearranged fibroblastic tumor, neurotrophic tyrosine receptor kinase (NTRK)–rearranged spindle cell neoplasm, superficial CD34-positive fibroblastic tumor, and CRTC1::TRIM11 cutaneous tumor. Diagnostic terminology has also been refined; for example, the term ‘atypical intradermal smooth muscle neoplasm’ replaces ‘cutaneous leiomyosarcoma’ for lesions confined to the dermis, whereas the designation leiomyosarcoma is reserved for tumors with overt subcutaneous infiltration. In addition, epithelioid fibrous histiocytoma has been reassigned to the family of tumors of uncertain differentiation. This review summarizes the key updates and newly recognized entities in the chapter on cutaneous soft tissue tumors in the 5th edition of the WHO classification of skin tumors, emphasizing their clinicopathological and molecular implications.
Original Articles
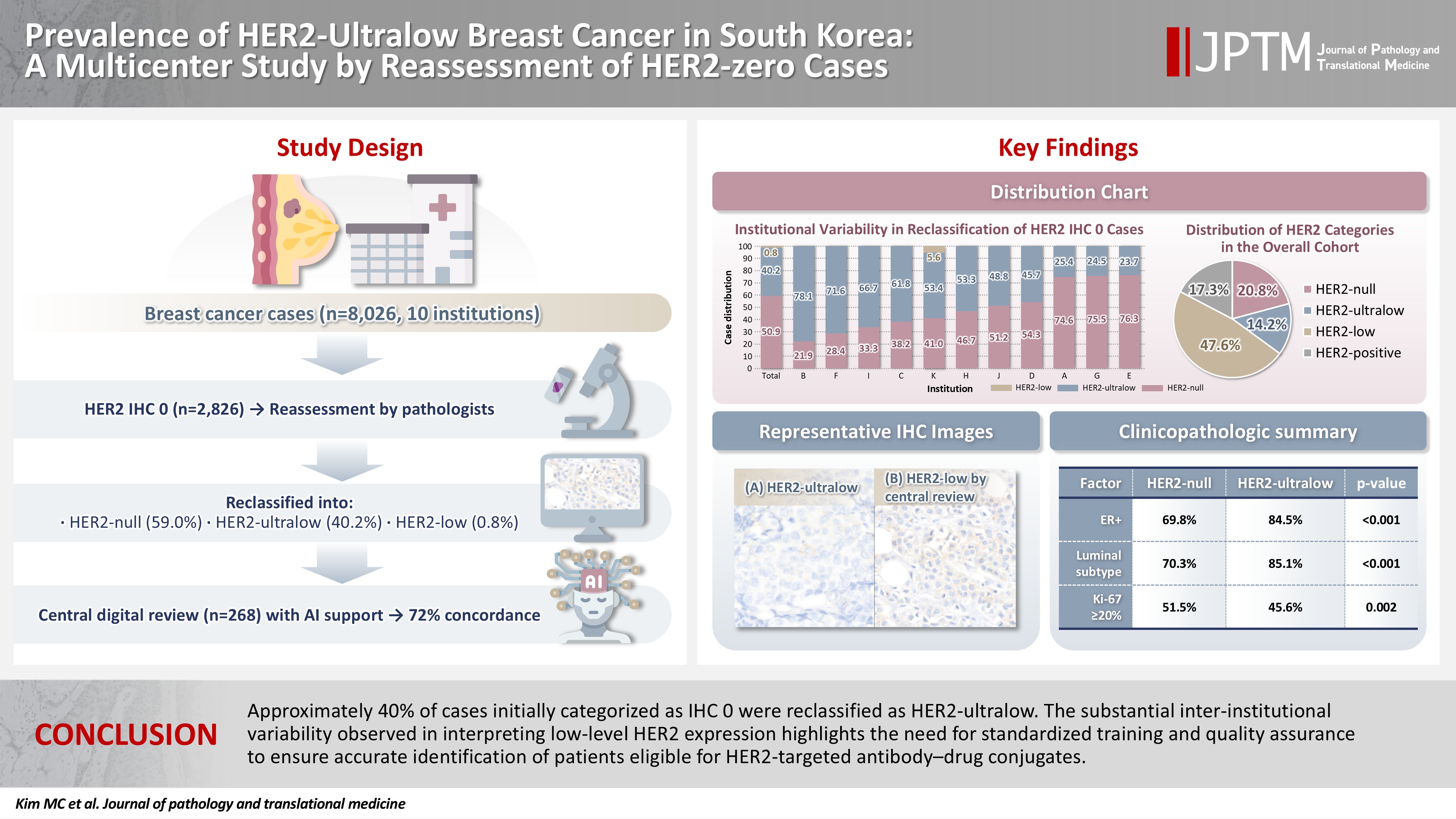
- Prevalence of HER2-ultralow breast cancer in South Korea: a multicenter study by reassessment of HER2-zero cases
- Min Chong Kim, Eun Yoon Cho, Hee Jin Lee, Ji Shin Lee, Jee Yeon Kim, Wan Seop Kim, Chungyeul Kim, Sun-Young Jun, Hye Jeong Choi, So Mang Lee, Ahrong Kim, Ji-Young Kim, Jeong Yun Shim, Gyungyub Gong, Young Kyung Bae
- J Pathol Transl Med. 2026;60(2):184-192. Published online February 23, 2026
- DOI: https://doi.org/10.4132/jptm.2025.10.22
- 685 View
- 63 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material - Background
This study aimed to determine the prevalence of human epidermal growth factor receptor 2 (HER2)–ultralow breast cancer among cases initially classified as HER2 immunohistochemistry (IHC) 0 and assess interobserver variability in interpreting low-level HER2 expression. Methods: In this multicenter retrospective study, all invasive breast cancer cases diagnosed between January and December 2022 across 10 Korean institutions were retrieved. Institutional pathologists reexamined HER2 IHC slides originally reported as IHC 0 according to the 2018 American Society of Clinical Oncology/College of American Pathologists guidelines and reclassified them as HER2-null (0), HER2-ultralow (0+), or HER2-low (1+). Slides from 10% of HER2-null and HER2-ultralow cases were digitized for central review and independently assessed by two pathologists, with discrepancies resolved by consensus. Results: Among 8,026 cases, 2,836 cases (35.5%) were initially reported as IHC 0. Upon re-review, 1,673 (59.0%), 1,139 (40.2%), and 24 (0.8%) cases were reclassified as HER2-null, HER2-ultralow, and HER2-low, respectively. The prevalence of HER2-ultralow breast cancer varied considerably across institutions (23.7%–78.1%). Central review of 268 digitized cases showed concordance in 193 cases (72.0%). Among the 75 discordant cases, 54 tumors (72.0%) were upgraded from HER2-null to HER2-ultralow, and 18 (24.0%) tumors were upgraded from HER2-ultralow to HER2-low. Furthermore, two tumors (2.7%) were downgraded from HER2-ultralow to HER2-null. Conclusions: Approximately 40% of cases initially categorized as IHC 0 were reclassified as HER2-ultralow. The substantial inter-institutional variability observed in interpreting low-level HER2 expression highlights the need for standardized training and quality assurance to ensure accurate identification of patients eligible for HER2-targeted antibody–drug conjugates.
- Correlation between HER2 gene copy number and immunohistochemistry categories in HER2-negative breast cancer: diagnostic utility for differentiating HER2-null, ultralow, and low tumors
- Min Chong Kim, Young Kyung Bae
- J Pathol Transl Med. 2026;60(2):193-201. Published online February 25, 2026
- DOI: https://doi.org/10.4132/jptm.2025.11.07
- 495 View
- 64 Download
-
 Abstract
Abstract
 PDF
PDF - Background
The recent recognition of human epidermal growth factor receptor 2 (HER2)–low and HER2-ultralow breast cancers (BCs) has expanded the therapeutic relevance of HER2 testing in the antibody-drug conjugate era. However, the biological continuum of HER2 expression measured by immunohistochemistry (IHC) and its relationship with the HER2 gene copy number remain unclear. Methods: We retrospectively analyzed 135 HER2-negative invasive BCs and reclassified them as HER2-null (IHC 0), HER2-ultralow (0+), or HER2-low (1+ or 2+ without amplification). HER2 gene copy number was determined using silver-enhanced in situ hybridization. Statistical analyses were performed to compare HER2 copy number among IHC categories and evaluate the discriminatory value of HER2 copy number for distinguishing IHC subgroups. Results: The mean HER2 copy number increased stepwise across IHC categories: 1.95 ± 0.54 (null), 2.03 ± 0.43 (ultralow), 2.25 ± 0.65 (low, 1+), and 3.29 ± 1.05 (low, 2+). Significant differences were observed between the ultralow and low groups (p = .003) and between the null and low groups (p < .001), but not between the null and ultralow groups or between the ultralow and 1+ groups. Conclusions: HER2 gene copy number was positively correlated with protein expression as reflected by IHC categories. Although HER2 gene copy number was statistically higher in HER2-low than in HER2-null tumors, the substantial overlap in copy number ranges likely limits its utility in distinguishing HER2-low from HER2- null BCs.
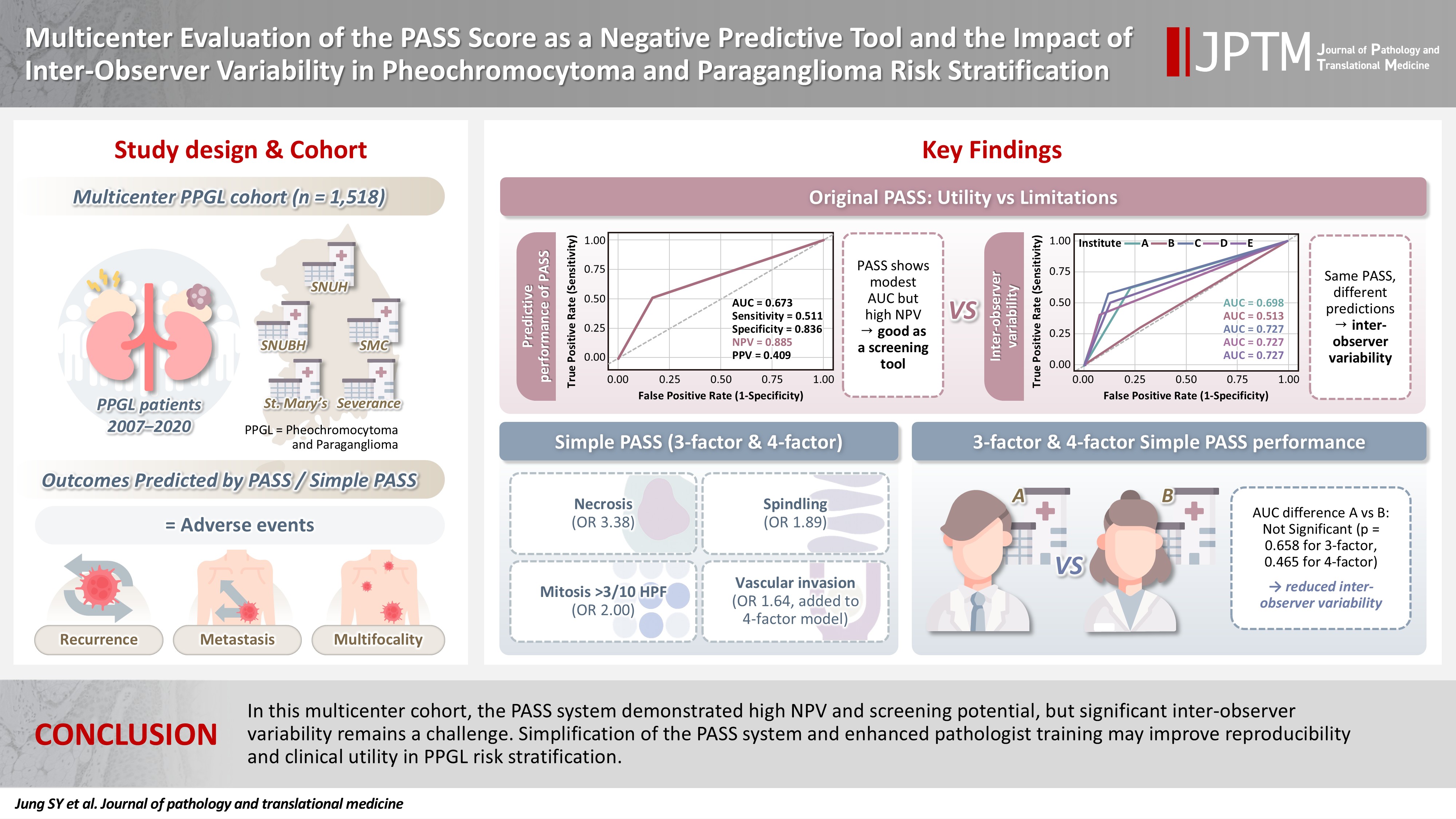
- Multicenter evaluation of the PASS score as a negative predictive tool and the impact of inter-observer variability in pheochromocytoma and paraganglioma risk stratification
- Sungyeon Jung, Hye-Ri Shin, Su-Jin Shin, Hee Young Na, Soon-Won Hong, So Yeon Park, Chan Kwon Jung, Kyeong Cheon Jung, Young Lyun Oh, Jae-Kyung Won
- J Pathol Transl Med. 2026;60(2):202-213. Published online February 23, 2026
- DOI: https://doi.org/10.4132/jptm.2025.11.05
- 654 View
- 41 Download
-
 Abstract
Abstract
 PDF
PDF - Background
The Pheochromocytoma of the Adrenal Gland Scaled Score (PASS) is widely used for risk stratification in pheochromocytoma and paraganglioma (PPGL), but its clinical utility is limited by inter-observer variability of its parameters and inconsistent predictive performance. Methods: We conducted a multicenter retrospective study of 1,518 patients with PPGL from five tertiary referral centers in Korea. Prognostic utility of PASS system was assessed using logistic regression, Kaplan-Meier analysis, and receiver operating characteristic (ROC) curve analysis. Inter-observer variability was inferred by comparing area under the ROC curve (AUCs) across institutions. Simplified PASS systems were developed based on multivariable analysis of key histopathological parameters. Results: The PASS system was a significant predictor of adverse events and recurrence-free survival. Although the PASS system demonstrated only modest discriminative ability (AUC, 0.673), it showed a high negative predictive value (NPV, 0.885), supporting its usefulness as a screening tool for benign behavior. However, there was significant inter-institutional variability in PASS performance (AUC; range, 0.513 to 0.727; p < .05). The 3-factor Simple PASS, which incorporates necrosis, spindling, and mitotic figures, exhibited less inter-observer variation. The 4-factor Simple PASS, which adds vascular invasion to the 3-factor model, also showed reduced inter-observer variability and improved AUC and NPV compared to the original PASS system. Conclusions: In this multicenter cohort, the PASS system demonstrated high NPV and screening potential, but significant inter-observer variability remains a challenge. Simplification of the PASS system and enhanced pathologist training may improve reproducibility and clinical utility in PPGL risk stratification.
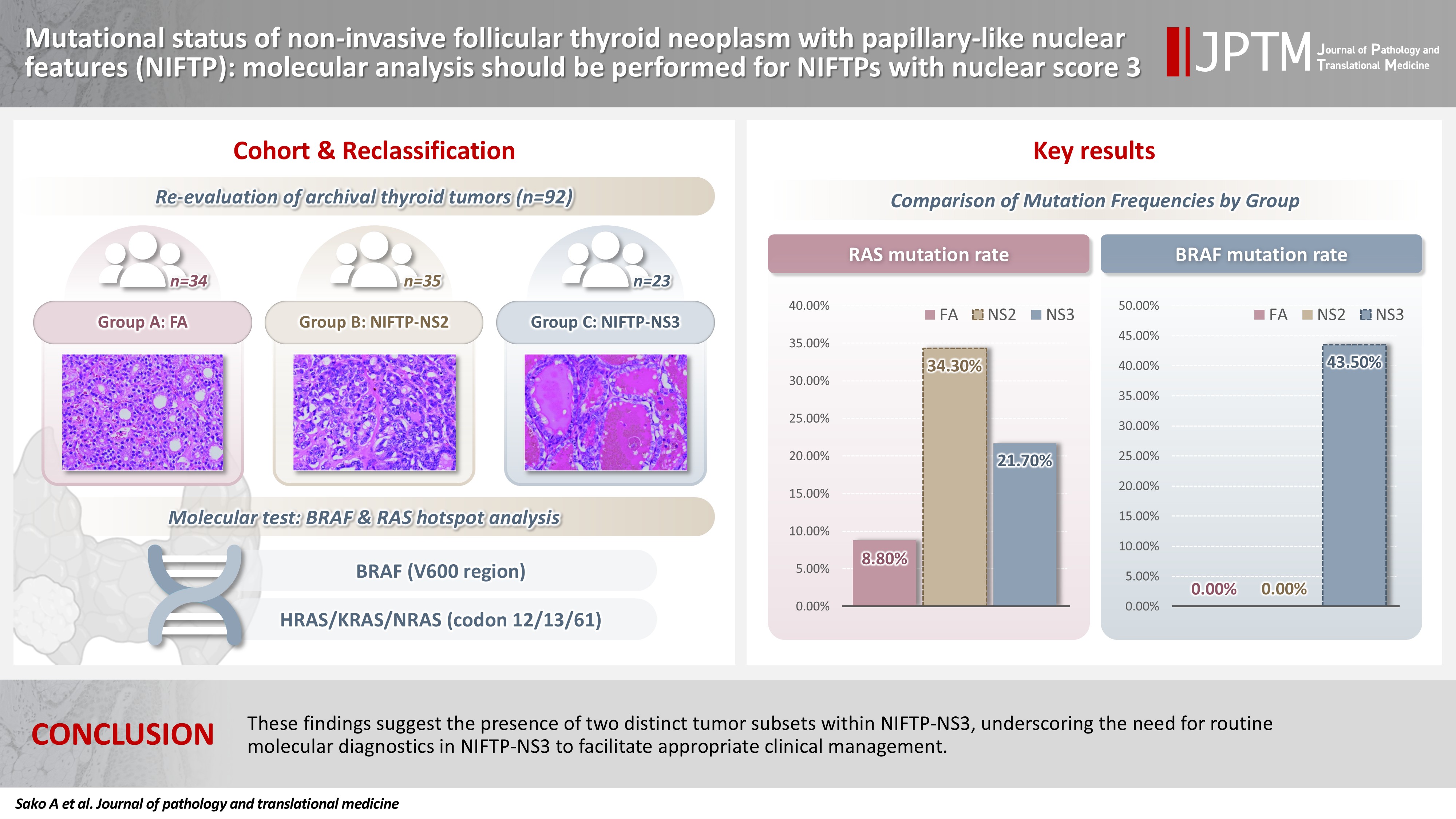
- Mutational status of non-invasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP): molecular analysis should be performed for NIFTPs with nuclear score 3
- Ayaka Sako, Mitsuyoshi Hirokawa, Michiko Matsuse, Miyoko Higuchi, Akira Miyauchi, Takashi Akamizu, Atsushi Kawakami, Norisato Mitsutake
- J Pathol Transl Med. 2026;60(2):214-219. Published online February 23, 2026
- DOI: https://doi.org/10.4132/jptm.2025.12.06
- 641 View
- 70 Download
-
 Abstract
Abstract
 PDF
PDF - Background
The classification of non-invasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP) was introduced to prevent the overtreatment of indolent tumors that were formerly diagnosed as non-invasive encapsulated follicular variant papillary thyroid carcinomas (NIEFV-PTCs). Although NIFTP was initially estimated to account for 10%–20% of papillary thyroid carcinomas in Western populations, its incidence is substantially lower in Asian cohorts. However, a multi-institutional Japanese study revealed that 31.0% of tumors previously diagnosed as follicular adenomas (FAs) were reclassified as NIFTPs. NIFTP diagnosis requires a nuclear score (NS) of 2–3, and according to the recent World Health Organization criteria, molecular analysis is recommended, but not mandatory, to exclude high-risk subtypes, namely cases with the BRAFV600E mutation, particularly for NS3 tumors. Methods: We performed genetic analysis on 92 archival thyroid tumor samples, including 69 previously diagnosed as FA, of which 34 remained as FA upon re-evaluation (group A) and 35 were reclassified as NIFTP with NS2 (group B). Additional 23 tumors previously diagnosed as NIEFV-PTC were reclassified as NIFTP with NS3 (group C). Results: RAS mutations were detected in 8.8%, 34.3%, and 21.7% of the tumor samples in groups A, B, and C, respectively, whereas BRAF mutations were present in 43.5% of the tumor samples in group C only. Conclusions: These findings suggest the presence of two distinct tumor subsets within NIFTP-NS3, underscoring the need for routine molecular diagnostics in NIFTP-NS3 to facilitate appropriate clinical management.
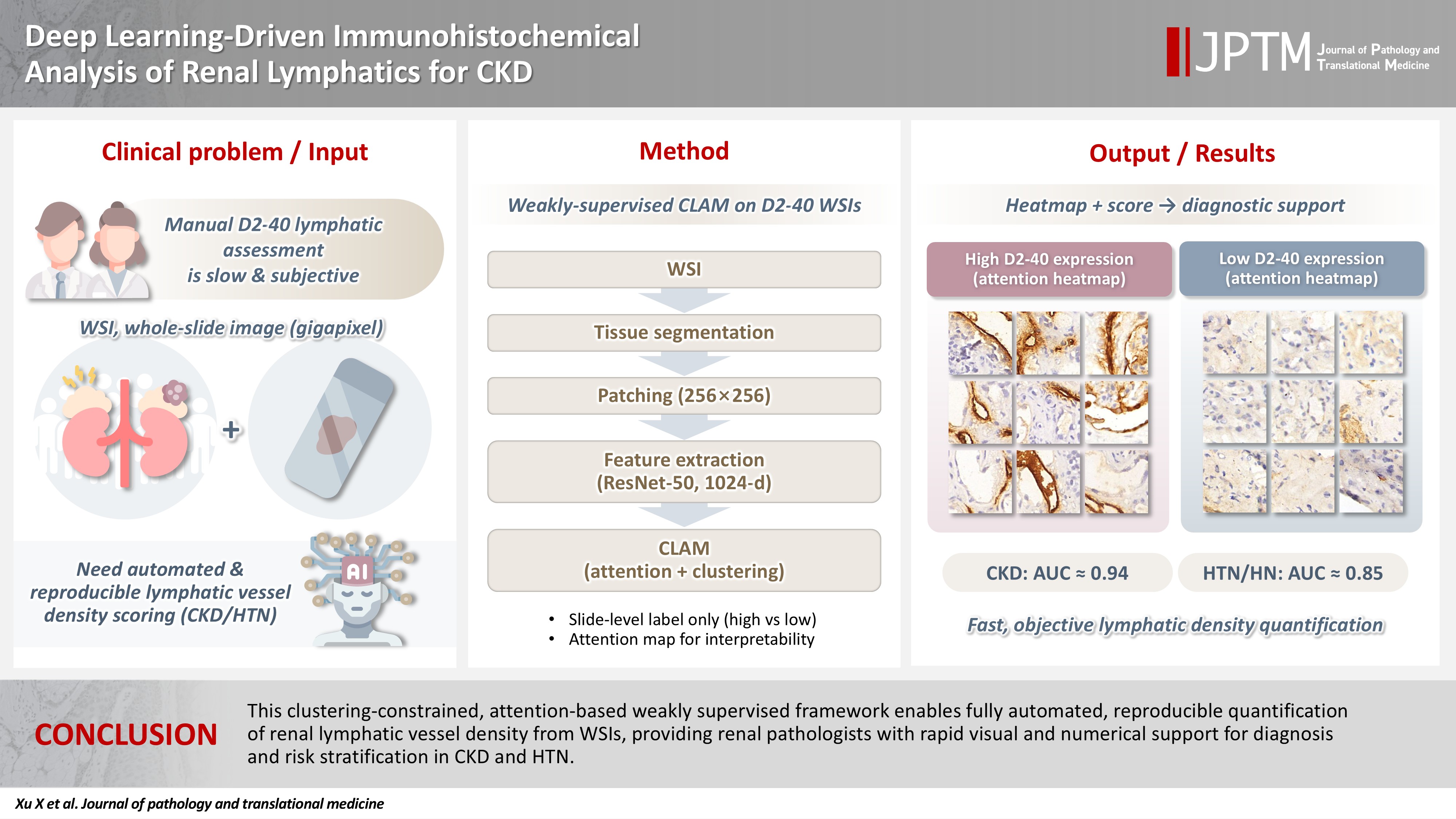
- Deep learning–driven immunohistochemical analysis of renal lymphatics for chronic kidney disease: bioinformatic and histopathological study
- Xin Xu, YanPing Lin, Guangchang Pei, Rui Zeng, Gang Xu
- J Pathol Transl Med. 2026;60(2):220-230. Published online March 13, 2026
- DOI: https://doi.org/10.4132/jptm.2025.12.15
- 373 View
- 19 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material - Background
Renal lymphatic vessel density is clinically relevant in kidney disease but is still assessed by slow, subjective visual estimation. We evaluated a weakly supervised, attention-based multiple-instance learning framework for automated detection and quantification of renal lymphatic vessel density on D2-40-stained whole-slide images (WSIs). Methods: Two independent internal datasets from Tongji Hospital were collected, including 198 cases of chronic kidney disease (CKD) and 50 cases of hypertensive nephropathy (HTN). All biopsies were immunohistochemically stained for D2-40 and digitized as WSIs. Pathologists provided only slide-level labels (D2-40 high vs. D2-40 low). Tissue regions were automatically segmented, tiled into patches, and encoded using a pretrained convolutional neural network. Patch embeddings were then analyzed with a clustering-constrained attention multiple-instance learning (CLAM) model. Unlike conventional multiple-instance learning (MIL) methods that only weight instances, CLAM jointly performs attention-based instance selection and instance-level clustering to distinguish positive from negative evidence within each slide, yielding more discriminative slide-level features and interpretable attention maps. Performance was compared with a classic MIL model trained on the same features. Results: CLAM achieved area under the receiver operating characteristic curves of 0.942 and 0.858 on the CKD and HTN datasets, respectively, outperforming classic MIL (0.866 and 0.801). Attention maps highlighted lymphatic-rich regions consistent with renal pathologists’ assessments. Conclusions: This clustering-constrained, attention-based weakly supervised framework enables fully automated, reproducible quantification of renal lymphatic vessel density from WSIs, providing renal pathologists with rapid visual and numerical support for diagnosis and risk stratification in CKD and HTN.
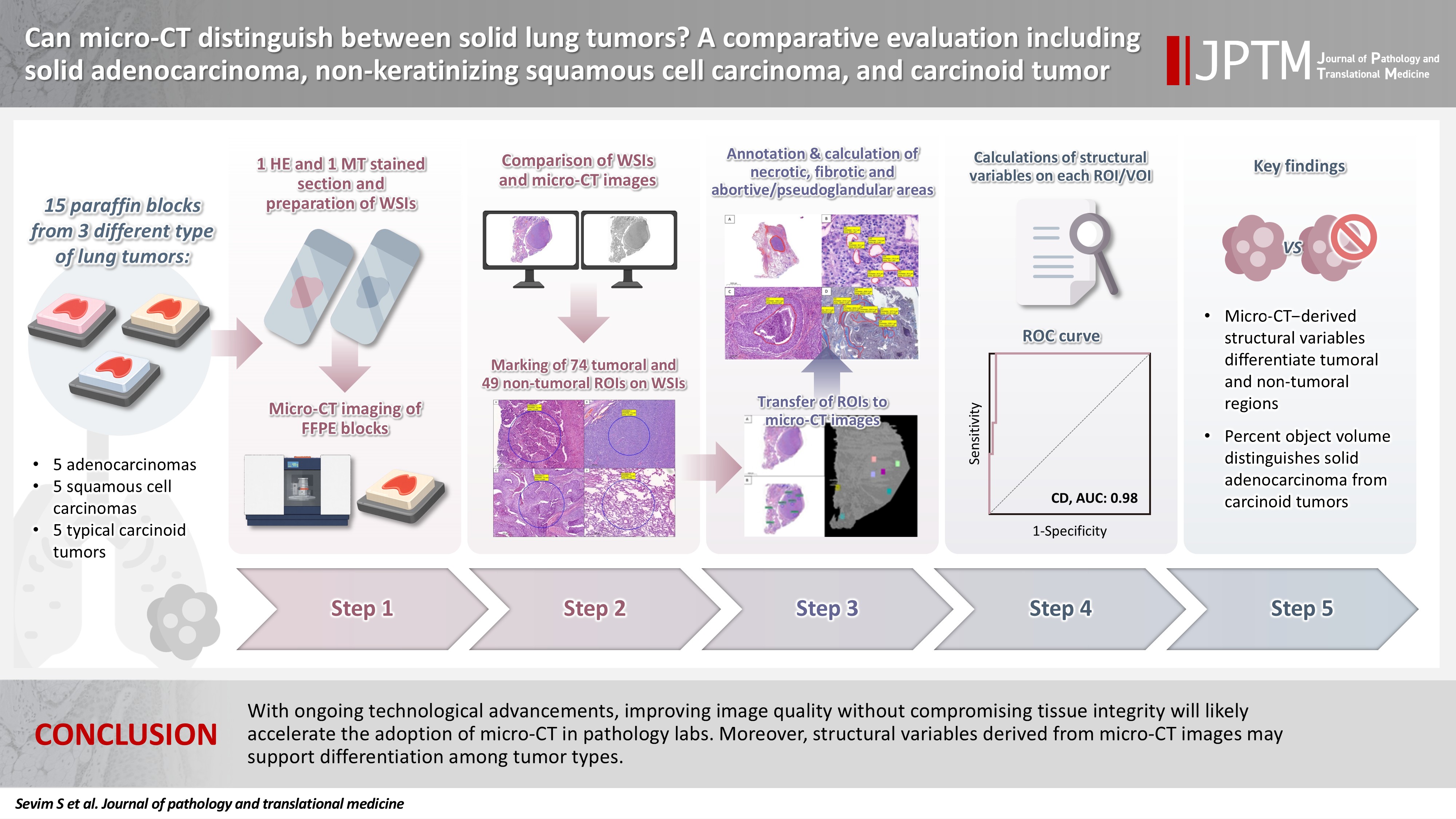
- Can micro-CT distinguish between solid lung tumors? A comparative evaluation including solid adenocarcinoma, non-keratinizing squamous cell carcinoma, and carcinoid tumor
- Selim Sevim, Serpil Dizbay Sak, Kaan Orhan, Arda Buyuksungur, Duru Karasoy, Hilal Ozakinci, Ayten Kayi Cangir
- J Pathol Transl Med. 2026;60(2):231-245. Published online March 10, 2026
- DOI: https://doi.org/10.4132/jptm.2025.12.16
- 350 View
- 40 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material - Background
Some pulmonary carcinomas display a solid pattern, and immunohistochemistry is commonly used for tumor differentiation. Micro–computed tomography (micro-CT), with its ability to produce detailed three-dimensional images using small voxel sizes, may offer additional insights. This study investigates whether three solid tumor types, solid adenocarcinoma (sAC), non-keratinizing squamous cell carcinoma, and carcinoid tumor (CaT), can be differentiated using micro-CT. Methods: Fifteen paraffin blocks, five for each type, were scanned with micro-CT (Skyscan 1275, Bruker). These images were compared to whole slide images (WSIs) of the same tumors. Consequently, tumoral (n = 74) and non-tumoral (n = 49) regions of interest (tumor ROIs [tROIs] and non-tumor ROIs [ntROIs]) were selected on the micro-CT images and evaluated in terms of certain structural variables (percent object volume, structure model index, structure thickness, structure linear density, connectivity, connectivity density, open porosity, closed porosity) to investigate whether tumors can be differentiated from normal parenchyma and from each other. Results: Although detailed images comparable to WSIs could not be obtained, it was considered an important advantage to be able to examine the entire depth of the paraffin blocks. tROIs and ntROIs could be distinguished based on all variables (p < .001). Additionally, sAC showed a notable difference from CaT in “percent object volume” (p = .011). Conclusions: With ongoing technological advancements, improving image quality without compromising tissue integrity will likely accelerate the adoption of micro-CT in pathology labs. Moreover, structural variables derived from micro-CT images may support differentiation among tumor types.
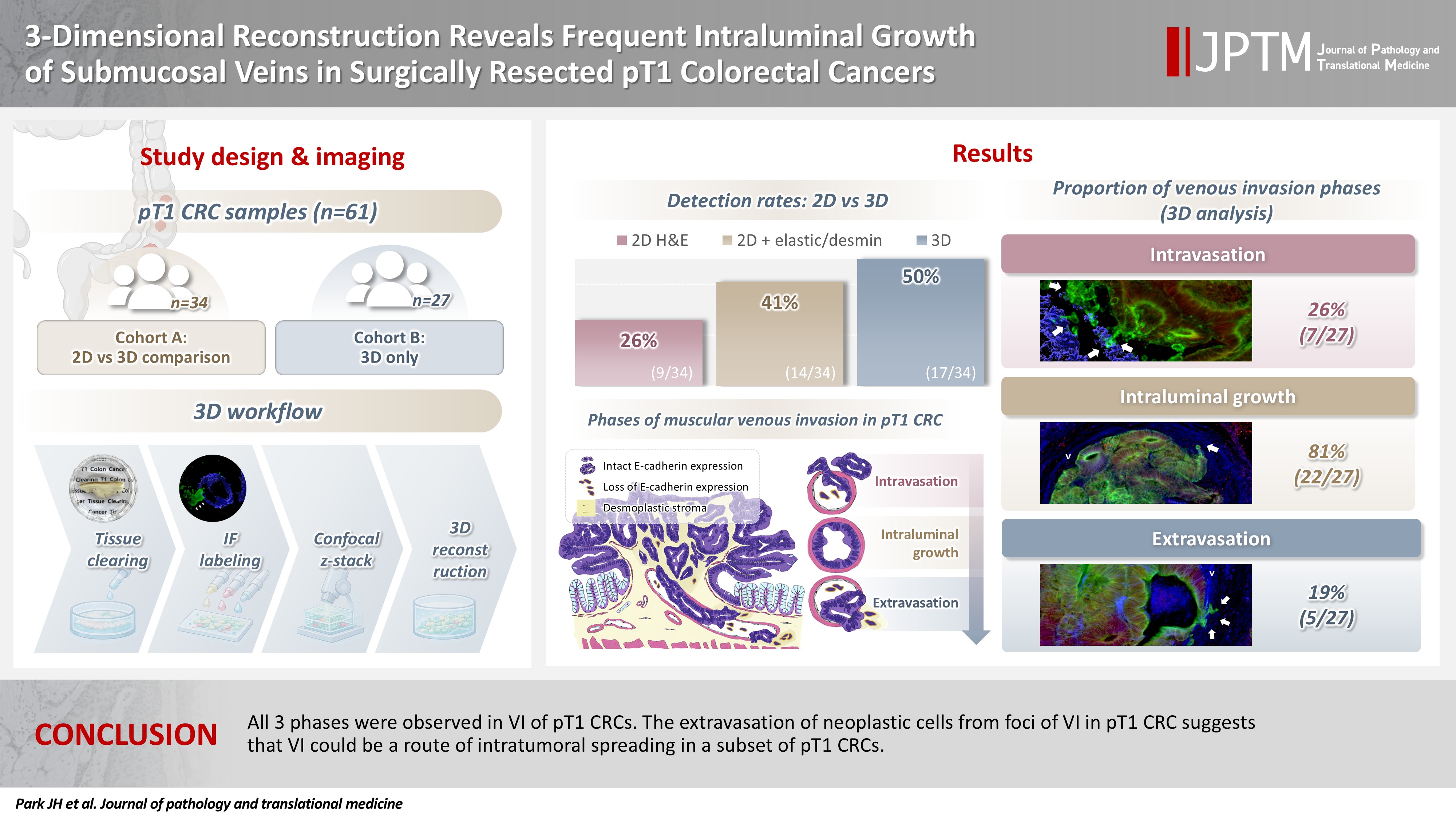
- 3-Dimensional reconstruction reveals frequent intraluminal growth of submucosal veins in surgically resected pT1 colorectal cancers
- Jihyun Park, Mi-Ju Kim, Yeon Wook Kim, Byong-Wook Lee, Junyoung Shin, Jinho Shin, Chan-Gi Pack, Dong-Hoon Yang, Jihun Kim, In Ja Park, Ralph H. Hruban, Seung-Mo Hong
- J Pathol Transl Med. 2026;60(2):246-262. Published online March 10, 2026
- DOI: https://doi.org/10.4132/jptm.2025.12.19
- 298 View
- 28 Download
-
 Abstract
Abstract
 PDF
PDF - Background
Although venous invasion (VI) is associated with distant metastasis and observed in >50% of pT2–4 colorectal cancers (CRCs), the role of VI in pT1 CRCs is not well-defined. Methods: Thirty-four surgically resected pT1 CRCs were reevaluated for 2-dimensional (2D) VI using hematoxylin and eosin (H&E)–stained slides with additional elastic and desmin immunohistochemical staining (cohort A). Additionally, 27 pT1 CRCs without knowing VI status were selected for 3-dimensional (3D) VI evaluation only (cohort B). All 61 cases (cohorts A and B) were studied in 3D using tissue clearing. Results: VI was detected more commonly in 3D (17/34, 50.0%) than in 2D H&E slide evaluation (9/34, 26.5%, p = .047). When VI was identified in 3D (27/61, 44.3%), the most common phase was that of intraluminal growth (22/27, 81.5%), followed by intravasation (7/27, 25.9%) and extravasation (5/27, 18.5%). E-cadherin expression was characterized in 3D in foci of VI and varied in each phase of invasion. Conclusions: All three phases were observed in VI of pT1 CRCs. The extravasation of neoplastic cells from foci of VI in pT1 CRC suggests that VI could be a route of intratumoral spreading in a subset of pT1 CRCs.
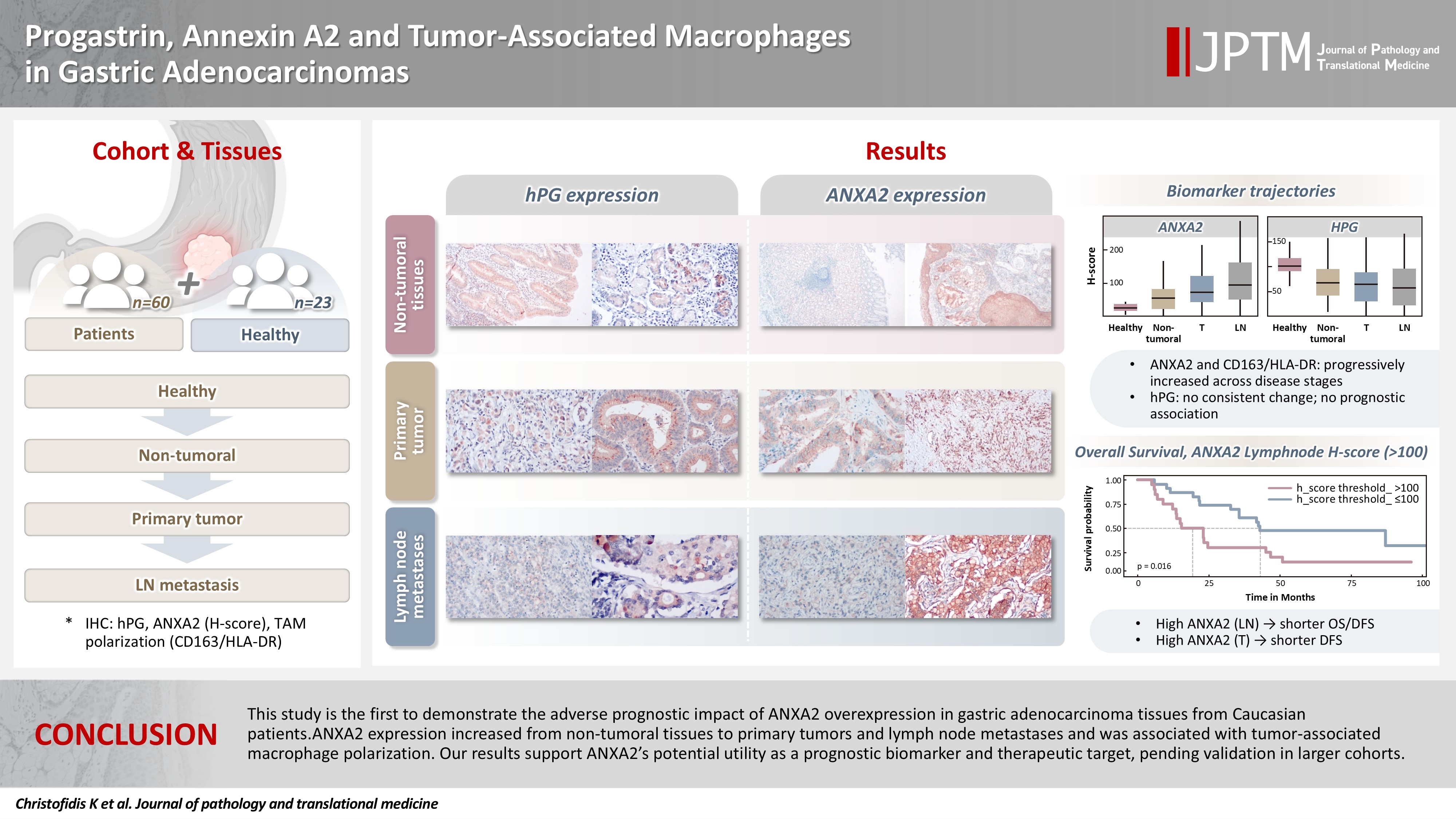
- Progastrin, annexin A2, and tumor-associated macrophages in gastric adenocarcinoma
- Konstantinos Christofidis, Rodanthi Fioretzaki, Stylianos Mavropoulos Papoudas, Nikolaos Charalampakis, Nikolaos Kavantzas, Dimitrios Schizas, Stratigoula Sakellariou
- J Pathol Transl Med. 2026;60(2):263-279. Published online March 10, 2026
- DOI: https://doi.org/10.4132/jptm.2025.12.20
- 331 View
- 43 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material - Background
Gastric adenocarcinoma is a major cause of cancer mortality worldwide, and reliable biomarkers remain insufficient. This study investigates the immunohistochemical expression of progastrin (hPG) and annexin A2 (ANXA2) and the polarization of tumor-associated macrophages in gastric adenocarcinoma to explore their potential prognostic and biological significance. Methods: A retrospective analysis was conducted on formalin-fixed, paraffin-embedded tissue samples from 60 patients with gastric adenocarcinoma (primary tumors, lymph node metastases, and non-tumoral gastric mucosa) and gastric biopsies from 23 healthy controls. The expression of hPG and ANXA2 was quantified using the H-score, and the CD163/human leukocyte antigen–DR (HLA-DR) ratio was used to represent macrophage polarization (M2/M1). Statistical analyses included non-parametric tests, Spearman correlations, Kaplan-Meier survival curves, and Cox proportional-hazards models. Results: ANXA2 expression was significantly elevated in cancer cells from primary tumors and lymph node metastases, compared with the non-tumoral gastric mucosa tissues and gastric mucosa tissues from healthy controls. ANXA2 expression increased with the tumor grade. High ANXA2 levels were associated with shorter overall and disease-free survival, but they did not have independent prognostic value. Although hPG expression correlated positively with ANXA2, it showed no significant prognostic association. The CD163/HLA-DR ratio increased with tumor progression and negatively correlated with ANXA2, but it did not influence survival outcomes. Conclusions: This study is the first to demonstrate the adverse prognostic impact of ANXA2 overexpression in gastric adenocarcinoma tissues from Caucasian patients. Our results suggest that ANXA2 might have utility as a prognostic biomarker and therapeutic target, if further large-scale studies validate and expand our findings.
Newsletter
- What's new in molecular genetic pathology 2026: emerging biomarkers for personalized cancer therapies
- Umberto Maccio
- J Pathol Transl Med. 2026;60(2):280-283. Published online January 3, 2026
- DOI: https://doi.org/10.4132/jptm.2026.01.03
- 1,229 View
- 202 Download
-
 Abstract
Abstract
 PDF
PDF - New and emerging biomarkers and current molecular assays for the most prevalent and lethal cancers worldwide—breast, lung, prostate, and colorectal cancer—are described. Notably, HER2-low breast cancer and HER2-mutated non-small cell lung cancer have recently been recognized as targetable entities. In addition, various tissue-based analyses are now available to assess prognosis and the risk of relapse in prostate cancer.

 E-submission
E-submission



 First
First Prev
Prev



