Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 55(1); 2021 > Article
-
Original Article
Interobserver diagnostic reproducibility in advanced-stage endometrial carcinoma -
Ho Jin Jung1
 , Soo Yeon Lee1
, Soo Yeon Lee1 , Jin Hwa Hong2
, Jin Hwa Hong2 , Yi Kyeong Chun1
, Yi Kyeong Chun1
-
Journal of Pathology and Translational Medicine 2021;55(1):43-52.
DOI: https://doi.org/10.4132/jptm.2020.10.04
Published online: December 3, 2020
1Department of Pathology, Korea University Guro Hospital, Korea University College of Medicine, Seoul, Korea
2Department of Obstetrics and Gynecology, Korea University Guro Hospital, Korea University College of Medicine, Seoul, Korea
- Corresponding Author: Yi Kyeong Chun, MD, Department of Pathology, Korea University Guro Hospital, Korea University College of Medicine, 148 Gurodong-ro, Guro-gu, Seoul 08308, Korea, Tel: +82-2-2626-1472, Fax: +82-2-2626-1486, E-mail: ykcmd@naver.com
© 2021 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (https://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Figure & Data
References
Citations

- Diagnostic Accuracy of Endometrial Sampling Methods for Determining Histologic Type and Grade in Endometrial Cancer: A Retrospective Cohort Study
Dina Gumin, Avishalom Sharon, Susana Mustafa Mikhail, Inshirah Sgayer, Raneen Abushqara, Lior Lowenstein, Ala Aiob
Cureus.2025;[Epub] CrossRef - Accuracy of endometrial sampling in the diagnosis of endometrial cancer: a multicenter retrospective analysis of the JAGO-NOGGO
Zaher Alwafai, Maximilian Heinz Beck, Sepideh Fazeli, Kathleen Gürtler, Christine Kunz, Juliane Singhartinger, Dominika Trojnarska, Dario Zocholl, David Johannes Krankenberg, Jens-Uwe Blohmer, Jalid Sehouli, Klaus Pietzner
BMC Cancer.2024;[Epub] CrossRef - Deep Learning for Grading Endometrial Cancer
Manu Goyal, Laura J. Tafe, James X. Feng, Kristen E. Muller, Liesbeth Hondelink, Jessica L. Bentz, Saeed Hassanpour
The American Journal of Pathology.2024; 194(9): 1701. CrossRef - Application of NGS molecular classification in the diagnosis of endometrial carcinoma: A supplement to traditional pathological diagnosis
Qunxian Rao, Jianwei Liao, Yangyang Li, Xin Zhang, Guocai Xu, Changbin Zhu, Shengya Tian, Qiuhong Chen, Hui Zhou, Bingzhong Zhang
Cancer Medicine.2023; 12(5): 5409. CrossRef - Risk Stratification of Endometrial Cancer Patients: FIGO Stage, Biomarkers and Molecular Classification
Jenneke C. Kasius, Johanna M. A. Pijnenborg, Kristina Lindemann, David Forsse, Judith van Zwol, Gunnar B. Kristensen, Camilla Krakstad, Henrica M. J. Werner, Frédéric Amant
Cancers.2021; 13(22): 5848. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
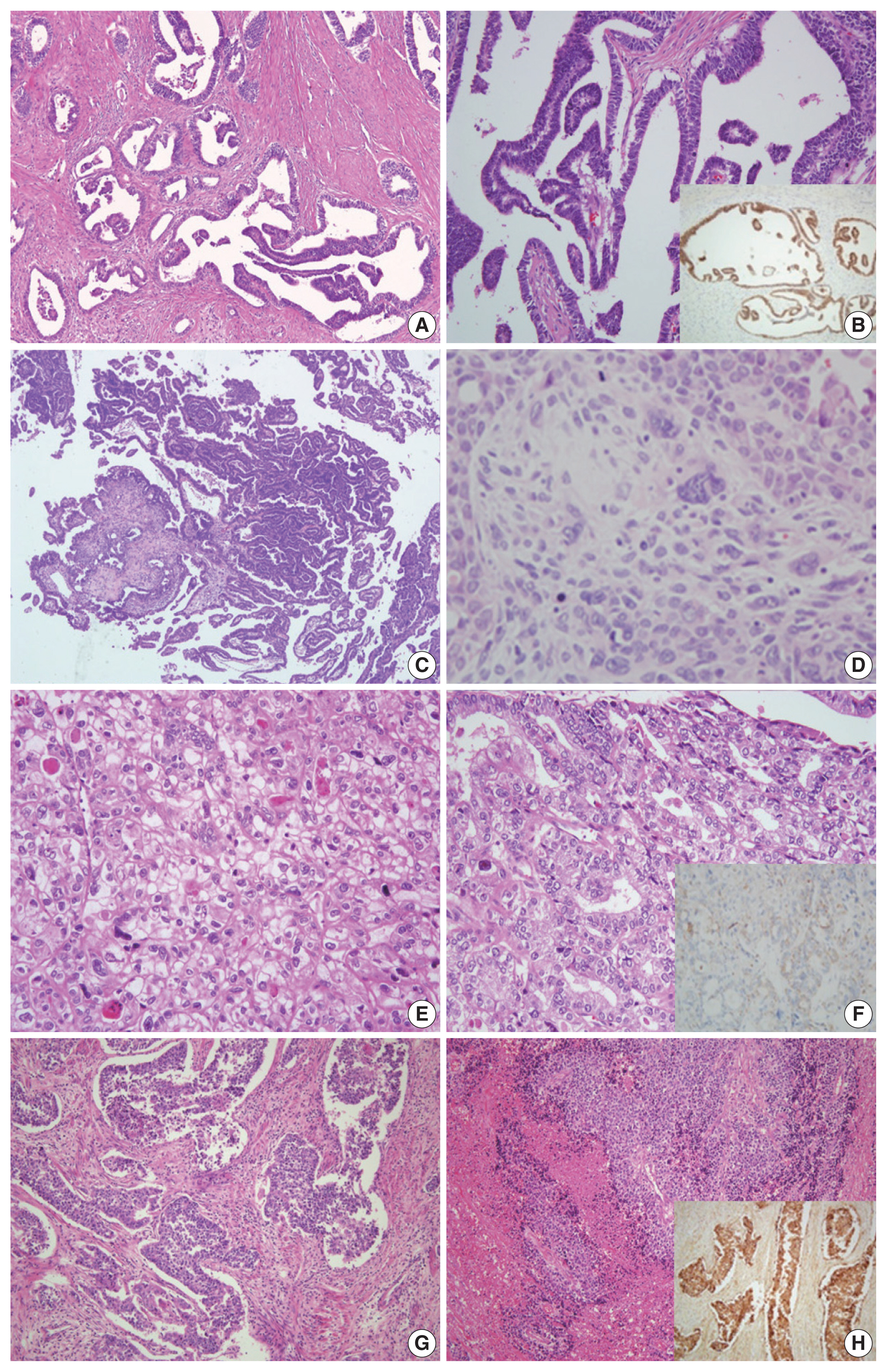
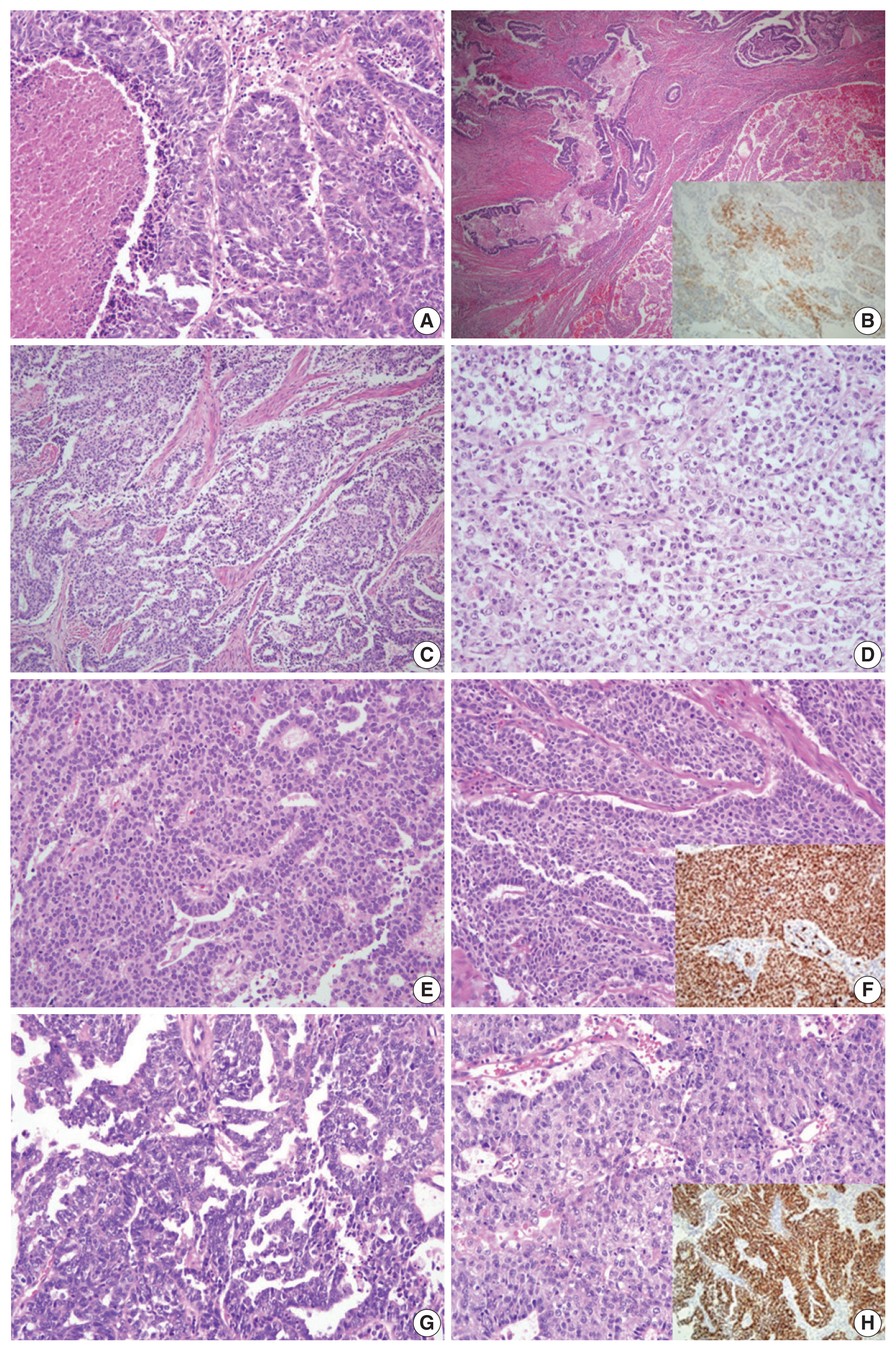
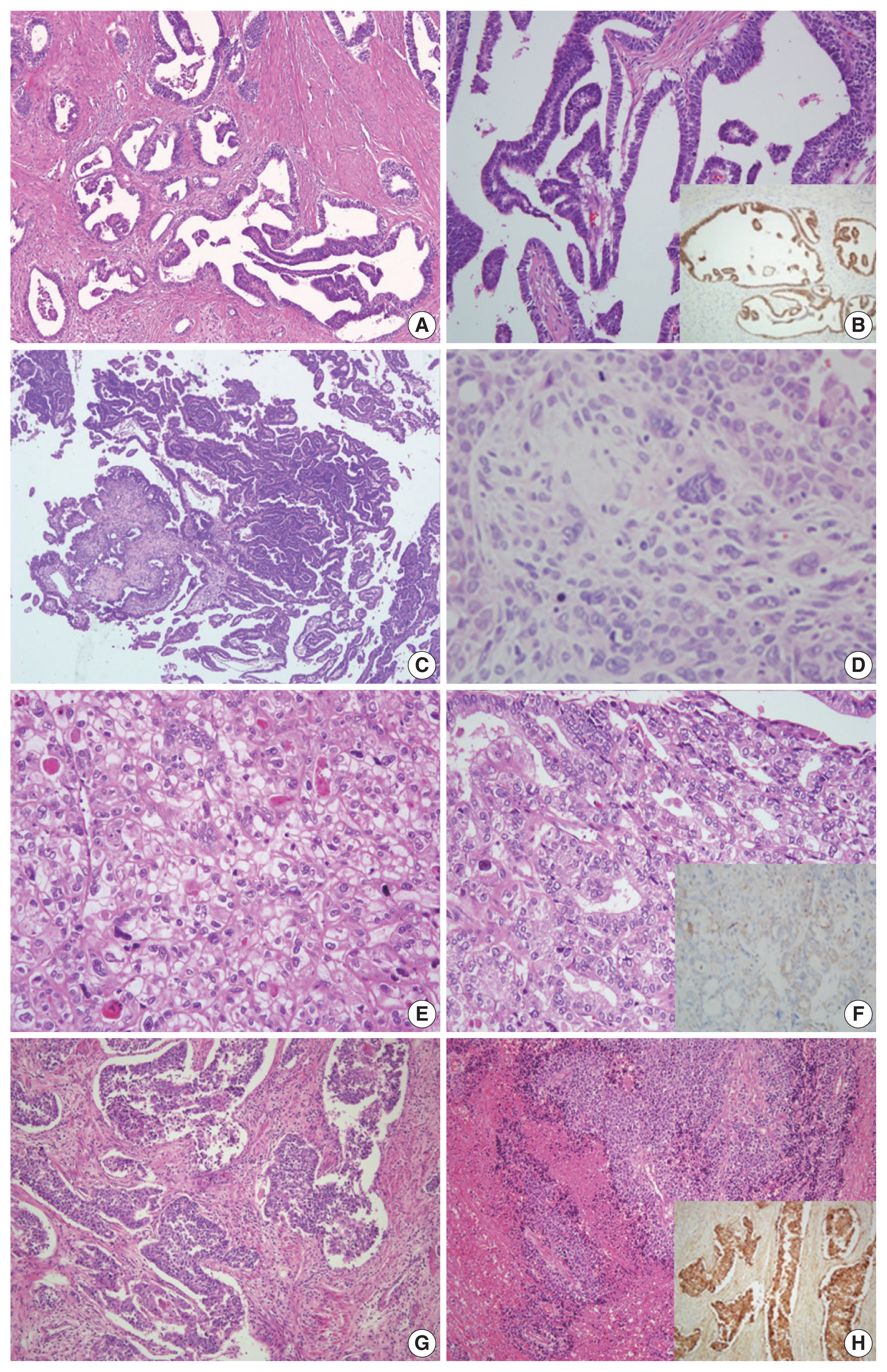
Fig. 1
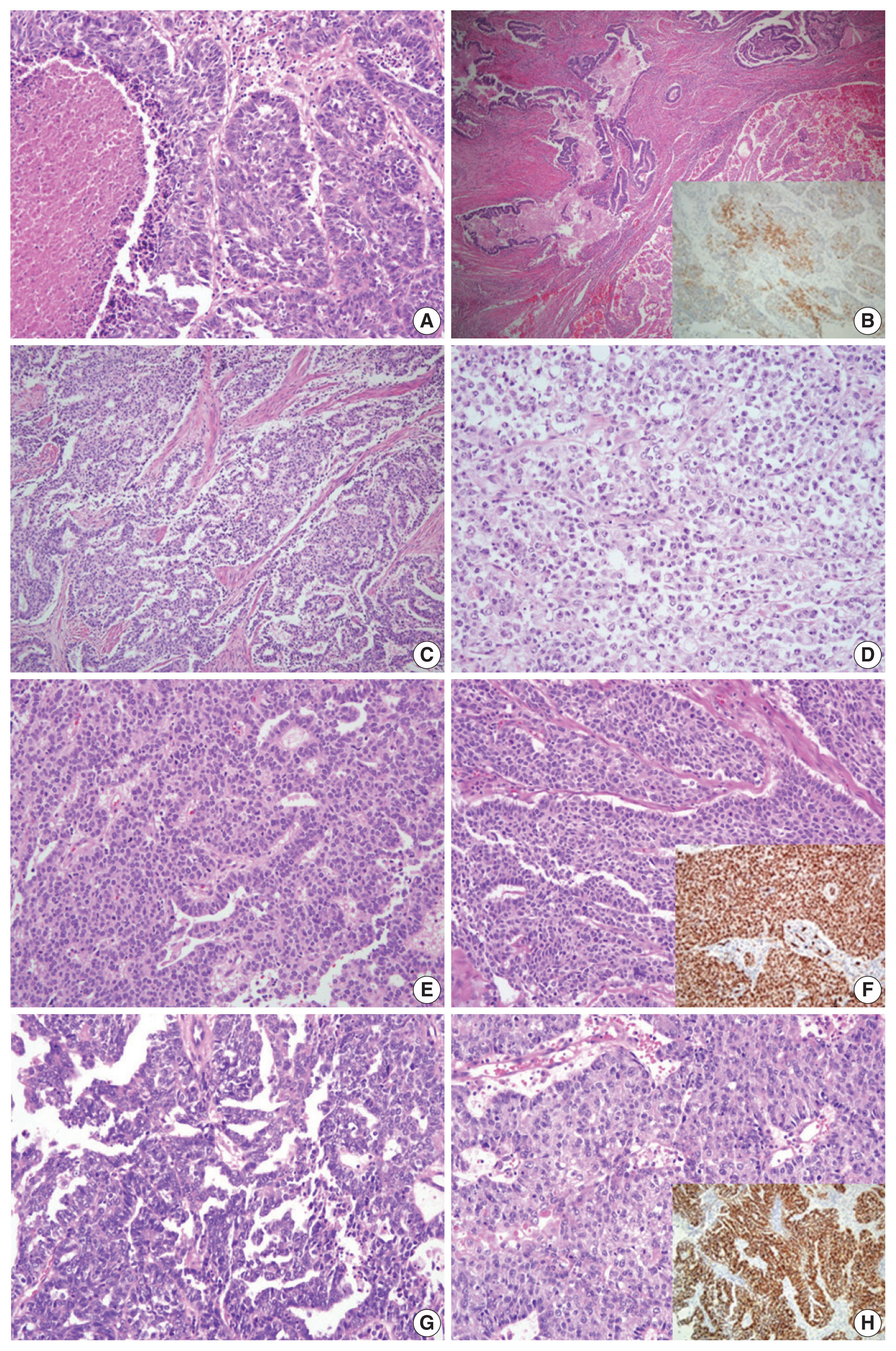
Fig. 2
| Case No. | Original diagnosis | Primary diagnosis | ||
|---|---|---|---|---|
|
| ||||
| Reviewer 1 | Reviewer 2 | Reviewer 3 | ||
| 1 | G1 EC | MesL | Low-grade EC | Low-grade EC |
| 2 | SC | SC vs. CS | SC | SC |
| 3 | G3 EC | G3 EC | CC | G3 EC |
| 4 | G3 EC | LCNEC | G3 EC | LCNEC |
| 5 | G3 EC | Mixed (G3 EC + CC + LCNEC) | G3 EC | SC |
| 6 | G3 EC | DD | DD | SC |
| 7 | G3 EC | G3 EC vs. SC | SC | G3 EC |
| 8 | SC | G3 EC vs. SC | SC | SC |
| 9 | G1 EC | Low-grade EC | Low-grade EC | Low-grade EC |
| 10 | G2 EC | Low-grade EC | Low-grade EC | Low-grade EC |
| 11 | G2 EC | Low-grade EC | Low-grade EC | Low-grade EC |
| 12 | G2 EC | Low-grade EC | Low-grade EC | Low-grade EC |
| 13 | G3 EC | G3 EC | G3 EC | G3 EC |
| 14 | G3 EC | G3 EC | G3 EC | G3 EC |
| 15 | G3 EC | G3 EC | G3 EC | G3 EC |
| 16 | G3 EC | G3 EC | G3 EC | G3 EC |
| 17 | G3 EC | G3 EC | G3 EC | G3 EC |
| 18 | SC | SC | SC | SC |
| 19 | SC | SC | SC | SC |
| 20 | SC | SC | SC | SC |
| 21 | CC | CC | CC | CC |
| Case No. | Original diagnosis | p53 | ER | PR | Others |
|---|---|---|---|---|---|
| 1 | G1 EC | Wild type | (−) | (+), 30% | GATA3 (+), TTF-1 (+), CD10 (−) |
| 2 | SC | Aberrant | (+), 10% | (+), 5% | p16 (+, diffuse in carcinoma) CK (+, diffuse in carcinoma, focal in sarcoma) Vimentin (+ in sarcoma) WT1 (+ in carcinoma) |
| 3 | G3 EC | Wild type | (−) | (−) | Napsin A (+), HNF-1β (−) |
| 4 | G3 EC | Wild type | (+), 30% | (−) | CD56 (+), chromogranin (+), synaptophysin (−) |
| 5 | G3 EC | Wild type | (+), 20% | (+), 15% | CD56 (+), synaptophysin (+), chromogranin (+, focal) PTEN (+), WT1 (−) |
| 6 | G3 EC | Wild type | (+), 10% | (+), 5% | pMMR |
| 7 | G3 EC | Null | (−) | (−) | p16 (+, diffuse), pMMR, WT1 (+), PTEN (+) |
| 8 | SC | Aberrant | (−) | (−) | p16 (+, diffuse), pMMR, WT1 (+, focal) |
| Case No. | Original diagnosis | Primary diagnosis | 2nd diagnosis | Consensus diagnosis | ||||
|---|---|---|---|---|---|---|---|---|
|
|
| |||||||
| Reviewer 1 | Reviewer 2 | Reviewer 3 | Reviewer 1 | Reviewer 2 | Reviewer 3 | |||
| 1 | G1 EC | MesL | Low-grade EC | Low-grade EC | MesL | MesL | MesL | MesL |
| 2 | SC | SC vs. CS | SC | SC | CS | CS | CS | CS |
| 3 | G3 EC | G3 EC | CC | G3 EC | CC | CC | CC | CC |
| 4 | G3 EC | LCNEC | G3 EC | LCNEC | LCNEC | LCNEC | LCNEC | LCNEC |
| 5 | G3 EC | Mixed (G3 EC + CC + LCNEC) | G3 EC | SC | LCNEC | LCNEC | Mixed (low-grade EC + LCNEC) | LCNEC |
| 6 | G3 EC | DD | DD | SC | DD | DD | G3 EC | DD |
| 7 | G3 EC | G3 EC vs. SC | SC | G3 EC | G3 EC vs. SC | SC | SC | SC |
| 8 | SC | SC vs. G3 EC | SC | SC | G3 EC | SC | G3 EC | SC |
| Case No. | Original diagnosis | Consensus diagnosis |
|---|---|---|
| 1 | G1 EC | MesL |
| 2 | SC | CS |
| 3 | G3 EC | CC |
| 4 | G3 EC | LCNEC |
| 5 | G3 EC | LCNEC |
| 6 | G3 EC | DD |
| 7 | G3 EC | SC |
| 8 | SC | SC |
| 9 | G1 EC | Low-grade EC |
| 10 | G2 EC | Low-grade EC |
| 11 | G2 EC | Low-grade EC |
| 12 | G2 EC | Low-grade EC |
| 13 | G3 EC | G3 EC |
| 14 | G3 EC | G3 EC |
| 15 | G3 EC | G3 EC |
| 16 | G3 EC | G3 EC |
| 17 | G3 EC | G3 EC |
| 18 | SC | SC |
| 19 | SC | SC |
| 20 | SC | SC |
| 21 | CC | CC |
G1, grade 1; G2, grade 2; G3, grade 3; EC, endometrioid carcinoma; MesL, mesonephric-like adenocarcinoma; SC, serous carcinoma; CS, car-cinosarcoma; CC, clear cell carcinoma; LCNEC, large cell neuroendocrine carcinoma; Mixed, mixed cell adenocarcinoma; DD, dedifferentiated carcinoma.
ER; estrogen receptor; PR, progesterone receptor; EC, endometrioid carcinoma; G1, grade 1; G3, grade 3; TTF-1, thyroid transcription factor-1; SC, serous carcinoma; CK, cytokeratin; pMMR, proficient mismatch repair.
G1, grade 1; G2, grade 2; G3, grade 3; EC, endometrioid carcinoma; MesL, mesonephric-like adenocarcinoma; SC, serous carcinoma; CS, carcinosarcoma; LCNEC, large cell neuroendocrine carcinoma; Mixed, mixed cell adenocarcinoma; CC, clear cell carcinoma; DD, dedifferentiated carcinoma. Consensus diagnosis was made after next-generation sequencing analysis.
G1, grade 1; G2, grade 2; G3, grade 3; EC, endometrioid carcinoma; MesL, mesonephric-like adenocarcinoma; SC, serous carcinoma; CS, carcinosarcoma; CC, clear cell carcinoma; LCNEC, large cell neuroendocrine carcinoma; DD, dedifferentiated carcinoma.

 E-submission
E-submission