Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 49(1); 2015 > Article
-
Brief Case Report
Squamous Cell Carcinoma of the Seminal Vesicle from Zinner Syndrome: A Case Report and Review of Literature - Younghoon Kim, Hae Woon Baek, Eunoh Choi, Kyung Chul Moon
-
Journal of Pathology and Translational Medicine 2015;49(1):85-88.
DOI: https://doi.org/10.4132/jptm.2014.10.28
Published online: January 15, 2015
Department of Pathology, Seoul National University College of Medicine, Seoul, Korea
- Corresponding Author: Kyung Chul Moon, M.D. Department of Pathology, Kidney Research Institute, Medical Research Center, Seoul National University College of Medicine, 103 Daehak-ro, Jongno-gu, Seoul 110-799, Korea Tel: +82-2-2072-1767, Fax: +82-2-743-5530, E-mail: blue7270@snu.ac.kr
• Received: June 30, 2014 • Revised: October 22, 2014 • Accepted: October 24, 2014
© 2015 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- A 41-year-old male was admitted to our hospital due to gross hematuria for two and a half months. Twelve years previous, the patient underwent transurethral resection, suprapubic cystostomy, and urethral sounds to cure a right seminal vesicle cyst with multiple stones and obstruction of the right ejaculatory duct. At that time, a biopsy diagnosed the seminal vesicle cyst as an epidermal cyst, a benign cyst lined with a thin layer of squamous epithelium. A week after his current admission, perineal and scrotal pains newly developed. Digital rectal examination detected tenderness with a hard and highly elevated posterior prostate compressing the rectal wall. No prostate-specific antigen elevation was detected and urine cytology was negative for malignant cells. Computed tomography (CT) urography revealed hypoplastic change in the right kidney, a 4.9-cm-sized right seminal vesicle cyst with a thickened wall, and benign prostate hyperplasia (Fig. 1A). The patient was diagnosed with Zinner syndrome with a seminal vesicle cyst. Transrectal sonography detected a 3.8-cm-sized hypoechoic lesion at the left transitional zone of the prostate showing a bulging contour and prominent vascularity, which favored chronic prostatitis over malignancy. Treatment included not only palliative medication but also aggressive procedures such as nerve blocking for pain control. Six months after the initial onset of gross hematuria, another transrectal biopsy was done and the specimen was pathologically diagnosed as poorly differentiated carcinoma. CT urography and magnetic resonance imaging demonstrated a bulging mass at the left prostate gland, which had increased in size compared with images taken three months previous (Fig. 1B). The mass showed signs of internal necrosis and anorectal adhesion, but did not show anorectal invasion. A 4.7-cm-sized seminal vesicle cyst at the right side and necrotic lymph nodes at the left external iliac area were also observed. Through surgery, the urinary bladder, prostate, and bilateral seminal vesicles were removed en bloc. On gross examination, the specimen consisted of an 11.0×6.0×5.0-cm-sized multinodular mass with a tan-white cut surface accompanied by hemorrhage and necrosis. Adjacent to the tumor, the seminal vesicle cyst had a smooth mucosa measuring 5.0×3.0 cm. Under the microscope, squamous metaplasia was observed in the seminal vesicle cyst lining (Fig. 2A). Microscopically observed, the tumor was a poorly differentiated carcinoma (Fig. 2B). Immunohistochemical analysis of the tumor was positive for p63 (Fig. 3A) and negative for cytokeratin 7 (Fig. 3B), cytokeratin 20 (Fig. 3C), and carcinoembryonic antigen (Fig. 3D). Although the specimen displayed some immunoactivity for vimentin (Fig. 3E), the staining was positive in macrophages, as reported in murine seminal vesicle carcinoma [5]. Histochemical studies (periodic acid–Schiff [PAS] and mucicarmine) were positive for mucin (Fig. 3F). The tumor was pathologically diagnosed as poorly differentiated squamous cell carcinoma arising from the seminal vesicle cyst. The involvement of carcinoma was detected in rectal and right ureter tissues, but no lymph node metastasis was observed. The postoperative course had no further events. The patient was discharged and scheduled for adjuvant concurrent chemoradiation therapy.
CASE REPORT
- Our patient suffered from Zinner syndrome, a male counterpart of Mayer-Rokitansky-Küster-Hauser (MRKH) syndrome which occurs in females. MRKH syndrome is reportedly associated with ovarian cancer and renal cell carcinoma in the genitourinary tract [6,7]. Two cases of adenocarcinoma arising from a seminal vesicle cyst in Zinner syndrome patients have been previously reported [8,9]. Table 1 summarizes those cases as well as the present case.
- In the current case, no diagnostic tool was able to detecting the hidden malignancy until the inflammation subsided. Moreover, poorly differentiated and wide-spread features of the malignancy made it difficult to determine the exact origin of the tumor before surgery. In this case, squamous metaplasia was pathologically proven 12 years prior to the discovery of the malignancy, which clarifies the origin of the tumor. By contrast, in all three previously reported cases of squamous carcinoma arising from the seminal vesicle, squamous metaplastic foci were diagnosed simultaneously with the carcinoma [2-4]. Although tumor cells in the present case were positive for mucin in PAS and mucicarmine staining, it is not uncommon for a squamous cell carcinoma to express mucin content [10].
- To the best of our knowledge, this is the first report of a squamous cell carcinoma of the seminal vesicle developed in a Zinner syndrome patient. Our case indicates that long-term observation and thorough evaluation are mandatory for patients with Zinner syndrome expressing nonspecific but rapid and progressive urogenital symptoms.
DISCUSSION
Fig. 1.Computed tomography urography. (A) Images taken three months before the surgery demonstrate a 4.9-cm-sized right seminal vesicle cyst (arrowhead) and an exophytic tumor measuring up to 3 cm (arrow). (B) Images taken three months later show a 4.7-cm-sized seminal vesicle cyst (arrowhead) with an enlarged mass (arrow) bulging to the left prostate gland (6.1 cm).
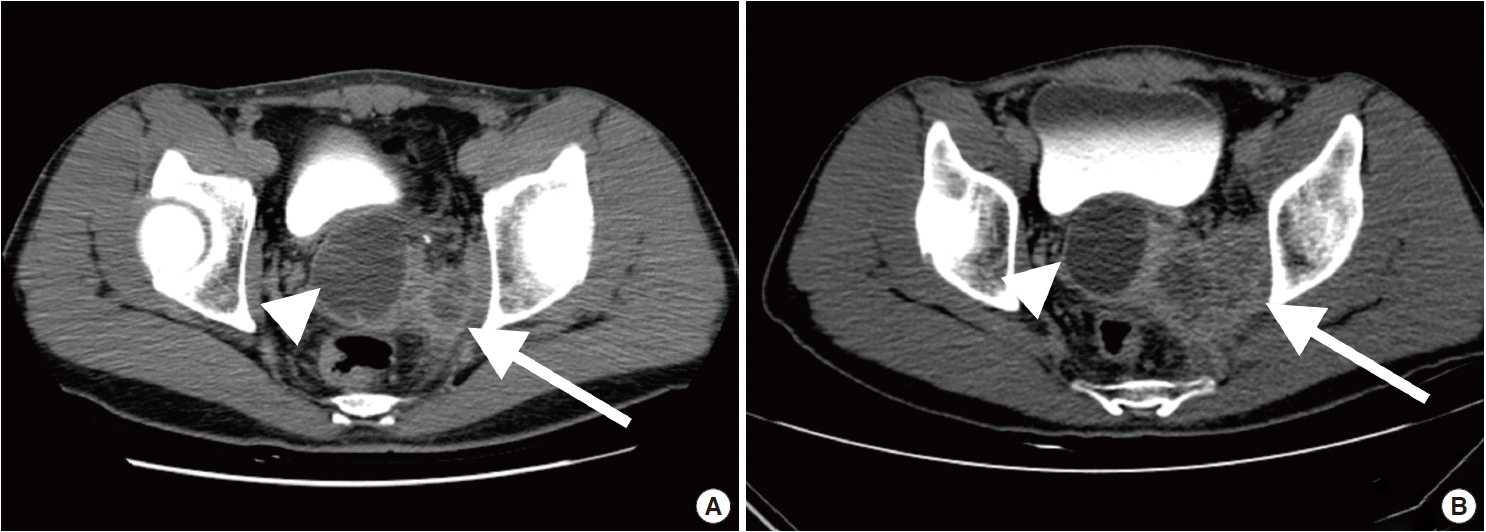

Fig. 2.Histopathologic findings of the right seminal vesicle cyst. The specimen shows squamous metaplasia (insert), glandular tissues (A), and a poorly differentiated carcinoma (B).
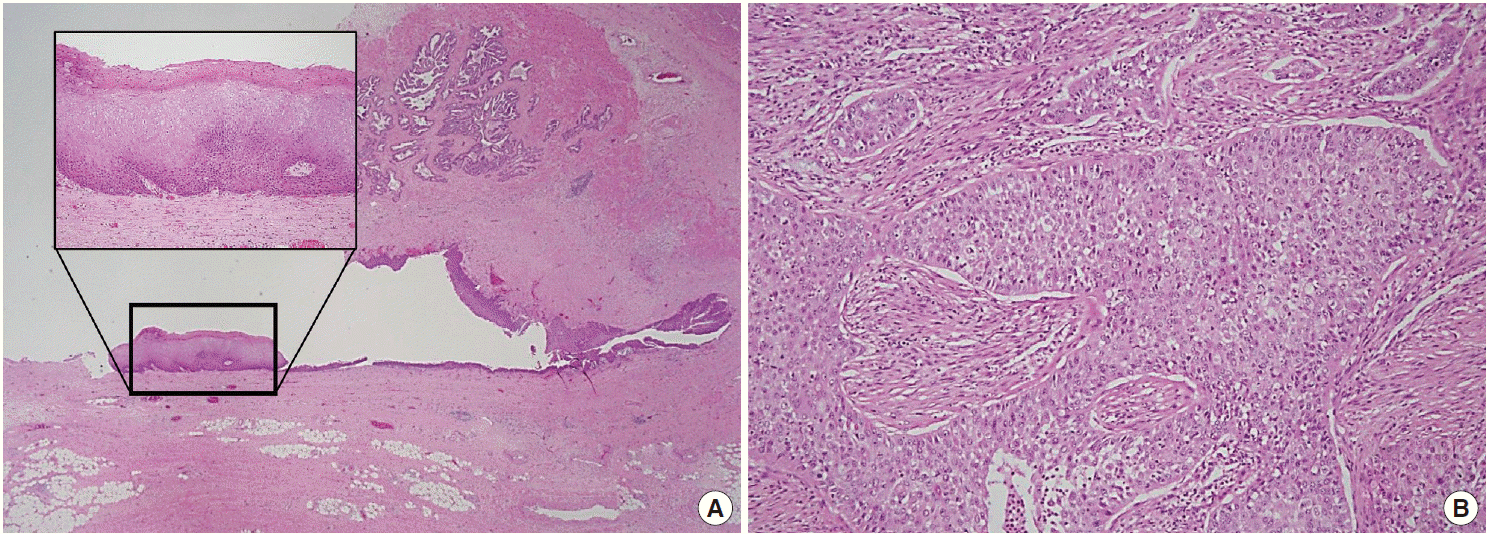

Fig. 3.Immunohistochemistry and histochemical staining for phenotyping of the carcinoma. Tumor cells are positive for p63 (A), and negative for cytokeratin 7 (B), cytokeratin 20 (C), and carcinoembryonic antigen (D). Vimentin shows focal positivity in macrophages (E), and periodic acid–Schiff staining is positive for mucin (F).

Table 1.Summary of reported cases of carcinomas arising from a seminal vesicle cyst in Zinner syndrome patients
| Okada et al. (1992) [8] | Lee et al. (2007) [9] | Present case | |
|---|---|---|---|
| Age (yr) | 17 | 41 | 41 |
| Chief complaint | Lower abdominal mass and dysuria | Hematuria | Hematuria |
| Past medical history | None | Prostatitis | Squamous metaplasia, stone formation and chronic inflammation of the seminal vesicle |
| Seminal vesicle cyst detection | Latest admission | Latest admission | Twelve years ago |
| Location | Right seminal vesicle cyst | Left seminal vesicle cyst | Right seminal vesicle cyst |
| Pathologic diagnosis | Papillary adenocarcinoma | Mucinous adenocarcinoma | Poorly differentiated squamous cell carcinoma |
| Prostate-specifi antigen | Normal | Normal | Normal |
- 1. Lorber G, Pizov G, Gofrit ON, Pode D. Seminal vesicle cystadenoma: a rare clinical perspective. Eur Urol 2011; 60: 388-91. ArticlePubMed
- 2. Tabata K, Irie A, Ishii D, Yanagisawa N, Iwamura M, Baba S. Primary squamous cell carcinoma of the seminal vesicle. Urology 2002; 59: 445.Article
- 3. Yanagisawa N, Saegusa M, Yoshida T, Okayasu I. Squamous cell carcinoma arising from a seminal vesicular cyst: possible relationship between chronic inflammation and tumor development. Pathol Int 2002; 52: 249-53. ArticlePubMed
- 4. Wang J, Yue X, Zhao R, Cheng B, Wazir R, Wang K. Primary squamous cell carcinoma of seminal vesicle: an extremely rare case report with literature review. Int Urol Nephrol 2013; 45: 135-8. ArticlePubMedPDF
- 5. Shoda T, Mitsumori K, Imazawa T, et al. A spontaneous seminal vesicle adenocarcinoma in an aged F344 rat. Toxicol Pathol 1998; 26: 448-51. ArticlePubMedPDF
- 6. Ghirardini G, Magnani A. Mayer-Rokitansky-Küster-Hauser syndrome and ovarian cancer: report of a case. Clin Exp Obstet Gynecol 1995; 22: 247-8. PubMed
- 7. Mermerkaya M, Burgu B, Hamidi N, et al. Mayer-Rokitansky-Küster-Hauser syndrome accompanied by renal cell carcinoma: a case report. J Pediatr Hematol Oncol 2013; 35: e309-10. PubMed
- 8. Okada Y, Tanaka H, Takeuchi H, Yoshida O. Papillary adenocarcinoma in a seminal vesicle cyst associated with ipsilateral renal agenesis: a case report. J Urol 1992; 148: 1543-5. ArticlePubMed
- 9. Lee BH, Seo JW, Han YH, Kim YH, Cha SJ. Primary mucinous adenocarcinoma of a seminal vesicle cyst associated with ectopic ureter and ipsilateral renal agenesis: a case report. Korean J Radiol 2007; 8: 258-61. ArticlePubMedPMC
- 10. Ocque R, Tochigi N, Ohori NP, Dacic S. Usefulness of immunohistochemical and histochemical studies in the classification of lung adenocarcinoma and squamous cell carcinoma in cytologic specimens. Am J Clin Pathol 2011; 136: 81-7. ArticlePubMed
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- Zinner Syndrome: Case report of atypical symptoms and literature
Alejandro Acuña-Pacheco, Eduardo González-Rojas, Pedro Iván Aguilar-Ordaz, Joel Porfirio Rodelo-López, Benjamin Bueno-Mendoza, Israel Hernández-Rivera, Jose Rene Jungfermann-Guzman, Jesús Rodolfo Favela-Camacho
Urology Case Reports.2025; 60: 102986. CrossRef - Zinner syndrome in pediatric patients: rare disease leading to challenging management
Ottavio Adorisio, Cinzia Orazi, Lorenzo Maria Gregori, Francesco De Peppo, Massimiliano Silveri
Frontiers in Pediatrics.2024;[Epub] CrossRef - Zinner syndrome: a rare diagnosis in infancy
Joanne Michelle Oida Rose, Ravi Banthia, Zain Tamboli, Hira Lal
BMJ Case Reports.2022; 15(5): e248558. CrossRef - Classifying seminal vesicle cysts in the diagnosis and treatment of Zinner syndrome: A report of six cases and review of available literature
Zhengwu Tan, Bing Li, Lan Zhang, Ping Han, Haitao Huang, Andrew Taylor, Xin Li
Andrologia.2020;[Epub] CrossRef - Incidental imaging findings suggesting Zinner syndrome in a young patient with pulmonary embolism: A case report
Benedikt Hergan, Franz A. Fellner, Kaveh Akbari
Radiology Case Reports.2020; 15(4): 437. CrossRef - Ectopic ureter associated with Zinner’s syndrome in a kidney recipient: case report and literature review
Korhan Tuncer, Gizem Kilinc, Ismail Sert, Goksever Akpinar, Cem Tugmen
Revista da Associação Médica Brasileira.2020; 66(5): 692. CrossRef - Zinner’s Syndrome: A Rare Diagnosis of Dysuria Based on Imaging
Ahmed Ibrahimi, Abdelmoughit Hosni, Idriss Ziani, Fatima Zahra Laamrani, Hachem El Sayegh, Laila Jroundi, Lounis Benslimane, Yassine Nouini, Apul Goel
Case Reports in Urology.2020; 2020: 1. CrossRef - Primary squamous cell carcinoma of the seminal vesicle
Lu Fang, Qian Hong, Lei Chen, Yi Wang, Liang-Kuan Bi, Dong-Dong Xie, De-Xin Yu
Medicine.2019; 98(12): e14788. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
Squamous Cell Carcinoma of the Seminal Vesicle from Zinner Syndrome: A Case Report and Review of Literature
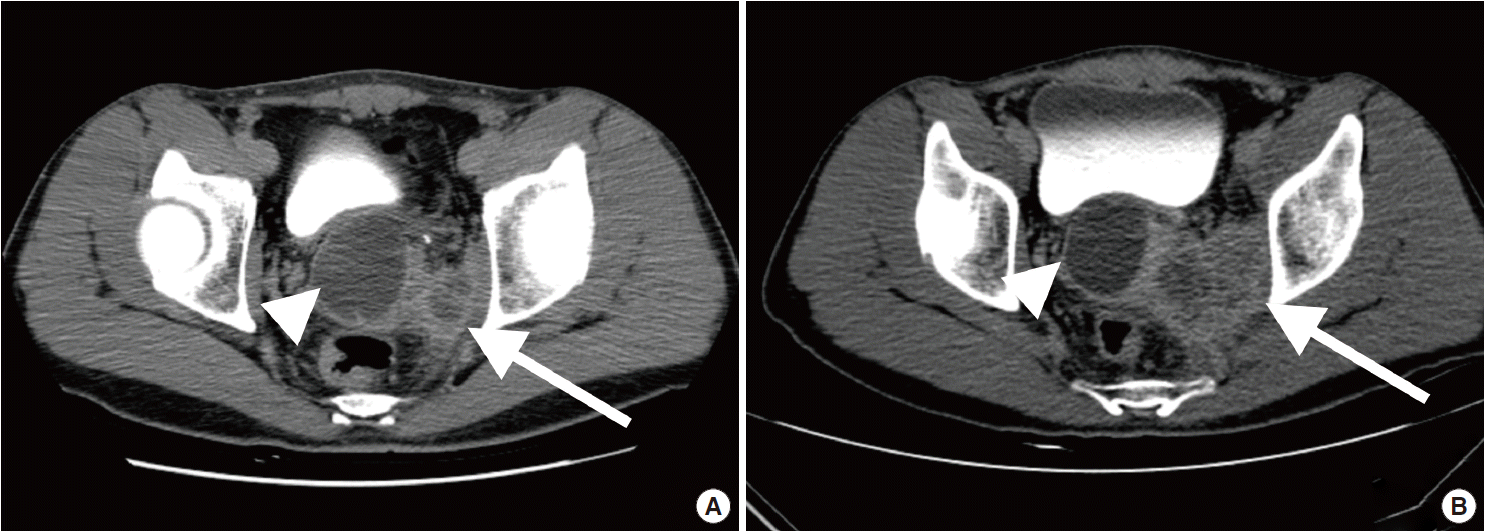
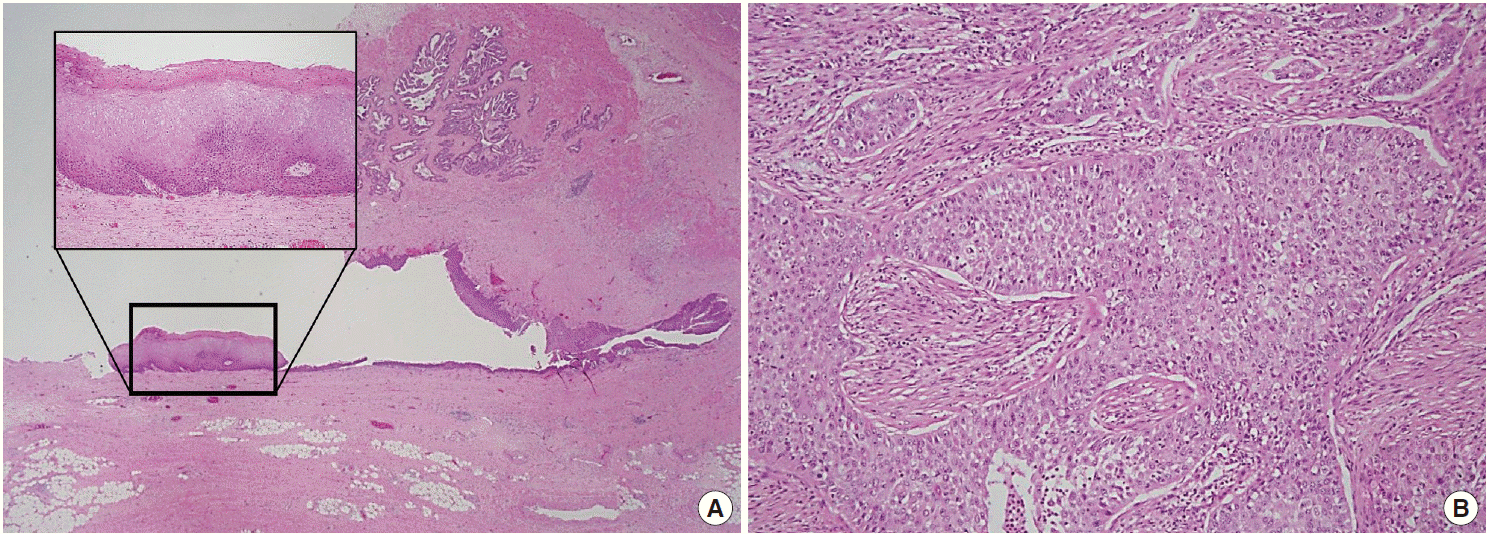
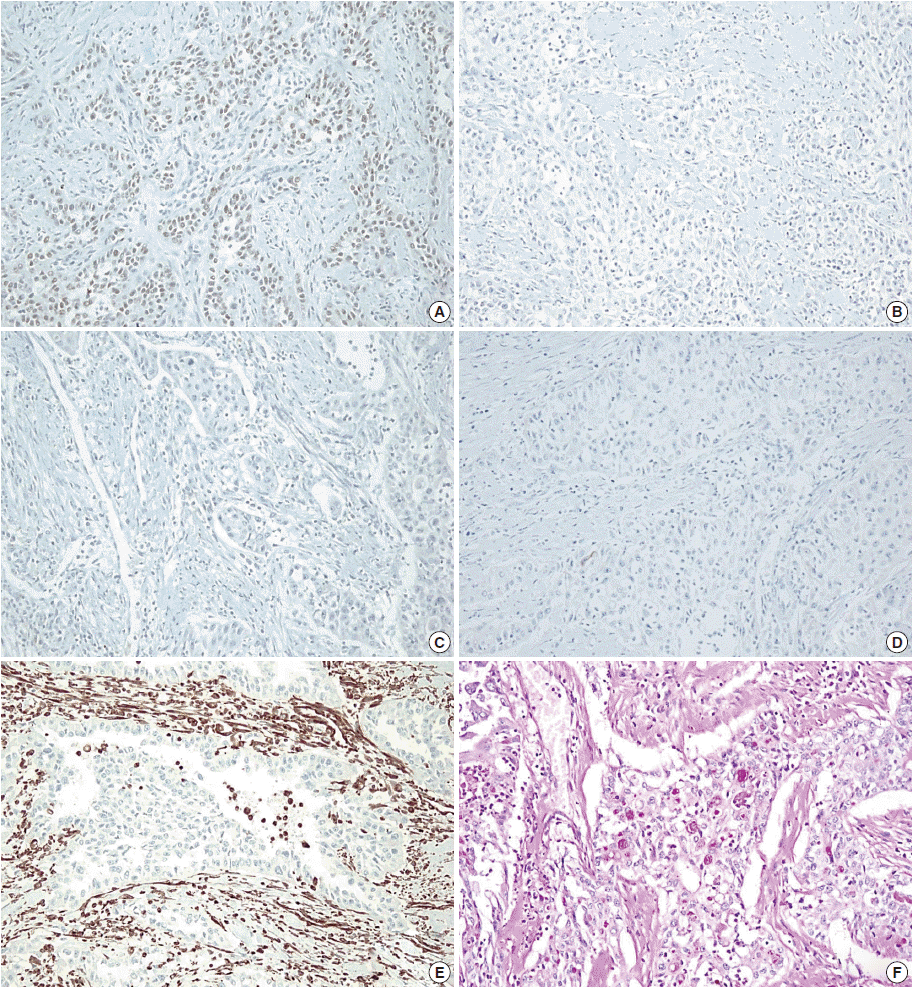
Fig. 1. Computed tomography urography. (A) Images taken three months before the surgery demonstrate a 4.9-cm-sized right seminal vesicle cyst (arrowhead) and an exophytic tumor measuring up to 3 cm (arrow). (B) Images taken three months later show a 4.7-cm-sized seminal vesicle cyst (arrowhead) with an enlarged mass (arrow) bulging to the left prostate gland (6.1 cm).
Fig. 2. Histopathologic findings of the right seminal vesicle cyst. The specimen shows squamous metaplasia (insert), glandular tissues (A), and a poorly differentiated carcinoma (B).
Fig. 3. Immunohistochemistry and histochemical staining for phenotyping of the carcinoma. Tumor cells are positive for p63 (A), and negative for cytokeratin 7 (B), cytokeratin 20 (C), and carcinoembryonic antigen (D). Vimentin shows focal positivity in macrophages (E), and periodic acid–Schiff staining is positive for mucin (F).
Fig. 1.
Fig. 2.
Fig. 3.
Squamous Cell Carcinoma of the Seminal Vesicle from Zinner Syndrome: A Case Report and Review of Literature
| Okada et al. (1992) [8] | Lee et al. (2007) [9] | Present case | |
|---|---|---|---|
| Age (yr) | 17 | 41 | 41 |
| Chief complaint | Lower abdominal mass and dysuria | Hematuria | Hematuria |
| Past medical history | None | Prostatitis | Squamous metaplasia, stone formation and chronic inflammation of the seminal vesicle |
| Seminal vesicle cyst detection | Latest admission | Latest admission | Twelve years ago |
| Location | Right seminal vesicle cyst | Left seminal vesicle cyst | Right seminal vesicle cyst |
| Pathologic diagnosis | Papillary adenocarcinoma | Mucinous adenocarcinoma | Poorly differentiated squamous cell carcinoma |
| Prostate-specifi antigen | Normal | Normal | Normal |
Table 1. Summary of reported cases of carcinomas arising from a seminal vesicle cyst in Zinner syndrome patients

 E-submission
E-submission





