Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 48(5); 2014 > Article
-
Brief Case Report
Detection of Human Papillomavirus Type 39 in a Seborrheic Inclusion Cyst of the Buttock - Dae Hyun Song1, Sang-Guk Lee2,3, Dong Chul Kim4,5, Jeong Hee Lee4,5, Gyung Hyuck Ko4,5, Jong Sil Lee4,5
-
Korean Journal of Pathology 2014;48(5):398-400.
DOI: https://doi.org/10.4132/KoreanJPathol.2014.48.5.398
Published online: October 27, 2014
1Departments of Pathology, The Armed Forces Capital Hospital, Seongnam, Korea
2Departments of Laboratory Medicine, The Armed Forces Capital Hospital, Seongnam, Korea
3Department of Laboratory Medicine, Yonsei University College of Medicine, Seoul, Korea
4Department of Pathology, Gyeongsang National University School of Medicine, Jinju, Korea
5Gyeongsang Institute of Health Science, Jinju, Korea
- Corresponding Author: Jong Sil Lee, M.D. Department of Pathology, Gyeongsang National University School of Medicine, 15 Jinju-daero 816beon-gil, Jinju 660-751, Korea Tel: +82-55-751-8763, Fax: +82-55-759-7952, E-mail: jongsil25@dreamwiz.com
© 2014 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 8,800 Views
- 49 Download
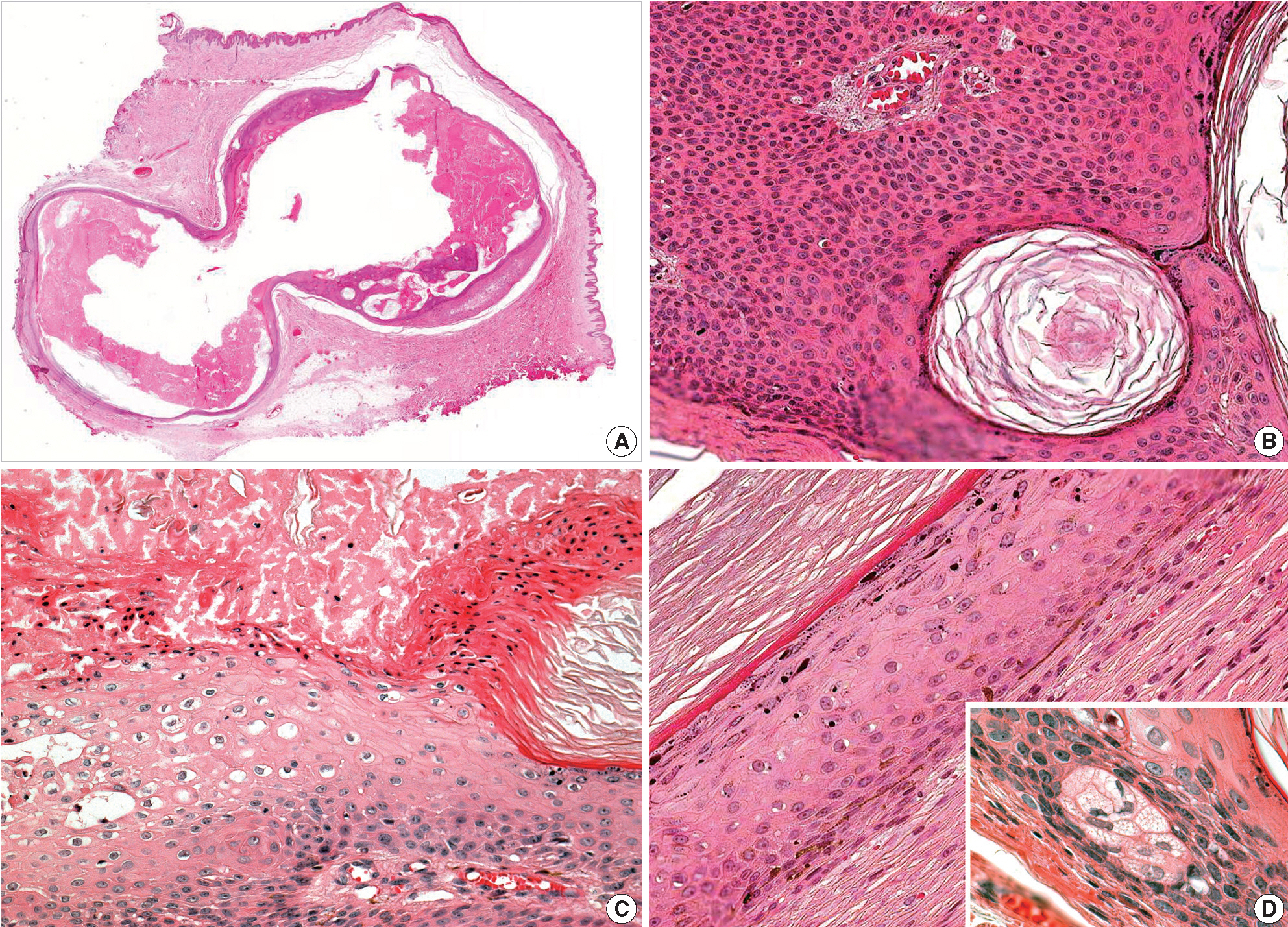
- A 21-year-old Korean man presenting with a subcutaneous mass in the left buttock was admitted to the Armed Forces Capital Hospital. An excisional biopsy of the lesion was performed, and gross examination of skin tissue revealed a 2 cm unilocular cyst that was filled with a soft, yellow substance. Microscopic examination of the lesion identified a cyst containing keratins (Fig. 1A). Approximately one half of the cyst exhibited features of a dermoid cyst with a mature squamous epithelium with granular layers and small clusters of sebaceous cells (Fig. 1D), while the other half demonstrated basaloid cell proliferation and pseudohorn cysts (Fig. 1B). In addition, squamous eddies, vacuolated cells and parakeratosis were observed in the cyst wall (Fig. 1C). No significant area of inflammation as might be present in a foreign body reaction was observed in the adjacent soft tissue. There was also no evidence of viral infection, such as vacuolated cells or parakeratosis, at the skin surface.
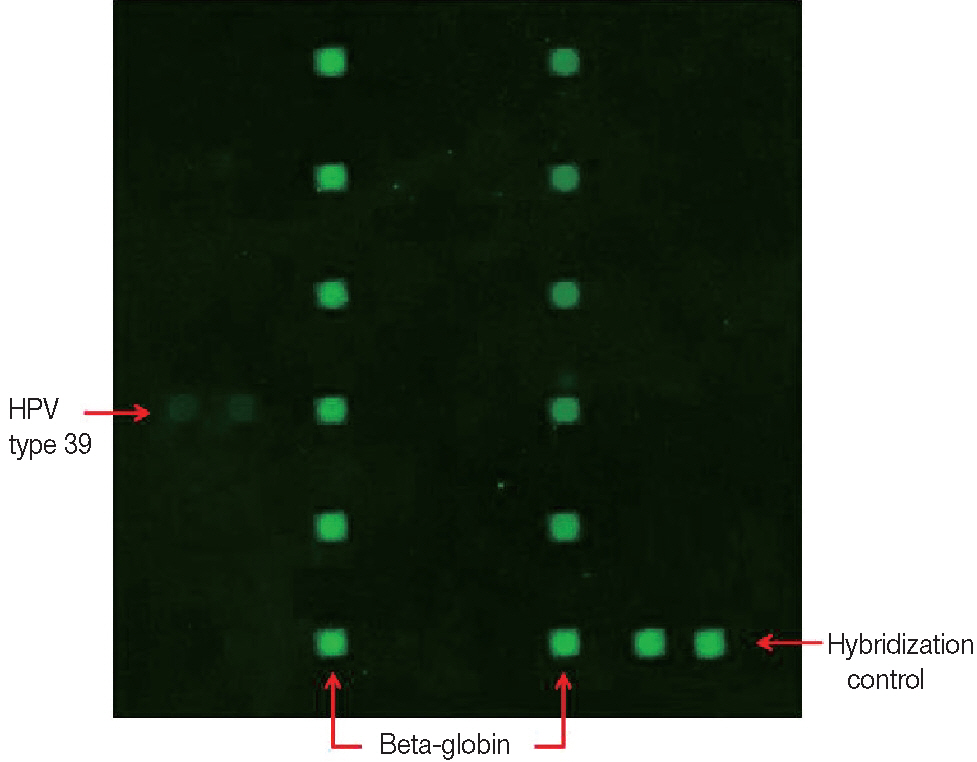
- HPV detection and genotyping were performed using the commercial HPV Genotyping Chip Kit provided by the Biomedlab Company (Seoul, Korea). DNA was isolated from the paraffin-embedded tissue using the DNA extraction kit that was supplied as a component of the HPV Genotyping Kit. Target HPV DNA and beta globin (as an internal control) were amplified by polymerase chain reaction (PCR) and labeled with cyanine 3 for signal detection. The amplified cyanine 3-labeled products were mixed with a hybridization buffer and applied to the DNA chip. Hybridization was performed at 60°C for 1 hour, and then the chip was washed for 2 minutes with distilled water and air-dried at room temperature. HPV DNA hybridization was detected using the DNA Chip Scanner (NimbleGen MS200, Roche, Basel, Switzerland). Of the 32 HPV genotypes tested, the HPV genotype 39 probe was hybridized with the DNA amplified from our specimen (Fig. 2).
CASE REPORT
- SIC generally has been considered an EC variant due to the histological similarities. However, our case showed dermoid cyst-like features rather than EC, mostly because of the presence of sebaceous gland cells and the soft, yellowish content of the cyst. The content of ECs has a whitish toothpaste-like appearance rather than the yellowish atheroma-like feature of a dermoid cyst. Sebaceous cells of dermoid cysts product a lipid component, which gives their content a yellowish appearance. To our knowledge, the three previously reported SIC cases described the presence of soft, yellowish content[1,4,5]; however, sebaceous cells in SIC have never been described. We suggest the possibility that undetected sebaceous cells were present in previously reported cases on the basis of their yellowish content[1,4,5], and SIC should be defined as a variant of cutaneous cysts rather than epidermal, epidermoid, or infundibular cysts.
- Previous studies have questioned the association of SIC with viral infection since microscopic observations have revealed wart-like features, such as eosinophilic cytoplasmic inclusions, vertical tiers of parakeratotic cells, and vacuolated cells[4]. In the present case of SIC, we also observed vacuolated cells and parakeratosis, which are features suggestive of viral infection, and we identified the HPV genotype 39 using DNA chip analysis. Two previous reports have examined the association between HPV infection and SIC (Table 1). In a previous study, Terada[1] described microscopic observations indicating HPV infection and confirmed HPV infection by immunohistochemical staining. However, no such microscopic observations were mentioned in a case reported by Fernandez-Flores[4]. Instead, Fernandez-Flores[4] attempted HPV genotyping using PCR, but no HPV DNA was detected. Although few data have been published in regards to SIC with HPV, ECs of cutaneous cysts have been relatively well studied in regards to HPV infection. HPV types 57 and 60 were recently implicated in palmoplantar ECs[6]. However, HPV type 39, which was detected in our case, has not been identified in EC. This high-risk HPV type was primarily cloned from bowenoid papules of the penis, and it comprised 3.9% of the biopsy specimens collected from female patients infected with HPV[7]. The clinical implications of high-risk HPV infection in non-genital areas have not been fully determined. Kim et al.[8] identified the high-risk HPV type 59 in a verrucous cyst of the flank and suggested that closer attention will be needed to characterize the clinical behavior associated with that type of cyst. Therefore, further studies will be required in order to determine the clinical implications of skin lesions associated with high-risk HPV infection.
- The pathogenesis of SIC with HPV infection is unclear. However, it has been mentioned that the development of EC along with HPV in areas lacking follicles, such as the palms and soles, could possibly be caused by the traumatic implantation of the epidermis. Park et al.[6] described the development of EC in weight-bearing surfaces using three stages: first, a pre-existing wart is implanted into the dermis by weight; second, the implanted portion forms a cyst in the dermis. Finally, the pre-existing wart undergoes regression, leaving only the dermal cyst behind. Since the buttock, which was the location in our case, can also be considered a weight-bearing area, this theory could explain the pathogenesis of the cyst described in the present case. This theory was also supported by the association of nongenital seborrheic keratosis with HPV infection, which was described by Tardío et al.[9].
- In conclusion, we report a case of SIC associated with HPV type 39, which we identified using DNA chip analysis. This is the first report of successful HPV genotyping associated with SIC.
DISCUSSION
| Case No. | Age (yr) | Sex | Country | Location | Microscopic indications of viral infection | Squamous eddies | Analysis tool | HPV detection | Reference |
| 1 | 84 | M | Spain | Left thigh | × | O | PCR | × | Fernandez-Flores[4] |
| 2 | 41 | M | Japan | Neck | Eosinophilic cytoplasmic inclusion | × | IHS | O | Terada[1] |
| 3 | 21 | M | Korea | Left buttock | Vacuolated cell, parakeratosis | O | DNA chip | O | Present case |
- 1. Terada T. Seborrheic inclusion cyst of the skin positive for cytoplasmic inclusion bodies and HPV antigen. Int J Clin Exp Pathol 2012; 5: 455-7. PubMedPMC
- 2. Uchi H, Yasukawa F, Kiryu H, Hashimoto K, Furue M. Infundibular cyst with seborrheic verruca-like cyst walls in a patient with Yusho disease exposed to dioxins. Eur J Dermatol 2012; 22: 687-8. ArticlePubMed
- 3. Kwittken J. Seborrheic keratosis with epidermal cyst. Mt Sinai J Med 1980; 47: 258-60. PubMed
- 4. Fernandez-Flores A. Seborrheic inclusion cysts: a study of human papillomavirus infection by polymerase chain reaction. Am J Dermatopathol 2009; 31: 310-2. ArticlePubMed
- 5. Pusiol T, Zorzi MG, Morichetti D. Extracutaneous seborrheic inclusion cyst: an unusual presentation. Pathologica 2010; 102: 420-2. PubMed
- 6. Park HS, Kim WS, Lee JH, et al. Association of human papillomavirus infection with palmoplantar epidermal cysts in Korean patients. Acta Derm Venereol 2005; 85: 404-8. ArticlePubMed
- 7. Volpers C, Streeck RE. Genome organization and nucleotide sequence of human papillomavirus type 39. Virology 1991; 181: 419-23. ArticlePubMed
- 8. Kim H, Seok JY, Kim SH, et al. Human papillomavirus type 59 identified in a verrucous cyst of the flank. Eur J Dermatol 2006; 16: 254-7. PubMed
- 9. Tardío JC, Bancalari E, Moreno A, Martín-Fragueiro LM. Genital seborrheic keratoses are human papillomavirus-related lesions: a linear array genotyping test study. APMIS 2012; 120: 477-83. ArticlePubMed
REFERENCES
Figure & Data
References
Citations

 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
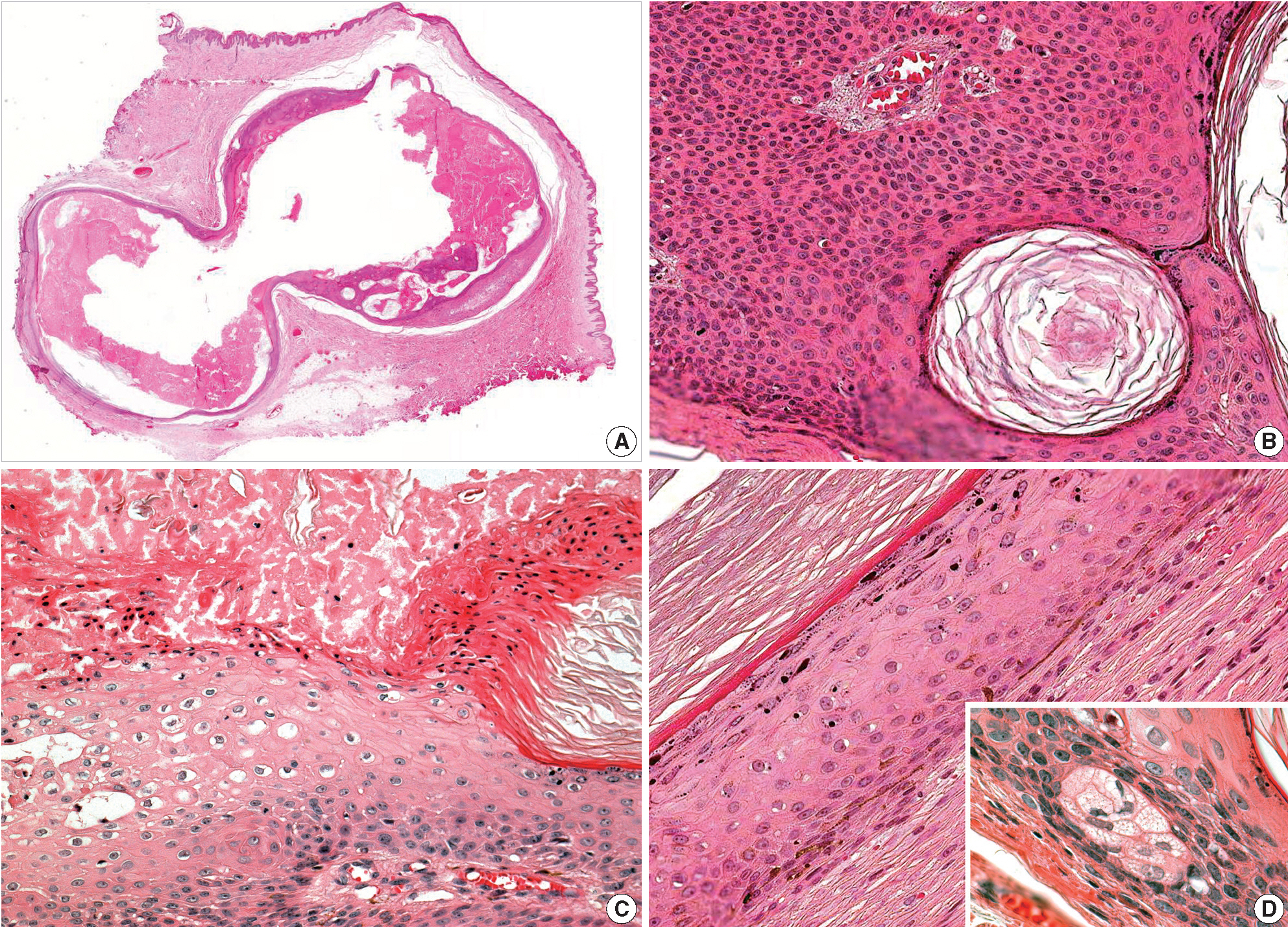
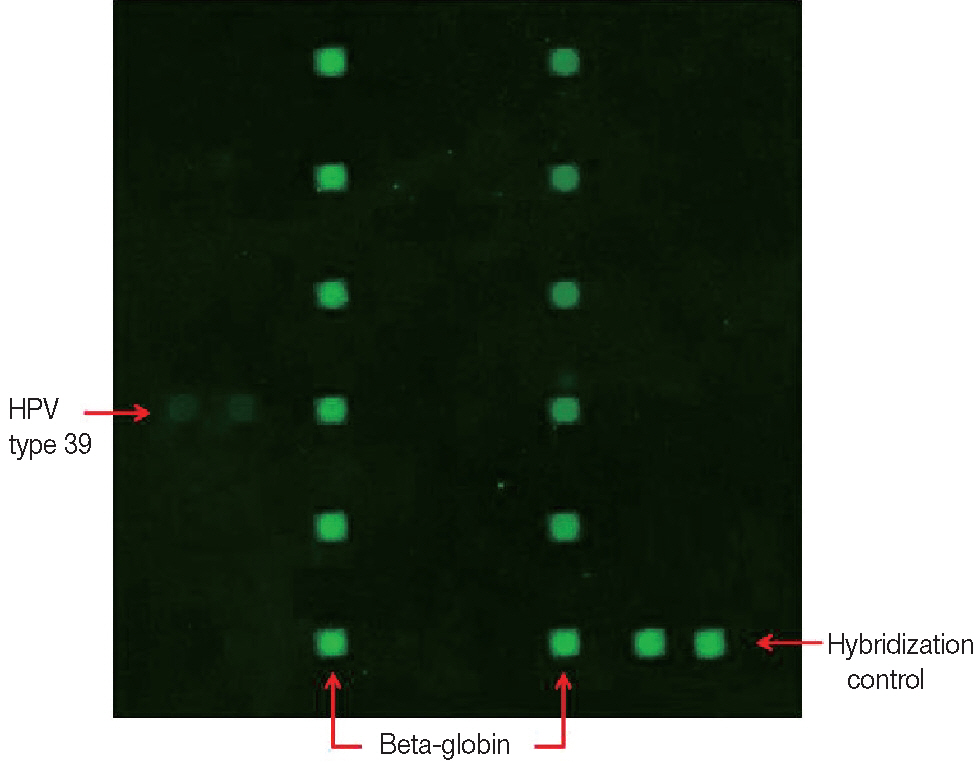
Fig. 1.
Fig. 2.
| Case No. | Age (yr) | Sex | Country | Location | Microscopic indications of viral infection | Squamous eddies | Analysis tool | HPV detection | Reference |
| 1 | 84 | M | Spain | Left thigh | × | O | PCR | × | Fernandez-Flores[4] |
| 2 | 41 | M | Japan | Neck | Eosinophilic cytoplasmic inclusion | × | IHS | O | Terada[1] |
| 3 | 21 | M | Korea | Left buttock | Vacuolated cell, parakeratosis | O | DNA chip | O | Present case |
M, male; PCR, polymerase chain reaction; IHS, immunohistochemical stain.

 E-submission
E-submission




