Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 48(5); 2014 > Article
-
Brief Case Report
Solid Form of Epithelioid Hemangioma: A Case Report - Jin Roh, Min Jeong Song, Mi Woo Lee1, Chan-Sik Park
-
Korean Journal of Pathology 2014;48(5):394-397.
DOI: https://doi.org/10.4132/KoreanJPathol.2014.48.5.394
Published online: October 27, 2014
Departments of Pathology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
1Departments of Dermatology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- Corresponding Author: Chan-Sik Park, M.D. Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, 88 Olympic-ro 43-gil, Songpa-gu, Seoul 138-736, Korea Tel: +82-2-3010-5838, Fax: +82-2-472-7898, E-mail: csikpark@amc.seoul.kr
• Received: October 14, 2013 • Revised: December 15, 2013 • Accepted: December 20, 2013
© 2014 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- A 35-year-old female patient presented with multiple consecutive dermal nodules in her left forearm. On the initial physical examination, there were approximately ten well-defined, erythematous, round nodules up to 0.6 cm in size. The lesions were nontender, nonpruritic, and without ulceration (Fig. 1). The lesions slowly grew in size over the course of 4 months. The patient was otherwise healthy with no significant medical conditions. Excisional biopsies were performed in three nodules in the left forearm and elbow area. Microscopic examination of skin biopsies of the lesions revealed ill-defined lobular architecture with focally infiltrative borders in the superficial dermis (Fig. 2A). Most of the lesions consisted of solid sheets of epithelioid and spindle cells. Those cells had a moderate amount of eosinophilic cytoplasm, and some had cytoplasmic vacuoles (Fig. 2B). They had moderately pleomorphic nuclei and prominent nucleoli. Some of the epithelioid cells formed immature vessels. Vascular structures of variable size were identified at the periphery of the lesion (Fig. 2D). The vascular spaces were canalized and lined by plump epithelioid endothelial cells. Few inflammatory cell infiltrations were visualized and extravasated red blood cells were prominent. No necrosis was present. Up to five mitotic figures per high-powered field were identified with no atypical mitoses. These cells were focally positive for CD31 and CD34, but negative for human herpesvirus-8 (HHV-8) on immunohistochemical staining (Fig. 2C, E). Resection margins were clear. The initial presumptive diagnosis was epithelioid vascular tumor of borderline malignancy.
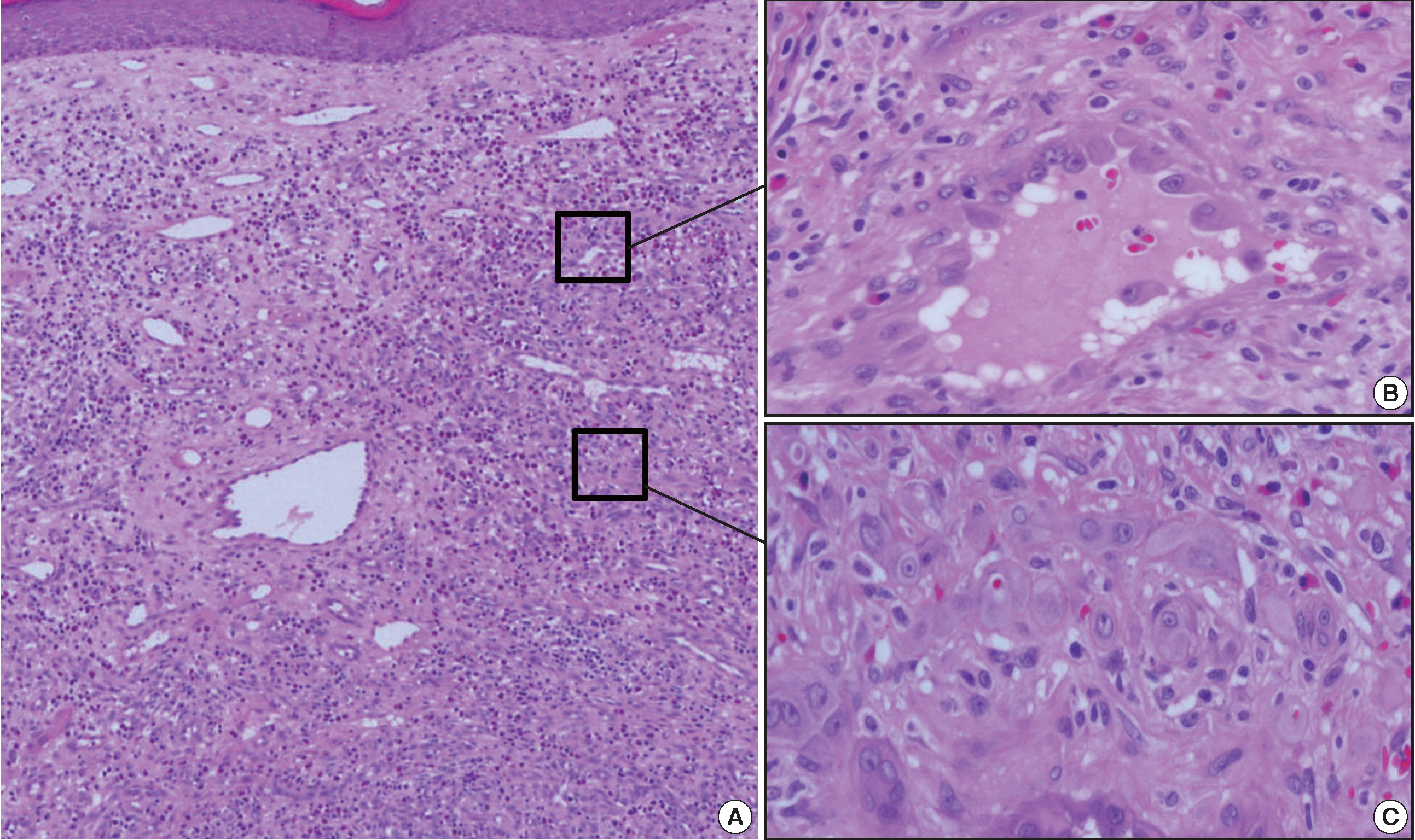
- Approximately 1 year later, a 0.7-cm-sized nodule with the same clinical features as the nodules seen on initial physical exam developed. Histologically, this was a superficial ill-defined lesion with remarkably different morphologies to previous lesions (Fig. 3A). Most of the lesion demonstrated typical histologic features of EH (Fig. 3B). Vascular structures had prominent endothelial cells protruding deeply into the lumen, creating “tombstones” appearance. Mixed inflammatory cells, including many eosinophils, were dispersed between the tumor cells. A central, focal, and solid component similar to the previous biopsy was present (Fig. 3C). The epithelioid endothelial cells were also immunopositive for CD31. The cells expressed Factor VIII and CD34 to a much lesser extent than CD31. D2-40 immunostaining was negative (data not shown). The patient has survived more than 30 months since the first biopsy. There has not been any recurrence of the lesions after application of a topical corticosteroid.
CASE REPORT
- In this paper, we present the case of a patient with an EH comprised almost entirely from epithelioid endothelial cells with a solid growth pattern. The differential diagnoses for cutaneous lesions that mostly consist of epithelioid cells include poorly differentiated squamous cell carcinoma, melanoma, epithelioid vascular tumor[6], atypical fibroxanthoma, cutaneous leiomyosarcoma, epithelioid fibrous histiocytoma, and epithelioid sarcoma. Immunohistochemical stains are crucial in classifying the lineage of the tumor. However, immunohistochemical stains do have limitations. For instance, certain melanomas, atypical fibroxanthomas, and even carcinomas can express CD31[7] and some vascular tumors such as epithelioid angiosarcoma (EA) express vascular markers only focally or weakly, but may express cytokeratin[7].
- Among the epithelioid vascular tumors, major differential diagnoses are anaplastic Kaposi sarcoma (KS), epithelioid hemangioendothelioma (EHE), and EA. Anaplastic KS is rare variant of KS. Histologically it shows a typical haphazard proliferation of epithelioid and spindle cells with moderate pleomorphism. Unlike EH, it displays marked cellular atypia and brisk mitosis. A few reports have indicated its aggressive clinical behavior[8]. Immunohistochemical staining for HHV-8 confirm the diagnosis. The tumor in this case was negative in HHV-8 immunostaining and exhibited benign clinical behavior. EHE usually arises as a solitary mass on the extremities. The tumor cells arrange in cords or chains in distinctive myxohyaline stroma. Most cases of EHE follow an indolent clinical course, with 20% to 30% of cases showing risk of metastasis[9]. In this patient, diagnosis was potentially confusing owing to the proliferation of solid endothelial cells. However, the mass did not have the typical myxohyaline or sclerotic stroma of EHE. EA can also display solid sheets of epithelioid or spindled endothelial cells without obvious vasoformation. Although EA is most prevalent during the seventh decade of life, it can affect individuals of all ages. Patients often present with a rapidly enlarging mass deep within the muscles of the lower extremities. Important histologic characteristics to discriminate EA from other vascular tumors are severe nuclear atypia, brisk mitotic counts, coagulative necrosis, and extensive hemorrhage. EA is a highly malignant tumor and more than half of patients die within the first year. In our case, histologic features such as a lobular architecture, peripheral maturation, absence of necrosis or atypical mitotic figures, and the lack of significant nuclear atypia decreased the index of suspicion for high-grade malignancy. Its clinical behavior was also indolent.
- In summary, a lesion composed of epithelioid endothelial cells in a solid growth pattern can be considered EH when there are none of the above mentioned evidences of malignancy, characteristic myxohyaline stroma, or immunoreactivity for HHV-8.
- An EH showing an entirely solid growth pattern, as in this case, is extremely rare. A review of the published literature written in English identified, only one study on the solid form of EH, which described these lesions as an atypical, immature, or exuberant form of EH[10]. However, EH with focal solid growth is not uncommon if the mass is scrutinized carefully. One study reported that 80% of solid form of EH cases were interpreted as malignant vascular tumors by at least one contributing pathologist[10]. Therefore, using a punch biopsy that may include only part of the solid mass can cause a diagnostic challenge for pathologists who are not aware of solid form of EH. To our knowledge, our present study is the first report of a solid form of EH in the Korean pathologic literature. It is important that the solid form of EH be distinguished from malignant vascular tumors to avoid overly aggressive intervention.
DISCUSSION
Fig. 1.(A) Multiple well-defined erythematous papules on the forearm, each measuring 0.3–0.4 cm. (B) A recurrent erythematous, round protruding papule in the forearm, measuring 0.7 cm.
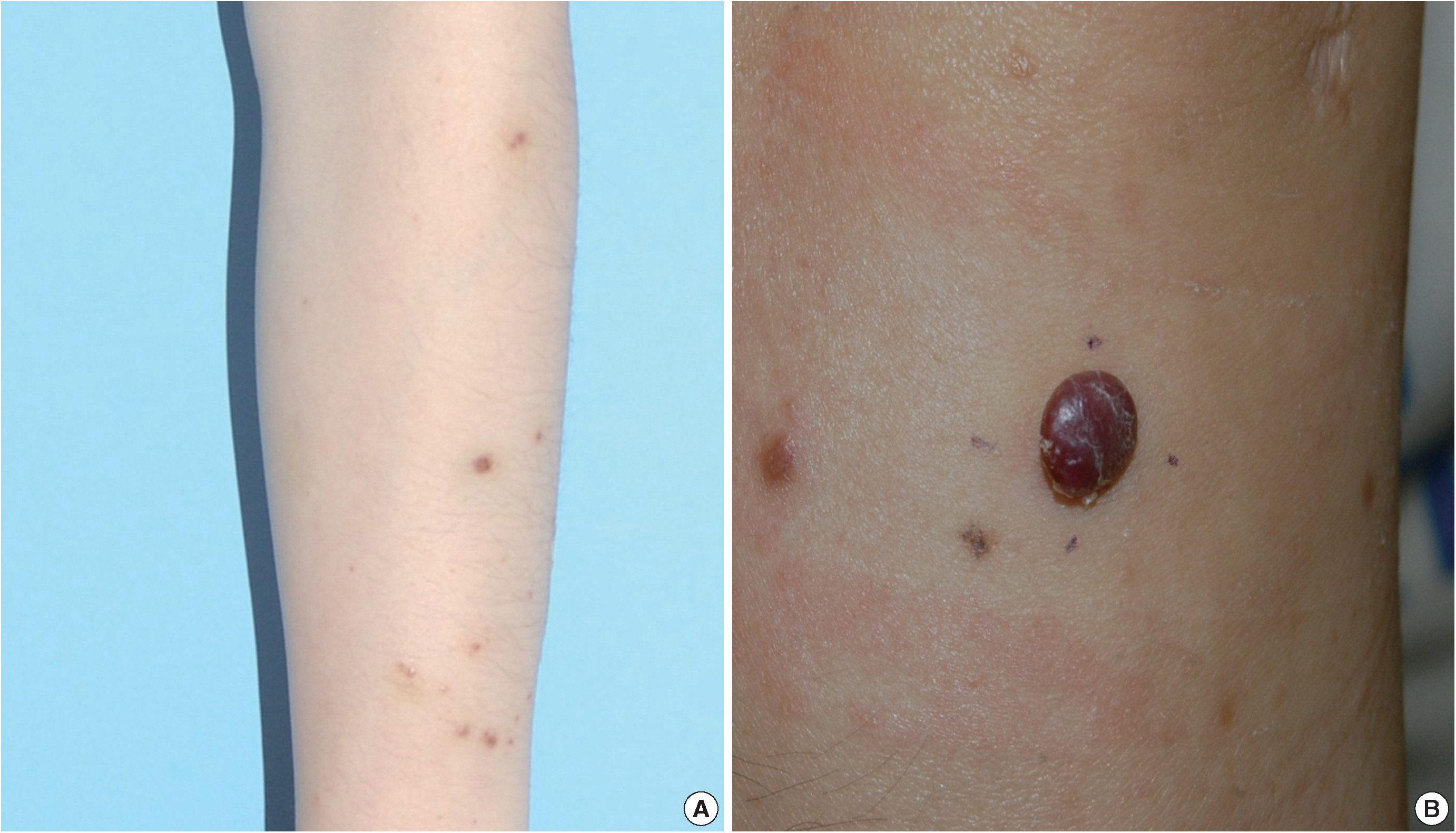

Fig. 2.Histologic features of the first lesion. (A) The lesion is a poorly differentiated cellular tumor with focally infiltrating borders in the superficial dermis. (B) Most of the lesion shows solid proliferation of epithelioid endothelial cells. (C) Epithelioid endothelial cells are focally positive for CD34 immunohistochemical staining. (D) At the periphery, well-canalized vessels are observed. (E) Human herpesvirus-8 related antigen is negative.
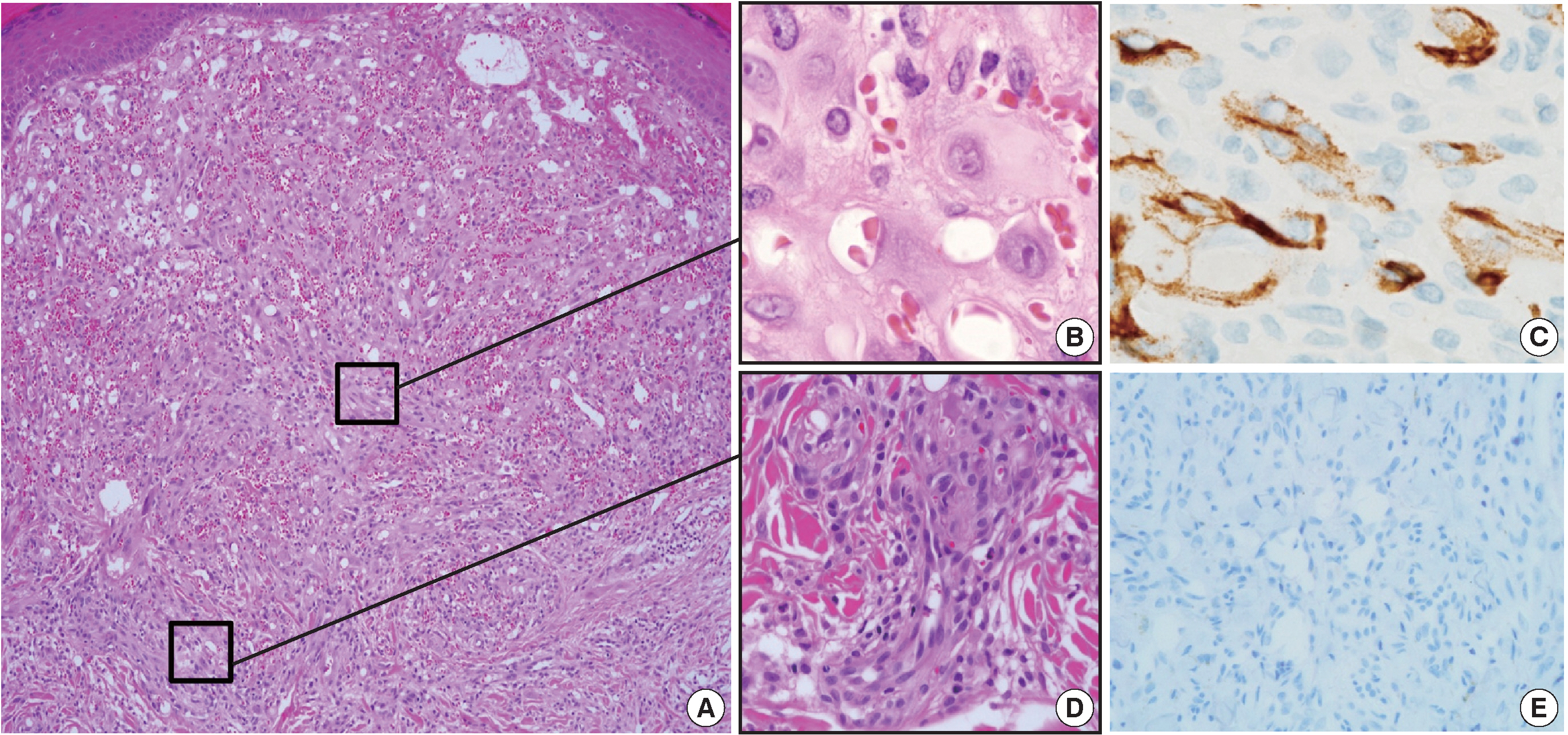

Fig. 3.Histologic features of the second lesion. (A) A recurrent lesion shows typical histologic characteristics of epithelioid hemangioma. (B) Vascular structures that comprise the lesion are lined by prominent epithelioid endothelial cells. The stroma contains mixed inflammatory cells, including many eosinophils. (C) A focal solid component is present in the center of the lesion.

- 1. Olsen TG, Helwig EB. Angiolymphoid hyperplasia with eosinophilia: a clinicopathologic study of 116 patients. J Am Acad Dermatol 1985; 12(5 Pt 1): 781-96. PubMed
- 2. Wells GC, Whimster IW. Subcutaneous angiolymphoid hyperplasia with eosinophilia. Br J Dermatol 1969; 81: 1-14. ArticlePubMed
- 3. Eady RA, Cowen T, Jones EW. Proceedings: pseudopyogenic granuloma: the histopathogenesis in the light of ultrastructural studies. Br J Dermatol 1976; 95 Suppl 14: 13.
- 4. Eady RA, Jones EW. Pseudopyogenic granuloma: enzyme histochemical and ultrastructural study. Hum Pathol 1977; 8: 653-68. ArticlePubMed
- 5. Rosai J, Gold J, Landy R. The histiocytoid hemangiomas: a unifying concept embracing several previously described entities of skin, soft tissue, large vessels, bone, and heart. Hum Pathol 1979; 10: 707-30. PubMed
- 6. Buonaccorsi JN, Plaza JA. Role of CD10, wide-spectrum keratin, p63, and podoplanin in the distinction of epithelioid and spindle cell tumors of the skin: an immunohistochemical study of 81 cases. Am J Dermatopathol 2012; 34: 404-11. PubMed
- 7. Ordóñez NG. Immunohistochemical endothelial markers: a review. Adv Anat Pathol 2012; 19: 281-95. PubMed
- 8. Huwait H, Meneghetti A, Nielsen TO. Kaposi sarcoma of the adrenal gland resembling epithelioid angiosarcoma: a case report. Sarcoma 2011; 2011: 898257. ArticlePubMedPMCPDF
- 9. Mentzel T, Beham A, Calonje E, Katenkamp D, Fletcher CD. Epithelioid hemangioendothelioma of skin and soft tissues: clinicopathologic and immunohistochemical study of 30 cases. Am J Surg Pathol 1997; 21: 363-74. ArticlePubMed
- 10. Fetsch JF, Sesterhenn IA, Miettinen M, Davis CJ Jr. Epithelioid hemangioma of the penis: a clinicopathologic and immunohistochemical analysis of 19 cases, with special reference to exuberant examples often confused with epithelioid hemangioendothelioma and epithelioid angiosarcoma. Am J Surg Pathol 2004; 28: 523-33. PubMed
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- Clinicopathologic and radio-genomic insights into hepatic epithelioid hemangioma: An illustrated review
Elijah Brown, Karen Schoedel, Adarsh Barwad, Alessandro Furlan, Paul J.L. Zhang, Nigar Anjuman Khurram
Human Pathology.2026; 170: 105955. CrossRef - Epithelioid Haemangioma of Bone: A Case Series and Comprehensive Literature Review Reappraising the Diagnostic Classification of All Epithelioid Vascular Neoplasms of Bone
Subramaniam Ramkumar
Cureus.2021;[Epub] CrossRef - Update on cutaneous epithelioid vascular tumours
Boštjan Luzar, Eduardo Calonje
Diagnostic Histopathology.2018; 24(8): 273. CrossRef - Epithelioid Hemangioma (Angiolymphoid Hyperplasia With Eosinophilia) of the Heart With Peripheral Eosinophilia and Nephrotic Syndrome
Isidro Machado, Agustín Chong, Anisia Serrano, Alfredo Mario Naranjo Ugalde, Damian Pineda, Laynes Savón, Ever Olivera, Antonio Llombart-Bosch
International Journal of Surgical Pathology.2016; 24(1): 59. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
Solid Form of Epithelioid Hemangioma: A Case Report
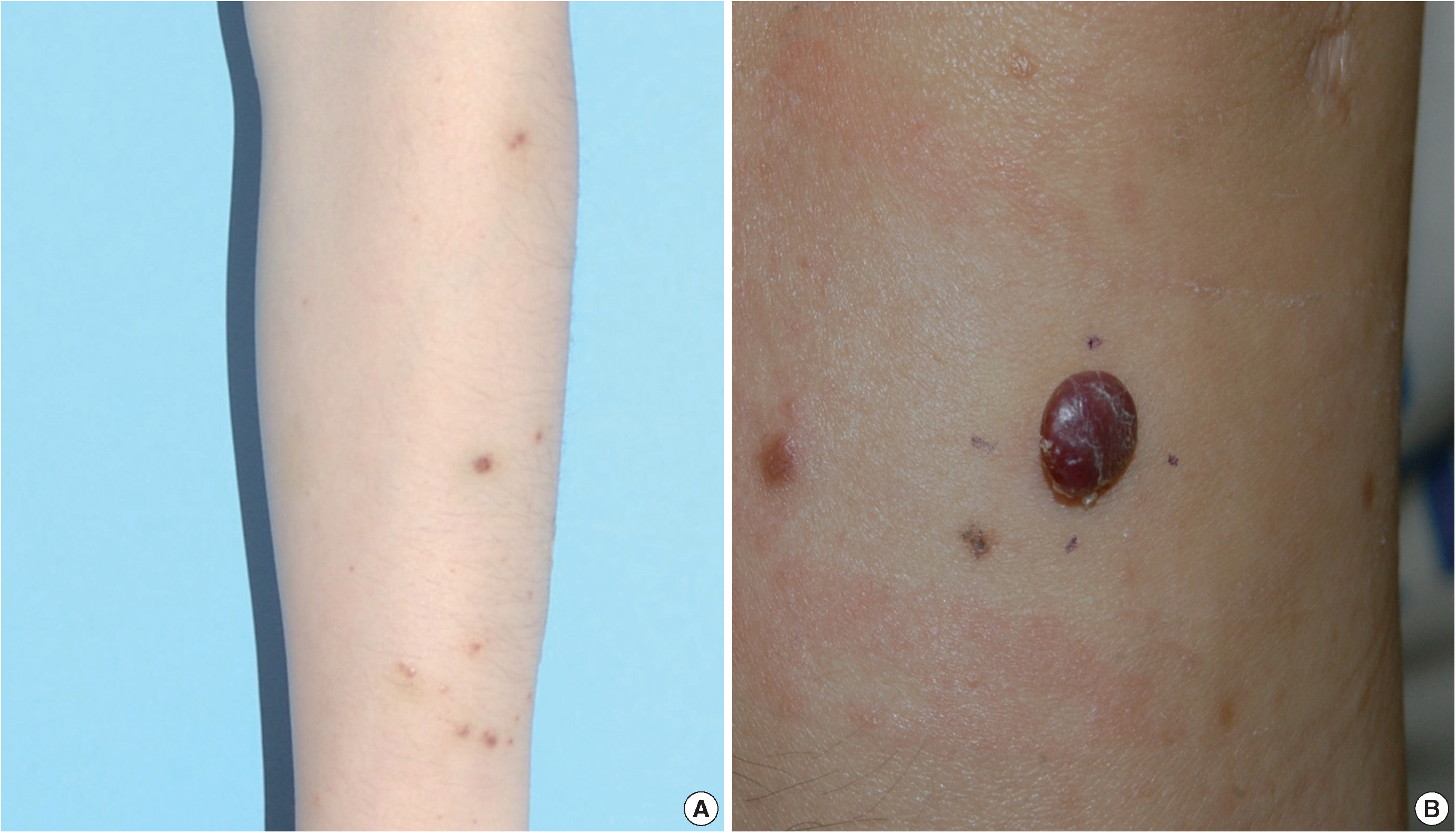
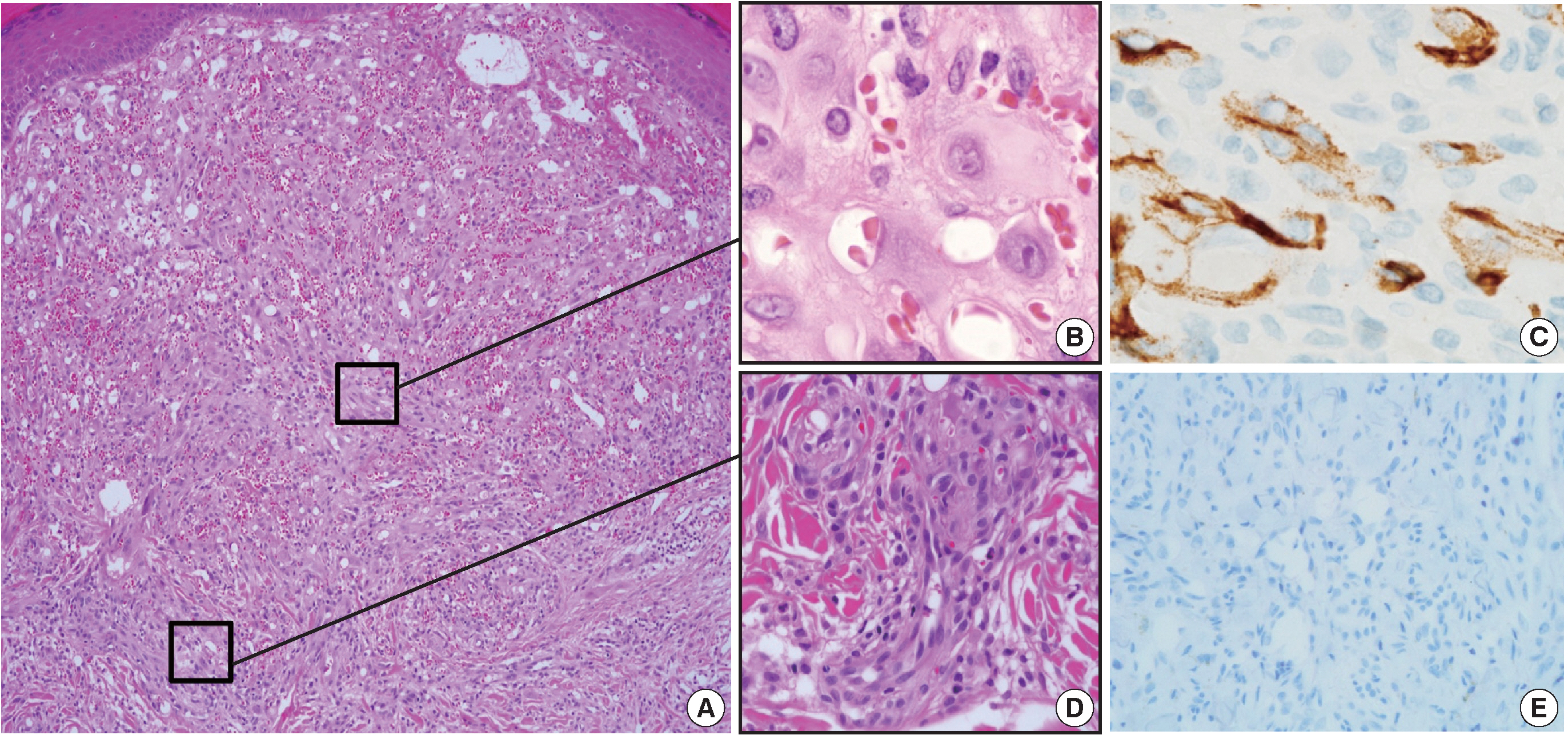
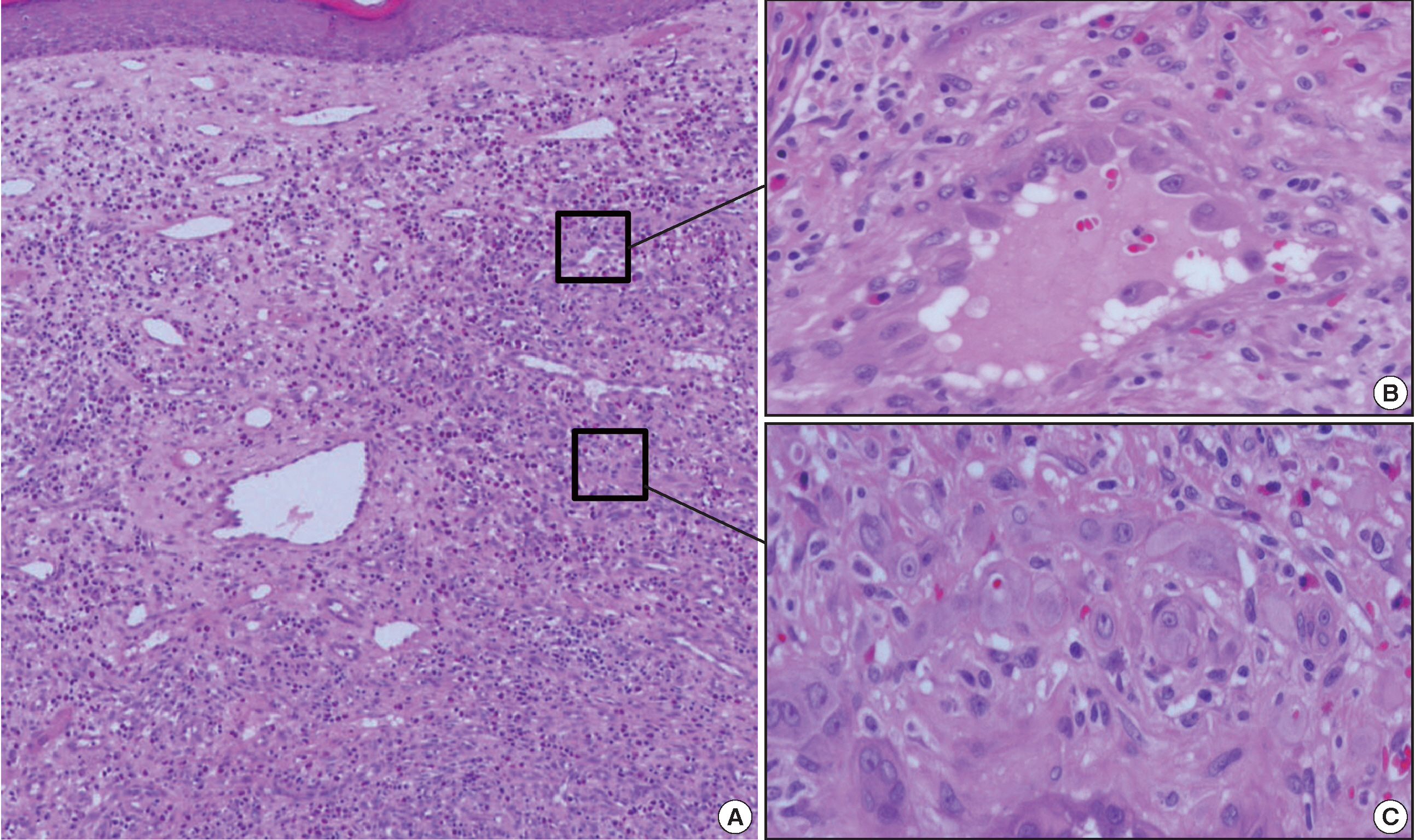
Fig. 1. (A) Multiple well-defined erythematous papules on the forearm, each measuring 0.3–0.4 cm. (B) A recurrent erythematous, round protruding papule in the forearm, measuring 0.7 cm.
Fig. 2. Histologic features of the first lesion. (A) The lesion is a poorly differentiated cellular tumor with focally infiltrating borders in the superficial dermis. (B) Most of the lesion shows solid proliferation of epithelioid endothelial cells. (C) Epithelioid endothelial cells are focally positive for CD34 immunohistochemical staining. (D) At the periphery, well-canalized vessels are observed. (E) Human herpesvirus-8 related antigen is negative.
Fig. 3. Histologic features of the second lesion. (A) A recurrent lesion shows typical histologic characteristics of epithelioid hemangioma. (B) Vascular structures that comprise the lesion are lined by prominent epithelioid endothelial cells. The stroma contains mixed inflammatory cells, including many eosinophils. (C) A focal solid component is present in the center of the lesion.
Fig. 1.
Fig. 2.
Fig. 3.
Solid Form of Epithelioid Hemangioma: A Case Report

 E-submission
E-submission





