Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(6); 2013 > Article
-
Brief Case Report
Micropapillary Mucinous Adenocarcinoma of the Lung: A Brief Case Report - In Ho Choi, Boin Lee, Joungho Han, Chin A Yi1, Yong Soo Choi2, Jin Seok Ahn3
-
Korean Journal of Pathology 2013;47(6):603-605.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.6.603
Published online: December 24, 2013
Department of Pathology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
1Department of Radiology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
2Department of Thoracic Surgery, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
3Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- Corresponding Author: Joungho Han, M.D. Department of Pathology, Samsung Medical Center, Sungkyunkwan University School of Medicine, 81 Irwon-ro, Gangnam-gu, Seoul 135-710, Korea. Tel: +82-2-3410-2800, Fax: +82-2-3410-0025, hanjho@skku.edu
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- A 69-year-old woman presented with persistent cough for one month previously. The patient had a 13 pack-year smoking history and hypertension. Chest computed tomography revealed predominant ground glass opacity with patchy consolidation occupying almost the entire left lower lobe of the lung (Fig. 1A). It was histologically confirmed as adenocarcinoma of the lung by transbronchial lung and lymph node biopsy. Analysis of EGFR and K-ras mutation by polymerase chain reaction-sequencing was performed on tissue from the lymph node biopsy, which revealed missense mutation of K-ras in the 12th codon, c.34G>T (p.G12C), without EGFR mutation. After neoadjuvant concurrent chemoradiation, a left lower lobectomy was performed. The gross examination revealed a poorly defined, large, mass-like lesion with scattered dilated bronchus, which was similar in appearance to lobar pneumonia and well correlated with radiologic findings (Fig. 1B).
- Microscopically, the tumor demonstrated a predominantly micropapillary growth pattern in the alveolar cavity and a lepidic growth pattern along the septa with hobnail features (Fig. 1C). Neoplastic cells with abundant clear or amphophilic cytoplasm intermingled with each other (Fig. 1D). Special stains (periodic acid Schiff, mucicarmine, and Alcian blue pH 1.0 and 2.5) revealed positivity in the cytoplasm of a few tumor cells (Fig. 1D, inset). Tumor cells had well-defined cell borders and large, pleomorphic nuclei (Fig. 1E), with occasional horse-shoe shaped nuclei and perinuclear clearing. Their nuclei had a contorted prominent membrane, similar to the raisinoid appearance induced by human papillomavirus infection, and had frequent prominent macronucleoli (Fig. 1F).
- No complications were noted in the two-month follow-up period after discharge.
CASE REPORT
- Due to the scarcity of publications regarding the micropapillary subtype of lung adenocarcinoma and the problem of the undefined threshold for classification of micropapillary adenocarcinoma, the incidence rate of this disease is not well known, but is estimated to represent approximately 6% of all lung adenocarcinomas (6.8% in Warth et al.6 and 6.0% in Solis et al.7) with no available data in the Korean population. Recent reports have described it as having a poor prognosis similar to solid-type lung adenocarcinomas.7
- Considering the usefulness of EGFR-tyrosine kinase inhibitors, the importance of confirming the existence of EGFR or K-ras mutations cannot be emphasized enough. EGFR and K-ras mutations are virtually mutually-exclusive and directly related to targets of different subsets of lung adenocarcinomas.8 Therefore, it is important to decide, using relevant findings, which test is the priority for the limited specimens obtained from percutaneous needle biopsy or bronchoscopic biopsy. For example, if histologic findings are more indicative of K-ras mutation, we can potentially prevent an unnecessary test for EGFR mutation.
- De Oliveira Duarte Achcar et al.5 analyzed the molecular profile of 15 primary micropapillary adenocarcinomas for K-ras, EGFR, and BRAF mutations after dividing them into three groups by histologic features: nonmucinous hobnail configuration or terminal reserve unit-type micropapillary adenocarcinoma, tumors with predominant columnar or polygonal cell configuration, and tumors of mixed type. In their study, tumors with nonmucinous hobnail pattern and those with columnar or polygonal pattern showed no significant differences in EGFR, K-ras, and BRAF mutation tests, and both K-ras and EGFR mutations could occur in mucin-producing tumors. Therefore, they concluded that there was no clear association between these mutations and morphologic features.
- However, Inamura et al.4 reported a significant association between the hobnail cell type and EGFR mutation, in the setting of lung adenocarcinomas categorized into hobnail, columnar, and polygonal cell type. Ninomiya et al.9 also reported that the hobnail cell type and micropapillary morphology predicted a higher incidence of EGFR mutation in lung adenocarcinomas and speculated that distinct micropapillary features reflect a step of tumor progression from a well-differentiated papillary adenocarcinoma of the hobnail cell type to a less differentiated state.
- When cytological features, rather than growth pattern, are a priority for consideration, micropapillary adenocarcinoma with mucinous features should be regarded as mucinous adenocarcinoma. This might also correspond with the results of other studies that suggest K-ras mutation is related to mucinous BAC of the lung.2
- Considering our case, which demonstrated a hobnail or lepidic growth pattern with mucinous differentiation in combination with K-ras mutation, further study is needed to clarify the relationship between the presence of mucinous features of micropapillary predominant adenocarcinoma and K-ras mutation.
DISCUSSION
- 1. Travis WD, Brambilla E, Noguchi M, et al. International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society international multidisciplinary classification of lung adenocarcinoma. J Thorac Oncol 2011; 6: 244-285. PubMedPMC
- 2. Finberg KE, Sequist LV, Joshi VA, et al. Mucinous differentiation correlates with absence of EGFR mutation and presence of KRAS mutation in lung adenocarcinomas with bronchioloalveolar features. J Mol Diagn 2007; 9: 320-326. ArticlePubMedPMC
- 3. Kakegawa S, Shimizu K, Sugano M, et al. Clinicopathological features of lung adenocarcinoma with KRAS mutations. Cancer 2011; 117: 4257-4266. ArticlePubMed
- 4. Inamura K, Ninomiya H, Ishikawa Y, Matsubara O. Is the epidermal growth factor receptor status in lung cancers reflected in clinicopathologic features? Arch Pathol Lab Med 2010; 134: 66-72. ArticlePubMedPDF
- 5. De Oliveira Duarte Achcar R, Nikiforova MN, Yousem SA. Micropapillary lung adenocarcinoma: EGFR, K-ras, and BRAF mutational profile. Am J Clin Pathol 2009; 131: 694-700. PubMed
- 6. Warth A, Muley T, Meister M, et al. The novel histologic International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society classification system of lung adenocarcinoma is a stage-independent predictor of survival. J Clin Oncol 2012; 30: 1438-1446. ArticlePubMed
- 7. Solis LM, Behrens C, Raso MG, et al. Histologic patterns and molecular characteristics of lung adenocarcinoma associated with clinical outcome. Cancer 2012; 118: 2889-2899. ArticlePubMed
- 8. Gazdar AF, Shigematsu H, Herz J, Minna JD. Mutations and addiction to EGFR: the Achilles ‘heal’ of lung cancers? Trends Mol Med 2004; 10: 481-486. ArticlePubMed
- 9. Ninomiya H, Hiramatsu M, Inamura K, et al. Correlation between morphology and EGFR mutations in lung adenocarcinomas: significance of the micropapillary pattern and the hobnail cell type. Lung Cancer 2009; 63: 235-240. ArticlePubMed
REFERENCES
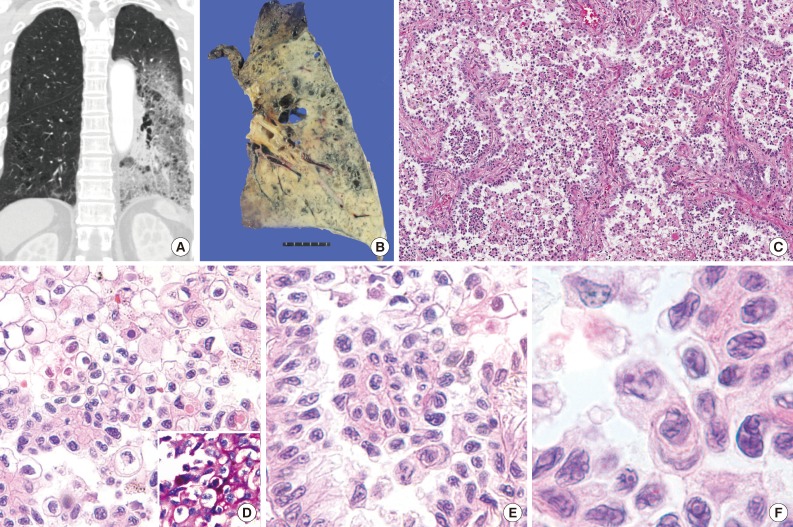
Figure & Data
References
Citations

- Molecular Pathology of Lung Cancer
James J. Saller, Theresa A. Boyle
Cold Spring Harbor Perspectives in Medicine.2022; 12(3): a037812. CrossRef - The clinicopathological significance of ALK rearrangements and KRAS and EGFR mutations in primary pulmonary mucinous adenocarcinoma
Yang Qu, Nanying Che, Dan Zhao, Chen Zhang, Dan Su, Lijuan Zhou, Lili Zhang, Chongli Wang, Haiqing Zhang, Lixin Wei
Tumor Biology.2015; 36(8): 6417. CrossRef - Cytomorphological identification of advanced pulmonary adenocarcinoma harboring KRAS mutation in lymph node fine‐needle aspiration specimens: Comparative investigation of adenocarcinoma with KRAS and EGFR mutations
Dae Hyun Song, Boram Lee, Yooju Shin, In Ho Choi, Sang Yun Ha, Jae Jun Lee, Min Eui Hong, Yoon‐La Choi, Joungho Han, Sang‐Won Um
Diagnostic Cytopathology.2015; 43(7): 539. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure

 E-submission
E-submission



