Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 46(6); 2012 > Article
-
Original Article
Diagnostic Difficulties in Fine Needle Aspiration of Benign Salivary Glandular Lesions - Hye Jung Jo, Hyo Jung Ahn, Soojin Jung, Hye-Kyoung Yoon
-
Korean Journal of Pathology 2012;46(6):569-575.
DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.6.569
Published online: December 26, 2012
Department of Pathology, Busan Paik Hospital, Inje University College of Medicine, Busan, Korea.
- Corresponding Author: Hye-Kyoung Yoon, M.D. Department of Pathology, Busan Paik Hospital, Inje University College of Medicine, 75 Bokji-ro, Busanjin-gu, Busan 614-735, Korea. Tel: +82-51-890-6628, Fax: +82-51-891-1834, pathyoon@inje.ac.kr
• Received: July 23, 2012 • Revised: September 24, 2012 • Accepted: October 15, 2012
© 2012 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- The diagnostic accuracy of fine needle aspiration cytology (FNAC) of salivary lesions is relatively high, but cytologic interpretation might be confusing if the sample is lacking typical cytologic features.
-
Methods
- There were 77 cases of benign salivary lesions, consisting of pleomorphic adenoma (PA) in 61 cases, Warthin's tumor (WT) in 12 cases, and other benign lesions in 4 cases. The causes of the discrepancies between the FNAC and the histologic diagnoses were evaluated.
-
Results
- Major discrepancies were noted in 4 of the 61 PA cases, and in 1 of 12 WT cases. The causes of the major discrepancies were a mislabeled site in 1 PA and 1 WT case, and an interpretation error in 3 PA cases. Minor discrepancies were more common in the WT cases (7 of 12 cases) than in the PA cases (11 of 61 cases). The causes of the minor discrepancies were a mislabeled site in 1 PA and 1 WT case, an inadequate sample in 7 PA and 2 WT cases, a lack of typical cytomorphology in 2 PA and 2 WT cases, and an interpretation error in 1 PA and 2 WT cases.
-
Conclusions
- To increase the diagnostic accuracy in the benign salivary lesions, recognition of both characteristic and less typical cytomorphology is needed.
- There were 77 cases of histologically-proven benign salivary glandular lesions among 238 FNAC cases of salivary glandular lesions during the most recent 5 years at Pusan Paik Hospital. Of the 77 cases, PA was found in 61 cases, WT in 12 cases, monomorphic adenoma in 1 case, lipoma in 1 case and mucocele in 2 cases.
- The diagnoses of FNAC and the subsequent operation were compared, and were categorized as concordant, minor discrepancy, or major discrepancy between them. The cases diagnosed simply as benign or other benign tumor were defined as minor discrepancy, and the cases diagnosed as atypical, malignant or metastasis as major discrepancy.
- A review of the FNAC smears and the histologic sections was performed in the cases showing diagnostic discrepancies. The cytologic slides were routinely stained with Papanicolaou methods. The cytologic findings were reviewed based on the specimen adequacy and the presence or lack of typical cytomorphology. Evaluation of the causes of discrepancies was performed by comparing the histologic features and cytomorphology.
MATERIALS AND METHODS
- Among the 77 cases of benign salivary glandular lesions, major diagnostic discrepancies between the FNAC and histologic diagnoses were noted in 4 of the 61 PA cases, and in 1 of the 12 WT cases, Minor diagnostic discrepancies were found in 7 of the 12 WT cases and in 11 of the 61 PA cases. One monomorphic adenoma case, one lipoma case and 2 cases of mucocele showed diagnostic concordances between the FNAC and histologic diagnoses.
- Of the 61 PA cases, 46 cases showed concordance between the FNAC and histologic diagnoses (Table 1). The cytologic diagnoses of 4 PA cases showing major discrepancies were as follows: mucoepidermoid carcinoma 1 case, malignant or atypical in 2 cases, and metastatic carcinoma 1 case the last due to mislabeling of the aspiration site as a lymph node. The case that was misdiagnosed as mucoepidermoid carcinoma revealed a low cellular yield and a few epithelial cell clusters that consisted of basaloid, squamoid and mucous cell-like cells, which were misinterpreted as mucoepidermoid carcinoma (Fig. 1A). Histologically, this case showed occasional squamous metaplasia and microcystic change (Fig. 1B). Another case that was overdiagnosed as malignant showed less conspicuous stromal tissue, and suspicious cytologic atypia. Histologically this tumor showed a spindle cellular mass with very scanty epithelial and stromal elements. One case, diagnosed as atypical, revealed low cellularity with a lack of both epithelial and stromal elements and suspicious nuclear atypia. Another case was diagnosed as metastatic carcinoma due to mislabeling as a lymph node instead of a salivary gland, and as a result, the smeared epithelial components were misinterpreted as metastasis.
- The cytologic diagnoses of the 11 PA cases showing minor discrepancies were simply benign in 7 cases, spindle cell lesion in 2 cases, suppurative inflammation in 1 case, and a benign condition in the 1 case that was mislabeled as a lymph node. The major causes of misinterpretation as simply a benign epithelial lesion or tumor was an inadequately sized sample. On review, a poor cellular yield and a small quantity of stromal components were noted. Two cases were misdiagnosed as a spindle cell tumor (ruling out schwannoma), and these cases showed predominant spindle cell populations cytologically and histologically. Another case that has been interpreted as ruling out myoepithelioma or monomorphic adenoma, showed degenerated epithelial or spindle cells on the cytologic smears, and the tumor revealed variegated histologic features with degenerative changes. One case that was misinterpreted as suppurative inflammation showed cell debris in a dirty background instead of the typical cytologic features suggesting pleomorphic adenoma. The cell debris in the dirty background was misinterpreted as degenerated inflammatory cells (Fig. 2).
- Among the 12 WT cases, only 4 cases showed a diagnostic concordance between the FNAC diagnosis and histologic diagnosis (Table 2). In these 4 cases, oncocytic cell clusters and lymphocytes were observed in the necrotic background (Fig. 3A). Of the 12, 6 cases showed minor discrepancies, and 4 cases among the 6 were diagnosed as simply benign, 1 case as an inflammatory condition, and the other case as ruling out monomorphic adenoma. Two of these 4 cases, that were diagnosed as simply benign, were misdiagnosed due to inadequate aspirates. On review of the FNAC smears, lymphoid cells with rare epithelial or stromal components were noted. The other 2 of the 4 cases that were diagnosed as simply benign were misdiagnosed due to inadequate aspirates associated with cystic change in the WT. Aspirates from the cystic WT revealed a relatively poor cellular yield with lack of a oncocytic cell clusters and somewhat squamoid features (Fig. 3B), or revealed cell debris in the necrotic background with scanty spindle or squamoid cells mixed with the cell debris (Fig. 3C, D). One case that was diagnosed as an inflammatory condition, showed cell debris or variable sized or shaped, degenerated cells mixed with various inflammatory cells in the necrotic background, instead of epithelial cell clusters with oncocytic change or rich lymphoid cells, on review of the FNAC smears. The other case that was diagnosed as ruling out monomorphic adenoma was confirmed to be interpretation error. On cytologic-histologic correlation, diagnostic discrepancies in the WT cases were related to inadequate aspirates in 2 cases, were poor or rare epithelial cells associated with cystic change in 2 cases, and there were interpretation errors in 2 of the 6 cases. The remaining 2 cases were mislabeled as lymph nodes clinically, and one case was misdiagnosed as ruling out metastasis based on the presence of squamoid cells but it was described as not so atypical, and the other case was diagnosed as a benign condition, and the smeared cells were interpreted as benign follicular cells.
RESULTS
- FANC was introduced as a diagnostic tool by Martin and Ellis11 in the 1930s,5 and FNAC of salivary glands was developed by Eneroth et al. between the 1950s and the 1960s.5 In the salivary gland lesions, FNAC is widely used as an initial diagnostic tool for further planning because it is inexpensive, rapid, well-tolerated, less invasive, and has been proven to have a high diagnostic accuracy.1,6,10,12-15
- According to the literatures since 1994, sensitivity, specificity, and diagnostic accuracies of FNAC in the salivary glandular lesions has been reported as 70-100%, 91-100%, and 84-98.3%, respectively.1-6,9,10,13,14,16 Singh Nanda et al.5 reported that sensitivity, specificity, and positive and negative predictive values were 84.61%, 91.66%, 91.60%, and 85.00%, respectively, in benign salivary gland lesions, but 84.61%, 86.48%, 68.75%, and 94.11%, respectively, in malignant salivary gland lesions. There were no differences in sensitivity, but specificity and the positive predictive values were higher in the benign salivary gland lesions while the negative predictive value was higher in malignant lesions. Brennan et al.6 reported that there were few differences in the sensitivity and specificity of between initial FNAC and repeated FNAC as 70% and 93% and 84% and 95%, respectively.
- A PA is the most common salivary gland neoplasm accounting for about 60% of all salivary gland tumor,17 thus PA should always be first considered when diagnosing the FNAC smears of a salivary gland.18 Histologically, PA shows a mixture of epithelial and mesenchymal elements. The epithelial components of a PA consist of cuboidal, basaloid, squamous, spindle, plasmacytoid and clear cells, and show trabeculae, ductal, cystic, and solid patterns. The mesenchymal components are mucoid, myxoid, cartilaginous, or hyalinized.19
- In the aspirates from PA, various combinations of three main elements, myoepithelial cells, ductal cells, and chondromyxoid matrix were observed. The epithelial components consisted of myoepithelial cells and ductal cells forming sheets or scattered single cells. The myoepithelial cells were more conspicuous in the FNAC smears than in the histologic section, and they appeared as loose clusters or singly dispersed cells. The myoepithelial cells showed various types of cytomorphology such as plasmacytoid, spindled, stellate or clear cells.10,20 In air-dried preparations, the myoepithelial cells had a pale smudgy blue outline.18 The ductal cells were less conspicuous than the myoepithelial cells and presented as 3-dimensional clusters of cuboidal cells with round nuclei. In both myoepithelial cells and ductal cells, nucleoli were rarely seen. The chondromyxoid stroma of PA was generally cyanophilic and fibrillar in the Pap stains, and can obscure the epithelial component when mixed with the epithelial component. Infrequently, metaplastic squamous cells, cystic fluid, sebaceous cells, inflammatory cells, tyrosine crystals can be observed.10,20
- The diagnostic accuracy of FNAC in PA has been reported to be 81.6% (40/49) by Paik et al.,3 84% (16/19) by Kim and Lee10 and 86.7% (26/30) by Zhang et al.4 In this study, the major diagnostic discrepancies between the FNAC and histologic diagnoses were noted in 4 (6.6%) of the 61 PA cases, and minor diagnostic discrepancies were found in 11 (18.0%) of the 61 PA cases. The diagnostic accuracy in this study was 75.4% (46/61), which was relatively low compared with the previous studies.
- In cases showing the typical three components of PA, the cytologic diagnosis can be easily performed,18,20 however, it is sometimes problematic. The cases showing dominant mucoid background and less cellular smear could be diagnosed as cystic lesions such as mucocele or mucoepidermoid carcinoma, and the cases showing cellular smears of exclusively epithelial or myoepithelial cells could be misinterpreted as monomorphic adenoma, or overdiagnosed as mucoepidermoid carcinoma or adenoid cystic carcinoma. The most helpful cytomorphologic features in distinguishing PA from malignancies are the presence of plasmacytoid myoepithelial cells and fibrillary stromal tissues. In adenoid cystic carcinoma cases, mainly dense clusters of basaloid cells and spherical hyaline globules have been observed.10,16,18,21 In cases of carcinoma ex PA, it is important to identify nuclear atypia.18,22 A neurogenic tumor should always be included in the differential diagnosis, and presence of metachromatic dense fibrillary materials is suggestive of PA.18
- One case wrongly diagnosed as mucoepidermoid carcinoma revealed a low cellular yield and a few epithelial cell clusters containing squamoid and mucous cell-like cells. These cells might be related to squamous metaplasia and microcystic change based on the histological findings. However, on review of the FNAC slide, low cellularity, no obvious mucous or squamoid cells, and inconspicuous nuclear atypia were not compatible with the cytologic features of the mucoepidermoid carcinoma. In the 2 cases overdiagnosed as malignant or atypical, the cause of discrepancy was related to a lack of typical cytomorphology, and the FNAC smears showed low cellularity; however, the nuclear atypia was overestimated. Histologically, one of the cases revealed a spindle cellular mass with very scant epithelial elements. To avoid diagnostic pitfalls in PA cases, identification of the three main cytologic components is required.16,18 In one case that was misdiagnosed as metastatic carcinoma, it was due to mislabeling as a lymph node instead of a salivary gland; therefore, recognition of the clinical information and radiological findings is essential before examination of FNAC smears.
- Of the 11 PA cases showing minor discrepancies, 7 cases were diagnosed as simply benign, and the major cause of the misinterpretations as a simply benign epithelial lesion or tumor was an inadequate sample. On cytologic review, a poor cellular yield and a small quantity of stromal components were associated. Two of the 11 cases were misdiagnosed as a spindle cell tumor (ruling out schwannoma), and these tumors showed predominant spindle cell populations histologically, but focal typical epithelial cells or stromal cells were observed on review of the FNAC smears. Another case, that was interpreted as ruling out myoepithelioma or monomorphic adenoma, showed degenerated epithelial or spindle cells, and histologically this case showed variegated histologic features with degenerative changes. In a case misinterpreted as suppurative inflammation, the small sized naked cells in the dirty background were misinterpreted as degenerated inflammatory cells.
- In summary, the major causes showing diagnostic discrepancies in PA were poor cellularity, a lack of the three main cytologic components and the presence of a lack of typical cytomorphology related to degenerative change.
- A WT is the second common benign tumor of the salivary gland after PA,23 and a WT occurs mainly in the parotid gland bilaterally in about 10% of the cases, has a slight male dominance, and is related with cigarette smoking.12,20,23,24 Histologically, WT characteristically consists of cystic and solid areas. The cystic area is lined with two layers of tall and columnar oncocytic luminal cells and flattened or cuboidal basal cells, while the stroma consists of lymphoid tissues.12,20,25 Cytologically a WT shows variable cellularity, and consists of 3 components including epithelial cells, lymphocytes, and a fluidy background. Epithelial cells are oncocytic cells with a low nuclear/cytoplasmic (N/C) ratio, large bland nuclei, prominent nucleoli, abundant eosinophilic and granular cytoplasm, and a sharply demarcated cytoplasmic border. The epithelial cells present as monolayered sheets or as multilayered cohesive sheets with a 2 or 3 cell thickness. Most of the lymphocytes are small and mature, and dispersed single cells. A dirty background containing cell debris can be observed, and might show a mucoid substance or calcifications and crystals. Infrequently mast cells, siderophages, giant cells, sebaceous gland cells, metaplastic squamous cells, and spindle cells can be observed.15,20,26
- In cases showing the three components, oncocytic epithelial cells or sheets, scattered lymphocytes, and a dirty background, the diagnosis of a WT can be made without difficulty. However, the above-mentioned three components are not characteristics of only WT.20 Differential diagnoses of WT include chronic sialadenitis, and a lymphoepithelial cyst, in which oncocytic epithelial cells and lymphocytes appear together.16,20 Oncocytoma and oncocytic carcinoma may also contain lymphocytes, but the oncocytic cell layers of these neoplasm are larger than that of oncocytic cells of WT, and has a thickness of 3 or more cells.15,20 The eosinophilic cells of acinic cell carcinoma may be confused with WT, but zymogen granules, cellular pleomorphism, and prominent nucleoli are seen in cases of acinic cell carcinoma.16,20 Intermediate squamoid cells of mucoepidermoid carcinoma, or metaplastic squamous or oncocytic cells of PA can also be confused with WT.10,15,16 In particular, one of the epithelial and lymphoid components has been predominant in WT, and diagnostic difficulties have frequently occurred in that situation.15
- In this study, only 4 of the 12 WT cases showed diagnostic concordance between the FNAC diagnosis and histologic diagnosis. The diagnostic accuracy of WT was 33.3% (4/12), which was a relatively low concordance rate compared with the previous studies, which had 33-100% accuracies.3,10,15,27 Of the 12 WT cases, 6 showed minor discrepancies, and 4 cases of the 6 were diagnosed as simply benign, and the other 2 cases were diagnosed as an the inflammatory condition or ruling out monomorphic adenoma. The 2 cases that were diagnosed as simply benign were misdiagnosed due to inadequate aspirates. In both of the cases, the smeared cells were mostly lymphoid cells with rare epithelial or stromal components. Cystic change in a WT can occur, and aspirates from this cystic tumor in our cases revealed a relatively poor cellular yield and individual degenerated epithelial cells with an occasional squamoid feature, lacking oncocytic cells. Interpretation errors were noted in 2 cases, and one of the cases that was diagnosed as an inflammatory condition showed cell debris or a variable sized or shaped degenerated cells mixed with the inflammatory cells in the necrotic background instead of epithelial or oncocytic cells and rich lymphoid cells. The other case that was diagnosed as ruling out monomorphic adenoma was regarded as an interpretation error. On cytologic-histologic review, the diagnostic discrepancies in the WT cases were due to inadequate aspirates in 2 cases, poor or rare oncocytic cells associated with cystic change in 2 cases, and interpretation error in 2 cases. Another 2 cases were mislabeled as lymph nodes, clinically, and one of the cases was misdiagnosed as rule out metastasis based on the presence of squamoid cells, but the smeared epithelial cells were described as not so atypical, and the other case that was diagnosed as a benign condition had smeared cells that were interpreted as benign follicular lymphoid cells.
- To increase the diagnostic accuracy in benign salivary glandular lesions, triple assessment consisting of cytologic features, clinical information, and radiologic findings is essential. Recognition of aspiration sites and the correlation with radiologic findings are important, and a detailed cytologic examination based on both typical and not typical cytologic features will be needed.
DISCUSSION
- 1. Kechagias N, Ntomouchtsis A, Valeri R, et al. Fine-needle aspiration cytology of salivary gland tumours: a 10-year retrospective analysis. Oral Maxillofac Surg 2012; 16: 35-40. ArticlePubMedPDF
- 2. Layfield LJ, Gopez EV. Cystic lesions of the salivary glands: cytologic features in fine-needle aspiration biopsies. Diagn Cytopathol 2002; 27: 197-204. ArticlePubMed
- 3. Paik I, Lee HK, Lee YD. The role of fine needle aspiration cytology of salivary gland tumors. J Korean Surg Soc 1999; 57: 533-540.
- 4. Zhang S, Bao R, Bagby J, Abreo F. Fine needle aspiration of salivary glands: 5-year experience from a single academic center. Acta Cytol 2009; 53: 375-382. PubMed
- 5. Singh Nanda KD, Mehta A, Nanda J. Fine-needle aspiration cytology: a reliable tool in the diagnosis of salivary gland lesions. J Oral Pathol Med 2012; 41: 106-112. ArticlePubMed
- 6. Brennan PA, Davies B, Poller D, et al. Fine needle aspiration cytology (FNAC) of salivary gland tumours: repeat aspiration provides further information in cases with an unclear initial cytological diagnosis. Br J Oral Maxillofac Surg 2010; 48: 26-29. ArticlePubMed
- 7. Colella G, Cannavale R, Flamminio F, Foschini MP. Fine-needle aspiration cytology of salivary gland lesions: a systematic review. J Oral Maxillofac Surg 2010; 68: 2146-2153. ArticlePubMed
- 8. David O, Blaney S, Hearp M. Parotid gland fine-needle aspiration cytology: an approach to differential diagnosis. Diagn Cytopathol 2007; 35: 47-56. ArticlePubMed
- 9. Hughes JH, Volk EE, Wilbur DC. Cytopathology Resource Committee, College of American Pathologists. Pitfalls in salivary gland fine-needle aspiration cytology: lessons from the College of American Pathologists Interlaboratory Comparison Program in Nongynecologic Cytology. Arch Pathol Lab Med 2005; 129: 26-31. ArticlePubMedPDF
- 10. Kim MJ, Lee TS. A cytologic study of fine needle aspiration biopsy of salivary gland diseases. Korean J Cytopathol 1994; 5: 120-129.
- 11. Martin HE, Ellis EB. Biopsy by needle puncture and aspiration. Ann Surg 1930; 92: 169-181. ArticlePubMedPMC
- 12. Veder LL, Kerrebijn JD, Smedts FM, den Bakker MA. Diagnostic accuracy of fine-needle aspiration cytology in Warthin tumors. Head Neck 2010; 32: 1635-1640. ArticlePubMed
- 13. Stewart CJ, MacKenzie K, McGarry GW, Mowat A. Fine-needle aspiration cytology of salivary gland: a review of 341 cases. Diagn Cytopathol 2000; 22: 139-146. ArticlePubMed
- 14. Daneshbod Y, Daneshbod K, Khademi B. Diagnostic difficulties in the interpretation of fine needle aspirate samples in salivary lesions: diagnostic pitfalls revisited. Acta Cytol 2009; 53: 53-70. PubMed
- 15. Parwani AV, Ali SZ. Diagnostic accuracy and pitfalls in fine-needle aspiration interpretation of Warthin tumor. Cancer 2003; 99: 166-171. ArticlePubMed
- 16. Park A, Kim HK, Kim DW, Jin SY, Lee DW. Fine needle aspiration cytology of the salivary gland: an analysis of 221 cases. Korean J Cytopathol 1999; 10: 133-143.
- 17. Spiro RH. Salivary neoplasms: overview of a 35-year experience with 2,807 patients. Head Neck Surg 1986; 8: 177-184. ArticlePubMed
- 18. Gahine R, Sudarshan V, Hussain N, Krishnani C. Pleomorphic adenoma: a diagnostic pitfall in the diagnosis of salivary gland lesions on FNAC: case reports with review of the literature. Cytojournal 2010; 7: 17.ArticlePubMedPMC
- 19. Ito FA, Jorge J, Vargas PA, Lopes MA. Histopathological findings of pleomorphic adenomas of the salivary glands. Med Oral Patol Oral Cir Bucal 2009; 14: E57-E61. PubMed
- 20. Mukunyadzi P. Review of fine-needle aspiration cytology of salivary gland neoplasms, with emphasis on differential diagnosis. Am J Clin Pathol 2002; 118(Suppl): S100-S115. ArticlePubMed
- 21. Lee SS, Cho KJ, Jang JJ, Ham EK. Differential diagnosis of adenoid cystic carcinoma from pleomorphic adenoma of the salivary gland on fine needle aspiration cytology. Acta Cytol 1996; 40: 1246-1252. ArticlePubMed
- 22. Kim KM, Lee A, Yoon SH, Kang JH, Shim SI. Carcinoma ex pleomorphic adenoma of the palate: a case report. J Korean Med Sci 1997; 12: 63-66. ArticlePubMedPMC
- 23. Daguci L, Simionescu C, Stepan A, Munteanu C, Dăguci C, Bătăiosu M. Warthin tumor: morphological study of the stromal compartment. Rom J Morphol Embryol 2011; 52: 1319-1323. PubMed
- 24. Chedid HM, Rapoport A, Aikawa KF, Menezes Ados S, Curioni OA. Warthin's tumor of the parotid gland: study of 70 cases. Rev Col Bras Cir 2011; 38: 90-94. ArticlePubMed
- 25. Faur A, Lazăr E, Cornianu M, Dema A, Vidita CG, Găluşcan A. Warthin tumor: a curious entity: case reports and review of literature. Rom J Morphol Embryol 2009; 50: 269-273. PubMed
- 26. Flezar M, Pogacnik A. Warthin's tumour: unusual vs. common morphological findings in fine needle aspiration biopsies. Cytopathology 2002; 13: 232-241. ArticlePubMedPDF
- 27. Ballo MS, Shin HJ, Sneige N. Sources of diagnostic error in the fine-needle aspiration diagnosis of Warthin's tumor and clues to a correct diagnosis. Diagn Cytopathol 1997; 17: 230-234. ArticlePubMed
REFERENCES
Fig. 1Cytologic and histologic findings of pleomorphic adenoma case misdiagnosed as mucoepidermoid carcinoma. The fine needle aspiration cytology smear shows a few cellular clusters with squamoid features (A), and histologically squamous metaplasia and microcystic changes are observed (B).
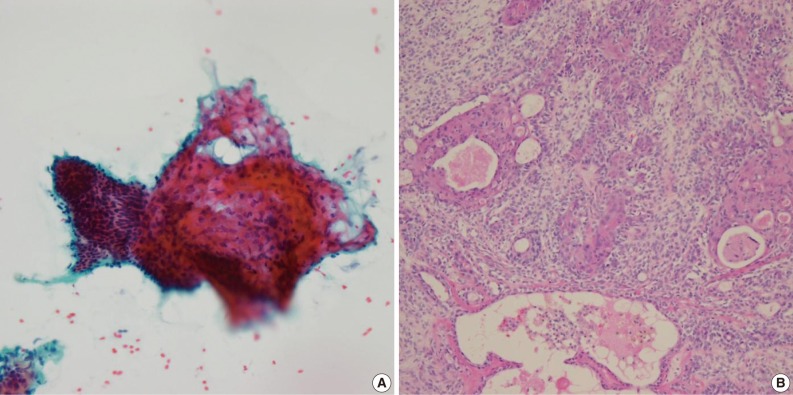

Fig. 2Cytologic findings of pleomorphic adenoma cases misdiagnosed as an inflammatory condition. The fine needle aspiration cytology smear displays low cellularity with a lack of three cellular components such as epithelial, myoepithelial and stromal origin (A), and the nuclear debris in the necrotic background that are misinterpreted as inflammatory cells (B).
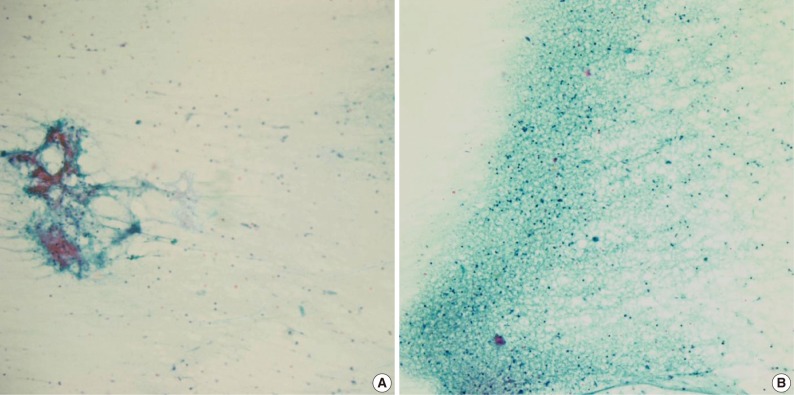

Fig. 3Cytologic findings of the Warthin's tumor cases showing diagnostic discrepancies. The typical cytologic features of Warthin's tumor consist of oncocytic cell clusters and mature lymphocytes in the necrotic background (A). A case, diagnosed as simply benign, shows degenerated or squamoid cells mixed with inflammatory cells instead of oncocytic cell clusters (B). Another Warthin's tumor case with cystic change reveals cell debris in the necrotic background (C), and scanty spindled or squamoid cells mixed with cell debris (D).
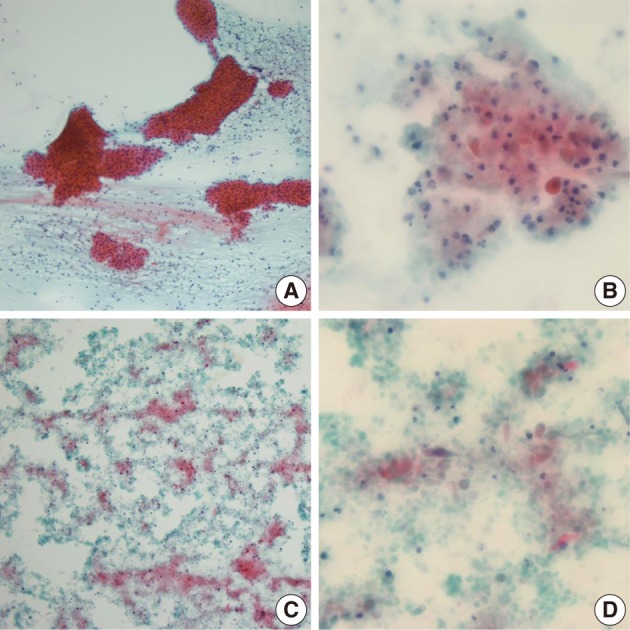

Figure & Data
References
Citations
Citations to this article as recorded by 

- Deep Learning Based on Ultrasound Images Differentiates Parotid Gland Pleomorphic Adenomas and Warthin Tumors
Yajuan Li, Mingchi Zou, Xiaogang Zhou, Xia Long, Xue Liu, Yanfeng Yao
Ultrasonic Imaging.2025; 47(3-4): 107. CrossRef - Clinical decision making when cytology indicates a Warthin tumor
Minna Sirviö, Katri Aro, Mira Naukkarinen, Antti Mäkitie, Jussi Tarkkanen, Jetta Kelppe, Timo Atula
Scientific Reports.2024;[Epub] CrossRef - Different MRI-based radiomics models for differentiating misdiagnosed or ambiguous pleomorphic adenoma and Warthin tumor of the parotid gland: a multicenter study
Jing Yang, Qiu Bi, Yiren Jin, Yong Yang, Ji Du, Hongjiang Zhang, Kunhua Wu
Frontiers in Oncology.2024;[Epub] CrossRef - Diagnostic Accuracy of Fine Needle Aspiration Cytology (FNAC) in Salivary Gland Lesions with Histopathological Examination (HPE) Correlation in a Tertiary Care Centre in Southern India
Banumathi Kasinathan, Babu Manohar, H. Ganapathy
Indian Journal of Otolaryngology and Head & Neck Surgery.2023; 75(2): 871. CrossRef - Diagnostic performance of qualitative and radiomics approach to parotid gland tumors: which is the added benefit of texture analysis?
Federica Vernuccio, Federica Arnone, Roberto Cannella, Barbara Verro, Albert Comelli, Francesco Agnello, Alessandro Stefano, Rosalia Gargano, Vito Rodolico, Giuseppe Salvaggio, Roberto Lagalla, Massimo Midiri, Antonio Lo Casto
The British Journal of Radiology.2021;[Epub] CrossRef - Improving the diagnosis of common parotid tumors via the combination of CT image biomarkers and clinical parameters
Dan Zhang, Xiaojiao Li, Liang Lv, Jiayi Yu, Chao Yang, Hua Xiong, Ruikun Liao, Bi Zhou, Xianlong Huang, Xiaoshuang Liu, Zhuoyue Tang
BMC Medical Imaging.2020;[Epub] CrossRef Evaluation of Salivary Gland Lesions by Fine Needle Aspiration Cytology at a Tertiary Care Hospital, Western Nepal
Anuj Poudel, Bigya Shrestha, Sudeep Regmi
Pathology and Laboratory Medicine International.2020; Volume 12: 9. CrossRef- Ultrasound-guided fine-needle capillary cytology of parotid gland masses coupled with a rapid-on-site evaluation improves results
R. Barats, S. Evrard, L. Collin, S. Vergez, S. Gellée, M. Courtade-Saïdi
Morphologie.2018; 102(336): 25. CrossRef - The Value of Ultrasound-Guided Fine-Needle Aspiration Cytology by Cytopathologists in the Diagnosis of Major Salivary Gland Tumors
Shahrzad Negahban, Sadegh Shirian, Bijan Khademi, Ahmad Oryan, Roshanak Sadoughifar, Mohammadian-Panah Mohammad, Azita Aledavood, Khosrow Daneshbod, Yayha Daneshbod
Journal of Diagnostic Medical Sonography.2016; 32(2): 92. CrossRef - Salivary Gland Tumors: A Diagnostic Dilemma!
Ranjit Kumar Peravali, H. Hari Kishore Bhat, Varsha H. Upadya, Anmol Agarwal, Sushma Naag
Journal of Maxillofacial and Oral Surgery.2015; 14(S1): 438. CrossRef - Diagnostic problems of salivary gland tumors
Ruchita Tyagi, Pranab Dey
Diagnostic Cytopathology.2015; 43(6): 495. CrossRef - Pleomorphic Adenoma of the Accessory Parotid Gland: Case Report and Reappraisal of Intraoral Extracapsular Dissection for Management
Tibebu M. Tsegga, Jennifer D. Britt, Aragon R. Ellwanger
Journal of Oral and Maxillofacial Surgery.2015; 73(3): 564. CrossRef
Diagnostic Difficulties in Fine Needle Aspiration of Benign Salivary Glandular Lesions
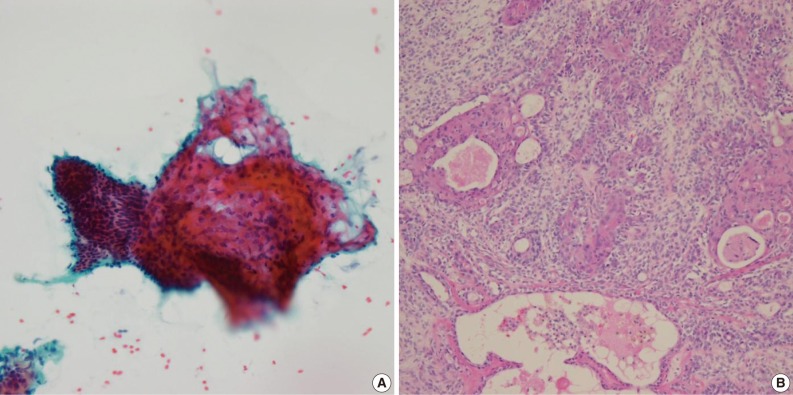
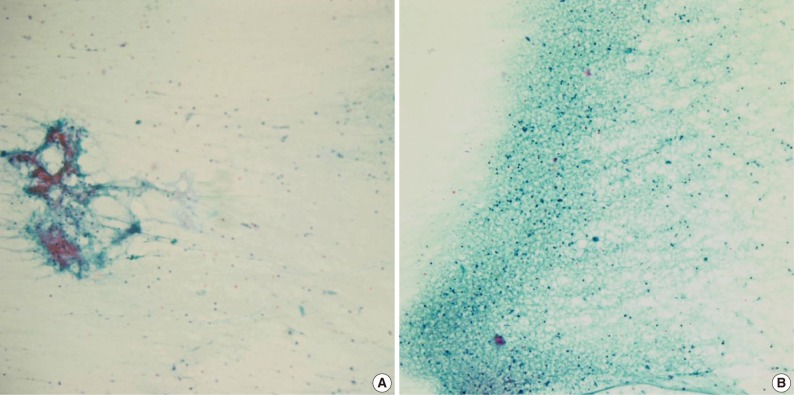
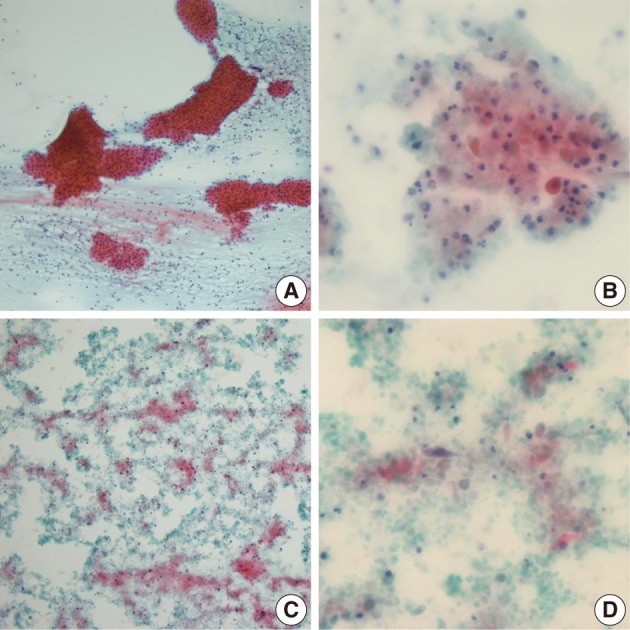
Fig. 1 Cytologic and histologic findings of pleomorphic adenoma case misdiagnosed as mucoepidermoid carcinoma. The fine needle aspiration cytology smear shows a few cellular clusters with squamoid features (A), and histologically squamous metaplasia and microcystic changes are observed (B).
Fig. 2 Cytologic findings of pleomorphic adenoma cases misdiagnosed as an inflammatory condition. The fine needle aspiration cytology smear displays low cellularity with a lack of three cellular components such as epithelial, myoepithelial and stromal origin (A), and the nuclear debris in the necrotic background that are misinterpreted as inflammatory cells (B).
Fig. 3 Cytologic findings of the Warthin's tumor cases showing diagnostic discrepancies. The typical cytologic features of Warthin's tumor consist of oncocytic cell clusters and mature lymphocytes in the necrotic background (A). A case, diagnosed as simply benign, shows degenerated or squamoid cells mixed with inflammatory cells instead of oncocytic cell clusters (B). Another Warthin's tumor case with cystic change reveals cell debris in the necrotic background (C), and scanty spindled or squamoid cells mixed with cell debris (D).
Fig. 1
Fig. 2
Fig. 3
Diagnostic Difficulties in Fine Needle Aspiration of Benign Salivary Glandular Lesions


Table 1 Causes of diagnostic discrepancy in the 61 cases of pleomorphic adenoma
Table 2 Causes of diagnostic discrepancy in the 12 cases of Warthin's tumors

 E-submission
E-submission


 PubReader
PubReader Cite this Article
Cite this Article




