Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 46(6); 2012 > Article
-
Original Article
Fine-Needle Aspiration Cytology of Pleomorphic Carcinomas of the Lung - Hee Seung Choi, Hyesil Seol, Il Yeong Heo, Chang Won Jung, Soo Youn Cho, Sunhoo Park, Jae Soo Koh, Seung-Sook Lee
-
Korean Journal of Pathology 2012;46(6):576-582.
DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.6.576
Published online: December 26, 2012
Department of Pathology, Korea Cancer Center Hospital, Korea Institute of Radiological and Medical Sciences, Seoul, Korea.
- Corresponding Author: Hyesil Seol, M.D. Department of Pathology, Korea Cancer Center Hospital, 75 Nowon-ro, Nowon-gu, Seoul 139-706, Korea. Tel: +82-2-970-2545, Fax: +82-2-970-2430, Hyesilseol@gmail.com
© 2012 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Pleomorphic carcinoma (PC) is a rare pulmonary malignancy. Because of its rarity and histological heterogeneity, cytopathologists might suspect PC only rarely on the basis of its cytological specimen. In addition, cytological findings from fine needle aspiration (FNA) specimens have rarely been described. Hence, we investigated the cytological features of FNA in the cases of PC.
-
Methods
- We reviewed 7 FNA specimens of PC. The patients had undergone surgical resection at the Korea Cancer Center Hospital between 2007 and 2011. The cytological features of PC were assessed and compared with the histopathological features of the corresponding surgical specimen. Immunocytochemical analysis with cytokeratin and vimentin was performed on the cell blocks.
-
Results
- The tumor cells were either dispersed or arranged in loose aggregates, and generally lacked any glandular or squamous differentiation. Pleomorphic or spindle shape tumor cells were observed, and mono-, bi-, or multi-nucleated giant cells were frequently observed. The background showed necrosis and contained numerous lymphocytes and neutrophils. Immunocytochemically, the tumor cells were positive for cytokeratin and vimentin.
-
Conclusions
- PC displays characteristic cytological features. It might therefore be possible to make an accurate diagnosis of PC by assessing the degree of nuclear atypia.
- Patients
- A retrospective review was performed for all the patients diagnosed with PC who had undergone surgery at the Korea Cancer Center Hospital from January 2007 to December 2011. Patients who were diagnosed with PC via the surgical specimen, and those who underwent preoperative FNA were included in this study. We excluded patients who had received preoperative chemotherapy or radiotherapy because of possible modifications of the shape and size of the tumor cells. In the end, 7 of the patients (5 men, 2 women) with a mean age of 67 years (range, 60 to 78 years) were included in this study, of which 6 were smokers. Clinicopathological parameters such as tumor size, tumor location, stage, and distant metastases were collected from the patients' medical records.
- FNA cytology
- FNA was performed by radiologists using a 23-gauge needle with or without anesthesia. The aspirated material was smeared onto 4 glass slides, immediately fixed in 95% alcohol and stained with a Papanicolaou stain. The cell blocks were prepared from the remainder or by rinsing the syringes. Immunocytochemical studies for cytokeratin and vimentin were performed on paraffin sections of the cell blocks in all cases. All of the surgical resection specimens had been fixed with 10% formalin and embedded in paraffin. A mean of 6.5 (range, 5 to 8) hematoxylin and eosin-stained slides per tumor was available. All the slides were reviewed with a multi-headed microscope by two pathologists. The cytological features were assessed and compared with the histopathological features of the corresponding surgical specimen. We defined the epithelial component of PC as malignant tumor cells with glandular or squamous differentiation. A component of large cell carcinoma was also included in the epithelial component of PC.
- We defined the large cell carcinoma component as tumor cells with a tendency to form loosely structured clusters composed of epithelial cells of unequal sizes without glandular or squamous differentiation. We defined the sarcomatoid component of PC as malignant giant and/or spindle cells.14
- The investigated cytological parameters were as follows: component of tumor cells, architectural pattern (fascicles, single cells, and cluster), adhesion (loose or tight), nuclei (pleomorphism, variability in size, number, and size of nucleoli, nuclear membrane, and chromatin texture), nature of the cytoplasm, nuclear-to-cytoplasmic ratio and background.
MATERIALS AND METHODS
- Clinicopathological features
- The clinico-pathological data and the original cytological diagnosis are summarized in Table 1. The mean tumor was 41 mm in diameter (range, 25 to 70 mm). Lobectomy with lymph node dissection was performed in 6 cases, and metastasectomy was performed in only 1 case (case 5). Case 1 had multiple bronchioloalveolar carcinomas of both lungs and renal cell carcinoma at the time of surgery. Two patients had pleura and chest wall invasion and four had regional lymph node metastasis. Three patients had distant metastases in the brain, adrenal glands, intestine, bone, and lung. The tumor stages were IIA in 1 case, IIB in 1 case, IIIA in 3 cases, and IV in 2 cases. Adjuvant chemotherapy was performed in 3 patients and adjuvant chemoradiation therapy was performed in 2 patients.
- Cytologic features
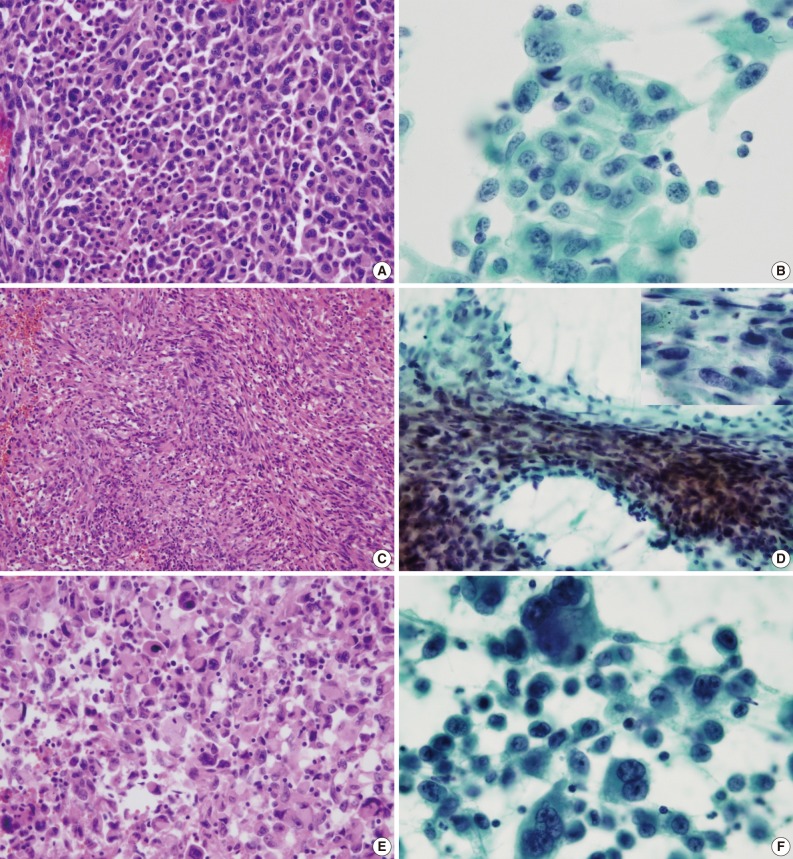
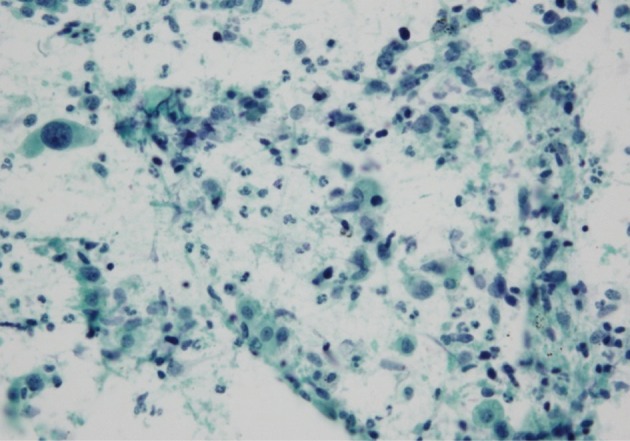
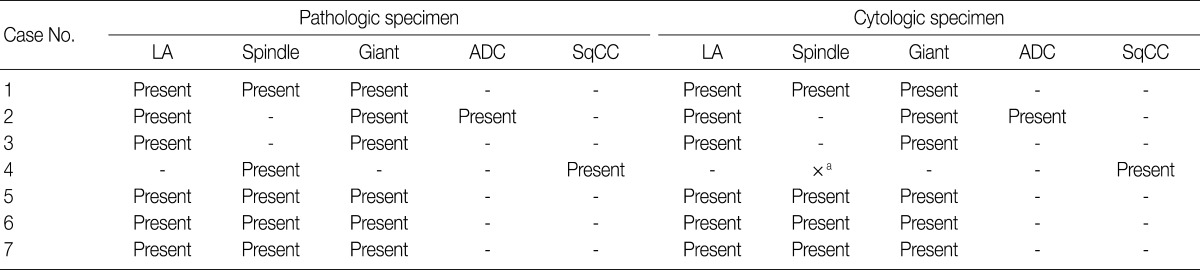
- The cell component of the cytological specimens usually reflected the components of the corresponding surgical specimen. The epithelial component indicated large cell carcinoma in 6 cases, and adenocarcinoma and squamous cell carcinoma in 1 case each. The spindle cell component was observed in 4 cases, and the GC component was observed in 6 cases (Fig. 1). The components of tumor cells in the pathological and cytological specimens are listed in Table 2.
- The cluster size of tumor cells varied. The tumor cells were arranged in monolayers, 3-dimensional clusters, or were scattered as single cells. Cohesive clusters were observed in some cases, but loose clusters were observed in most of the cases. The tumor cells were large, and the pleomorphism was marked. The shape of the tumor cells was epithelioid, spindle, or pleomorphic. The tumor cell size varied by more than 5-fold in 6 cases. The epithelial tumor cells had an irregular nuclear membrane and a coarsely granular chromatin (Fig. 1B). The chromatin was unevenly distributed in most of the cases. Most of the cells usually showed a prominent single nucleolus or nucleoli. The cytoplasm was abundant, thick, and well-demarcated. The malignant spindle cells were arranged singly or in fascicles (Fig. 1D). They had scant amount of cytoplasm with ill-defined margin and hyperchromatic nuclei. Mono-, bi-, and multi-nucleated GCs were frequently observed (Fig. 1F); they usually occurred singly. The nucleus of the giant tumor cells was hyperchromatic to vesicular, bizarre in shape, and more than 5 times the size of the resting lymphocytes, and often much larger. The nuclear size ranged from 15 µm to 65 µm. The background contained tumor diathesis with numerous polymorphonuclear leukocytes (Fig. 2). In the cell block, two different cellular patterns were seen: areas with aggregating pleomorphic sarcomatoid cells and/or entrapped spindle cells, and epithelioid tumor cells. Immunoreactivity to cytokeratin was present in the epithelial cells in all of the cases and in some of the spindle cells in 2 cases (focal, weakly positive). Vimentin was expressed in some of the epithelial components in 4 cases (focal, strongly positive) and in the sarcomatoid component of all the cases (Fig. 3). The summary of these cytological features of PC is listed in Table 3.
RESULTS
- There have been only a few reports of cytological analysis in cases of pulmonary PC.14-16 Most of the previous cytological reports have revealed similar findings. Hummel et al.15 reported that the cytological findings of PC included a conspicuous population of pleomorphic spindle cells arranged singly, in loose clusters, and in fascicles, and as micro-tissue fragments in a necrotic background. The myxoid stromal fragments were also present. In addition, cohesive clusters of atypical epithelial cells and multinucleated GCs were noted. Although malignant spindle cells and GCs can be helpful cytological features, PC of the lung is challenging to diagnose because it has very poor cellular differentiation, rare incidence and histological heterogeneity. In this context, histopathological correlation is an important prerequisite of a correct cytological evaluation. The surgical specimens in case 2 were composed of GC carcinoma, large cell carcinoma, and moderately differentiated adenocarcinoma. However, the cytologic specimen mainly consisted of clusters of adenocarcinoma and only a few scattered giant tumor cells. Because of the morphological continuum and overlapping features that exist between giant tumor cells and poorly differentiated large cell carcinoma, the original cytological diagnosis of this case was "non-small cell carcinoma (NSCC), favoring adenocarcinoma." The cytological specimen in case 4, which was misdiagnosed as "NSCC, favoring squamous cell carcinoma," mainly revealed tumor diathesis and some malignant squamoid cells, but not spindle cells, although squamous cell carcinoma and spindle tumor cells were marked in the surgical specimens. The sarcomatoid component was missed because of the low cellularity.
- GCs are one component of PC. However, there is no clear borderline between giant tumor cells and poorly differentiated large cell carcinoma. Fishback et al.2 reported that the single large pleomorphic nucleus of a GC was 4 times larger than that of small resting lymphocytes. Guillan and Zelman17 reported that the GCs varied in diameter from 50 µm to 120 µm, and Hellstrom and Fisher18 reported that the GCs measured from 80 µm to 100 µm. This vague definition of GCs has caused confusion among pathologists. In our study, the size of GCs varied, ranging from 15 µm to 65 µm. The nucleus of mononucleated giant tumor cells measured more than 5 times larger than that of small resting lymphocytes.
- Although the final diagnosis of PC relies in most cases on the close scrutiny of the hematoxylin and eosin stained sections, its immunohistochemical characterization may better highlight the different cell components occurring in these tumors. In several studies, the epithelial component has been stained to various extents with antibodies to cytokeratin, whereas the sarcomatous component is consistently immunoreactive for vimentin.2,10,19-22 In this current study, all tumors displayed dual expression of cytokeratin and vimentin by immunocytochemistry on the cell blocks. The sarcomatoid tumor cells were uniformly positive for vimentin and focally positive for cytokeratin. In contrast, the epithelioid tumor cells showed focal or diffuse positivity for cytokeratin and focally expressed vimentin.
- The prognosis for PC is generally thought to be worse than that for conventional NSCLC.2,9,10,12,23-25 A stage-adjusted comparison of 63 cases of PSC with an equivalent number of propensity score-matched cases of NSCLCs showed the 5-year survival to be significantly different between the 2 groups (24.5% for the PSC group and 46.3% for the NSCLC group),23 favoring the hypothesis that the prognosis of PSC may be poorer than that of NSCLC.24 The estimated 5-year survival rate in surgical series was up to 33% for PC (with the median overall survival ranging from 5 to 19 months).2,9,10,23-25
- PC has been reported to be highly metastatic. Chang et al.10 reported that 7 patients who had undergone surgical resections succumbed to early distant metastases in organs, such as the brain, bone, adrenal gland, and unusual sites including esophagus, jejunum, rectum, and kidney, within a few months. In the present study, metastasis was observed in 3 cases: one patient (case 2) had metastasis in both adrenal glands; one (case 5) had brain, small intestine, and mesentery metastasis at the time of diagnosis; and the third (case 7) had metastasis to the brain, bone, lung, and both adrenal glands. Two patients died of the disease, 1 patient survived with metastasis, and 4 patients survived without recurrence or metastasis.
- In conclusion, recognition of PC may be possible when adequate cytological material is present. The presence of loose clusters of poorly differentiated epithelial cells, malignant spindle cells, mono-, bi-, multi-nucleated GCs, and a necrotic background with lymphocytes and neutrophils is highly suggestive of PC.
DISCUSSION
- 1. Travis W, Brambilla E, Müller-Hermelink HK, Harris CC. World Health Organization classification of tumours: pathology and genetics of tumours of the lung, pleura, thymus and heart. 2004; Lyon: IARC Press, 53-58.
- 2. Fishback NF, Travis WD, Moran CA, Guinee DG Jr, McCarthy WF, Koss MN. Pleomorphic (spindle/giant cell) carcinoma of the lung: a clinicopathologic correlation of 78 cases. Cancer 1994; 73: 2936-2945. ArticlePubMed
- 3. Matsui K, Kitagawa M, Miwa A. Lung carcinoma with spindle cell components: sixteen cases examined by immunohistochemistry. Hum Pathol 1992; 23: 1289-1297. ArticlePubMed
- 4. Terzi A, Gorla A, Piubello Q, Tomezzoli A, Furlan G. Biphasic sarcomatoid carcinoma of the lung: report of 5 cases and review of the literature. Eur J Surg Oncol 1997; 23: 457.Article
- 5. Pelosi G, Sonzogni A, De Pas T, et al. Review article: pulmonary sarcomatoid carcinomas: a practical overview. Int J Surg Pathol 2010; 18: 103-120. ArticlePubMedPDF
- 6. Brambilla E, Travis WD, Colby TV, Corrin B, Shimosato Y. The new World Health Organization classification of lung tumours. Eur Respir J 2001; 18: 1059-1068. ArticlePubMed
- 7. Travis WD. Pathology of lung cancer. Clin Chest Med 2002; 23: 65-81. ArticlePubMed
- 8. Travis WD, Travis LB, Devesa SS. Lung cancer. Cancer 1995; 75(1 Suppl): 191-202. ArticlePubMed
- 9. Rossi G, Cavazza A, Sturm N, et al. Pulmonary carcinomas with pleomorphic, sarcomatoid, or sarcomatous elements: a clinicopathologic and immunohistochemical study of 75 cases. Am J Surg Pathol 2003; 27: 311-324. PubMed
- 10. Chang YL, Lee YC, Shih JY, Wu CT. Pulmonary pleomorphic (spindle) cell carcinoma: peculiar clinicopathologic manifestations different from ordinary non-small cell carcinoma. Lung Cancer 2001; 34: 91-97. ArticlePubMed
- 11. Wick MR, Ritter JH, Humphrey PA. Sarcomatoid carcinomas of the lung: a clinicopathologic review. Am J Clin Pathol 1997; 108: 40-53. ArticlePubMed
- 12. Raveglia F, Mezzetti M, Panigalli T, et al. Personal experience in surgical management of pulmonary pleomorphic carcinoma. Ann Thorac Surg 2004; 78: 1742-1747. ArticlePubMed
- 13. Franks TJ, Galvin JR. Sarcomatoid carcinoma of the lung: histologic criteria and common lesions in the differential diagnosis. Arch Pathol Lab Med 2010; 134: 49-54. ArticlePubMedPDF
- 14. Hiroshima K, Dosaka-Akita H, Usuda K, et al. Cytological characteristics of pulmonary pleomorphic and giant cell carcinomas. Acta Cytol 2011; 55: 173-179. ArticlePubMedPDF
- 15. Hummel P, Cangiarella JF, Cohen JM, Yang G, Waisman J, Chhieng DC. Transthoracic fine-needle aspiration biopsy of pulmonary spindle cell and mesenchymal lesions: a study of 61 cases. Cancer 2001; 93: 187-198. ArticlePubMed
- 16. Zafar N, Johns CD. Pleomorphic (sarcomatoid) carcinoma of lung: cytohistologic and immunohistochemical features. Diagn Cytopathol 2011; 39: 115-116. ArticlePubMed
- 17. Guillan RA, Zelman S. Giant-cell carcinoma of the lungs: an analysis of 12 cases. Am J Clin Pathol 1966; 46: 427-432. ArticlePubMed
- 18. Hellstrom HR, Fisher ER. Giant cell carcinoma of lung. Cancer 1963; 16: 1080-1088. ArticlePubMed
- 19. Ro JY, Chen JL, Lee JS, Sahin AA, Ordóñez NG, Ayala AG. Sarcomatoid carcinoma of the lung: immunohistochemical and ultrastructural studies of 14 cases. Cancer 1992; 69: 376-386. ArticlePubMed
- 20. Nakajima M, Kasai T, Hashimoto H, Iwata Y, Manabe H. Sarcomatoid carcinoma of the lung: a clinicopathologic study of 37 cases. Cancer 1999; 86: 608-616. ArticlePubMed
- 21. Humphrey PA, Scroggs MW, Roggli VL, Shelburne JD. Pulmonary carcinomas with a sarcomatoid element: an immunocytochemical and ultrastructural analysis. Hum Pathol 1988; 19: 155-165. ArticlePubMed
- 22. Pelosi G, Fraggetta F, Nappi O, et al. Pleomorphic carcinomas of the lung show a selective distribution of gene products involved in cell differentiation, cell cycle control, tumor growth, and tumor cell motility: a clinicopathologic and immunohistochemical study of 31 cases. Am J Surg Pathol 2003; 27: 1203-1215. PubMed
- 23. Martin LW, Correa AM, Ordonez NG, et al. Sarcomatoid carcinoma of the lung: a predictor of poor prognosis. Ann Thorac Surg 2007; 84: 973-980. ArticlePubMed
- 24. Mochizuki T, Ishii G, Nagai K, et al. Pleomorphic carcinoma of the lung: clinicopathologic characteristics of 70 cases. Am J Surg Pathol 2008; 32: 1727-1735. PubMed
- 25. Venissac N, Pop D, Lassalle S, Berthier F, Hofman P, Mouroux J. Sarcomatoid lung cancer (spindle/giant cells): an aggressive disease. J Thorac Cardiovasc Surg 2007; 134: 619-623. ArticlePubMed
REFERENCES

NSCCM, male; RUL, right upper lobe; Chemo, chemotherapy; LUL, left upper lobe; NSCC, non-small cell carcinoma; ADC, adenocarcinoma; LLL, left lower lobe; F, female; SqCC, squamous cell carcinoma; rad, radiotherapy; RLL, right lower lobe.
aThis size is assessed using computed tomography. Metastasectomy is performed in case 5.
Figure & Data
References
Citations

- Sarcomatoid carcinoma in cytology: Report of a rare entity presenting in pleural and pericardial fluid preparations
Atreyee Basu, Andre L. Moreira, Anthony Simms, Tamar C. Brandler
Diagnostic Cytopathology.2019; 47(8): 813. CrossRef - Cytological Evaluation of Pleomorphic Carcinoma of the Lung
Kevin Kuan, Samer N. Khader, Siba El Hussein
Diagnostic Cytopathology.2019; 47(9): 961. CrossRef - Combined small cell carcinoma with giant cell carcinoma component of the lung: A case successfully diagnosed by computed tomography‑guided fine‑needle aspiration cytology
Yusuke Ebisu, Mitsuaki Ishida, Tomohito Saito, Tomohiro Murakawa, Yoshiko Uemura, Koji Tsuta
Oncology Letters.2017;[Epub] CrossRef - Pulmonary Pleomorphic Carcinoma Detected as a Result of Pneumothorax and the Subsequent Occurrence of Multiple Cystic Metastases
Hideaki Yamakawa, Masahiro Yoshida, Masami Yabe, Yuri Baba, Emiri Baba, Hiroaki Katagi, Takeo Ishikawa, Masamichi Takagi, Takeo Nakada, Tadashi Akiba, Kazuyoshi Kuwano
Case Reports in Medicine.2014; 2014: 1. CrossRef - Pulmonary pleomorphic carcinoma with multiple metastases to the right posterior knee complicated by paraneoplastic hypercalcemia
PENG-FEI LI, CHENG-HSIANG LO, SHAN-HAN YANG, PING-YING CHUNG, CHING-LIANG HO
Oncology Letters.2014; 7(2): 452. CrossRef
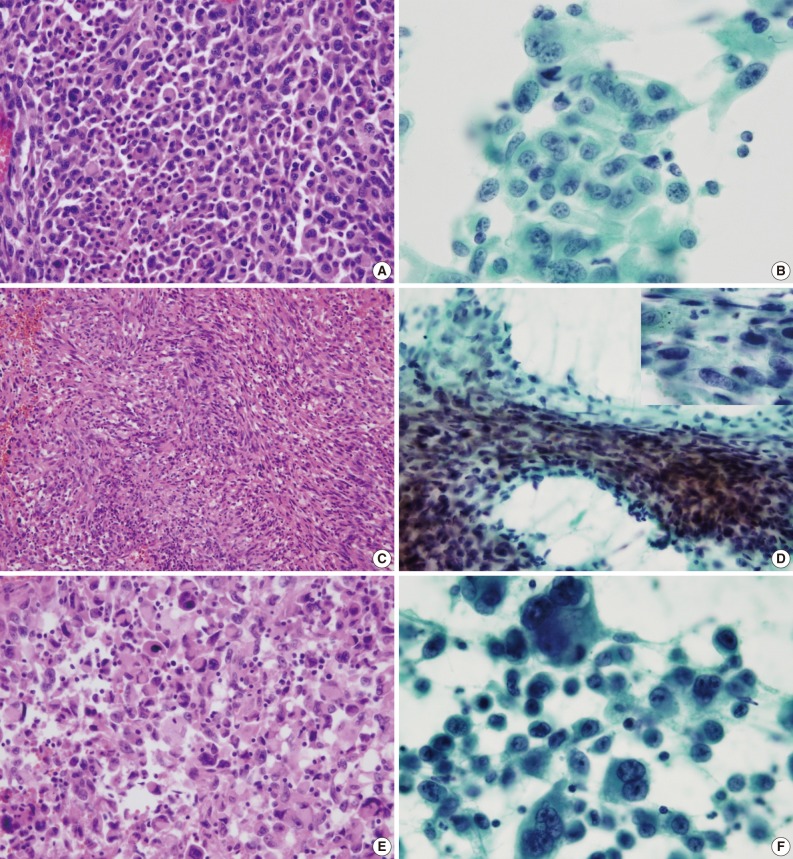
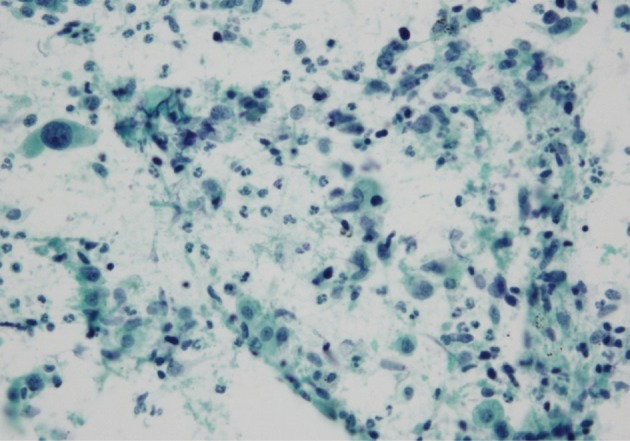

Fig. 1
Fig. 2
Fig. 3



NSCCM, male; RUL, right upper lobe; Chemo, chemotherapy; LUL, left upper lobe; NSCC, non-small cell carcinoma; ADC, adenocarcinoma; LLL, left lower lobe; F, female; SqCC, squamous cell carcinoma; rad, radiotherapy; RLL, right lower lobe. aThis size is assessed using computed tomography. Metastasectomy is performed in case 5.
LA, large cell carcinoma; ADC, adenocarcinoma; SqCC, squamous cell carcinoma. aThe spindle cell component is not observed in the cytological specimens from case 4, contrary to that observed in the corresponding case 4 pathological specimen.
±, positive in the variable minority of tumor cells; +, positive in the majority of tumor cells; -, negative in the tumor cells.

 E-submission
E-submission

 PubReader
PubReader Cite this Article
Cite this Article




