Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 46(5); 2012 > Article
-
Case Report
Extraskeletal Mesenchymal Chondrosarcoma in the Axillary Region: Reports of Two Cases - Chang-Young Seo, Sung-Taek Jung, Jae-Wook Byun
-
Korean Journal of Pathology 2012;46(5):483-488.
DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.5.483
Published online: October 25, 2012
Department of Orthopedic Surgery, Chonnam National University Hospital, Chonnam National University Medical School, Gwangju, Korea.
- Corresponding Author: Sung-Taek Jung, M.D. Department of Orthopedic Surgery, Chonnam National University Hospital, Chonnam National University Medical School, 42 Jebong-ro, Dong-gu, Gwangju 501-757, Korea. Tel: +82-62-227-1640, Fax: +82-62-225-7794, stjung@chonnam.ac.kr
• Received: March 6, 2011 • Revised: December 1, 2011 • Accepted: December 2, 2011
© 2012 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- Extraskeletal mesenchymal chondrosarcomas (EMCs) are relatively uncommon, and a location in the upper extremity, especially in the shoulder or axillary region, is rare. Furthermore, the radiographic findings of EMCs do not show any features that distinguish them from other neoplasms, and therefore, definitive diagnoses are made based on histological features. EMC is an aggressive tumor with a poor prognosis, and requires wide surgical excision. However, its treatment may involve peculiarities such as a difficulty in obtaining a proper surgical margin in the axillary region or shoulder. In this report, the authors present two rare cases of EMCs in the axillary region.
- Case 1
- A 35-year-old carpenter presented with an 8-month history of motor weakness of the left arm and a 3-month history of a palpable mass in the left axillary region. His weight was unchanged, and there was no relevant medical and no notable trauma history to the neck or upper limb.
- An examination of the left axillary region at presentation revealed a 5×5 cm sized, tender, soft tissue mass with a growing tendency. Atrophy of the left deltoid muscle and wrist drop were observed. No sensory deficit was observed in the left arm.
- Plain radiographs of the left shoulder and humerus were normal (Fig. 1A). However, computed tomography (CT) (Fig. 1B) and magnetic resonance imaging (MRI) revealed a well-defined soft tissue mass between the pectoralis major and subscapularis muscles without vascular encasement (Fig. 2). No peripheral or central calcification was observed. Electromyography was performed to evaluate motor weakness of left arm, and revealed incomplete left brachial plexus neuropathy around the posterior cord of the brachial plexus.
- Preoperative angiography depicted many feeding vessels via the thoracodorsal and left circumflex scapular arteries, and these vessels were embolized.
- An excisional biopsy was performed. Grossly the mass, which measured 7×5×4 cm and laid between the pectoralis major and subscapularis muscles, appeared to be well-encapsulated without neurovascular invasion. Sectioned surfaces of the tumor tissue were yellowish-white and glistening, and were basically hard and homogenous, except for a few small cystic and necrotic foci (Fig. 3). Histopathological assessment revealed an EMC with undifferentiated spindle cells and focal areas of well-differentiated cartilaginous tissues (Fig. 4). Postoperatively, there was no functional improvement of the left arm, and a course of radiotherapy (6,000 cGy over 30 treatment sessions) was initiated.
- At 6 weeks postoperatively, another well-defined, soft tissue tumor, measuring 3×3×3 cm, was detected by MRI in the left posterolateral chest wall and the anterior aspect of the latissimus dorsi muscle, which was apart from initial mass about 2 cm away proximally.
- Histologic findings after marginal excision showed intertwining fascicles of relatively uniform spindle cells and a collagen deposition which seemed like a fibrosarcoma. The fascicles are arranged at acute angles to each other, resulting in a herringbone appearance. The patient has been followed up periodically after postoperative cyclophosphamide, vincristine, doxorubicin, and dacarbazine chemotherapy, and had experienced no recurrence or metastasis at his 18-month postoperative follow-up.
- Case 2
- A 52-year-old man presented with a 6-month history of a soft tissue mass around the right axillary region, which appeared to be gradually increasing in size. He had no history of any major illness or trauma. Physical examination showed a nontender mass measuring 3×3 cm with a hard-elastic consistency that was palpable over the right axillary area. The mass was well demarcated from the surrounding structures, and no skin changes or neurovascular deficit were present.
- Plain radiographs showed soft tissue swelling with stippled calcification on the right medial axillary region (Fig. 5A). Pathologic findings showed no osteolysis or cortical destruction around the lesion. CT also depicted calcification without an osteolytic lesion, and without thinning or expansion of the cortex of bone around the lesion (Fig. 5B). In MR images, the peripheral area exhibited high intensity in both T1-weighted and T2-weighted images, whereas the central area exhibited low intensity in both (Fig. 6A, B). Enhanced MRI revealed peripheral and mild speculated enhancement around the lesion, which was regarded to be due to calcification (Fig. 6C). Chest and abdominal CT and a bone scan showed no evidence of metastasis.
- The mass, lying on the latissimus dorsi muscle was excised. The tumor measured 3×3×3 cm in dimensions and appeared to be well-encapsulated. The longitudinal section of the resected mass revealed tan-white tissue with central and focal areas of hemorrhage and necrosis (Fig. 7). The histologic examination was consistent with a diagnosis of mesenchymal chondrosarcoma. The tumor consisted of undifferentiated small neoplastic cells, varying from round to spindle-shaped, and interspersed with well differentiated foci of cartilaginous tissue (Fig. 8).
- The patient underwent a course of postoperative external beam radiation (6,000 cGy over 30 treatment sessions). At 1 year after surgery, the patient had remained free of local recurrence or metastasis.
CASE REPORTS
- Extraskeletal chondrosarcomas are relatively rare neoplasms, and are far less common than their intraosseous counterparts.7 Histological subtypes of extraskeletal chondrosarcomas include myxoid and mesenchymal, with the former being the most common.8 EMC was first reported by Lightenstein and Bernstein in 1959.9 Compared with the myxoid type, EMC is rare and more aggressive, and has a poor prognosis.3 In contrast to conventional chondrosarcoma, for which only 1% of tumors are extraskeletal, mesenchymal chondrosarcoma developed in soft tissues in almost half of reported cases. These tumors were located mainly in the orbit, the cranial and spinal meningeal coverings, and the lower limbs, particularly the thigh.10 Other sites of involvement, including the upper extremity, and especially the shoulder and axillary regions, are rare. A literature search, limited to the English language, unearthed few cases of EMC of the upper arm,11 although Weiss and Goldblum10 reviewed cases in which metastasis from a primary mesenchymal chondrosarcoma of bone mimicked a tumor of extraskeletal origin. Accordingly, a skeletal survey is essential, particularly when a tumor is encountered in an unusual location.
- EMCs differ from typical chondrosarcomas in several respects. Chondrosarcomas show a predilection for middle-aged to elderly males, whereas EMCs have a slight female preponderance and occur in the nervous system in patients aged 20-30 years and in soft tissues in patients age 40 years and older.12 Chondrosarcomas tend to be more indolent tumors than MCs, which are notably aggressive, and have respective 5- and 10-year survival rates of 54.6% and 27.3%.5
- Plain radiography of EMC shows a soft-tissue mass with mineralized chondroid matrix due to arc-shaped or stippled calcification in over 50% of cases,10 and CT commonly depicts coarse calcification and predominantly peripheral enhancement.13 We observed plain radiographic abnormalities in case 2 of our study, but not in case 1. Furthermore, CT and MRI findings did not enable differentiation of EMC from other neoplasms. Thus, a definitive diagnosis was made based on histological features. Microscopically, the most common feature of mesenchymal chondrosarcoma is a biphasic pattern composed of sheets of undifferentiated round, oval, or spindle-shaped cells and small, usually well-defined, islets of well-differentiated, benign-appearing cartilaginous tissue.5,10 The undifferentiated cells have ovoid or elongated hyperchromatic nuclei and scant, poorly outlined cytoplasm, and are arranged in small aggregates or in a hemangiopericytoma-like pattern about sinusoidal vascular channels. Solid cellular and richly vascular patterns may be present in different portions of the same neoplasm. Cartilaginous foci are usually well-defined, but there are also poorly circumscribed cartilaginous areas that blend with undifferentiated tumor cells. Spindle cell areas are present in some cases but are rarely a prominent feature.10,14 However, there is no specific immunohistochemical marker for EMC.10,13
- A diagnosis of EMC can be difficult using small biopsy or fine-needle aspiration biopsy specimens that demonstrate only one of the two tissue elements. Therefore, the entities that should be considered in the differential diagnosis include Ewing sarcoma, hemangiopericytoma, and synovial sarcoma and other variants of chondrosarcoma, especially in cases without a cartilaginous element.10,14
- Because of its rarity and the lack of studies on the efficacies of treatment methods, no specific treatment guidelines based on scientific evidence have been established for EMC.2 Furthermore, no reliable correlation has been established between the histological appearance and biological behavior of EMCs, but all should be considered high-grade tumors due to the presence of small round cells.13 To decide upon treatment protocols, Huvos et al.15 divided patients into two histologic categories, namely, undifferentiated small cell types and hemangiopericytomatoid variants. However, these protocols are applicable to skeletal lesions, and no specific guidelines regarding the surgical resection of extraskeletal lesions were mentioned. Some agreement has been reached that resection with wide margins is the optimal treatment.5,16,17 However, the roles of adjuvant chemotherapy and radiotherapy remain obscure due to an absence of objective evidence.18 In case 1, because of the lesions location, potentially curative surgery would have involved amputation of the upper limb, which was not accepted by the patient. Therefore, the tumor was resected along with normal-appearing tissues surrounding its borders. However, we were able to obtain a substantially wider margin.
- EMCs are usually associated with a poor prognosis.1,2,4 Mesenchymal chondrosarcoma is a highly malignant tumor that is characterized by the presence of solid, highly cellular areas composed of round or slightly spindled primitive mesenchymal cells,19 showing a propensity to metastasize, most commonly to the lungs.5 Therefore, it is important for clinicians to realize that long-term follow-up is essential. Furthermore, wide surgical excision is required in the treatment of EMCs. However, it is usually difficult to achieve a proper margin, when a tumor arises around the shoulder or axillary region, because complete or partial limb loss results in significant impairment. Therefore, the accurate diagnosis of EMC is the most important step, and clinicians must consider clinical, radiologic, and histopathologic features to confirm a diagnosis.
DISCUSSION
- 1. Wu KK, Collon DJ, Guise ER. Extra-osseous chondrosarcoma: report of five cases and review of the literature. J Bone Joint Surg Am 1980; 62: 189-194. ArticlePubMed
- 2. Anderson JT, Lucas GL. Extraskeletal mesenchymal chondrosarcoma of the forearm: a case report. J Hand Surg Am 2007; 32: 389-392. ArticlePubMed
- 3. Bertoni F, Picci P, Bacchini P, et al. Mesenchymal chondrosarcoma of bone and soft tissues. Cancer 1983; 52: 533-541. ArticlePubMed
- 4. Steffen CM, Kennedy PJ, Page J, Dan N. An extraskeletal chondrosarcoma arising in the brachial plexus. J Surg Oncol 1992; 50: 58-61. ArticlePubMed
- 5. Nakashima Y, Unni KK, Shives TC, Swee RG, Dahlin DC. Mesenchymal chondrosarcoma of bone and soft tissue: a review of 111 cases. Cancer 1986; 57: 2444-2453. ArticlePubMed
- 6. Huvos AG. In: Mitchell J, ed. Mesenchymal chondrosarcoma. Bone tumors: diagnosis, treatment, and prognosis. 1991; Philadelphia: W. B. Saunders, 382-393.
- 7. Kransdorf MJ, Meis JM. From the archives of the AFIP. Extraskeletal osseous and cartilaginous tumors of the extremities. Radiographics 1993; 13: 853-884. ArticlePubMed
- 8. Resnick D, Kransdorf MJ. Bone and joint imaging. 2005; Philadelphia: Elsevier Saunders, 1225-1226.
- 9. Lightenstein L, Bernstein D. Unusual benign and malignant chondroid tumors of bone: a survey of some mesenchymal cartilage tumors and malignant chondroblastic tumors, including a few multicentric ones, as well as many atypical benign chondroblastomas and chondromyxoid fibromas. Cancer 1959; 12: 1142-1157. ArticlePubMed
- 10. Weiss SW, Goldblum JR. Enzinger and Weiss's soft tissue tumors. 2001; St. Louis: Mosby, 1380-1385.
- 11. Sato N, Minase T, Yoshida Y, et al. An ultrastructural study of extraskeletal mesenchymal chondrosarcoma. Acta Pathol Jpn 1984; 34: 1355-1363. ArticlePubMed
- 12. Louvet C, de Gramont A, Krulik M, et al. Extraskeletal mesenchymal chondrosarcoma: case report and review of the literature. J Clin Oncol 1985; 3: 858-863. ArticlePubMed
- 13. Jamal W, Rhys Evans P, Sheppard MN. Mesenchymal chondrosarcoma of the vagus nerve. J Laryngol Otol 2002; 116: 644-646. ArticlePubMed
- 14. Chung MJ, Oh SY, Kang MJ, Lee DG, Choi HY, Kim SH. Fine needle aspiration cytology of extraskeletal mesenchymal chondrosarcoma: a case report. Korean J Cytopathol 1997; 8: 194-198.
- 15. Huvos AG, Rosen G, Dabska M, Marcove RC. Mesenchymal chondrosarcoma: a clinicopathologic analysis of 35 patients with emphasis on treatment. Cancer 1983; 51: 1230-1237. ArticlePubMed
- 16. Knott PD, Gannon FH, Thompson LD. Mesenchymal chondrosarcoma of the sinonasal tract: a clinicopathological study of 13 cases with a review of the literature. Laryngoscope 2003; 113: 783-790. ArticlePubMed
- 17. Rushing EJ, Armonda RA, Ansari Q, Mena H. Mesenchymal chondrosarcoma: a clinicopathologic and flow cytometric study of 13 cases presenting in the central nervous system. Cancer 1996; 77: 1884-1891. ArticlePubMed
- 18. Dantonello TM, Int-Veen C, Leuschner I, et al. Mesenchymal chondrosarcoma of soft tissues and bone in children, adolescents, and young adults: experiences of the CWS and COSS study groups. Cancer 2008; 112: 2424-2431. ArticlePubMed
- 19. Chow WA. Update on chondrosarcomas. Curr Opin Oncol 2007; 19: 371-376. ArticlePubMed
REFERENCES
Fig. 1Case 1: (A) Anteroposterior radiographs of the left shoulder. Plain radiography findings are normal. (B) Coronal plane reconstructed computed tomography scan, showing a well-defined soft tissue mass around the left axillary region with no abnormal bony structure. Neither peripheral nor central calcifications are observed.
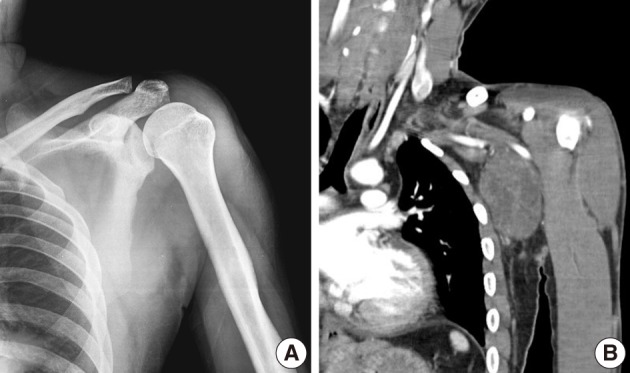

Fig. 2Case 1: Preoperative magnetic resonance imaging. (A) A coronal plane T1-weighted (T1W) image. (B) A coronal plane T2-weighted (T2W) image. (C) A coronal plane Gd-enhanced image. (D) An axial plane T2W image. A well-defined soft tissue mass between the pectoralis major and subscapularis muscles without vascular encasement. The peripheral area shows high intensity in both T1W and T2W images, whereas the central area exhibits heterogenous intensity in T1W and T2W images. The tumor shows peripheral enhancement on Gd-enhanced images.
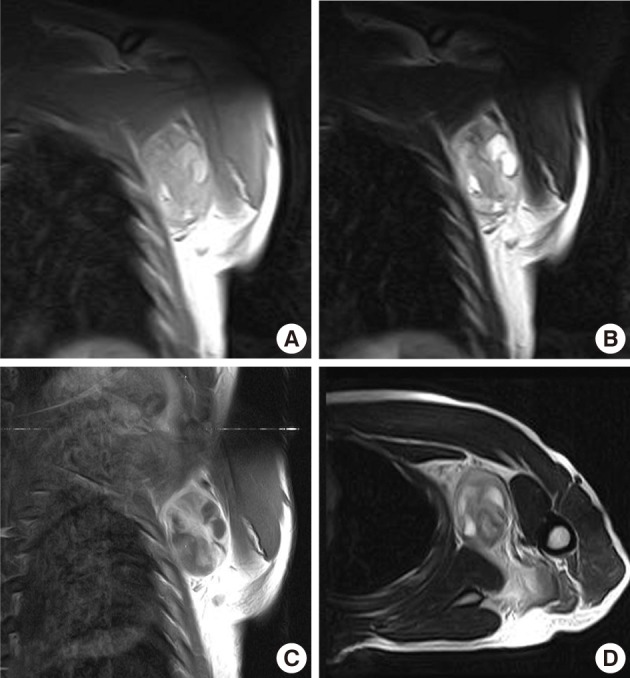

Fig. 3Case 1: Gross appearance of the tumor. (A) The mass appears well-encapsulated and has an elastic-hard consistency. (B) Longitudinal section of the resected mass reveals a yellowish-white, glistening surface. The mass is homogenous except for a few small focal cystic and necrotic changes.
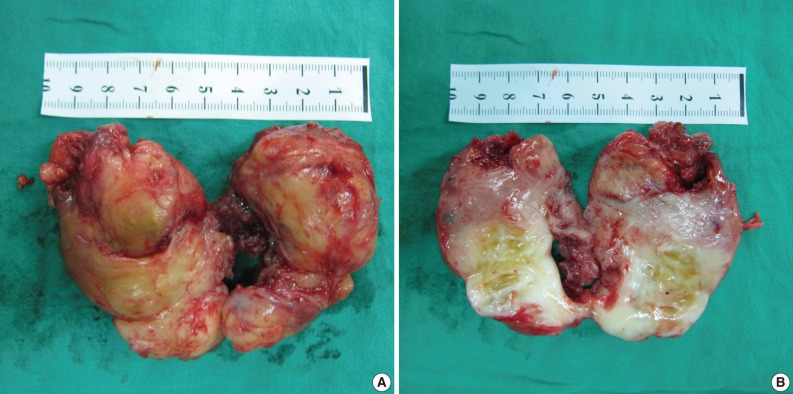

Fig. 4Case 1: Histologic examination. (A) Histologic features showing scattered patterns with undifferentiated cells and focal areas of well-differentiated cartilaginous tissue. (B,C) Intimate admixture of islands of cartilaginous tissue and undifferentiated small neoplastic cells. (D) Immunohistochemically, the cartilaginous portion of the tumor shows strong S-100 protein positivity.
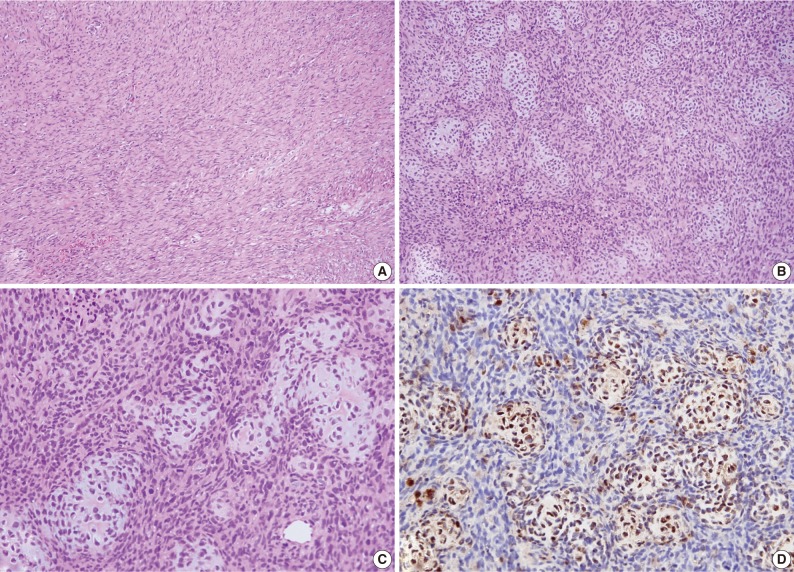

Fig. 5Case 2: (A) Anteroposterior radiographs of right medial axilla. Plain radiograph showing stippled calcification. (B) Axial computed tomography image showing calcification but no bony abnormality.
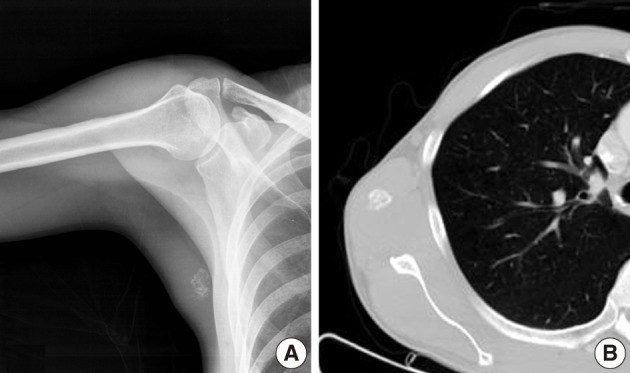

Fig. 6Case 2: Preoperative magnetic resonance images. (A) Coronal plane T1-weighted (T1W) image. (B) Coronal T2-weighted (T2W) image. (C) Coronal plane Gd-enhanced image. The peripheral area has high intensity in T1W and T2W images and enhancement in Gd-enhanced images, whereas the central area has lower intensity.
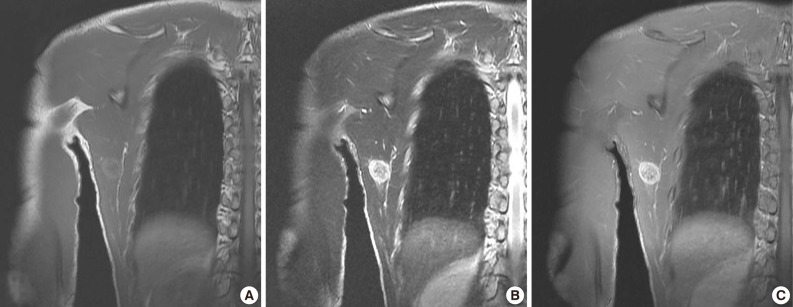

Fig. 7Case 2: Gross appearance of the tumor. Longitudinal section shows tan-white tissue with focal hemorrhage and necrosis.
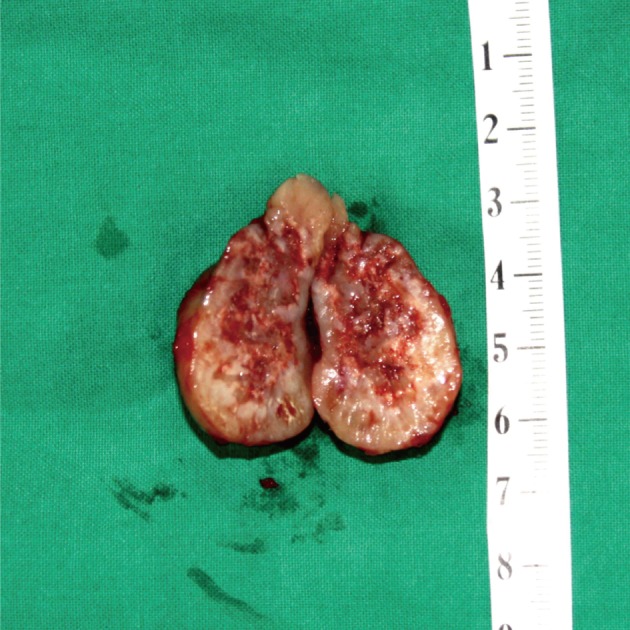

Fig. 8Case 2: Histologic findings. Histologic features showing (A) gradual transition between hypercellular stroma and focal areas of well-differentiated cartilaginous tissue, (B) tumors consisted of undifferentiated small neoplastic cells, (C) the coexistence of nests of well-defined cartilaginous tissue.
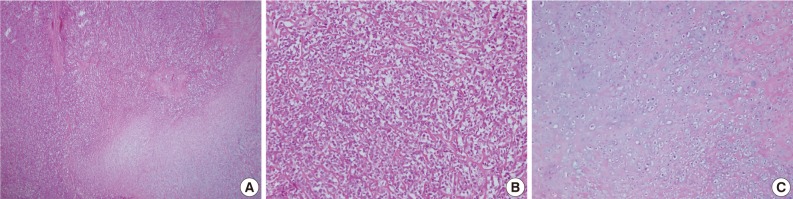

Figure & Data
References
Citations
Citations to this article as recorded by 

- Extraskeletal Mesenchymal Chondrosarcoma of the Arm: A Rare Case Report with a Review of Literature
Anubhuti Chaturvedi, Nimisha Dhankar, Swapnil Suman, Nita Khurana, Chandra Bhushan Singh, Sapna Singh
Journal of Bone and Joint Diseases.2025; 40(3): 170. CrossRef - Complete surgical excision of a high‐grade extraskeletal mesenchymal chondrosarcoma, resulting in a longer survival than previously reported
Hannah Reeves, Tesh Smalle
Veterinary Record Case Reports.2022;[Epub] CrossRef - Locally recurrent extraskeletal myxoid chondrosarcoma of the shoulder: a case of complete neoadjuvant radiotherapy response
Luca Improta, Sergio Valeri, Rossana Alloni, Chiara Pagnoni, Francesco Mallozzi Santa Maria, Beniamino Brunetti, Carlo Greco, Irene Aprile, Mirella Maselli, Bruno Vincenzi, Alessandro Gronchi
Clinical Sarcoma Research.2020;[Epub] CrossRef - Primary extraskeletal chondrosarcoma in the axillary region of a dog with review of the literature
Catrina Pennington, Josep Monne Rodriguez, Marlene Finck, Ben Walton
Veterinary Record Case Reports.2018;[Epub] CrossRef - Mesenchymal Chondrosarcoma of Bone and Soft Tissue: A Systematic Review of 107 Patients in the Past 20 Years
Jie Xu, Dasen Li, Lu Xie, Shun Tang, Wei Guo, David M Loeb
PLOS ONE.2015; 10(4): e0122216. CrossRef - Management of renal extraskeletal mesenchymal chondrosarcoma
Vitalie Gherman, Ciprian Tomuleasa, Catalina Bungardean, Nicolae Crisan, Victor-Dan Ona, Bogdan Feciche, Alexandru Irimie, Ioan Coman
BMC Surgery.2014;[Epub] CrossRef
Extraskeletal Mesenchymal Chondrosarcoma in the Axillary Region: Reports of Two Cases
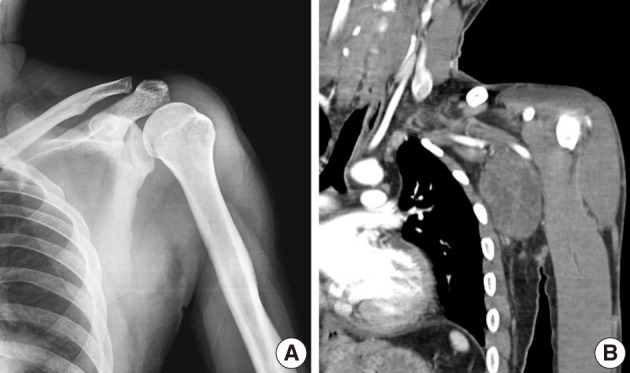

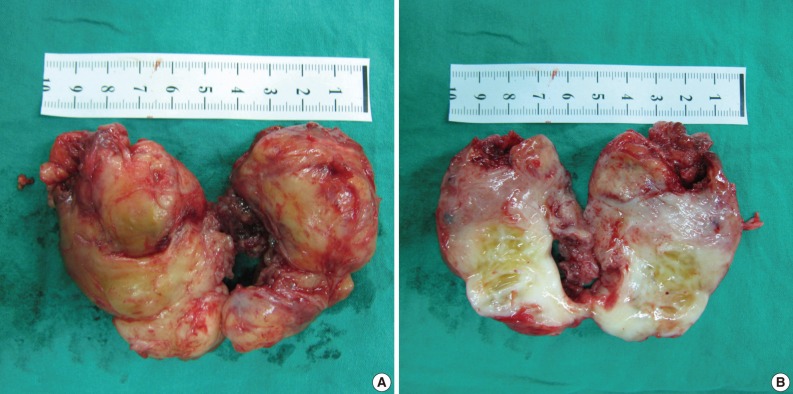
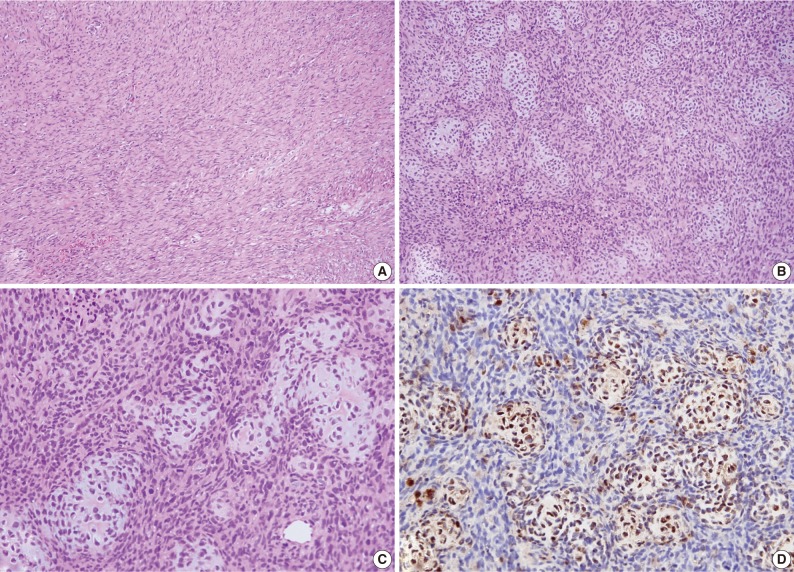
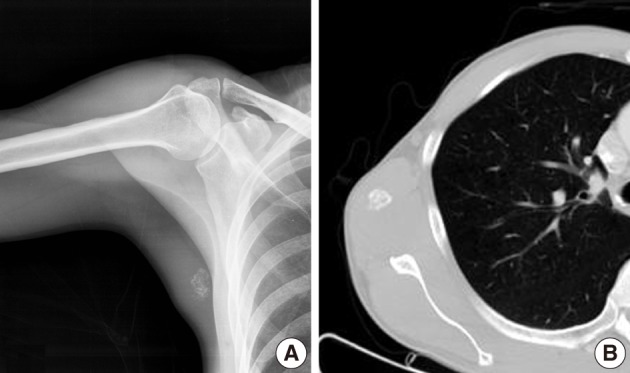
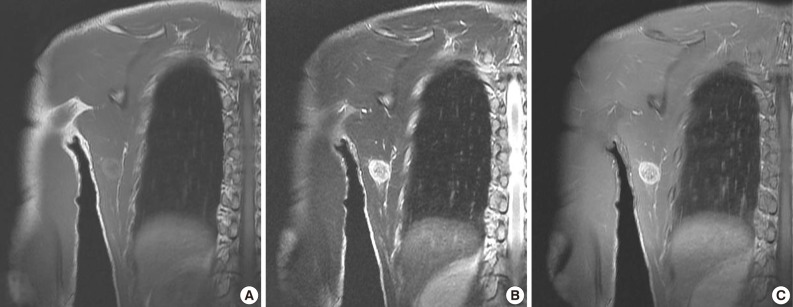
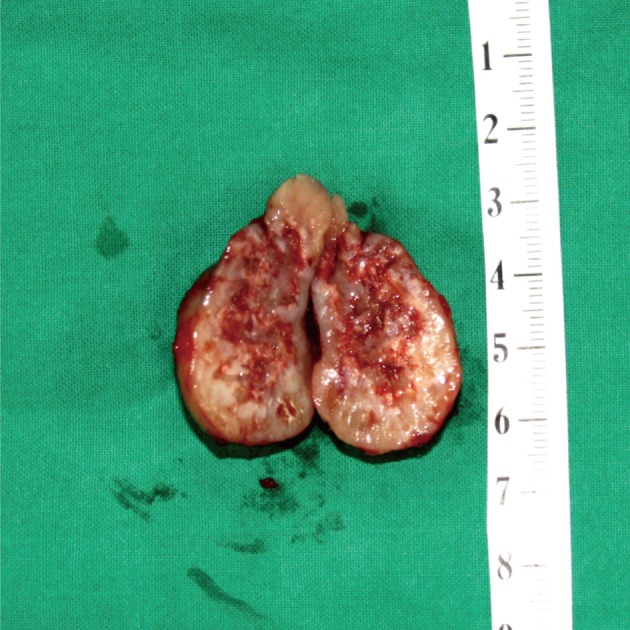
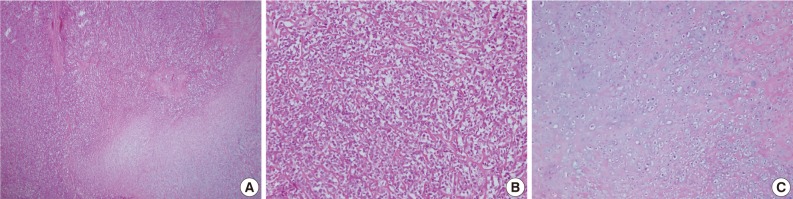
Fig. 1 Case 1: (A) Anteroposterior radiographs of the left shoulder. Plain radiography findings are normal. (B) Coronal plane reconstructed computed tomography scan, showing a well-defined soft tissue mass around the left axillary region with no abnormal bony structure. Neither peripheral nor central calcifications are observed.
Fig. 2 Case 1: Preoperative magnetic resonance imaging. (A) A coronal plane T1-weighted (T1W) image. (B) A coronal plane T2-weighted (T2W) image. (C) A coronal plane Gd-enhanced image. (D) An axial plane T2W image. A well-defined soft tissue mass between the pectoralis major and subscapularis muscles without vascular encasement. The peripheral area shows high intensity in both T1W and T2W images, whereas the central area exhibits heterogenous intensity in T1W and T2W images. The tumor shows peripheral enhancement on Gd-enhanced images.
Fig. 3 Case 1: Gross appearance of the tumor. (A) The mass appears well-encapsulated and has an elastic-hard consistency. (B) Longitudinal section of the resected mass reveals a yellowish-white, glistening surface. The mass is homogenous except for a few small focal cystic and necrotic changes.
Fig. 4 Case 1: Histologic examination. (A) Histologic features showing scattered patterns with undifferentiated cells and focal areas of well-differentiated cartilaginous tissue. (B,C) Intimate admixture of islands of cartilaginous tissue and undifferentiated small neoplastic cells. (D) Immunohistochemically, the cartilaginous portion of the tumor shows strong S-100 protein positivity.
Fig. 5 Case 2: (A) Anteroposterior radiographs of right medial axilla. Plain radiograph showing stippled calcification. (B) Axial computed tomography image showing calcification but no bony abnormality.
Fig. 6 Case 2: Preoperative magnetic resonance images. (A) Coronal plane T1-weighted (T1W) image. (B) Coronal T2-weighted (T2W) image. (C) Coronal plane Gd-enhanced image. The peripheral area has high intensity in T1W and T2W images and enhancement in Gd-enhanced images, whereas the central area has lower intensity.
Fig. 7 Case 2: Gross appearance of the tumor. Longitudinal section shows tan-white tissue with focal hemorrhage and necrosis.
Fig. 8 Case 2: Histologic findings. Histologic features showing (A) gradual transition between hypercellular stroma and focal areas of well-differentiated cartilaginous tissue, (B) tumors consisted of undifferentiated small neoplastic cells, (C) the coexistence of nests of well-defined cartilaginous tissue.
Fig. 1
Fig. 2
Fig. 3
Fig. 4
Fig. 5
Fig. 6
Fig. 7
Fig. 8
Extraskeletal Mesenchymal Chondrosarcoma in the Axillary Region: Reports of Two Cases

 E-submission
E-submission
 PubReader
PubReader Cite this Article
Cite this Article









