Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 49(3); 2015 > Article
-
Brief Case Report
Traumatic Bowel Perforation and Inguinal Hernia Masking a Mesenteric Calcifying Fibrous Tumor - Dong Hyun Kim,*, Kyueng-Whan Min1,*, Dong-Hoon Kim, Seoung Wan Chae, Jin Hee Sohn, Jung-Soo Pyo, Sung-Im Do, Kyungeun Kim, Hyun Joo Lee
-
Journal of Pathology and Translational Medicine 2015;49(3):267-269.
DOI: https://doi.org/10.4132/jptm.2015.03.20
Published online: May 15, 2015
Department of Pathology, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, Seoul, Korea
1Department of Pathology, Hallym University Sacred Heart Hospital, Hallym University College of Medicine, Anyang, Korea
- Corresponding Author Dong-Hoon Kim, M.D., Ph.D. Department of Pathology, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, 29 Saemunan-ro, Jongno-gu, Seoul 110-746, Korea Tel: +82-2-2001-2392, Fax: +82-2-2001-2398, E-mail: idavid.kim@samsung.com
- *Dong Hyun Kim and Kyueng-Whan Min contributed equally to this work.
• Received: December 31, 2014 • Revised: March 11, 2015 • Accepted: March 20, 2015
© 2015 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- A 71-year-old man visited our hospital for progressive abdominal pain after a fall. He also complained of nausea, vomiting, and abdominal discomfort for the previous 2 hours and had a medical history of hypertension, diabetes, and stroke. Physical examination revealed abdominal tenderness with mild rigidity. Abdominal computerized tomography revealed diffuse wall thickening of the distal ileum with free air and fluid collections and two inguinal hernias (Fig. 1). Additionally, there was a small amount of fluid collection and an air bubble in the right inguinal canal, suspicious for abscess or fecal spillage. An explorative laparoscopy was performed under the impression of bowel perforation associated with inguinal hernia. Laparoscopy revealed a 2-cm-sized bowel perforation located at 15 cm above the ileocecal valve in the right inguinal herniated lesion. Intriguingly, a hard mass was noted near the perforation site. The patient underwent small bowel resection and herniorrhaphy.
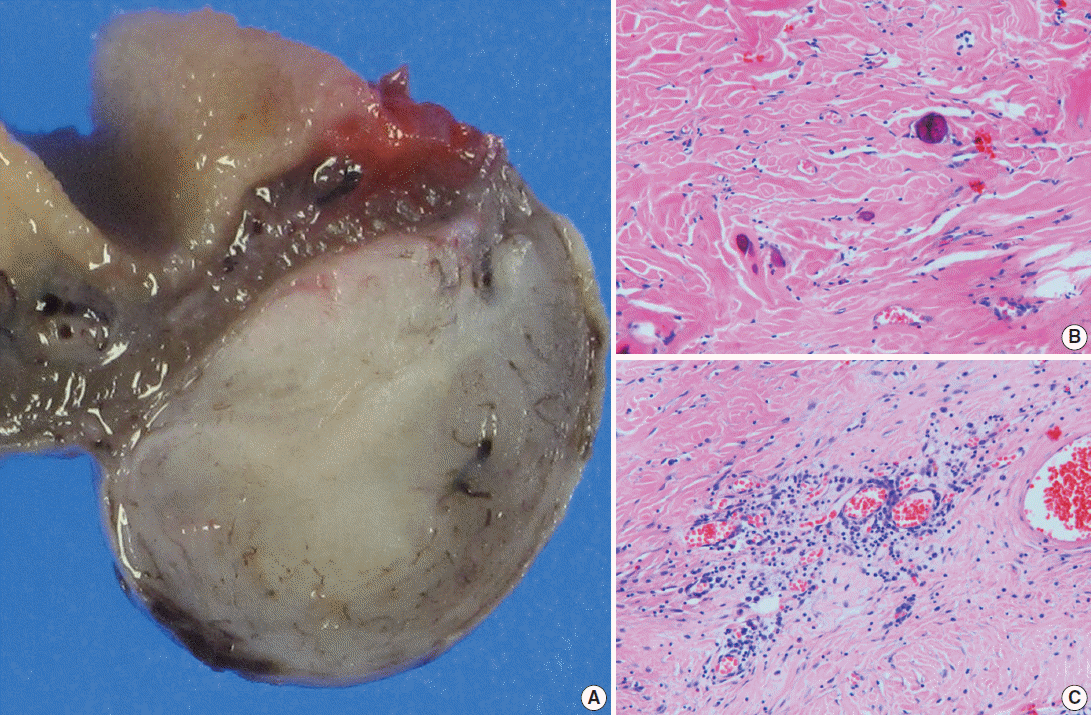
- Segmental resection revealed a firm, 1.1×1.1×0.7-cm-sized mass located at 2 cm from the perforation. The cut surface demonstrated a solid, well-demarcated, gray-brown mass in the mesentery. The remaining mucosal surface was edematous with congestion (Fig. 2A). Microscopically, the mass showed hypocellular sclerosis with wavy collagenous stroma, microcalcifications, and scattered inflammatory cells (Fig. 2B, C). Immunohistochemical staining results were negative for c-kit, smooth muscle actin, desmin, S-100 protein, and CD34 in the stromal cells. The Ki-67 labeling index was less than 1%. Pathological diagnosis thus confirmed a CFT. IgG and IgG4 immunohistochemical stains were also performed for this lesion to determine if the tumor was associated with an IgG4-related disease. IgG stain was positive, but IgG4 stain was negative. At the 6-month postoperative follow-up visit, the patient remained well without complications.
CASE REPORT
- CFT can occur in a wide range of ages and may arise from different sites. It is often detected incidentally, but visceral CFT has occasionally presented as a painful mass due to mass effect. Most cases of CFT in the small intestine have been reported as pain inducing lesion. Emanuel et al. [1] described presentation with abdominal symptoms (intussusception, abdominal pain) in four patients. Mesenteric CFT presenting with acute peritonitis has also been documented [2]. CFT needs to be distinguished from gastrointestinal stromal tumors, desmoid tumors and myomas, which have varying clinical outcomes. The typical radiographic findings in CFT are a well circumscribed, homogeneous mass, but these findings are nonspecific, so biopsy with histologic confirmation is needed prior to treatment [3].
- The mechanism of CFT development is thought to be reactive pseudotumoral. Previous reports suggested that CFT is a late sclerosing stage of inflammatory myofibroblastic tumors (IMT) [4,5]. However, Sigel et al. [6] reported that anaplastic lymphoma kinase (ALK) stain, which is positive in IMT, was negative in their CFT patients. Nascimento et al. [7] also reported that CD34 was positive and ALK-1 was negative in their CFT patients. These studies do not support a relationship between CFT and IMT. Other studies described CFT associated with IgG4+ plasma cells. IgG4+ plasma cells were increased in a case of disseminated abdominal CFT associated with sclerosing angiomatoid nodular transformation of the spleen [8]. A study of gastric CFT reported that IgG4+ plasma cells were seen in this tumor [9]. Larson et al. [10] also suggested that CFT could be an IgG4 related disease. Although these reports support that CFT might be IgG4 related, our case was negative for IgG4 stain. Thus, more work needs to be done to better understand the mechanism of CFT development.
- In the present case, the mesenteric CFT was slow-growing and asymptomatic, but abdominal symptoms appeared abruptly after trauma. Bowel perforation and hernia masked the solitary mass from radiological detection, but laparoscopy revealed a solid mass 2 cm from the bowel perforation. Considering the solid mass near the perforation and the patient’s history of trauma, bowel perforation may have been caused by herniation of the CFT. Although it is rare for inguinal herniation and bowel perforation due to trauma to occur simultaneously with CFT, this case indicates that unusual clinical findings can be important for early detection and presurgical planning.
DISCUSSION
Fig. 1.Abdominal contrast-enhanced computed tomography scan reveals diffuse enhancing wall thickening (arrow) without an obvious mass-like lesion.
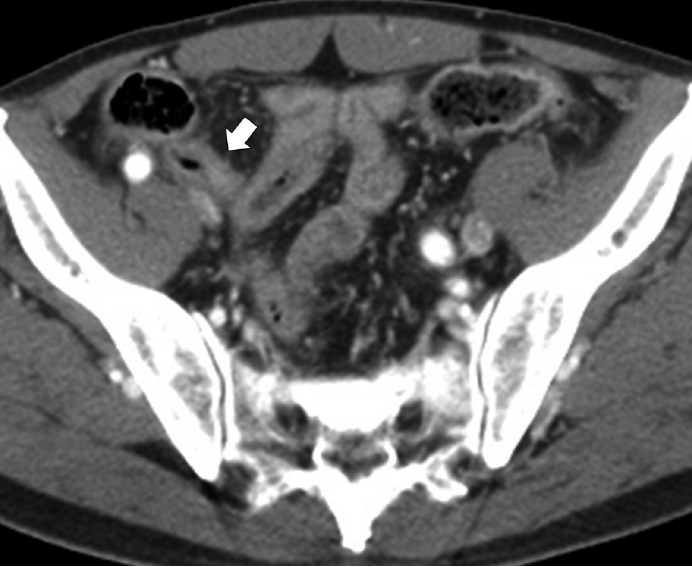

Fig. 2.(A) Viewed grossly, a well-demarcated gray-brown firm solid mass is confined to the mesenteric fat (right). (B) Microscopically, there are dispersed sparse spindle cells and occasional dystrophic calcification among thick wavy collagen bundles (left upper). (C) Patchy lymphoplasmacytic infiltrations are found throughout the tumor.

- 1. Emanuel P, Qin L, Harpaz N. Calcifying fibrous tumor of small intestine. Ann Diagn Pathol 2008; 12: 138-41. ArticlePubMed
- 2. Ben-Izhak O, Itin L, Feuchtwanger Z, Lifschitz-Mercer B, Czernobilsky B. Calcifying fibrous pseudotumor of mesentery presenting with acute peritonitis: case report with immunohistochemical study and review of literature. Int J Surg Pathol 2001; 9: 249-53. ArticlePubMedPDF
- 3. Giardino AA, Ramaiya NH, Shinagare AB, Jagannathan JP, Stachler MD, Raut CP. Case report: Calcifying fibrous tumor presenting as an asymptomatic pelvic mass. Indian J Radiol Imaging 2011; 21: 306-8. ArticlePubMedPMC
- 4. Van Dorpe J, Ectors N, Geboes K, D’Hoore A, Sciot R. Is calcifying fibrous pseudotumor a late sclerosing stage of inflammatory myofibroblastic tumor? Am J Surg Pathol 1999; 23: 329-35. ArticlePubMed
- 5. Pomplun S, Goldstraw P, Davies SE, Burke MM, Nicholson AG. Calcifying fibrous pseudotumour arising within an inflammatory pseudotumour: evidence of progression from one lesion to the other? Histopathology 2000; 37: 380-2. ArticlePubMedPDF
- 6. Sigel JE, Smith TA, Reith JD, Goldblum JR. Immunohistochemical analysis of anaplastic lymphoma kinase expression in deep soft tissue calcifying fibrous pseudotumor: evidence of a late sclerosing stage of inflammatory myofibroblastic tumor? Ann Diagn Pathol 2001; 5: 10-4. ArticlePubMed
- 7. Nascimento AF, Ruiz R, Hornick JL, Fletcher CD. Calcifying fibrous ‘pseudotumor’: clinicopathologic study of 15 cases and analysis of its relationship to inflammatory myofibroblastic tumor. Int J Surg Pathol 2002; 10: 189-96. ArticlePubMedPDF
- 8. Kuo TT, Chen TC, Lee LY. Sclerosing angiomatoid nodular transformation of the spleen (SANT): clinicopathological study of 10 cases with or without abdominal disseminated calcifying fibrous tumors, and the presence of a significant number of IgG4+ plasma cells. Pathol Int 2009; 59: 844-50. ArticlePubMed
- 9. Agaimy A, Bihl MP, Tornillo L, Wünsch PH, Hartmann A, Michal M. Calcifying fibrous tumor of the stomach: clinicopathologic and molecular study of seven cases with literature review and reappraisal of histogenesis. Am J Surg Pathol 2010; 34: 271-8. ArticlePubMed
- 10. Larson BK, Balzer B, Goldwasser J, Dhall D. Calcifying fibrous tumor: an unrecognized IgG4: related disease? APMIS 2015; 123: 72-6. ArticlePubMed
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- A rare cause of small intestinal obstruction in children: a case report of calcifying fibrous tumor
Zesheng Yang, Xiaoying Xie, Shicheng Wang, Weijun Xu, Guanghua Pei, Jun Wan
BMC Pediatrics.2025;[Epub] CrossRef - Calcifying Fibrous Tumor
Angeliki Chorti, Theodossis S. Papavramidis, Antonios Michalopoulos
Medicine.2016; 95(20): e3690. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
Traumatic Bowel Perforation and Inguinal Hernia Masking a Mesenteric Calcifying Fibrous Tumor
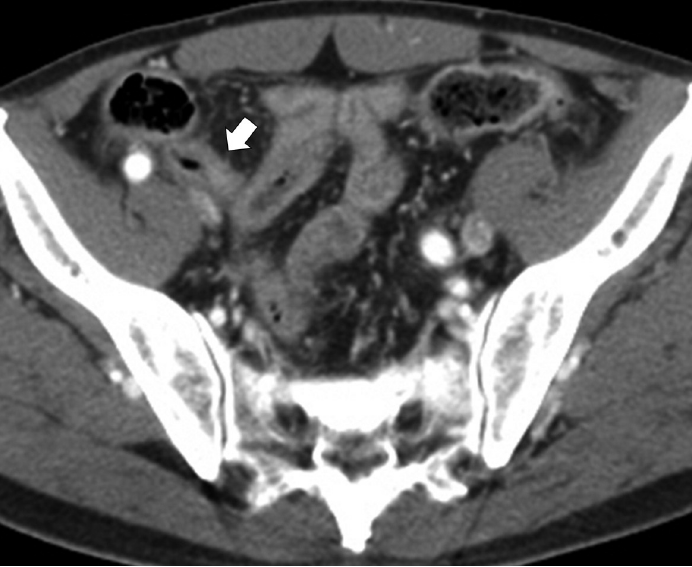
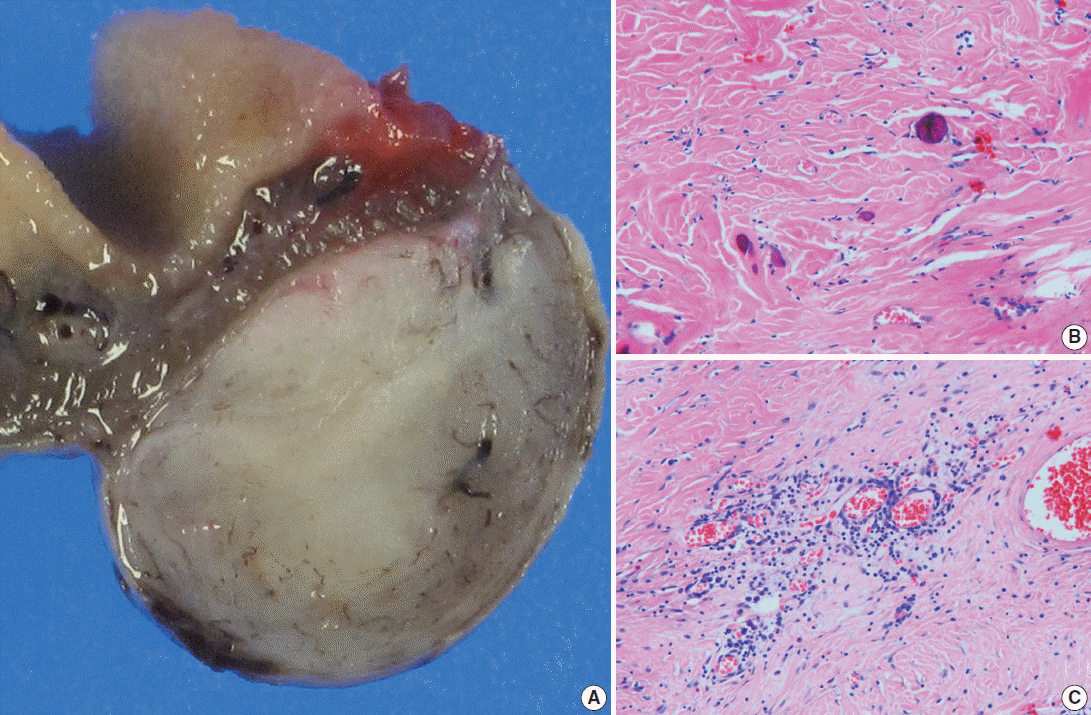
Fig. 1. Abdominal contrast-enhanced computed tomography scan reveals diffuse enhancing wall thickening (arrow) without an obvious mass-like lesion.
Fig. 2. (A) Viewed grossly, a well-demarcated gray-brown firm solid mass is confined to the mesenteric fat (right). (B) Microscopically, there are dispersed sparse spindle cells and occasional dystrophic calcification among thick wavy collagen bundles (left upper). (C) Patchy lymphoplasmacytic infiltrations are found throughout the tumor.
Fig. 1.
Fig. 2.
Traumatic Bowel Perforation and Inguinal Hernia Masking a Mesenteric Calcifying Fibrous Tumor

 E-submission
E-submission




