Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 48(1); 2014 > Article
-
Brief Case Report
Adenocarcinoma Arising in a Colonic Duplication Cyst: A Case Report and Review of the Literature - Myunghee Kang, Jungsuk An, Dong Hae Chung, Hyun Yee Cho
-
Korean Journal of Pathology 2014;48(1):62-65.
DOI: https://doi.org/10.4132/KoreanJPathol.2014.48.1.62
Published online: February 25, 2014
Department of Pathology, Gachon University Gil Medical Center, Incheon, Korea.
- Corresponding Author: Hyun Yee Cho, M.D. Department of Pathology, Gachon University Gil Medical Center, 21 Namdong-daero 774beon-gil, Namdong-gu, Incheon 405-760, Korea. Tel: +82-32-460-3073, Fax: +82-32-460-2394, hicho@gilhospital.com
• Received: March 20, 2013 • Revised: May 13, 2013 • Accepted: May 14, 2013
© 2014 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- A 23-year-old female patient was admitted with a palpable abdominal mass. The mass had been noted three years previously. On physical examination, a nontender and movable mass measuring approximately 7 cm was observed at the right lower quadrant.
- The patient's serum carcinoembryonic antigen (CEA) level was 6.51 ng/mL (normal range, <5.0 ng/mL), and that of cancer antigen 19-9 was 47.71 U/mL (normal range, <37 U/mL).
- Ultrasonography disclosed a hypoechoic cystic mass with an internal echogenic dot in the right lower quadrant. The echogenic dot was thought to be either an intraluminal secretion or necrotic debris (Fig. 1A). Computed tomography (CT) revealed a nonenhancing cystic mass measuring 7 cm, with linear calcification located adjacent to the medial side of the ascending colon (Fig. 1B). No enlarged regional lymph nodes, ascites, or other abnormalities were observed.
- A laparoscopic excision of the mass was performed. During the operation, the mass was observed to be attached to the ascending colon mesentery, having the blood supply by the right colic artery.
- On gross examination, the cystic mass measured 8.6×6.4×2.9 cm, and its outer surface was smooth; mesenteric fat adhered to the outer surface. The maximum thickness of the cystic wall was 1.5 cm, and focal calcification was noted within the wall. The cyst contained brownish-colored mucoid material (Fig. 2A).
- Microscopically, the cyst was found to have two well-organized layers of smooth muscle with an infiltrating adenocarcinoma forming irregular tubules (Fig. 2B, C). The tumor had invaded the pericystic mesenteric soft tissue and metastasized to eight of 16 mesenteric lymph nodes. Perineural and lymphovascular invasions were noted. Non-neoplastic glandular epithelium was not found; instead, a focal area of atypical columnar epithelium, which was compatible with low-grade dysplasia, was observed (Fig. 2D). On immunohistochemical study, tumor cells were positive for cytokeratin 7 (1:100, OV-TL 12/30, Dako, Glostrup, Denmark), cytokeratin 20 (1:100, KS20.8, Dako), CEA (prediluted, II-7, Dako), CDX2 (1:50, AMT 28, Novocastra, Newcastle upon Tyne, UK), and p53 (1:100, DO-7, Dako). The dysplastic epithelium was also positive for p53 (Fig. 2E).
- Positron emission tomography-CT was performed after surgery and revealed no hypermetabolic areas other than the previous operation site. Two months after surgery, the patient started adjuvant chemotherapy.
CASE REPORT
- Duplications are rare congenital anomalies that may occur anywhere along the alimentary tract.1 The most common site of involvement is the ileum. Colonic duplications comprise 4% to 18% of all duplications, and occur most often in the cecum. Strict morphologic criteria for the diagnosis of duplication have been established: 1) attachment to the alimentary tract, 2) presence of smooth muscle layers, and 3) presence of lining epithelium resembling that of the alimentary tract. Our case met all of these criteria.
- Malignant change in a duplication is very rare, and adenocarcinoma is the most common histologic type of malignancy found in these unusual cases.1-10 However, squamous cell carcinoma, carcinoid tumor, gastrointestinal stromal tumor, and leiomyosarcoma have also been reported.
- Thirteen cases of adenocarcinoma arising in a duplication of the colon have been reported to date in the English literature (Table 1).1-10 Among the 14 cases, including the current case, four patients were male and 10 patients were female. The mean age at diagnosis was 48.8 years (range, 23 to 72 years) and our patient was the youngest. Some of the patients, including our patient, were relatively young, suggesting that the epithelium of the duplication has a high risk of carcinogenesis. The most common symptoms were abdominal pain and a palpable mass, and the mean size was 10.2 cm (range, 3 to 20 cm). Calcifications in the cystic wall or calculi in the lumen were common findings and were present in six of the eight cases where relevant data were available. Peripheral calcifications were present in three cases and calculi were found in four of six cases. Calcification in a duplication can be a worrisome finding because calcification is extremely rare in benign duplications.6 The preoperative serum CEA level was available for five cases and was found to be elevated in four cases in which patients presented with advanced stage disease at the time of initial diagnosis. According to our survey, except for one case without relevant data, all cases had invasion beyond the muscular wall. Metastasis to a regional lymph node or to a distant organ was reported in three cases.3,9 Although there was insufficient data on the duration of follow-up, five of six cases involved uneventful clinical courses. One patient died 41 months after diagnosis due to extensive metastasis.9 No follow-up data was provided for the remaining eight patients. Due to its rarity and nonspecific symptoms, we thought that, in the present case, the diagnosis was made at an advanced stage. Calcification within the mass and elevated serum CEA level appear to be useful for preoperative prediction of malignant change in duplications.
- In conclusion, intestinal duplication is an uncommon congenital abnormality, and malignant change in a duplication is extremely rare. Differential diagnosis of a cystic mass located in or adjacent to the gastrointestinal tract should include duplication. When serum CEA level is high, and/or calcification within the mass is observed, the possibility of adenocarcinoma arising in a duplication cyst should be considered.
DISCUSSION
- 1. Orr MM, Edwards AJ. Neoplastic change in duplications of the alimentary tract. Br J Surg 1975; 62: 269-274. ArticlePubMedPDF
- 2. Tamoney HJ Jr, Testa RE. Carcinoma arising in a duplicated colon: case report. Cancer 1967; 20: 478-481. ArticlePubMed
- 3. Heiberg ML, Marshall KG, Himal HS. Carcinoma arising in a duplicated colon: case report and review of literature. Br J Surg 1973; 60: 981-982. ArticlePubMedPDF
- 4. Lee J, Jeon YH, Lee S. Papillary adenocarcinoma arising in a duplication of the cecum. Abdom Imaging 2008; 33: 601-603. ArticlePubMedPDF
- 5. Jung KH, Jang SM, Joo YW, et al. Adenocarcinoma arising in a duplication of the cecum. Korean J Intern Med 2012; 27: 103-106. ArticlePubMedPMC
- 6. Inoue Y, Nakamura H. Adenocarcinoma arising in colonic duplication cysts with calcification: CT findings of two cases. Abdom Imaging 1998; 23: 135-137. ArticlePubMedPDF
- 7. Hattori H. Adenocarcinoma occurring just at the attached site of colonic duplication in an adult man. Dig Dis Sci 2005; 50: 1754.ArticlePubMedPDF
- 8. Hsu H, Gueng MK, Tseng YH, Wu CC, Liu PH, Chen CC. Adenocarcinoma arising from colonic duplication cyst with metastasis to omentum: a case report. J Clin Ultrasound 2011; 39: 41-43. ArticlePubMed
- 9. Arkema KK, Calenoff L. Adenocarcinoma in tubular duplication of the sigmoid colon. Gastrointest Radiol 1977; 2: 137-139. ArticlePubMedPDF
- 10. Nakayama H, Akikusa B, Kondo Y, Saito N, Sarashina H, Okui K. Mucinous cystadenocarcinoma of the colon: report of a case. Dis Colon Rectum 1989; 32: 243-246. PubMed
REFERENCES
Fig. 1Radiologic findings. (A) A hypoechoic mass with an echogenic dot (arrow) in the right lower quadrant is observed on ultrasonography. (B) A cystic mass with linear calcification in the mesentery adjacent to the ascending colon is observed on computed tomography.


Fig. 2Gross and microscopic findings. (A) The cystic mass measured 8.6×6.4×2.9 cm and contained brownish-colored mucoid material. (B) Full-thickness of two smooth muscle layers and adjacent mesenteric soft tissue are invaded by tumor cells. (C) The tumor is moderately differentiated adenocarcinoma. (D) A focal area of atypical columnar epithelium, compatible with dysplasia, is noted on the surface. (E) The dysplastic epithelial cells are positive for p53.
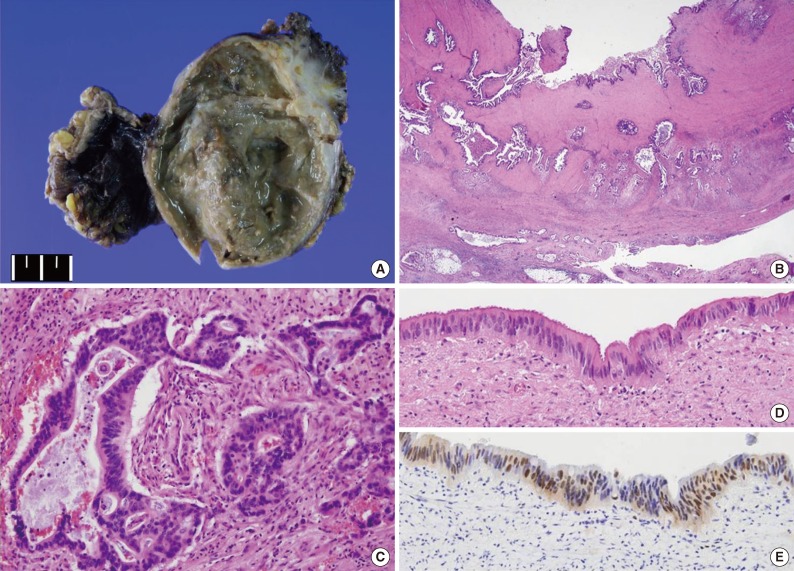

Table 1.Clinicopathologic summary of cases with adenocarcinoma arising in a colonic duplication cyst
| Case | Site | Age (yr)/Sex | Symptom | Serum CEA | Size (cm) | Calcification | Depth of invasion | Treatment | Prognosis |
|---|---|---|---|---|---|---|---|---|---|
| 1 [2] | Cecum | 61/F | Abdominal pain | ND | 9 | ND | Muscle layer | RHC, omentectomy, bilateral oophorectomy | NED |
| 2 [3] | Cecum | 50/F | Lower abdominal pain | ND | 8 | ND | Pericystic mesenteric soft tissue | RHC | ND |
| 3 [4] | Cecum | 40/F | Palpable mass | ↑ | 11 | N | Muscle layer | RHC | ND |
| 4 [5] | Cecum | 41/M | Palpable mass | Normal | 6 | Y | Pericystic mesenteric soft tissue | RHC, CT | NED |
| 5 [1] | Ascending | 41/F | RLQ pain | ND | 4 | Y | Thin fibrous wall (muscle layer) | Excision | NED |
| 6 [6] | Ascending | 38/M | Palpable mass | ↑ | ND | Y | Capsule (muscle layer) | RHC | ND |
| 7 [6] | Ascending | 59/F | Fever and lumbar pain | ↑ | ND | Y | Pericystic mesenteric soft tissue and right psoas muscle | Excision | ND |
| 8 [7] | Ascending | 59/M | Melena due to adenocarcinoma in the ascending colon | ND | 10 | ND | Subserosa | RHC | ND |
| 9 [1] | Transverse | 57/F | RLQ pain | ND | 11 | ND | Pericystic mesenteric soft tissue | RHC | NED |
| 10 [8] | Transverse | 40/M | Palpable mass | ND | 16 | ND | Pericystic mesenteric soft tissue and omentum | Excision, CT | ND |
| 11 [4] | Descending | 33/F | Palpable mass | ND | 15 | N | ND | Excision | ND |
| 12 [9] | Sigmoid | 69/F | Epigastric pain | ND | 15 | Y | Pericystic mesenteric soft tissue and retroperitoneum | Excision | Died 41 mos later |
| 13 [10] | Sigmoid | 72/F | Epigastric pain due to cholelithiasis | ND | 3 | ND | Subserosa | Sigmoid colectomy | ND |
| Present case | Ascending | 23/F | Palpable mass | ↑ | 8.6 | Y | Pericystic mesenteric soft tissue | Excision, CT | NED |
Figure & Data
References
Citations
Citations to this article as recorded by 

- Low-grade mucinous neoplasm originating from intestinal duplication: a case report and review of the literature
Huihui Yin, Jie Yu, Yunzhao Chen
World Journal of Surgical Oncology.2025;[Epub] CrossRef - Adenocarcinoma originating from a colonic duplication cyst: A case report
Jeehye Lee, Jung Wook Suh
World Journal of Gastrointestinal Surgery.2025;[Epub] CrossRef - Low-Grade Mucinous Neoplasm Arising in an Enteric Duplication Cyst of Pancreas: A Case Report and Literature Review
Mengjing Fan, Fang Yang
International Journal of Surgical Pathology.2024; 32(2): 422. CrossRef - Colonic duplication in an adult with chronic constipation: a case report and review of its surgical management
Muhammad Ash-Shafhawi Adznan, Hizami Amin Tai, Aras Emre Canda, Nevra Elmas, Mustafa Cem Terzi
Annals of Coloproctology.2024; 40(Suppl 1): S6. CrossRef - Tubular adenoma arising in tubular colonic duplication: a case report
Heonwoo Lee, Hyeong Rok An, Chan Wook Kim, Young Soo Park
Journal of Pathology and Translational Medicine.2024; 58(4): 198. CrossRef - Right-Sided Colonic Duplication Cyst with a Malignant Twist in a Young Adult — a Case Report
Laxmi Radhakrishnan, Joseph George, Latha K. Abraham
Journal of Gastrointestinal Cancer.2022; 53(3): 805. CrossRef - Endoscopic resection of a duodenal duplication cyst: A case report
Sayumi Kurita, Kazuo Kitagawa, Naoki Toya, Masahiko Kawamura, Muneo Kawamura, Ken Eto
DEN Open.2022;[Epub] CrossRef - Complete colonic duplication presenting as hip fistula in an adult with pelvic malformation: A case report
Xuan Cai, Jing-Tao Bi, Zhi-Xue Zheng, Ya-Qi Liu
World Journal of Clinical Cases.2022; 10(30): 11037. CrossRef - Sigmoid colon duplication seen as a rare cause of ileus in adult: case report
Barış BAYRAKTAR, Salih BOLUK, Sümeyra Emine BÖLÜK
Anatolian Current Medical Journal.2022; 4(3): 323. CrossRef - Successful management of tubular colonic duplication using a laparoscopic approach: A case report and review of the literature
Gan-Bin Li, Jia-Gang Han, Zhen-Jun Wang, Zhi-Wei Zhai, Yu Tao
World Journal of Clinical Cases.2020; 8(15): 3291. CrossRef - Sigmoid colon duplication with ectopic immature renal tissue in an adult: A case report
Hwan Namgung
World Journal of Clinical Cases.2020; 8(24): 6346. CrossRef - Retroperitoneal Mucinous Neoplasm Arising from Colonic Duplication Cyst
María M. Rojas-Rojas, Marcela Mejiah, Martha Mora, Jorge Otero, Fernando Arias-Amézquita, Eduardo Londoño-Schimmer, Paula A. Rodríguez-Urrego
Journal of Gastrointestinal Cancer.2019; 50(3): 583. CrossRef
Adenocarcinoma Arising in a Colonic Duplication Cyst: A Case Report and Review of the Literature
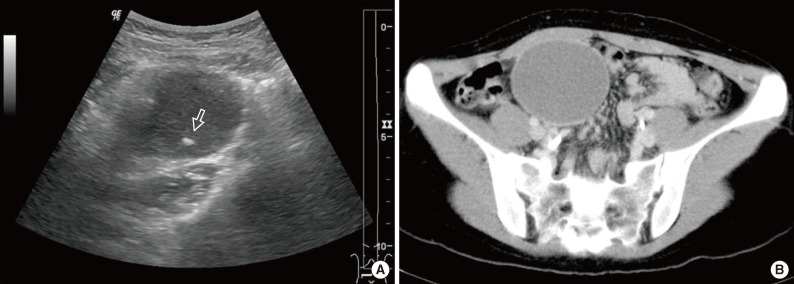
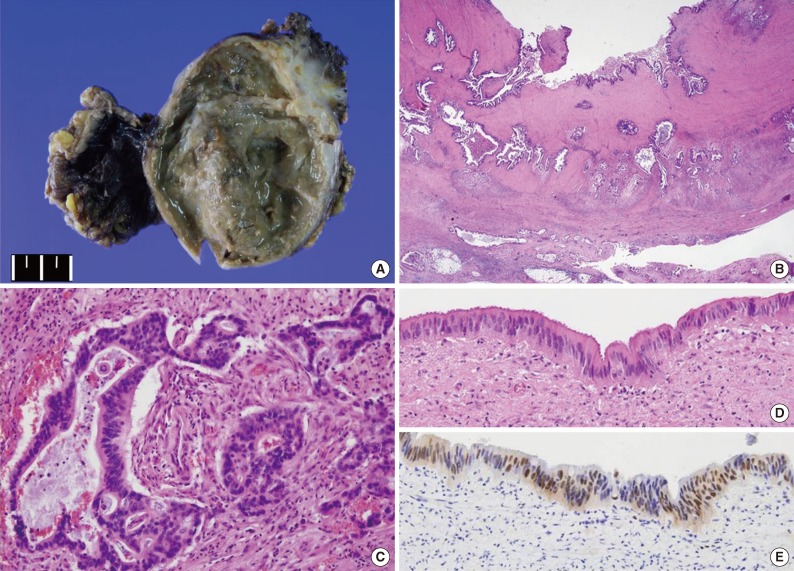
Fig. 1 Radiologic findings. (A) A hypoechoic mass with an echogenic dot (arrow) in the right lower quadrant is observed on ultrasonography. (B) A cystic mass with linear calcification in the mesentery adjacent to the ascending colon is observed on computed tomography.
Fig. 2 Gross and microscopic findings. (A) The cystic mass measured 8.6×6.4×2.9 cm and contained brownish-colored mucoid material. (B) Full-thickness of two smooth muscle layers and adjacent mesenteric soft tissue are invaded by tumor cells. (C) The tumor is moderately differentiated adenocarcinoma. (D) A focal area of atypical columnar epithelium, compatible with dysplasia, is noted on the surface. (E) The dysplastic epithelial cells are positive for p53.
Fig. 1
Fig. 2
Adenocarcinoma Arising in a Colonic Duplication Cyst: A Case Report and Review of the Literature
| Case | Site | Age (yr)/Sex | Symptom | Serum CEA | Size (cm) | Calcification | Depth of invasion | Treatment | Prognosis |
|---|---|---|---|---|---|---|---|---|---|
| 1 [2] | Cecum | 61/F | Abdominal pain | ND | 9 | ND | Muscle layer | RHC, omentectomy, bilateral oophorectomy | NED |
| 2 [3] | Cecum | 50/F | Lower abdominal pain | ND | 8 | ND | Pericystic mesenteric soft tissue | RHC | ND |
| 3 [4] | Cecum | 40/F | Palpable mass | ↑ | 11 | N | Muscle layer | RHC | ND |
| 4 [5] | Cecum | 41/M | Palpable mass | Normal | 6 | Y | Pericystic mesenteric soft tissue | RHC, CT | NED |
| 5 [1] | Ascending | 41/F | RLQ pain | ND | 4 | Y | Thin fibrous wall (muscle layer) | Excision | NED |
| 6 [6] | Ascending | 38/M | Palpable mass | ↑ | ND | Y | Capsule (muscle layer) | RHC | ND |
| 7 [6] | Ascending | 59/F | Fever and lumbar pain | ↑ | ND | Y | Pericystic mesenteric soft tissue and right psoas muscle | Excision | ND |
| 8 [7] | Ascending | 59/M | Melena due to adenocarcinoma in the ascending colon | ND | 10 | ND | Subserosa | RHC | ND |
| 9 [1] | Transverse | 57/F | RLQ pain | ND | 11 | ND | Pericystic mesenteric soft tissue | RHC | NED |
| 10 [8] | Transverse | 40/M | Palpable mass | ND | 16 | ND | Pericystic mesenteric soft tissue and omentum | Excision, CT | ND |
| 11 [4] | Descending | 33/F | Palpable mass | ND | 15 | N | ND | Excision | ND |
| 12 [9] | Sigmoid | 69/F | Epigastric pain | ND | 15 | Y | Pericystic mesenteric soft tissue and retroperitoneum | Excision | Died 41 mos later |
| 13 [10] | Sigmoid | 72/F | Epigastric pain due to cholelithiasis | ND | 3 | ND | Subserosa | Sigmoid colectomy | ND |
| Present case | Ascending | 23/F | Palpable mass | ↑ | 8.6 | Y | Pericystic mesenteric soft tissue | Excision, CT | NED |
Table 1. Clinicopathologic summary of cases with adenocarcinoma arising in a colonic duplication cyst
CEA, carcinoembryonic antigen; F, female; ND, no data available; RHC, right hemicolectomy; NED, no evidence of disease; N, no; M, male; Y, yes; CT, chemotherapy; RLQ, right lower quadrant.

 E-submission
E-submission
 PubReader
PubReader Cite this Article
Cite this Article



