Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(1); 2013 > Article
-
Brief Case Report
Actinomycotic Brain Abscess Developed 10 Years after Head Trauma - Ki Yong Na, Ji-Hye Jang1, Ji-Youn Sung, Youn Wha Kim, Yong-Koo Park
-
Korean Journal of Pathology 2013;47(1):82-85.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.1.82
Published online: February 25, 2013
Department of Pathology, Kyung Hee University School of Medicine, Seoul, Korea.
1Department of Radiology, Kyung Hee University School of Medicine, Seoul, Korea.
- Corresponding Author: Yong-Koo Park, M.D. Department of Pathology, Kyung Hee University Hospital, 26 Kyungheedae-ro, Dongdaemun-gu, Seoul 130-701, Korea. Tel: +82-2-958-8742, Fax: +82-2-958-8730, ykpark@khmc.or.kr
• Received: December 30, 2011 • Revised: February 6, 2012 • Accepted: February 22, 2012
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
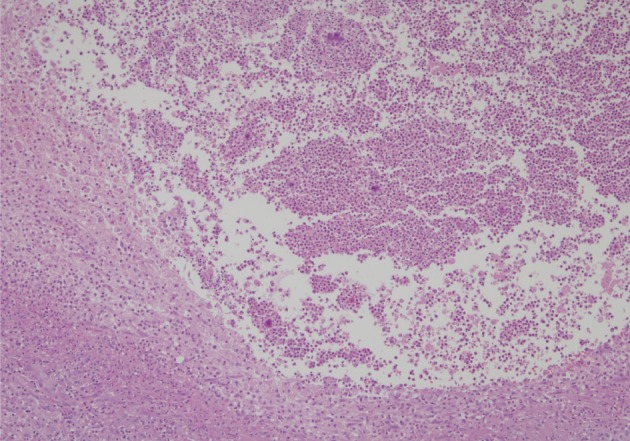
- A healthy 45-year-old man presented with 3 days of left-sided weakness, left-sided facial palsy, and dysarthria. His medical history was remarkable for a laceration head injury 10 years prior. He received a surgical suture and was admitted to the intensive care unit because mild intracranial hemorrhage was identified. He recovered with conservative management and the injury did not leave any neurologic sequelae. The patient and his family denied having any history of heavy alcohol consumption, dental carries, cancer, diabetes mellitus, or surgery on orofacial, thoracic, or abdominal areas. Physical examination revealed that the patient was drowsy but well oriented and afebrile. Laboratory examinations showed a white blood cell count of 9,900/mm3 with a differential count of 68% segmented cells and 23% lymphocytes. The erythrocyte sedimentation rate was 18 mm/hr and the C-reactive protein was 0.3 mg/dL. The initial non-enhanced brain computed tomography demonstrated a 3.5×2.5 cm irregular mass in the right frontal lobe with severe perilesional edema. Brain magnetic resonance imaging showed that the lesion was hypointense on a T1-weighted image and hyperintense on a T2-weighted image, with thick and irregular ring-like enhancement. A diffusion weighted image demonstrated high signal intensity, reflecting restricted diffusion (Fig. 1). For a definite diagnosis and treatment, the patient underwent open surgery and the mass was excised along with surrounding brain parenchyma. Grossly, the specimen included an abscess cavity containing yellowish green pus, surrounded by a thick fibrous capsule. Histological examination showed multiple filamentous organisms appearing as radiating rosettes in the central areas of the abscess (Fig. 2). These structures stained positively with periodic acid-Schiff, Grocott's methenamine silver, Gram stain, and negative with acid-fast stain (Fig. 3). These findings were consistent with the sulfur granules of actinomycoses.4 After the operation, the patient recovered consciousness and 4 weeks of intravenous ampicillin therapy was started (12 g/day). Streptococcus viridans was isolated from the infected brain during antibiotic treatment. However, Actinomyces species were not isolated, despite prolonged culture. The patient was discharged with a good outcome after 4 weeks and continued with 4 months of oral administration of ampicillin.
CASE REPORT
- In 1937, Frideman and Levy conducted a review of CNS actinomycoses by collecting 108 cases reported in the literature.5 However, as nocardiosis has been a recognized medical condition, many cases in Frideman and Levy's report5 were later considered to be nocardioses. Bolton and Ashenhurst6 described 17 cases of CNS actinomycoses. In addition, Smego7 performed a comprehensive analysis by collecting 70 cases of newly reported CNS actinomycoses and describing its main features. CNS actinomycoses have been reported to account for 1-15% of actinomycotic infections. A cerebral abscess is the most common presentation and typically appears as a single or multiple lesions involving the frontal or temporal lobe. In addition, CNS actinomycoses can present subdurally or epidurally. Common clinical manifestations are focal neurologic signs, headache, seizure, and diplopia. High fever and intracranial hypertension are not typical. The mean duration of symptoms prior to diagnosis is >2 months, which is longer than most causes of pyogeneic CNS infections. Most CNS actinomycoses are monomicrobial, although multiple organisms may be seen, which include Aggregatibacter, Capnocytophaga, Fusobacterium, Haemophilus, Staphylococcus, and Streptococcus species.6,7 These pathogens are believed to facilitate actinomycotic infection by inhibiting host defenses and establishing a microaerophilic environment.8
- Table 1 summarizes the features of CNS actinomycoses in Korea. Among them, one was found to be a cerebral abscess in a patient with 2 weeks of headache and diplopia, and another in a patient with a recent seizure episode. In these cases, Haemophilus and Streptococcus species were found coinfecting the patient.2,3 The clinical features of CNS actinomycoses in Korea, including the presenting case, seem to be typical.
- Predisposing conditions for CNS actinomycoses include dental infections, manipulations, oromaxillofacial trauma, gastrointestinal surgery, and the use of an intrauterine device. Less commonly, CNS actinomycoses develop in immunocompromised patients who have diabetes mellitus, neoplasm, or heavy alcohol consumption. These conditions are frequently associated with impairment of local defense barriers. Endogenous infection occurs when organisms enter into damaged tissue. CNS infection can result by direct extension or hematogenous spread from a primary focus. Although primary CNS actinomycoses have been suggested, it is more widely accepted that such cases should be classified as isolated CNS actinomycoses from an unknown primary focus.1,7 In this presenting case, the patient did not have any predisposing factors except for a history of a surgical procedure for a laceration head injury ten years earlier. CNS actinomycoses have been previously reported developing 8, 10, and 22 years after a surgical procedure for a penetrating head injury.4,8 The precise primary sites of infection in these cases are not well defined, however, past head trauma or surgical procedures might contribute to the development of CNS actinomycoses. Makhija et al.9 reported acute exacerbation of actinomycoses in a patient who had a windshield forehead injury. At the time of trauma, the lesion remained painless with only mild swelling, but the lesion increased and became tender with additional trauma after 16 years. It was speculated that actinomycotic implantation might have occurred at the time of initial trauma and the organism was in a chronic and slowly progressive phase, and later transformed from dormancy to produce acute actinomycoses after physical stimulation.9 This theory may be a possible explanation for the development of actinomycotic cerebral abscess an extended period of time after head injury.
- A definite diagnosis of actinomycoses is established by isolation of an actinomyces species in cultures. However, the cultures are negative in up to 70% of cases. Detection of sulfur granules on histological examination is a pathognomic clue to diagnosis. Sulfur granules are clusters of organisms that appear as round or oval basophilic masses with eosinophilic projections on hematoxylin and eosin staining.10 The differential diagnosis should include nocardiosis, which also can form sulfur granules and cause similar clinical syndromes involving the lungs, bones, joints, soft tissue, and the CNS. It is critical to distinguish between these two types of organisms because they respond differently to antibiotics. Microbiologically, Nocardia species are Gram-positive, acid-fast organisms with aerobic requirements, whereas Actinomycetes are Gram-positive, non-acid-fast organisms with anaerobic or microaerophilic requirements.1 Diagnosis of the presenting case was made by the detection of Gram-positive and acid-fast negative sulfur granules.
- In conclusion, we report a cerebral actinomycotic abscess in a patient presenting with motor weakness, which developed 10 years after laceration head trauma. Although CNS actinomycoses are rare, they should be considered causes of cerebral abscess if the patient has the associated risk factors.
DISCUSSION
- 1. Sullivan DC, Chapman SW. Bacteria that masquerade as fungi: actinomycosis/nocardia. Proc Am Thorac Soc 2010; 7: 216-221. ArticlePubMed
- 2. Song EH, Kwon HH, Jang EY, et al. A case of brain abscess caused by Actinomyces meyeri and Haemophilus aphrophilus. Korean J Med 2008; 74(Suppl 1): 144-148.
- 3. Park YM, Won IS, Kim JI, et al. A case of central nervous system actinomycosis presenting as brain abscess. Infect Chemother 2009; 41: 249-252. Article
- 4. Sundaram C, Purohit AK, Prasad VS, Meena AK, Reddy JJ, Murthy JM. Cranial and intracranial actinomycosis. Clin Neuropathol 2004; 23: 173-177. PubMed
- 5. Friedman ED, Levy HH. Actinomycotic infection of the central nervous system: report of a case and review of the literature. Int Clin 1937; 2: 36-61.
- 6. Bolton CF, Ashenhurst EM. Actinomycosis of the brain: case report and review of the literature. Can Med Assoc J 1964; 90: 922-928. PubMedPMC
- 7. Smego RA Jr. Actinomycosis of the central nervous system. Rev Infect Dis 1987; 9: 855-865. ArticlePubMed
- 8. Akhaddar A, Elouennass M, Baallal H, Boucetta M. Focal intracranial infections due to Actinomyces species in immunocompetent patients: diagnostic and therapeutic challenges. World Neurosurg 2010; 74: 346-350. ArticlePubMed
- 9. Makhija LK, Jha MK, Bhattacharya S, Bhardwaj M, Rai A, Mishra S. Dormant primary cutaneous actinomycosis: acute exacerbation after 16 years. J Plast Reconstr Aesthet Surg 2011; 64: 268-271. ArticlePubMed
- 10. Budenz CL, Tajudeen BA, Roehm PC. Actinomycosis of the temporal bone and brain: case report and review of the literature. Ann Otol Rhinol Laryngol 2010; 119: 313-318. ArticlePubMedPDF
REFERENCES
Fig. 1Magnetic resonance imaging of the brain. Gadolinium-enhanced T1-weighted images show a ring-like enhancing lesion in the right frontal lobe.
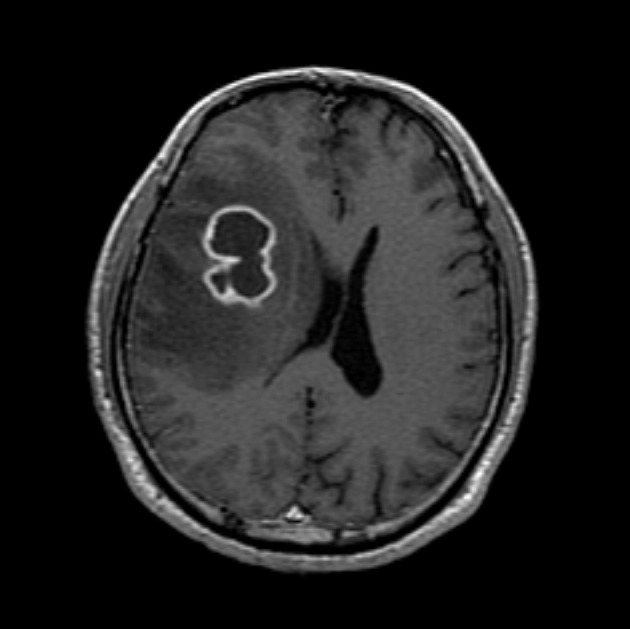

Fig. 3Gram-positive (A) and acid fast-negative (B) filamentous organisms consistent with Actinomyces.
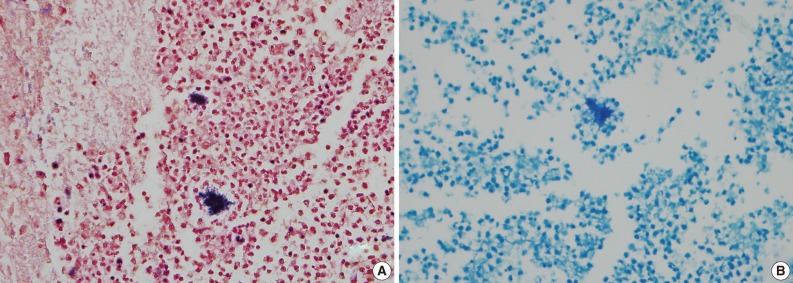

Table 1.Clinical features of central nervous system actinomycoses in Korea
Figure & Data
References
Citations
Citations to this article as recorded by 

- Unveiling a Rare Central Nervous System Infection: A Case of Primary Cerebral Actinomycosis
Shibashish Bhattacharjee, Mirant Doshi, Haryax Pathak
Neurology India Case Report.2025; 1(1): 74. CrossRef - Actinomyeces Cerebral Abscess Masquerading as Tuberculosis: Delayed Presentation following Head Trauma and Scalp Infection
Manish Kumar, Ankur Bajaj, Manjul Tripathi, BishanD Radotra, ManojK Tewari, ChiragK Ahuja
Neurology India.2019; 67(4): 1123. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
Actinomycotic Brain Abscess Developed 10 Years after Head Trauma
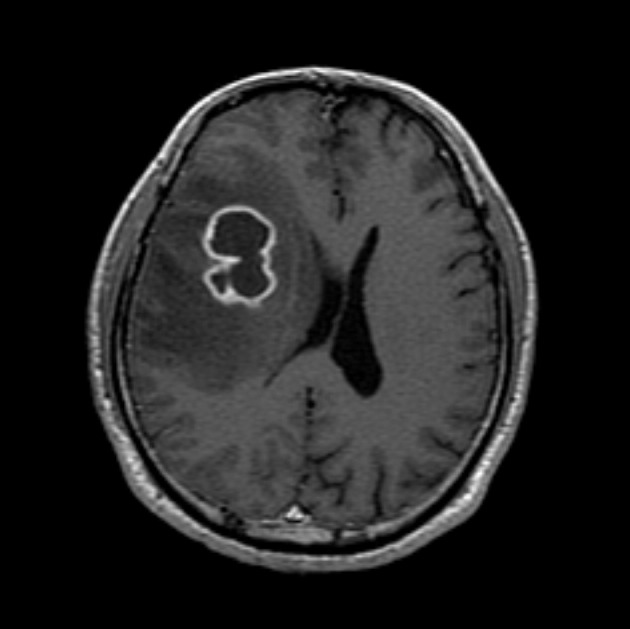
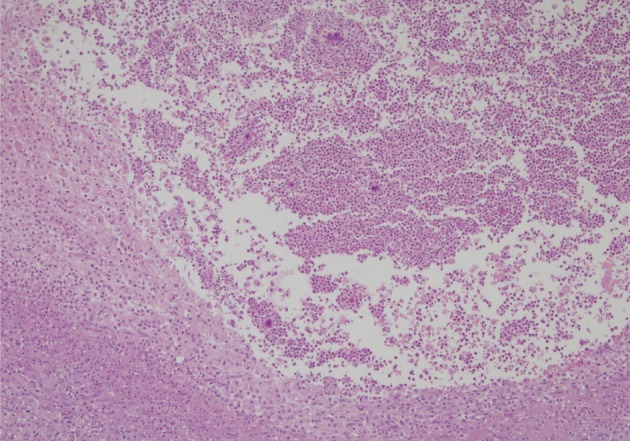
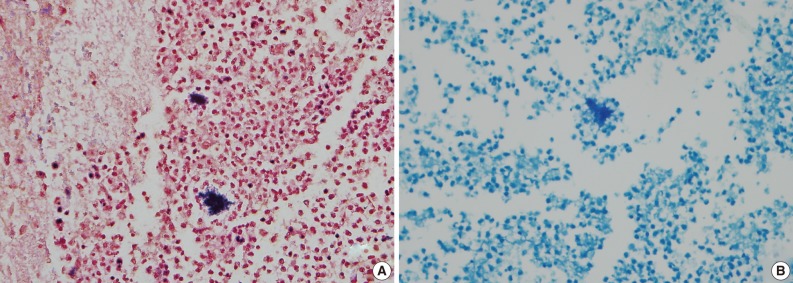
Fig. 1 Magnetic resonance imaging of the brain. Gadolinium-enhanced T1-weighted images show a ring-like enhancing lesion in the right frontal lobe.
Fig. 2 Section of the brain showing radiating bacterial colony in the pyogranuloma.
Fig. 3 Gram-positive (A) and acid fast-negative (B) filamentous organisms consistent with Actinomyces.
Fig. 1
Fig. 2
Fig. 3
Actinomycotic Brain Abscess Developed 10 Years after Head Trauma
| No. | Age (yr)/Sex | Risk factor | Clinical presentation | Image | Co-pathogen | Treatment |
|---|---|---|---|---|---|---|
| 1 | 27/F | None | Headache, diplopia | CT: 3.4 × 2.6 cm sized low density lesion with ring enhancement in right occipital lobe | Haemophilus species | I.V. ampicillin, metronidazole |
| 2 | 45/M | Dental carries | Generalized tonic-clonic seizure | MR: 2.2 × 1.8 cm sized mass in right frontal lobe. Iso signal intensity lesion on T1WI. Peripheral low signal intensity with peripheral enhancement on T2WI | Streptococcus species | I.V penicillin-G |
| Present case | 45/M | Head trauma | Motor weakness, drowsy consciousness | CT: 3.5 × 2.5 cm sized low attenuated lesion with rim enhancement in right frontal lobe | Streptococcus species | Open resection I.V ampicillin |
Table 1. Clinical features of central nervous system actinomycoses in Korea
CNS, central nervous system; F, female; CT, computed tomography; I.V., intravenous; M, male; MR, magnetic resonance; T1WI, T1-weighted image; T2WI, T2-weighted image.

 E-submission
E-submission