Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 54(4); 2020 > Article
-
Case Study
Gastric crystal-storing histiocytosis with concomitant mucosa-associated lymphoid tissue lymphoma -
Mee Joo1
 , Nam-Hoon Kim2
, Nam-Hoon Kim2
-
Journal of Pathology and Translational Medicine 2020;54(4):332-335.
DOI: https://doi.org/10.4132/jptm.2020.04.20
Published online: May 22, 2020
1Department of Pathology, Ilsan Paik Hospital, Inje University College of Medicine, Goyang, Korea
2Division of Gastroenterology and Hepatology, Department of Internal Medicine, Ilsan Paik Hospital, Inje University College of Medicine, Goyang, Korea
- Corresponding Author: Mee Joo, MD, PhD, Department of Pathology, Inje University Ilsan Paik Hospital, 170 Juhwa-ro, Ilsanseo-gu, Goyang 10380, Korea Tel: +82-31-910-7141, Fax: +82-31-910-7139, E-mail: mjoo@paik.ac.kr
© 2020 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- Crystal-storing histiocytosis (CSH) is a rare entity that is characterized by intrahistiocytic accumulation of crystallized immunoglobulins. CSH is not a malignant process per se, but the majority of CSH cases are associated with underlying lymphoproliferative disorder. Although CSH can occur in a variety of organs, gastric CSH is very rare. We present a localized gastric CSH with concomitant mucosaassociated lymphoid tissue (MALT) lymphoma, manifesting as an ulcer bleeding in a 56-year-old man. Histologically, the biopsied gastric mucosa demonstrated expansion of the lamina propria by prominent collections of large eosinophilic mononuclear cells containing fibrillary crystalloid inclusions. Immunohistochemical studies revealed that the crystal-storing cells were histiocytes harboring kappa light chain-restricted immunoglobulin crystals. Within the lesion, atypical centrocyte-like cells forming lymphoepithelial lesions were seen, consistent with MALT lymphoma. Since this entity is rare and unfamiliar, difficulties in diagnosis may arise. Particularly, in this case, the lymphomatous area was obscured by florid CSH, making the diagnosis more challenging.
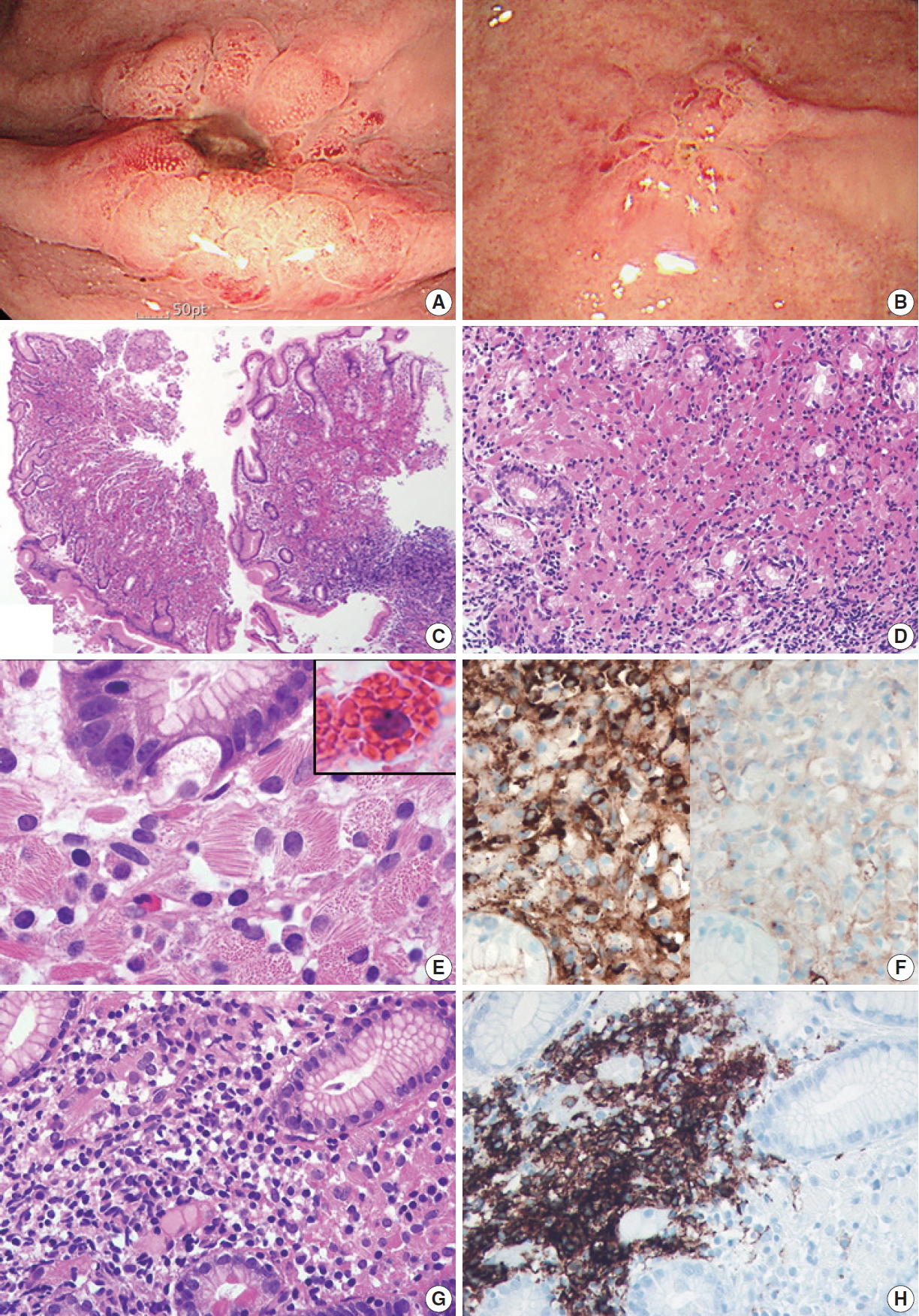
- A 79-year-old man was admitted to our hospital with melena and hematemesis lasting for 5 hours. He denied any previous history of gastrointestinal bleeding. His medical history included lacunar infarct, hypertension, and dementia. He was taking Aspirin Protect (100 mg/day). Laboratory test demonstrated anemia (hemoglobin, 9 g/dL; hematocrit, 26.7%), but otherwise normal complete blood counts. Emergency endoscopy and colonoscopy were performed. Colonoscopy showed no bleeding focus. Esophagogastroduodenoscopy revealed a 1 cm-sized active ulcer with an eroded vessel in the high body (Fig. 1A), which was treated with epinephrine injection and argon plasma coagulation. The surrounding mucosa was hyperemic and friable. A flat nodular lesion with irregular margin and discoloration was also seen in the low body (Fig. 1B). Multiple biopsy specimens were obtained from the margin, the surrounding area of the ulcer, and the flat nodular lesion. Microscopically, all biopsy specimens showed marked expansion of the lamina propria by sheets of large mononuclear cells with brightly eosinophilic cytoplasm and eccentric small bland nuclei (Fig. 1C, D). At higher magnification, abundant eosinophilic cytoplasm contained fibrillary, needle-like, non-refractile crystalloid inclusions (Fig. 1E). Immunohistochemical studies revealed that these crystal-storing cells were histiocytes harboring kappa light chain-restricted immunoglobulin crystals; they were diffusely positive for CD68 and kappa light chain (Fig. 1F, left), but negative for lambda light chain (Fig. 1F, right), desmin, smooth muscle actin, S100 protein, CD117, CD1a, and cytokeratin. Also, there were multifocal aggregates of atypical lymphocytes and plasma cells, which also showed kappa light chain restriction. Atypical lymphocytes forming lymphoepithelial lesions were CD20-positive B cells (Fig. 1G, H), and the diagnosis of CSH associated with MALT lymphoma was rendered accordingly. It was not easy to identify the lymphoepithelial lesions due to overwhelming accumulation of crystal-storing histiocytes. Helicobacter pylori–like organisms were not observed histologically, and the result of Campylobacter-like organism test was also negative. The patient refused to undergo bone marrow biopsy. Abdominal and pelvic computed tomography revealed no lymph node enlargement or organomegaly.
CASE REPORT
- To date, about 130 cases of CSH have been reported in a wide variety of organs, including bone marrow, lymph nodes, liver, spleen, lungs, gastrointestinal tract, kidney, central nervous system, and skin [1-5]. It may present as either localized (a single deposit involving only one organ or site) or generalized (multiple deposits involving more than one organ or site) forms [2]. Localized CSH is more frequent than generalized CSH (about 70% versus 30%) [2-3,5]. The vast majority of the reported CSH cases had underlying lymphoproliferative or plasma cell disorder with monotypic kappa light chain [2-4]. CSH is extremely rare in the stomach, and until now, 14 cases of gastric CSH (including the present one) have been described in the English literature [1,4,6-10]. The detailed clinical and pathological findings of these patients are summarized in Table 1. The 14 patients reported included 8 men and 6 women with a mean age at diagnosis of 61 years (range, 35 to 82 years). There were one generalized (7.1%) and 13 localized (92.9%) forms. Among them, eight patients (57%) had a concomitant or subsequent lymphoplasmacytic malignancy: four MALT lymphomas with kappa-restriction, one mantle cell lymphoma with lambda-restriction, one diffuse large B cell lymphoma with kappa-restriction, one multiple myeloma with lambda-restriction, and one metachronous thymic lymphoma. Five patients (35.7%) had no associated disease except H. pylori infection, and the remaining one patient showed no etiologic condition. Four of the patients who had H. pylori infection alone did not develop other gastric lesion or symptoms during the follow up period [7-9]. Compared to other organs, gastric CSH predominantly manifested as a localized form, and about half of the cases were not related to clonal lymphoproliferative disorders: instead, they were frequently associated with H. pylori–associated gastritis.
- The differential diagnosis of CSH may include a variety of conditions characterized by collections of large eosinophilic tumor cells (adult rhabdomyoma, granular cell tumor, and oncocytic neoplasms) or histiocytic aggregation (Langerhans cell histiocytosis, fibrous histiocytoma, xanthogranuloma, Gaucher’s disease, malakoplakia, and mycobacterial or fungal infection) [2-4,6]. However, considering the distinctive intracytoplasmic immunoglobulin crystal and gastric location, Russell body gastritis (RBG) may be the most difficult differential diagnosis. RBG is another rare entity characterized by aberrant immunoglobulin deposits in the stomach [11]. Unlike CSH consisting of predominantly histiocytes with crystallized immunoglobulin in the lysosome, RBG is composed of plasma cells with small condensed spherical immunoglobulin surrounded by endoplasmic reticulum membrane (called Mott cells) [12]. Besides histological picture displaying diffuse proliferation of large eosinophilic mononuclear cells in the lamina propria, RBG cases also share similar features, such as frequent kappa light chain restriction of accumulated immunoglobulin (43%) and strong association with H. pylori infection (67%) [11]. There were two cases of RBG related to lymphoplasmacytic neoplasm: one with concomitant MALT lymphoma and another with metachronous multiple myeloma three years after RBG diagnosis [13,14]. However, so far, RBG has been considered a unique inflammatory reaction rather than a paraneoplastic phenomenon. Thus, gastric CSH seems to be more significant than RBG in the aspect of association with lymphoproliferative disorder.
- In conclusion, although CSH rarely manifests in the stomach, the recognition of CSH is important to initiate a clinical workup searching for the underlying neoplasm or associated cause. Therefore, once the diagnosis of CSH is rendered, pathologists have to provide prompt notification to the clinician. Sometimes, CSH can be so extensive as to obscure the concomitant neoplasm. Thus, pathologists should be aware of the detailed histological features of CSH to avoid misdiagnosis and also should have a high level of suspicion for the presence of accompanying lymphoproliferative disorder.
DISCUSSION
Ethics Statement
This study was approved by the Institutional Review Board of Inje University Ilsan Paik Hospital with a waiver of informed consent (IRB No. ISPAIK 2020-02-004) and performed in accordance with the principles of the Declaration of Helsinki.
Author contributions
Conceptualization: MJ, NHK. Investigation: MJ. Visualization: MJ, NHK.
Writing—original draft: MJ. Writing—review & editing: MJ, NHK
Conflict of Interest
The authors declare that they have no potential conflicts of interest.
Funding
No funding to declare.
| Year | Study | Sex/Age (yr) | Endoscopic finding | Helicobacter pylori infection | Associated neoplasm | Ig light chain | Follow-up data |
|---|---|---|---|---|---|---|---|
| 1999 | Jones et al. [1] | F/35 | NS | NS | Thymic lymphoma | Polyclonal | Persist |
| 2006 | Stewart and Spagnolo [7] | M/82 | Gastritis | Yes | No | Lambda | Died of unrelated cause |
| 2006 | Stewart and Spagnolo [7] | M/81 | Gastritis | Yes | No | Insufficient | No symptoms or lesion |
| 2006 | Stewart and Spagnolo [7] | F/52 | Erosion | Yes | No | Lambda | No symptoms or lesion |
| 2007 | Joo et al. [8] | F/56 | Polyps | Yes | No | Polyclonal | No residual lesion |
| 2013 | Yano et al. [9] | F/55 | Discoloration with granularity | Yes | No | Polyclonal | Alive without disease |
| 2014 | Vaid et al. [10] | M/NS | Discolored patch | NS | No | Kappa | No |
| 2016 | Kanagal-Shamanna et al. [4] | M/43 | Nodule | NS | MALT lymphoma | Kappa | Alive without disease |
| 2016 | Kanagal-Shamanna et al. [4] | M/51 | NS | NS | Multiple myeloma | Lambda | No |
| 2018 | Arnold et al. [6] | a | Discoloration with granularity | Yes | MALT lymphoma | Kappa | Persist |
| 2018 | Arnold et al. [6] | a | Discoloration with granularity | Yes | MALT lymphoma | Kappa | Persist |
| 2018 | Arnold et al. [6] | a | Malignant-appearing mass | No | Mantle cell lymphoma | Lambda | Died of lymphoma |
| 2018 | Arnold et al. [6] | a | Malignant-appearing mass | No | DLBCL | Kappa | Died of lymphoma |
| 2020 | Present case | M/79 | Ulcer, flat nodularity | No | MALT lymphoma | Kappa | No |
- 1. Jones D, Bhatia VK, Krausz T, Pinkus GS. Crystal-storing histiocytosis: a disorder occurring in plasmacytic tumors expressing immunoglobulin kappa light chain. Hum Pathol 1999; 30: 1441-8. ArticlePubMed
- 2. Dogan S, Barnes L, Cruz-Vetrano WP. Crystal-storing histiocytosis: report of a case, review of the literature (80 cases) and a proposed classification. Head Neck Pathol 2012; 6: 111-20. ArticlePubMedPMCPDF
- 3. Fang H, Chiu A, Reichard KK. Crystal-storing histiocytosis in bone marrow: a clinicopathologic study of eight cases and review of the literature. Am J Clin Pathol 2018; 149: 148-63. PubMedPDF
- 4. Kanagal-Shamanna R, Xu-Monette ZY, Miranda RN, et al. Crystalstoring histiocytosis: a clinicopathological study of 13 cases. Histopathology 2016; 68: 482-91. ArticlePubMed
- 5. Galeano-Valle F, Díaz-Crespo FJ, Melero-Martín R, Apaza-Chávez JE, Del-Toro-Cervera J, Demelo-Rodríguez P. Massive generalized crystal-storing histiocytosis associated with extracellular crystalline nephropathy: clinical, immunohistochemical, and ultrastructural studies of a unique disorder and review of the literature. CEN Case Rep 2019; 8: 166-72. ArticlePubMedPMCPDF
- 6. Arnold CA, Frankel WL, Guo L, et al. Crystal-storing histiocytosis in the stomach: a clue to subtle hematolymphoid malignancies. Am J Surg Pathol 2018; 42: 1317-24. PubMed
- 7. Stewart CJ, Spagnolo DV. Crystalline plasma cell inclusions in helicobacter-associated gastritis. J Clin Pathol 2006; 59: 851-4. ArticlePubMedPMC
- 8. Joo M, Kwak JE, Chang SH, et al. Localized gastric crystal-storing histiocytosis. Histopathology 2007; 51: 116-9. ArticlePubMed
- 9. Yano Y, Nagahama T, Matsui T, et al. Gastric crystal-storing histiocytosis detected with asymptomatic Sjogren’s syndrome: report of a case and summary. Clin J Gastroenterol 2013; 6: 237-42. ArticlePubMedPDF
- 10. Vaid A, Caradine KD, Lai KK, Rego R. Isolated gastric crystal-storing histiocytosis: a rare marker of occult lymphoproliferative disorders. J Clin Pathol 2014; 67: 740-1. ArticlePubMed
- 11. Joo M. Rare Gastric lesions associated with Helicobacter pylori infection: a histopathological review. J Pathol Transl Med 2017; 51: 341-51. ArticlePubMedPMCPDF
- 12. Hasegawa H. Aggregates, crystals, gels, and amyloids: intracellular and extracellular phenotypes at the crossroads of immunoglobulin physicochemical property and cell physiology. Int J Cell Biol 2013; 2013: 604867.ArticlePubMedPMCPDF
- 13. Joo M. Gastric mucosa-associated lymphoid tissue lymphoma masquerading as Russell body gastritis. Pathol Int 2015; 65: 396-8. PubMed
- 14. Altindag SD, Cakir E, Ekinci N, Avci A, Dilek FH. Analysis of clinical and histopathological findings in Russell body gastritis and duodenitis. Ann Diagn Pathol 2019; 40: 66-71. ArticlePubMed
REFERENCES
Figure & Data
References
Citations

- Crystal-Storing Histiocytosis of the Stomach: An Unusual Clinical Context of a Rare Entity
Jenna Magri, Katsiaryna Khatskevich, Lauren Shealy, David Lewin, Chadi Hajar
International Journal of Surgical Pathology.2026; 34(1): 222. CrossRef - Sunny side up
João Pedro Pereira, Joanne Lopes, Fatima Carneiro, Francisco Baldaque-Silva
Frontline Gastroenterology.2025; 16(4): 344. CrossRef - Crystal-storing histiocytosis in the stomach: A case report and review of the literature
Linghong Kong, Liyan Xue, Yanfeng Zhong, Shenglan Wang, Danfeng Zheng, Lining Wang, Yang Jiao, Xinpeng Zhang, Huizhong Xue, Xiaogang Liu
Frontiers in Oncology.2022;[Epub] CrossRef - Lambda-Restricted Crystal-Storing Histiocytosis of Stomach: A Case Report and Review of Literature
Nalini Bansal, Pankaj Puri, Nishant Nagpal, Rahul Naithani, Rahul Gupta
Cureus.2021;[Epub] CrossRef - Immunoglobulin-Storing Histiocytosis: A Case Based Systemic Review
Hanne Wiese-Hansen, Friedemann Leh, Anette Lodvir Hemsing, Håkon Reikvam
Journal of Clinical Medicine.2021; 10(9): 1834. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
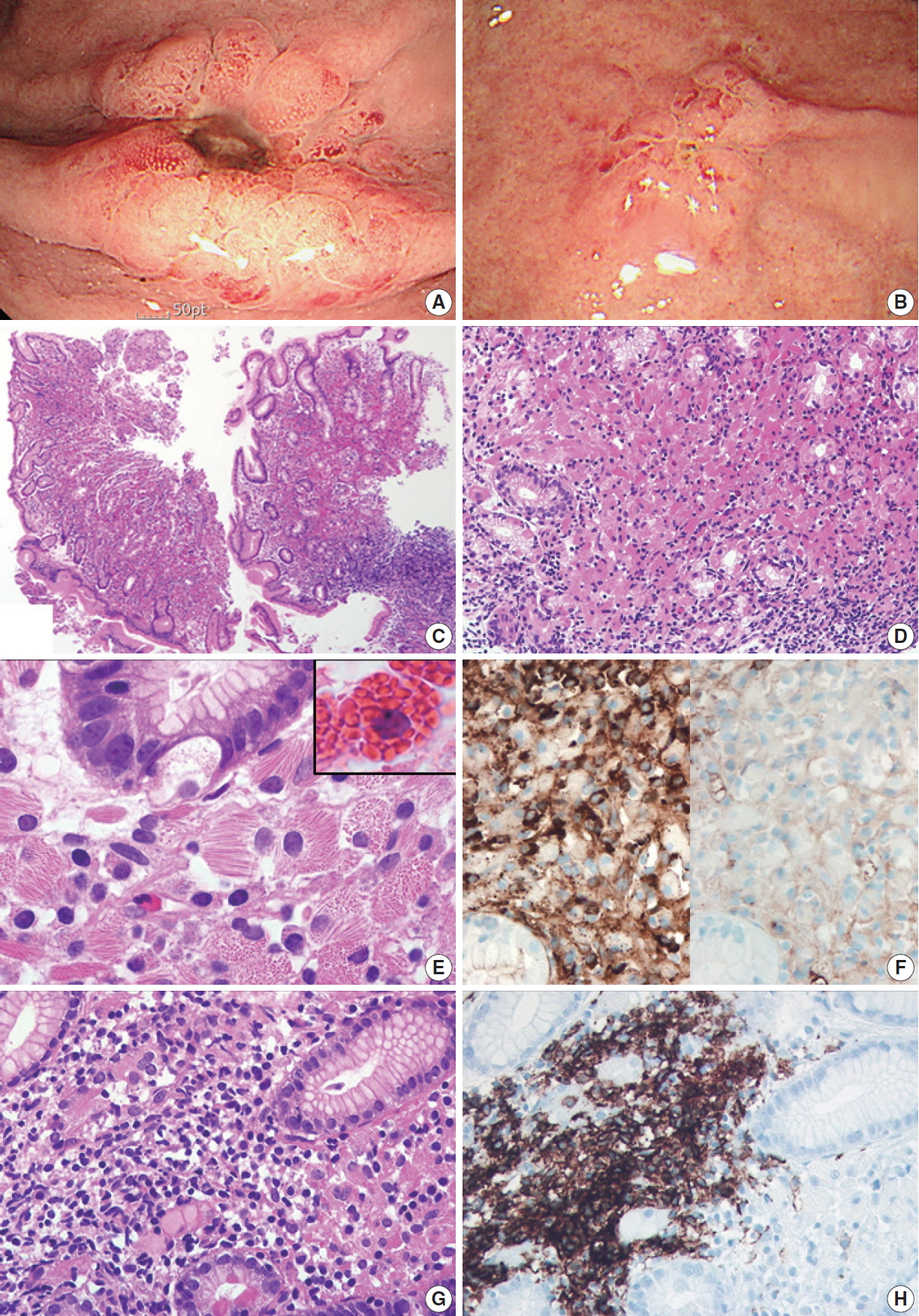
Fig. 1.
| Year | Study | Sex/Age (yr) | Endoscopic finding | Helicobacter pylori infection | Associated neoplasm | Ig light chain | Follow-up data |
|---|---|---|---|---|---|---|---|
| 1999 | Jones et al. [1] | F/35 | NS | NS | Thymic lymphoma | Polyclonal | Persist |
| 2006 | Stewart and Spagnolo [7] | M/82 | Gastritis | Yes | No | Lambda | Died of unrelated cause |
| 2006 | Stewart and Spagnolo [7] | M/81 | Gastritis | Yes | No | Insufficient | No symptoms or lesion |
| 2006 | Stewart and Spagnolo [7] | F/52 | Erosion | Yes | No | Lambda | No symptoms or lesion |
| 2007 | Joo et al. [8] | F/56 | Polyps | Yes | No | Polyclonal | No residual lesion |
| 2013 | Yano et al. [9] | F/55 | Discoloration with granularity | Yes | No | Polyclonal | Alive without disease |
| 2014 | Vaid et al. [10] | M/NS | Discolored patch | NS | No | Kappa | No |
| 2016 | Kanagal-Shamanna et al. [4] | M/43 | Nodule | NS | MALT lymphoma | Kappa | Alive without disease |
| 2016 | Kanagal-Shamanna et al. [4] | M/51 | NS | NS | Multiple myeloma | Lambda | No |
| 2018 | Arnold et al. [6] | Discoloration with granularity | Yes | MALT lymphoma | Kappa | Persist | |
| 2018 | Arnold et al. [6] | Discoloration with granularity | Yes | MALT lymphoma | Kappa | Persist | |
| 2018 | Arnold et al. [6] | Malignant-appearing mass | No | Mantle cell lymphoma | Lambda | Died of lymphoma | |
| 2018 | Arnold et al. [6] | Malignant-appearing mass | No | DLBCL | Kappa | Died of lymphoma | |
| 2020 | Present case | M/79 | Ulcer, flat nodularity | No | MALT lymphoma | Kappa | No |
F, female; NS, not stated; M, male; MALT, mucosa-associated lymphoid tissue; DLBCL, diffuse large B cell lymphoma. Including 2 females and 2 males with age range from 56 to 82.

 E-submission
E-submission



