Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 53(5); 2019 > Article
-
Case Study
Diffuse Involvement of Primary Colorectal Lymphoma Simulating Ulcerative Colitis -
Ji-Ye Kim
 , Sun Hee Chang
, Sun Hee Chang , Han Seong Kim
, Han Seong Kim , Mee Joo
, Mee Joo
-
Journal of Pathology and Translational Medicine 2019;53(5):332-336.
DOI: https://doi.org/10.4132/jptm.2019.07.12
Published online: August 2, 2019
Department of Pathology, Inje University Ilsan Paik Hospital, Goyang, Korea
- Corresponding Author: Mee Joo, MD, PhD, Department of Pathology, Inje University Ilsan Paik Hospital, 170 Juhwa-ro, Ilsanseo-gu, Goyang 10380, Korea Tel: +82-31-910-7141, Fax: +82-31-910-7139, E-mail: mjoo@paik.ac.kr
© 2019 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 7,171 Views
- 93 Download
Abstract
- Diffuse involvement of colorectal lymphoma masquerading as colitis is a very rare presentation of primary colorectal lymphoma. Detecting occult lymphoma is difficult in the setting of diffuse colonic involvement with no definite mass and inflammatory mucosal changes. We encountered a case of diffuse-type primary colorectal lymphoma simulating ulcerative colitis in a previously healthy 31-year-old woman. Despite multiple mucosal biopsies, the biopsy diagnosis was not made due to unawareness of atypical lymphocytes admixed with dense lymphoplasmacytic infiltration. The present case emphasizes the importance of being aware of this rare presentation of primary colorectal lymphoma in order to avoid misdiagnosis.
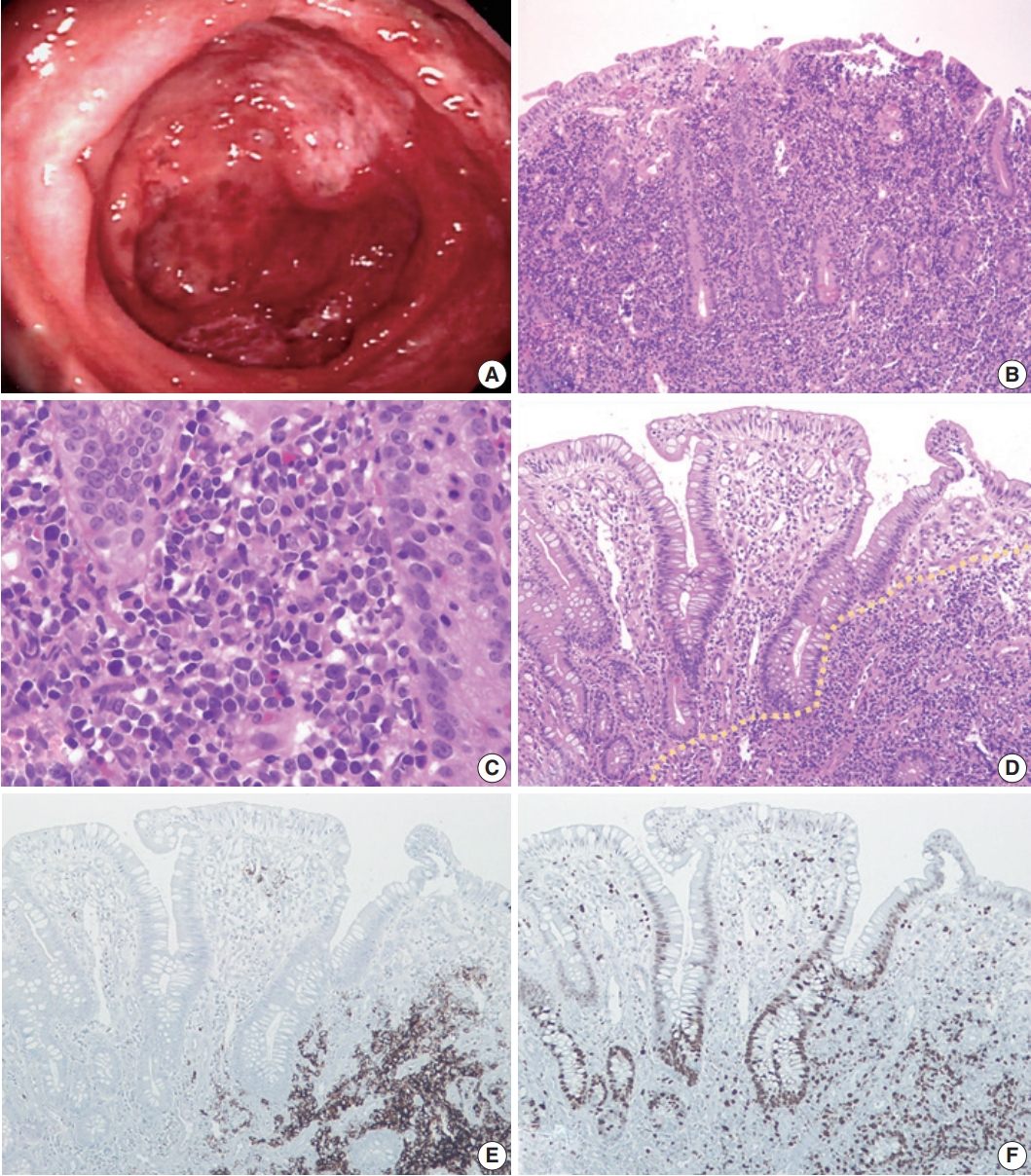
- A 31-year-old woman with no prior history of IBD presented with intermittent abdominal pain and mucoid diarrhea. Five months later, she developed hematochezia and was diagnosed with UC on the basis of endoscopic biopsy. She was treated with oral Pentasa and Pentasa enema. However, she continued to be symptomatic with marked weight loss (20 kg in 4 months). Follow-up colonoscopy revealed continuous mucosal changes from the rectum to the cecum, such as diffuse hyperemia, edematous changes, friability, loss of normal vascularity, and multiple variable-sized ulcers, consistent with UC (Fig. 1A). No mass-forming lesion was present throughout. Abdominal and pelvic computed tomography revealed diffuse edematous wall thickening in the whole colon and multiple enlargements of mesenteric lymph nodes. No hepatosplenomegaly was observed. Multiple biopsies were taken from the terminal ileum, cecum, transverse colon, sigmoid colon, and rectum. All biopsy specimens revealed diffuse infiltration of large, atypical lymphoid cells with a high nucleus-to-cytoplasmic ratio, occasional prominent nucleoli, and mitotic figures (Fig. 1B, C). They expanded the lamina propria, pushed the crypts apart, and invaded the submucosa. By immunochemistry, the tumor cells were diffusely positive for CD20, bcl2, and p53, and negative for CD3, CD5, CD10, and cyclin D1. The Ki-67 labeling index was about 60%. The overall histology and immunophenotype supported a diagnosis of diffuse large B-cell lymphoma. There was no detectable evidence of extra-abdominal lymphadenopathy or lymphomatous involvement elsewhere. Bone marrow evaluation was also negative for lymphoma. As no extraintestinal disease was found, we considered it as a primary colorectal lymphoma. The patient died while receiving the second cycle of R-CHOP chemotherapy.
- We retrospectively reviewed the colonoscopic biopsy specimens that were initially diagnosed with UC. At low magnification view, the background mucosa displayed crypt architectural distortion and dense inflammatory cell infiltration, resembling chronic colitis. However, we found some lymphoma cells, which were admixed with other inflammatory cells or showed focal aggregation in the basal portion of the mucosa (Fig. 1D). Those lymphoma cells were more distinguishable on immunostaining for CD20 and Ki-67 (Fig. 1E, F).
- Ethics statment
- This study was approved by the Institutional Review Board of Inje University Ilsan Paik Hospital with a waiver of informed consent (IRB No. ISPAIK 2019-05-005) and performed in accordance with the principles of the Declaration of Helsinki.
CASE REPORT
- Twenty cases (including the present one) of “colitis-like” diffuse-type colorectal lymphoma were reported in the English literature [2-17]. We excluded cases of colorectal lymphoma developed in patients with a longstanding IBD or prior established diagnosis of extracolonic lymphoma. The clinical and pathological findings of these patients are summarized in Table 1. There were 10 men, 8 women, and two children. Their mean age at presentation was 53.2 years (range, 6 to 82 years). The main symptoms were diarrhea/hematochezia (90%), loss of weight (50%), abdominal pain (25%), and fever (10%). Endoscopic findings were consistent with UC (55%), Crohn disease (20%), diffuse colitis (20%), and multiple ulcers (5%): no localized mass-like lesion was present. All cases were non-Hodgkin’s lymphoma: eight were B-cell type (four mantle cell lymphomas, two mucosa-associated lymphoid tissue lymphomas, one diffuse large B-cell lymphoma, and one follicular lymphoma), seven were T-cell type, one was natural killer cell lymphoma, and four other unclassified lymphoreticular malignancies (one malignant lymphoma, one reticulum cell sarcoma, one lymphosarcoma, and one lymphocytic lymphoma). Of note, there were nine patients in whom colitis was histologically confirmed with multiple biopsy or colectomy but were subsequently found to have lymphoma involvement in a retrospective review of previous slides. Taken together, diffuse type primary colorectal lymphoma is a very rare disease and is easily misdiagnosed, particularly in reliance on endoscopic biopsy examination alone. The possibility of a hidden lymphomatous involvement would have to be considered in patients with medically refractory and rapidly progressive colitis.
- Lymphoma involvement of the gastrointestinal (GI) tract may occur either as an isolated primary neoplasm or as a manifestation of systemic generalized lymphoma. To distinguish between primary and secondary GI lymphoma, Dawson’s criteria is generally applied: (1) no palpable superficial lymphadenopathy at initial presentation, (2) a normal chest X-ray with no mediastinal lymphadenopathy, (3) no evidence of leukemia, (4) a predominant mass in the bowel with only local lymphadenopathy, and (5) no hepatosplenomegaly [19]. When the diagnostic criteria of primary colonic lymphoma are strictly applied, only seven of 20 cases of “colitis-like” colorectal lymphoma are eligible for diffuse-type primary colorectal lymphoma: 13 cases were excluded due to synchronous other organ involvement at initial presentation, such as superficial or generalized lymphadenopathy (six cases), upper GI involvement (five cases), bone marrow involvement (four cases), or hepatosplenomegaly (three cases), etc. Consequently, it is a reflection that “colitis-like” diffuse involvement of colorectal lymphoma seems to be more often secondary. Accurate discrimination between primary and secondary colorectal lymphoma is important for proper staging and management. In conclusion, since primary colorectal lymphoma can rarely manifest as “colitis-like” diffuse colonic involvement, awareness of this rare presentation is important to ensure proper diagnosis and treatment.
DISCUSSION
Author contributions
Conceptualization: MJ.
Investigation: MJ.
Visualization: MJ.
Writing—original draft: JYK, MJ.
Writing—review & editing: JYK, MJ, SHC, HK.
Conflicts of Interest
The authors declare that they have no potential conflicts of interest.
Funding
No funding to declare.
| Year | Author | Sex/Age (yr) | Pathologic diagnosis | Endoscopic findings | Revision of initial diagnosis | Superficial LAP at presentation | Extracolonic involvement |
|---|---|---|---|---|---|---|---|
| 1968 | Friedman et al. [2] | M/37 | Reticulum cell sarcoma | UC | No | No | Liver |
| 1968 | Friedman et al. [2] | F/54 | Malignant lymphoma | UC | Yes (colectomy) | No | No |
| 1968 | Friedman et al. [2] | M/73 | Lymphosarcoma | UC | Yes (Bx) | No | No |
| 1980 | Weir et al. [3] | F/67 | Lymphocytic lymphoma | CD | Yes (Bx) | Yes, generalized | BM |
| 1992 | McCullough et al. [4] | M/44 | Mantle cell lymphoma | UC | Yes (Bx) | Yes, cervical axillary | Pancreas |
| 1995 | Lenzen et al. [5] | F/53 | MALT lymphoma | UC | No | No | Upper GI tract, BM |
| 1996 | Robert et al. [6] | F/71 | Mantle cell lymphoma | UC | Yes (colectomy) | No | No |
| 1996 | Hirakawa et al. [7] | M/47 | T-cell lymphoma | UC | No | No | Upper GI tract |
| 1997 | Son et al. [8] | F/40 | Peripheral T-cell lymphoma | CD | Yes (Bx) | No | No |
| 2003 | Isomoto et al. [9] | M/47 | Adult T-cell leukemia/lymphoma | UC | No | Yes, generalized | Stomach, skin |
| 2004 | Payne et al. [10] | F/76 | High-grade T-cell lymphoma | Colitis | No | Yes, generalized | No |
| 2004 | Tamura et al. [11] | M/61 | Mantle cell lymphoma | Colitis | No | No | Tonsil, upper GI tract |
| 2008 | Berkelhammer et al. [12] | F/82 | MALT lymphoma | CD | Yes (Bx) | No | No |
| 2014 | Koksal et al. [13] | M/73 | Mantle cell lymphoma | UC | No | No | Stomach |
| 2015 | Zaheen et al. [14] | M/74 | EBV-negative NK cell lymphoma | Colitis | No | No | BM, pleural effusion |
| 2015 | Wu et al. [15] | M/56 | T-cell lymphoma | Ulcers | Yes (Bx) | Yes, generalized | No |
| 2016 | Cheung et al. [16] | NA/12 | EBV-positive T-cell lymphoma | Colitis | No | No | Hepatosplenomegaly |
| 2016 | Cheung et al. [16] | NA/6 | EBV-positive T-cell lymphoma | CD | No | No | No |
| 2017 | Zenda et al. [17] | M/59 | Follicular lymphoma | UC | No | Yes, cervical inguinal | Spleen, BM |
| Present case | F/32 | Diffuse large B-cell lymphoma | UC | Yes (Bx) | No | No |
- 1. Wong MT, Eu KW. Primary colorectal lymphomas. Colorectal Dis 2006; 8: 586-91. ArticlePubMed
- 2. Friedman HB, Silver GM, Brown CH. Lymphoma of the colon simulating ulcerative colitis: report of four cases. Am J Dig Dis 1968; 13: 910-7. PubMedPDF
- 3. Weir AB, Poon MC, Groarke JF, Wilkerson JA. Lymphoma simulating Crohn’s colitis. Dig Dis Sci 1980; 25: 69-72. ArticlePubMedPDF
- 4. McCullough JE, Kim CH, Banks PM. Mantle zone lymphoma of the colon simulating diffuse inflammatory bowel disease: role of immunohistochemistry in establishing the diagnosis. Dig Dis Sci 1992; 37: 934-8. PubMedPDF
- 5. Lenzen R, Borchard F, Lübke H, Strohmeyer G. Colitis ulcerosa complicated by malignant lymphoma: case report and analysis of published works. Gut 1995; 36: 306-10. ArticlePubMedPMC
- 6. Robert ME, Kuo FC, Longtine JA, Sklar JL, Schrock T, Weidner N. Diffuse colonic mantle cell lymphoma in a patient with presumed ulcerative colitis: detection of a precursor monoclonal lymphoid population using polymerase chain reaction and immunohistochemistry. Am J Surg Pathol 1996; 20: 1024-31. PubMed
- 7. Hirakawa K, Fuchigami T, Nakamura S, et al. Primary gastrointestinal T-cell lymphoma resembling multiple lymphomatous polyposis. Gastroenterology 1996; 111: 778-82. ArticlePubMed
- 8. Son HJ, Rhee PL, Kim JJ, et al. Primary T-cell lymphoma of the colon. Korean J Intern Med 1997; 12: 238-41. ArticlePubMedPMCPDF
- 9. Isomoto H, Furusu H, Onizuka Y, et al. Colonic involvement by adult T-cell leukemia/lymphoma mimicking ulcerative colitis. Gastrointest Endosc 2003; 58: 805-8. ArticlePubMed
- 10. Payne S, Phillips M, Reffitt D, et al. An unusual case of colitis. Gut 2004; 53: 1824.ArticlePubMedPMC
- 11. Tamura S, Ohkawauchi K, Yokoyama Y, et al. Non-multiple lymphomatous polyposis form of mantle cell lymphoma in the gastrointestinal tract. J Gastroenterol 2004; 39: 995-1000. ArticlePubMedPDF
- 12. Berkelhammer C, Mohammed A, Zalzaleh G, Grela J, Blumstein A, Stein R. Spontaneous multiperforation in lymphomatous colitis masquerading as Crohn’s disease: 2 cases. Inflamm Bowel Dis 2008; 14: 1172-3. ArticlePubMed
- 13. Koksal AS, Taskiran I, Kalkan IH, Kayacetin E. A rare endoscopic appearance of primary gastrointestinal mantle cell lymphoma resembling ulcerative colitis. Bratisl Lek Listy 2014; 115: 800-1. ArticlePubMed
- 14. Zaheen A, Delabie J, Vajpeyi R, Frost DW. The first report of a previously undescribed EBV-negative NK-cell lymphoma of the GI tract presenting as chronic diarrhoea with eosinophilia. BMJ Case Rep 2015; 2015: bcr2015212103. ArticlePubMedPMC
- 15. Wu PH, Chu KE, Lin YM, Huang SH, Wu CC. T-cell lymphomas presenting as colon ulcers and eosinophilia. Case Rep Gastroenterol 2015; 9: 246-52. ArticlePubMedPMCPDF
- 16. Cheung FM, Tong Y, Wang Y, et al. Epstein-Barr virus-positive T-cell-associated colitis mimicking inflammatory bowel disease: clinicopathological study of two cases. Histopathology 2016; 68: 465-8. ArticlePubMed
- 17. Zenda T, Nakagawa N, Maruyama H, et al. Follicular lymphoma-related colitis resembling ulcerative colitis. Clin J Gastroenterol 2017; 10: 147-53. ArticlePubMedPDF
- 18. Farrell RJ, Ang Y, Kileen P, et al. Increased incidence of non-Hodgkin’s lymphoma in inflammatory bowel disease patients on immunosuppressive therapy but overall risk is low. Gut 2000; 47: 514-9. ArticlePubMedPMC
- 19. Dawson IM, Cornes JS, Morson BC. Primary malignant lymphoid tumours of the intestinal tract. Report of 37 cases with a study of factors influencing prognosis. Br J Surg 1961; 49: 80-9. ArticlePubMedPDF
REFERENCES
Figure & Data
References
Citations

 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
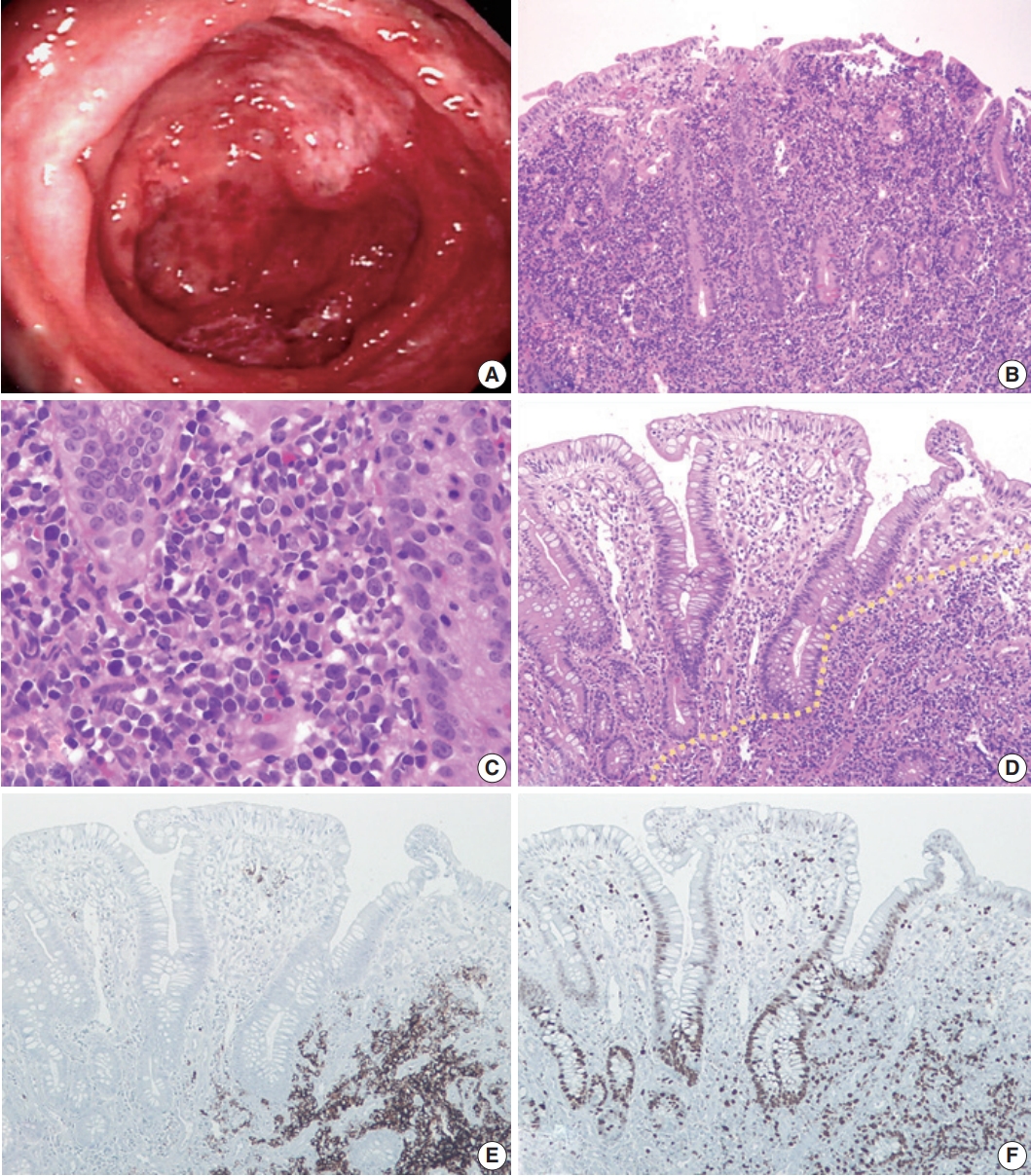
Fig. 1.
| Year | Author | Sex/Age (yr) | Pathologic diagnosis | Endoscopic findings | Revision of initial diagnosis | Superficial LAP at presentation | Extracolonic involvement |
|---|---|---|---|---|---|---|---|
| 1968 | Friedman et al. [2] | M/37 | Reticulum cell sarcoma | UC | No | No | Liver |
| 1968 | Friedman et al. [2] | F/54 | Malignant lymphoma | UC | Yes (colectomy) | No | No |
| 1968 | Friedman et al. [2] | M/73 | Lymphosarcoma | UC | Yes (Bx) | No | No |
| 1980 | Weir et al. [3] | F/67 | Lymphocytic lymphoma | CD | Yes (Bx) | Yes, generalized | BM |
| 1992 | McCullough et al. [4] | M/44 | Mantle cell lymphoma | UC | Yes (Bx) | Yes, cervical axillary | Pancreas |
| 1995 | Lenzen et al. [5] | F/53 | MALT lymphoma | UC | No | No | Upper GI tract, BM |
| 1996 | Robert et al. [6] | F/71 | Mantle cell lymphoma | UC | Yes (colectomy) | No | No |
| 1996 | Hirakawa et al. [7] | M/47 | T-cell lymphoma | UC | No | No | Upper GI tract |
| 1997 | Son et al. [8] | F/40 | Peripheral T-cell lymphoma | CD | Yes (Bx) | No | No |
| 2003 | Isomoto et al. [9] | M/47 | Adult T-cell leukemia/lymphoma | UC | No | Yes, generalized | Stomach, skin |
| 2004 | Payne et al. [10] | F/76 | High-grade T-cell lymphoma | Colitis | No | Yes, generalized | No |
| 2004 | Tamura et al. [11] | M/61 | Mantle cell lymphoma | Colitis | No | No | Tonsil, upper GI tract |
| 2008 | Berkelhammer et al. [12] | F/82 | MALT lymphoma | CD | Yes (Bx) | No | No |
| 2014 | Koksal et al. [13] | M/73 | Mantle cell lymphoma | UC | No | No | Stomach |
| 2015 | Zaheen et al. [14] | M/74 | EBV-negative NK cell lymphoma | Colitis | No | No | BM, pleural effusion |
| 2015 | Wu et al. [15] | M/56 | T-cell lymphoma | Ulcers | Yes (Bx) | Yes, generalized | No |
| 2016 | Cheung et al. [16] | NA/12 | EBV-positive T-cell lymphoma | Colitis | No | No | Hepatosplenomegaly |
| 2016 | Cheung et al. [16] | NA/6 | EBV-positive T-cell lymphoma | CD | No | No | No |
| 2017 | Zenda et al. [17] | M/59 | Follicular lymphoma | UC | No | Yes, cervical inguinal | Spleen, BM |
| Present case | F/32 | Diffuse large B-cell lymphoma | UC | Yes (Bx) | No | No |
LAP, lymphadenopathy; M, male; UC, ulcerative colitis; F, female; Bx, biopsy; CD, Crohn’s disease; BM, bone marrow; MALT, mucosa-associated lymphoid tissue; GI, gastrointestinal; EBV, Epstein-Barr virus; NA, not available.

 E-submission
E-submission



