Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 50(3); 2016 > Article
-
Case Study
Rare Case of Anal Canal Signet Ring Cell Carcinoma Associated with Perianal and Vulvar Pagetoid Spread - Na Rae Kim, Hyun Yee Cho, Jeong-Heum Baek1, Juhyeon Jeong, Seung Yeon Ha, Jae Yeon Seok, Sung Won Park1, Sun Jin Sym2, Kyu Chan Lee3, Dong Hae Chung
-
Journal of Pathology and Translational Medicine 2016;50(3):231-237.
DOI: https://doi.org/10.4132/jptm.2015.08.08
Published online: October 8, 2015
Department of Pathology, Gachon University Gil Medical Center, Incheon, Korea
1Department of General Surgery, Gachon University Gil Medical Center, Incheon, Korea
2Division of Hematology and Oncology, Department of Internal Medicine, Gachon University Gil Medical Center, Incheon, Korea
3Department of Radiation Oncology, Gachon University Gil Medical Center, Incheon, Korea
- Corresponding Author: Dong Hae Chung, MD Department of Pathology, Gachon University Gil Medical Center, 21 Namdong-daero 774beon-gil, Namdong-gu, Incheon 21565, Korea Tel: +82-32-460-3866 Fax: +82-32-460-2394 E-mail: dhchung@gilhospital.com
© 2016 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- A 61-year-old woman was referred to surgery for incidentally found colonic polyps during a health examination. Physical examination revealed widespread eczematous skin lesion without pruritus in the perianal and vulvar area. Abdominopelvic computed tomography showed an approximately 4-cm-sized, soft tissue lesion in the right perianal area. Inguinal lymph node dissection and Mils’ operation extended to perianal and perivulvar skin was performed. Histologically, the anal canal lesion was composed of mucin-containing signet ring cells, which were similar to those found in Pagetoid skin lesions. It was diagnosed as an anal canal signet ring cell carcinoma (SRCC) with perianal and vulvar Pagetoid spread and bilateral inguinal lymph node metastasis. Anal canal SRCC is rare, and the current case is the third reported case in the English literature. Seven additional cases were retrieved from the world literature. Here, we describe this rare case of anal canal SRCC with perianal Pagetoid spread and provide a literature review.
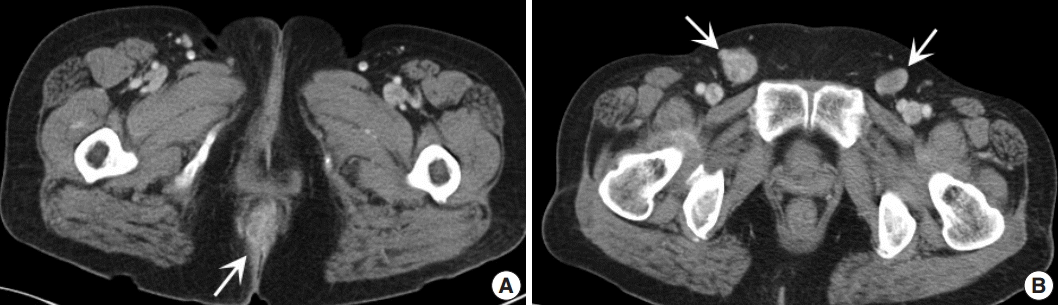
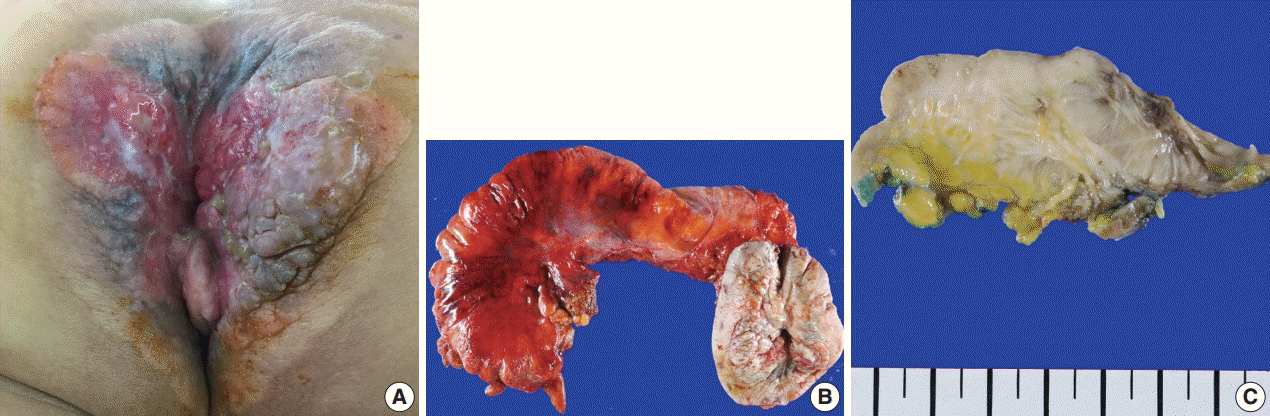
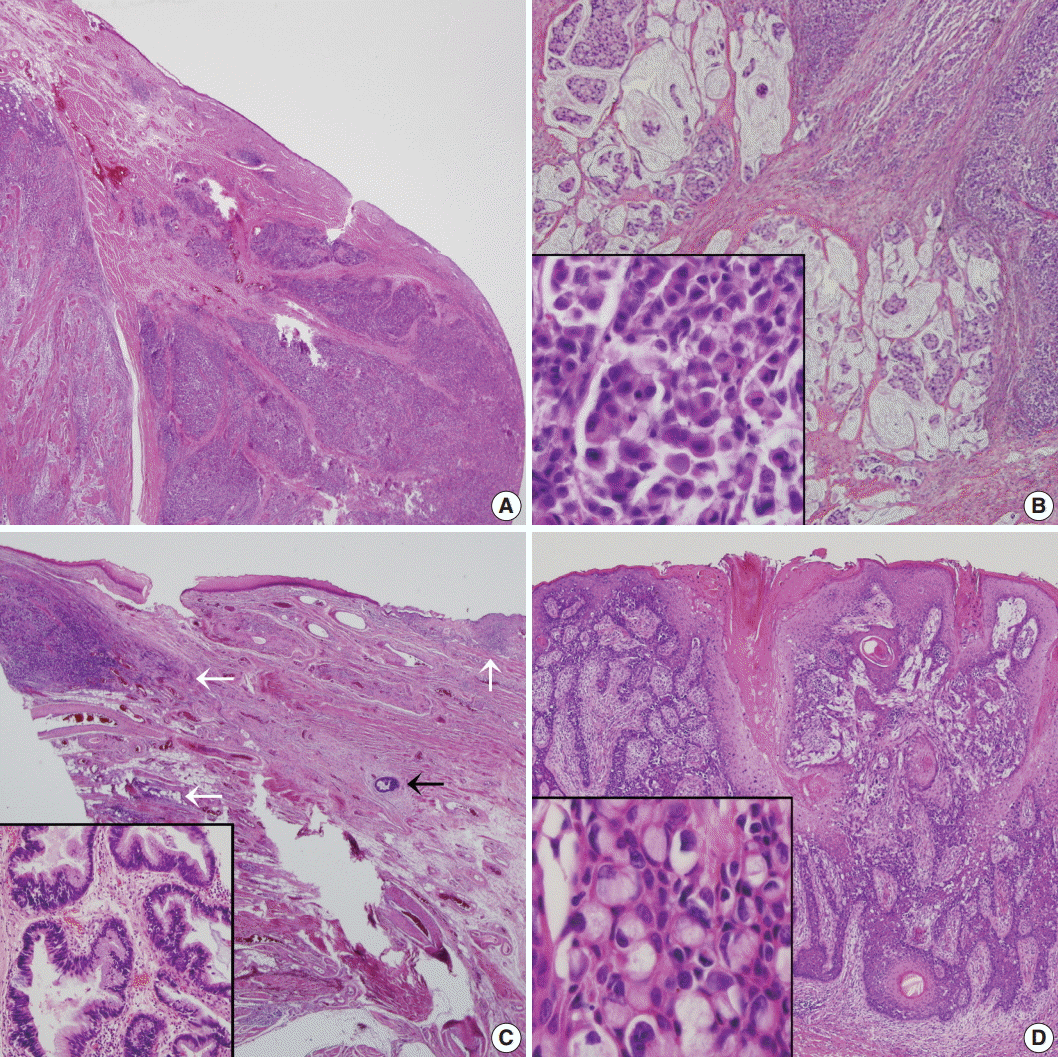
- A 61-year-old woman visited our hospital for alleged colon polyps found during a colonoscopy examination as part of a routine health examination. Endoscopic submucosal dissection was performed. Four polyps were diagnosed as tubular adenoma. Abdominopelvic computed tomography (CT) showed an approximately 4-cm-sized, well-enhancing soft tissue lesion in the right perianal area (Fig. 1A) and a well-enhancing enlarged lymph node in both inguinal areas, suggesting hypervascular tumor metastasis such as melanoma in those lymph nodes (Fig. 1B). Upon physical examination, however, a widespread erythematous lichenified skin lesion, measuring 12×9.7 cm, was noted in the perianal and vulvar area (Fig. 2A), which had developed over the previous 3 years according to the patient. She did not complain of pruritus. She had no history of anal fissure, fistula, or change in bowel habit. Skin biopsy was taken from the perianal and vulvar area, which showed infiltrating Pagetoid cells and SRCC cells. There was neither abnormal bowel wall thickening nor masses in the sigmoid colon. No other lesion was observed on upper esophagogastroscopy. The preoperative carcinoembryonic antigen (CEA) level was elevated to 39.75 ng/mL (reference, 0 to 5 ng/mL). Carbohydrate antigen 19-9 was within normal ranges. Inguinal lymph node dissection with an extended Miles’ operation was performed; traditional Miles’ operation with extended resection around bilateral labium majora, followed by skin reconstruction with gluteal flap, was performed. The resected specimen was composed of the anal canal and rectum with skin and soft tissue of the perianal region and vulva (Fig. 2B). Upon sectioning, the anal canal showed an ulcerative firm mass measuring 4.0×3.0×2.7 cm, which invaded the perianal sphincter muscle and subcutaneous fat of the anal skin (Fig. 2C). Light microscopy showed that the anal canal was totally replaced by singly scattered intracytoplasmic mucin-containing signet ring cells and some extracellular mucin (Fig. 3A, B). Focally, well-formed glands were also found in less than 5% of the tumor. Most of the surface epithelium was denuded. The focal residual transformation zone showed a focus of adenocarcinoma in situ (Fig. 3C), and was not contiguous with the anal glands. Anal canal SRCC was diagnosed. The erythematous skin around the anus and labium majora showed linear infiltration of Pagetoid cells as well as infiltration of signet ring cells in the dermis and subcutis (Fig. 3D). Involved skin area measured 8.2×8.0×0.5 cm. Lymphatic permeation was noted in the dermis. Enlarged bilateral inguinal lymph nodes also showed infiltration of the signet ring cells.
- Immunohistochemically, both signet ring cells and Pagetoid cells were positive for cytokeratin (CK) 20, CEA, MOC-31, and CK19. Intracellular and extracellular mucin was eosinophilically stained with mucicarmine. Both signet ring cells and Paget’s cells were positive for epidermal growth factor receptor and weakly positive for CK5/6. They were negative for CK7, human melanoma black 45, S-100 protein, caudal-related homeobox gene nuclear transcription factor (CDX2), p53, synaptophysin, chromogranin, and CD56. These results are shown in Table 1.
- A mutation study of the KRAS gene (codon 12 and codon 13) was performed by means of the peptide nucleic acid-mediated real-time polymerase chain reaction clamping method using genomic DNA isolated from formalin fixed paraffin-embedded tissue. Mutations in codon 12 and 13 were not detected in anal canal SRCC.
- Microsatellite instability (MSI) was tested using five Bethesda markers (D2S123, D5S346, D17S250, BAT25, and BAT26). MSI-high (MSI-H) was defined if they differed in at least two of the five markers and MSI-low was defined if they differed in only one of the five markers. Anal canal mass was microsatellite stable (MSS).
- Positron emission tomography CT and chest CT showed no evidence of systemic metastases. The case was diagnosed as anal canal SRCC with Pagetoid spread in perianal and vulva skin, stage IIIB according to the TNM staging system of the American Joint Committee on Cancer.
- The patient was scheduled to undergo chemotherapy with mitomycin-C and 5-fluorouracil (5-FU) and subsequent radiotherapy. The patient’s postoperative course was unremarkable.
- Institutional review board (IRB) approval was obtained for this case report.
CASE REPORT
- Colorectal SRCC is a rare histologic subtype of adenocarcinoma, accounting for 0.1% to 2.4% of all colorectal malignancies [1]. Anal canal SRCC is even rarer; only two cases of anal canal SRCCs have been reported in the English literature [4,5]. Seven additional cases have been retrieved from the Japanese literature [3,7-12].
- The current case showed anal canal SRCC. Anal canal carcinoma is histologically and pathogenetically divided into squamous cell carcinoma, cloacogenic (basaloid or transitional cell) carcinoma and adenocarcinoma [13]. Anal canal squamous cell carcinoma originates from the non-keratinizing squamous epithelium below the dentate line of the anal canal, while anal canal adenocarcinoma is regarded as arising from the upper part lined by the columnar epithelium. Anal canal adenocarcinomas are rare. According to the World Health Organization classification, anal canal adenocarcinomas are subclassified according to adenocarcinoma arising from anal mucosa and extramucosa, and adenocarcinoma arising from anorectal fistula or adenocarcinoma of anal glands [14]. No description of in situ lesion was found in the previously reported cases of anal canal SRCCs. Based on the findings of the overlying ulcerated anal surface mucosa, remnants of anal gland adenocarcinoma in situ without continuity to SRCC portion, the present anal canal SRCC belongs to extramucosal perianal adenocarcinoma with wide Pagetoid spread along the perianal soft tissues and skin.
- We have summarized the clinicopathologic characteristics in Table 2. Similar to the current case, eight out of 10 anal canal SRCCs (80%) were accompanied by perianal or vulvar Pagetoid spread [3-5,7-12]. Extramammary perianal Paget’s disease is a rare and heterogeneous neoplasm, which is frequently combined with underlying hidden adenocarcinoma [6]. Immunohistochemical panels such as CK7, CK20, CEA, gross cystic disease fluid protein-15 (GCDFP-15), MUC1, MUC2, MUC5AC, and CDX2 have been used to distinguish primary from secondary Paget’s disease [15]. In anal SRCCs with Pagetoid spread, i.e., not primary extramammary Paget’s disease, the tumor cells were positive for CK20, MUC1, or MOC-31, while they were negative for GCDFP-15, and negative for CK7, whereas primary Paget’s disease is commonly positive for CK7 [16]. However, none of these were a specific and confirmative diagnostic clue; further accumulative data may be needed. SRCC is most commonly encountered in the stomach, but it can also arise in various organs, such as breast, urinary bladder, or colon. Immunohistochemical profiles for CK7 and CK20 have also been used to differentiate the sites of origin for SRCCs; CK20 is normally expressed in the gastrointestinal epithelium, urothelium, and in Merkel’s cells. While CK7 is uncommonly expressed in the lower gastrointestinal tract, it is commonly expressed in lung, ovary, endometrium, and breast [15].
- As the term “signetring cells” implies, the tumor cells have intracellular mucin vacuoles displacing the nucleus to one side of the cells scattered as single cells or in loose clusters. This is caused by disrupted cell-to-cell adhesion and diffuse spreading, and results in more frequent lymphovascular invasion and node metastasis of SRCC as compared with other mucinous adenocarcinoma.
- In view of the clinical aspects, colorectal SRCC has a very high mortality rate compared to mucinous carcinoma and nonmucinous colorectal adenocarcinomas. Even a minor signet-ring cell component, i.e., 50% or less, in colorectal carcinomas was independently associated with a high mortality rate, regardless of molecular or other clinicopathological factors [17]. Anal canal SRCC grows insidiously underneath the mucosa, and thus it may not be found before the late advanced stage. It is uncertain whether anal SRCCs have an adverse prognosis like that of colorectal SRCC [18]. Inguinal nodes should be examined for anal cancer staging. In a review of 10 cases of anal canal SRCCs, the ages ranged from 46 to 87 years (mean, 63.4 years) [3-5,7-12]. Two cases were proven as primary anal canal SRCCs by autopsy [7,9]. SRCC has a shorter patient survival. During the follow-up period, ranging from 4 to 25 months, death caused by lymphatic or peritoneal carcinomatosis or distant metastases occurred in 50% of cases (5/10) [3,5,7,9,12]. The five remaining cases were alive with a follow-up period ranging from 2 months to 6 years. None of the previous cases underwent moleculocytogenetic application. For the past decade, molecular prognostic markers in colorectal carcinoma have been studied; MSI-H is a well-established reliable predictive marker for chemotherapeutic efficacy in colorectal carcinoma. However, contrary to colorectal SRCCs that take an aggressive course, colorectal mucinous carcinoma takes a favorable course, although both are associated with MSI-H [1]. Data on anal canal SRCC concerning MSI-H as a predictive factor is lacking. The current case showed MSS, albeit it is only one case. Due to the rarity of anal canal SRCCs, the MSI status of anal canal SRCC relating to prognosis has not been determined. Further accumulative investigation is required.
- Traditional treatment of anal canal carcinoma is surgery including abdominopelvic resection with or without chemoradiotherapy [19]. Radiation therapy for anal canal carcinoma reaches a cure rate of up to 70% in selected low-stage patients. However, there is no general agreement regarding the treatment of anal canal SRCC due to the extremely rare incidence and limited data on cancer treatment, as shown in Table 2. In a case reported by Yoshitani et al. [11], a patient with distant metastatic foci achieved complete remission after chemotherapy using bevacizumab/mFOLFOX6, FOLFIRI. Ioannidis et al. [4] reported on a long-survived stage II patient with no recurrent tumor who underwent chemoradiotherapy using 5-FU for a 6-year follow-up period.
- In summary, anal canal SRCC is extremely rare. If there is perianal Paget’s disease raising the possibility of underlying hidden adenocarcinoma, careful evaluation is necessary for early diagnosis of anal canal SRCC.
DISCUSSION
| Case No. | Author | Age(yr)/Sex | Presenting symptom | Gross and histopathology | Immunohistochemistry | TNM at the initial evaluation | Treatment | Clinical outcome (follow-up period) |
|---|---|---|---|---|---|---|---|---|
| 1 | Uchigasaki et al. (1992) [3] | 52/F | Anal ulcer and pruritus for 2 yra | Anal canal SRCC progressed from adenocarcinoma in situ of anal glands or mucinous glands (1.5 cm), Pagetoid spread (7 cm) | pCEA+ mCEA+ mucicarmine+ PAS+ D-PAS-resistant | T1N3M0, stage IIIB | Topical 5-FU cream, EB, ASR | Recurrence with systemic metastasis and died (7 mo) |
| 2 | Morihisa et al. (1999) [7] b | 75/F | Perianal erythema and erosion for 7 mo | Anal canal SRCC (size not described), Pagetoid spread (5 cm) | CEA+ mucicarmine+ PAS+ | N3M1, stage IV | Nephrostomy for ARF due to systemic metastasis | Systemic metastasis and died (4 mo) |
| 3 | Nagano et al. (1999) [8] | 46/F | Inguinal LN enlargement | Anal canal and rectal SRCC (3.5 cm), Pagetoid spread (7.5 cm) | Alcian blue+ PAS+ | T2N3M0, stage IIIB | APR, CTX (5-FU, LV) | NR (3 mo) |
| 4 | Naganuma et al. (2004) [9] | 85/F | Anal bleeding for 3 yr | Anal canal SRCC (unknown size of polyp, residual anal canal mass, 5 mm), Pagetoid spread (size not described) | CEA+ CA19-9+ CK20+ CK7– GCDFP-15– | N3M1, stage IV | Anal polypectomy with CTX (5-FU), RT | Systemic metastasis and died (25 mo) |
| 5 | Nishimura et al. (2004) [10] b | 76/F | Anal erosion and etythema for 2 yr | Anal canal SRCC (size not described), Pagetoid spread (6 cm) | CK20+ GCDFP-15- | Not availableb | APR, CTX (5-FU, cisplatin) | NR (5 mo) |
| 6 | Yoshitani et al. (2009) [11] | 50/M | Axillary and inguinal LN enlargement for 1 mo, erythematous perianal skin | Anal canal SRCC (size not precisely described; anal verge to dentate line), Pagetoid spread (size not described) | CK20+MUC-1+CK7- | N3M1, stage IV | CTX (bevacizumab/mFOLFOX6, FOLFIRI) | Alive with CR evaluated by PET-CT (6 mo) |
| 7 | Ikezawa et al. (2010) [12] | 53/M | Anal pruritus for 22 mo | Anal canal SRCC (1 cm), Pagetoid spread (2 cm) | pCEA+ CK20+ CK7– GCDFP-15– | T1N2M0, stage IIIB | APR, CTX (uracil-tegafur, LV, mFOLFOX6) | Recurrence with systemic metastasis and died (9 mo) |
| 8 | Ioannidis et al. (2012) [4] | 87/F | Anal discomfortness and pain for 8 mo | Anal canal SRCC (3 cm) | Not done | T2N0M0, stage II | Wide excision with chemotherapy (5-FU) and RT | NR (6yr) |
| 9 | Terada (2014) [5] | 49/M | Anal bleeding | Anal canal SRCC (< 2 cm) | AE1/3+ CK8+ CK19+ CEA+ CA19-9+ synaptophysin+ CDX2+ MUC1+ MUC2+ EMA+ CK7– CK20– CK5/6– MUC5AC– MUC6– CD56– NSE– chromogranin– vimentin– | T1N1M0, stage IIIA | Miles’ operation with LN dissection | Metastatic carcinomatosis and died (5 mo) |
| 10 | Kim et al. (2016) | 61/F | Erythematous perianal and vulvar skin for 3 yr, synchronous colon adenocarcinoma (0.2 cm) | Anal canal SRCC (4 cm), Pagetoid spread (8.2 cm) | pCEA+ CK20+ CK19+ MOC-31+ CDX2– CK7– HMB45– mucicarmine+ | T2N3M0, stage IIIB | Extended Miles’ operation and inguinal LN dissection, CTX (5-FU, MTC), RT (planned) | NR (2 mo) |
F, female; SRCC, signet ring cell carcinoma; pCEA, polyclonal carcinoembryonic antigen; mCEA, monoclonal carcinoembryonic antigen; PAS, periodic acid-Schiff; D-PAS, PAS diastase stain; 5-FU, 5-fluorouracil; EB, electron beam therapy; ASR, abdominosacral resection; CEA, carcinoembryonic antigen; ARF, acute renal failure; LN, lymph node; APR, abdominoperineal resection; CTX, chemotherapy; LV, leucovorin; NR, no recurrence; CA19-9, carbohydrate antigen 19-9; CK, cytokeratin; GCDFP-15, gross cystic disease fluid protein-15; RT, radiotherapy; M, male; mFOLFOX6, leucovorin, 5-FU, and oxaliplatin; FOLFIRI, irinotecan, 5-FU, and leucovorin; CR, complete remission; PET-CT, positron emission tomography-computed tomography; EMA, epithelial membrane antigen; NSE, neuron specific enolase; HMB-45, human melanoma black 45; MTC, mitomycin-C.
aThis case was diagnosed as Paget’s disease 15 years before the anal canal SRCC;
bThese articles were not available.
- 1. Inamura K, Yamauchi M, Nishihara R, et al. Prognostic significance and molecular features of signet-ring cell and mucinous components in colorectal carcinoma. Ann Surg Oncol 2015; 22: 1226-35. ArticlePubMedPMCPDF
- 2. Chu PG, Weiss LM. Immunohistochemical characterization of signet-ring cell carcinomas of the stomach, breast, and colon. Am J Clin Pathol 2004; 121: 884-92. ArticlePubMed
- 3. Uchigasaki S, Ochiai T, Miyakawa K, Suzuki H, Morishima T. Circumanal Paget’s disease which was complicated by anus cancers after 15 years. Skin Cancer 1992; 7: 254-8. Article
- 4. Ioannidis O, Papaemmanouil S, Paraskevas G, et al. Primary signet ring cell anal adenocarcinoma. J Gastrointest Cancer 2012; 43 Suppl 1: 168-70. ArticlePubMedPMCPDF
- 5. Terada T. Primary pure signet-ring cell carcinoma of the anus: a case report with immunohistochemical study. Endoscopy 2014; 46 Suppl 1 UCTN: E347. Article
- 6. Lopes Filho LL, Lopes IM, Lopes LR, Enokihara MM, Michalany AO, Matsunaga N. Mammary and extramammary Paget’s disease. An Bras Dermatol 2015; 90: 225-31. ArticlePubMedPMC
- 7. Morihisa A, Nobutoh T, Kohda M, Ueki H. An autopsy case of anal canal cancer showing Paget’s disease as the first sign. Jpn J Clin Dermatol 1999; 53: 758-60.
- 8. Nagano Y, Nanko M, Nagahori Y. Rectal signet ring cell carcinoma associated with perianal Paget’s disease: report of a case. J Jpn Surg Assoc 1999; 60: 473-6. Article
- 9. Naganuma H, Ottomo S, Takaya K, Sakai N, Tamahashi N, Takahashi T. An autopsy case of anal canal cancer (signet ring cell carcinoma) accompanied with perianal Pagetoid spread. Jpn J Diagn Pathol 2004; 21: 129-32.
- 10. Nishimura H, Kaneko T, Mizuki D, et al. A case of anal canal cancer presenting a pagetoid phenomenon in the perianal lesion. Jpn J Clin Dermatol 2004; 58: 674-7.
- 11. Yoshitani S, Hosokawa K, Yokoi M, et al. A case of anal canal carcinoma with systemic lymph node metastases successfully treated by bevacizumab+mFOLFOX6 therapy. Gan To Kagaku Ryoho 2009; 36: 2229-31. PubMed
- 12. Ikezawa F, Miura K, Shibata C, et al. Pagetoid spread with anal canal poorly differentiated adenocarcinoma: a case report. Jpn J Gastroenterol Surg 2010; 43: 678-84. Article
- 13. Moritani Y, Tomioka N, Izumi A, Takiue T, Kobayashi N, Shirakawa Y. A case of anal gland carcinoma accompanied by Pagetoid spread. Nippon Daicho Komonbyo Gakkai Zasshi 2009; 62: 121-6. Article
- 14. Welton ML, Lambert R, Bosman FT. Tumours of the anal canal. In: Bosman FT, Carneiro F, Hruban RH, Theise ND, eds. WHO classification of tumours of the digestive system. 4th ed. Lyon: IARC Press, 2010; 183-93.
- 15. Grelck KW, Nowak MA, Doval M. Signet ring cell perianal paget disease: loss of MUC2 expression and loss of signet ring cell morphology associated with invasive disease. Am J Dermatopathol 2011; 33: 616-20. ArticlePubMed
- 16. Ohnishi T, Watanabe S. The use of cytokeratins 7 and 20 in the diagnosis of primary and secondary extramammary Paget’s disease. Br J Dermatol 2000; 142: 243-7. ArticlePubMed
- 17. Ogino S, Nosho K, Kirkner GJ, et al. CpG island methylator phenotype, microsatellite instability, BRAF mutation and clinical outcome in colon cancer. Gut 2009; 58: 90-6. ArticlePubMedPMC
- 18. Kang H, O’Connell JB, Maggard MA, Sack J, Ko CY. A 10-year outcomes evaluation of mucinous and signet-ring cell carcinoma of the colon and rectum. Dis Colon Rectum 2005; 48: 1161-8. ArticlePubMed
- 19. Shridhar R, Shibata D, Chan E, Thomas CR. Anal cancer: current standards in care and recent changes in practice. CA Cancer J Clin 2015; 65: 139-62. ArticlePubMedPDF
REFERENCES
Figure & Data
References
Citations

- Primary Carcinomas of the Episiotomy Scar Site: A Systematic Literature Review
Andrea Palicelli, Federica Torricelli, Gabriele Tonni, Alessandra Bisagni, Eleonora Zanetti, Magda Zanelli, Venus Damaris Medina-Illueca, Beatrice Melli, Maurizio Zizzo, Andrea Morini, Maria Paola Bonasoni, Giacomo Santandrea, Giuseppe Broggi, Rosario Cal
Current Oncology.2025; 32(2): 65. CrossRef - Metastatic Carcinomas at the Episiotomy Site: A Systematic Literature Review
Andrea Palicelli, Gabriele Tonni, Federica Torricelli, Beatrice Melli, Vincenza Ylenia Cusenza, Sandra Martinelli, Eleonora Zanetti, Alessandra Bisagni, Magda Zanelli, Maria Paola Bonasoni, Teresa Rossi, Lucia Mangone, Venus Damaris Medina-Illueca, Mauriz
Cancers.2025; 17(17): 2801. CrossRef - A Case of Prostatic Signet-Ring Cell-like Carcinoma with Pagetoid Spread and Intraductal Carcinoma and Long-Term Survival: PD-L1 and Mismatch Repair System Proteins (MMR) Immunohistochemical Evaluation with Systematic Literature Review
Nektarios Koufopoulos, Argyro-Ioanna Ieronimaki, Andriani Zacharatou, Alina Roxana Gouloumis, Danai Leventakou, Ioannis Boutas, Dionysios T. Dimas, Adamantia Kontogeorgi, Kyparissia Sitara, Lubna Khaldi, Magda Zanelli, Andrea Palicelli
Journal of Personalized Medicine.2023; 13(6): 1016. CrossRef - Anal canal adenocarcinoma with neuroendocrine features accompanying secondary extramammary Paget disease, successfully treated with modified FOLFOX6: a case report
Masamichi Yamaura, Takeshi Yamada, Rei Watanabe, Hitomi Kawai, Suguru Hirose, Hiroki Tajima, Masashi Sato, Yuichi Uchida, Daisuke Suganuma, Yoshiyuki Yamamoto, Toshikazu Moriwaki, Ichinosuke Hyodo
BMC Cancer.2018;[Epub] CrossRef - Solitary left axillary lymph node metastasis after curative resection of carcinoma at the colostomy site: a case report
Ken Imaizumi, Shigenori Homma, Tadashi Yoshida, Tatsushi Shimokuni, Hideyasu Sakihama, Norihiko Takahashi, Hideki Kawamura, Emi Takakuwa, Akinobu Taketomi
Surgical Case Reports.2016;[Epub] CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
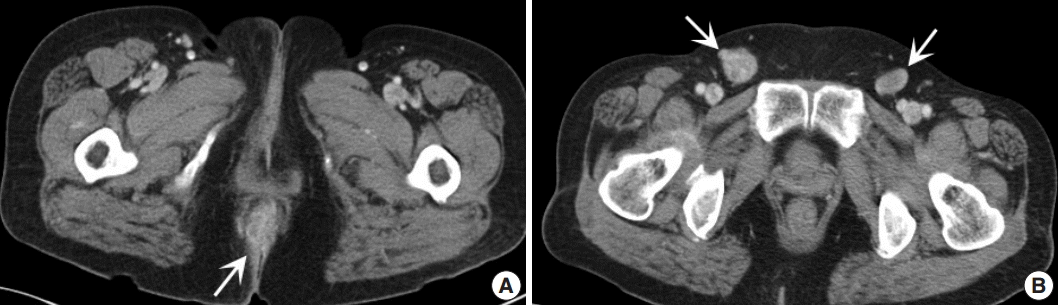
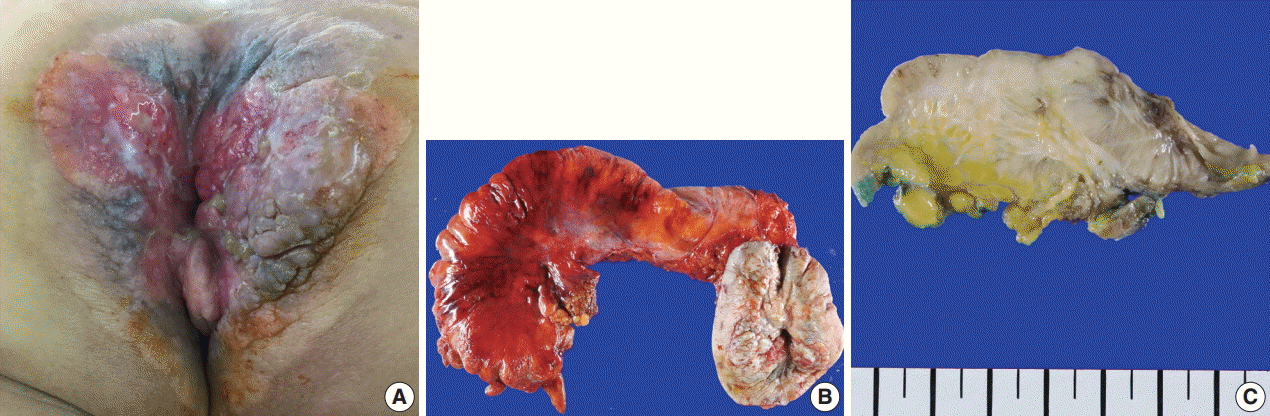
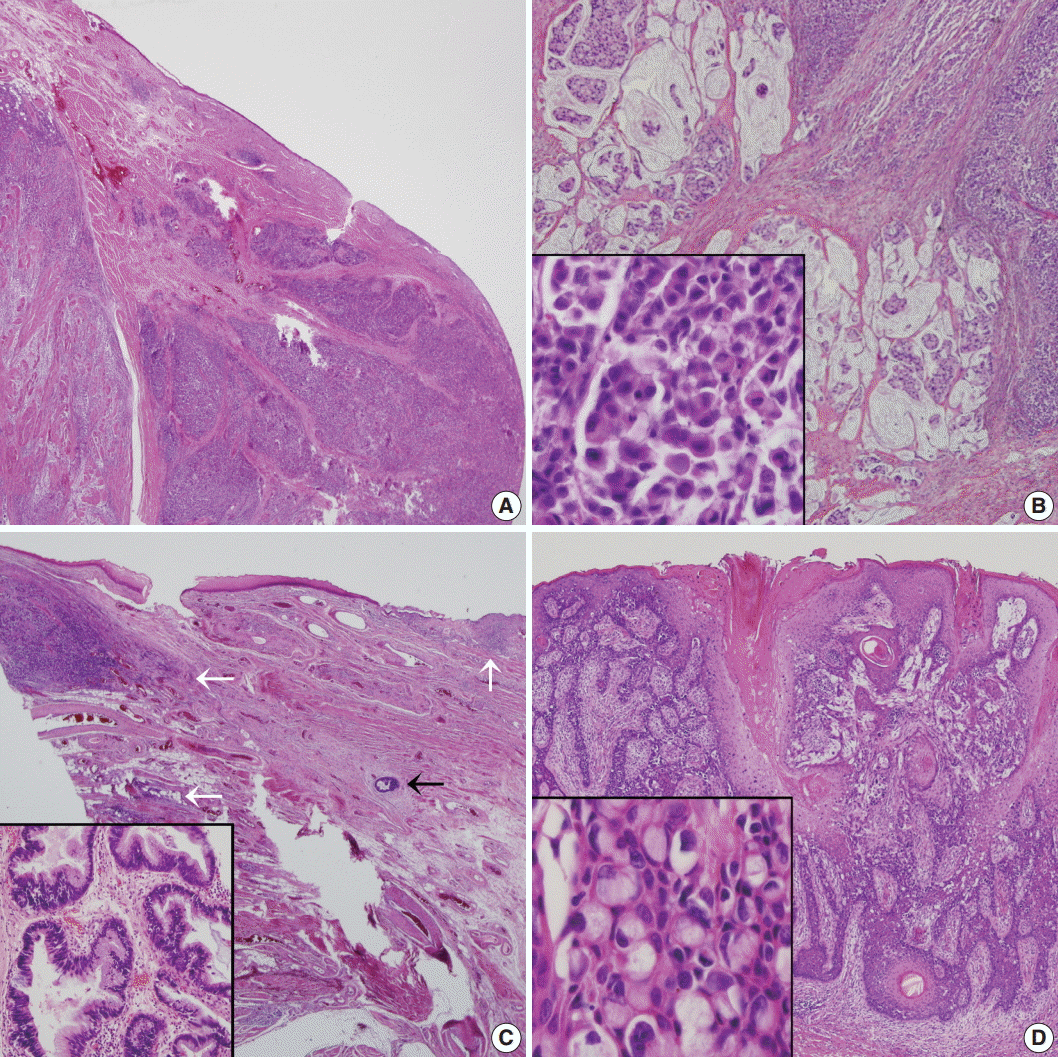
Fig. 1.
Fig. 2.
Fig. 3.
| Clone, dilution, company | Anal canal signet ring cell carcinoma | Pagetoid spread of vulvar and perianal skin | |
|---|---|---|---|
| CK7 | OV-TL 12/30; 1:100 dilution; Dako, Glostrup, Denmark | – | – |
| CK20 | Ks20.8; 1:100 dilution; Dako, Glostrup, Denmark | + | + |
| CK19 | A53-B/A2.26; 1:200 dilution; Thermo Scientific, Fremont, USA | + | + |
| CK5/6 | D5/16 B4; prediluted; Dako, Glostrup, Denmark | + weak | – |
| EGFR | SP84; 1:30 dilution; Novocastra, Newcastle upon Tyne, UK | + | + |
| CEA | Polyclonal; prediluted; Dako, Glostrup, Denmark | + | + |
| MOC-31 | MOC-31; 1:70 dilution; Novocastra, Newcastle upon Tyne, UK | + | + |
| CDX2 | AMT28; 1:50 dilution; Novocastra, Newcastle upon Tyne, UK | – | – |
| S-100 protein | Polyclonal; 1:1,200 dilution; Dako, Glostrup, Denmark | – | – |
| Synaptophysin | DAK-SYNAP; prediluted; Dako, Glostrup, Denmark | – | – |
| Chromogranin | polyclonal; prediluted; Dako, Glostrup, Denmark | – | – |
| CD56 | 123C3; prediluted; Dako, Glostrup, Denmark | – | – |
| HMB45 | HMB45; prediluted; Dako, Glostrup, Denmark | – | – |
| p53 | DO-7; 1:100 dilution; Dako, Glostrup, Denmark | + | + |
| Case No. | Author | Age(yr)/Sex | Presenting symptom | Gross and histopathology | Immunohistochemistry | TNM at the initial evaluation | Treatment | Clinical outcome (follow-up period) |
|---|---|---|---|---|---|---|---|---|
| 1 | Uchigasaki et al. (1992) [3] | 52/F | Anal ulcer and pruritus for 2 yr |
Anal canal SRCC progressed from adenocarcinoma in situ of anal glands or mucinous glands (1.5 cm), Pagetoid spread (7 cm) | pCEA+ mCEA+ mucicarmine+ PAS+ D-PAS-resistant | T1N3M0, stage IIIB | Topical 5-FU cream, EB, ASR | Recurrence with systemic metastasis and died (7 mo) |
| 2 | Morihisa et al. (1999) [7] |
75/F | Perianal erythema and erosion for 7 mo | Anal canal SRCC (size not described), Pagetoid spread (5 cm) | CEA+ mucicarmine+ PAS+ | N3M1, stage IV | Nephrostomy for ARF due to systemic metastasis | Systemic metastasis and died (4 mo) |
| 3 | Nagano et al. (1999) [8] | 46/F | Inguinal LN enlargement | Anal canal and rectal SRCC (3.5 cm), Pagetoid spread (7.5 cm) | Alcian blue+ PAS+ | T2N3M0, stage IIIB | APR, CTX (5-FU, LV) | NR (3 mo) |
| 4 | Naganuma et al. (2004) [9] | 85/F | Anal bleeding for 3 yr | Anal canal SRCC (unknown size of polyp, residual anal canal mass, 5 mm), Pagetoid spread (size not described) | CEA+ CA19-9+ CK20+ CK7– GCDFP-15– | N3M1, stage IV | Anal polypectomy with CTX (5-FU), RT | Systemic metastasis and died (25 mo) |
| 5 | Nishimura et al. (2004) [10] |
76/F | Anal erosion and etythema for 2 yr | Anal canal SRCC (size not described), Pagetoid spread (6 cm) | CK20+ GCDFP-15- | Not available |
APR, CTX (5-FU, cisplatin) | NR (5 mo) |
| 6 | Yoshitani et al. (2009) [11] | 50/M | Axillary and inguinal LN enlargement for 1 mo, erythematous perianal skin | Anal canal SRCC (size not precisely described; anal verge to dentate line), Pagetoid spread (size not described) | CK20+MUC-1+CK7- | N3M1, stage IV | CTX (bevacizumab/mFOLFOX6, FOLFIRI) | Alive with CR evaluated by PET-CT (6 mo) |
| 7 | Ikezawa et al. (2010) [12] | 53/M | Anal pruritus for 22 mo | Anal canal SRCC (1 cm), Pagetoid spread (2 cm) | pCEA+ CK20+ CK7– GCDFP-15– | T1N2M0, stage IIIB | APR, CTX (uracil-tegafur, LV, mFOLFOX6) | Recurrence with systemic metastasis and died (9 mo) |
| 8 | Ioannidis et al. (2012) [4] | 87/F | Anal discomfortness and pain for 8 mo | Anal canal SRCC (3 cm) | Not done | T2N0M0, stage II | Wide excision with chemotherapy (5-FU) and RT | NR (6yr) |
| 9 | Terada (2014) [5] | 49/M | Anal bleeding | Anal canal SRCC (< 2 cm) | AE1/3+ CK8+ CK19+ CEA+ CA19-9+ synaptophysin+ CDX2+ MUC1+ MUC2+ EMA+ CK7– CK20– CK5/6– MUC5AC– MUC6– CD56– NSE– chromogranin– vimentin– | T1N1M0, stage IIIA | Miles’ operation with LN dissection | Metastatic carcinomatosis and died (5 mo) |
| 10 | Kim et al. (2016) | 61/F | Erythematous perianal and vulvar skin for 3 yr, synchronous colon adenocarcinoma (0.2 cm) | Anal canal SRCC (4 cm), Pagetoid spread (8.2 cm) | pCEA+ CK20+ CK19+ MOC-31+ CDX2– CK7– HMB45– mucicarmine+ | T2N3M0, stage IIIB | Extended Miles’ operation and inguinal LN dissection, CTX (5-FU, MTC), RT (planned) | NR (2 mo) |
CK, cytokeratin; EGFR, epidermal growth factor receptor; CEA, carcinoembryonic antigen; CDX2, caudal-related homeobox gene nuclear transcription factor; HMB45, human melanoma black 45.
F, female; SRCC, signet ring cell carcinoma; pCEA, polyclonal carcinoembryonic antigen; mCEA, monoclonal carcinoembryonic antigen; PAS, periodic acid-Schiff; D-PAS, PAS diastase stain; 5-FU, 5-fluorouracil; EB, electron beam therapy; ASR, abdominosacral resection; CEA, carcinoembryonic antigen; ARF, acute renal failure; LN, lymph node; APR, abdominoperineal resection; CTX, chemotherapy; LV, leucovorin; NR, no recurrence; CA19-9, carbohydrate antigen 19-9; CK, cytokeratin; GCDFP-15, gross cystic disease fluid protein-15; RT, radiotherapy; M, male; mFOLFOX6, leucovorin, 5-FU, and oxaliplatin; FOLFIRI, irinotecan, 5-FU, and leucovorin; CR, complete remission; PET-CT, positron emission tomography-computed tomography; EMA, epithelial membrane antigen; NSE, neuron specific enolase; HMB-45, human melanoma black 45; MTC, mitomycin-C. This case was diagnosed as Paget’s disease 15 years before the anal canal SRCC; These articles were not available.

 E-submission
E-submission





