Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 49(2); 2015 > Article
-
Brief Case Report
Retiform Hemangioendothelioma of the Neck - Chin-Lung Kuo1,2,3,4, Paul Chih-Hsueh Chen5,6, Wing-Yin Li5,6, Pen-Yuan Chu1,3
-
Journal of Pathology and Translational Medicine 2015;49(2):171-173.
DOI: https://doi.org/10.4132/jptm.2013.10.14
Published online: March 12, 2015
1Department of Otorhinolaryngology-Head and Neck Surgery, Taipei Veterans General Hospital, Taipei, Taiwan
2Department of Otorhinolaryngology, Taoyuan Armed Forces General Hospital, Taoyuan, Taiwan
3Department of Otorhinolaryngology, National Yang-Ming University, Taipei, Taiwan
4Institute of Brain Science, National Yang-Ming University, Taipei, Taiwan
5Department of Pathology and Laboratory Medicine, Taipei Veterans General Hospital, Taipei, Taiwan
6Department of Pathology, National Yang-Ming University School of Medicine, Taipei, Taiwan
- Corresponding Author: Pen-Yuan Chu, M.D. Department of Otorhinolaryngology-Head and Neck Surgery, Taipei Veterans General Hospital, 201, Sec 2, Shih-Pai Road, Taipei 112, Taiwan Tel: +886-2-2875-7337, Fax: +886-2-2875-7338, E-mail: pychu@vghtpe.gov.tw
© 2015 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
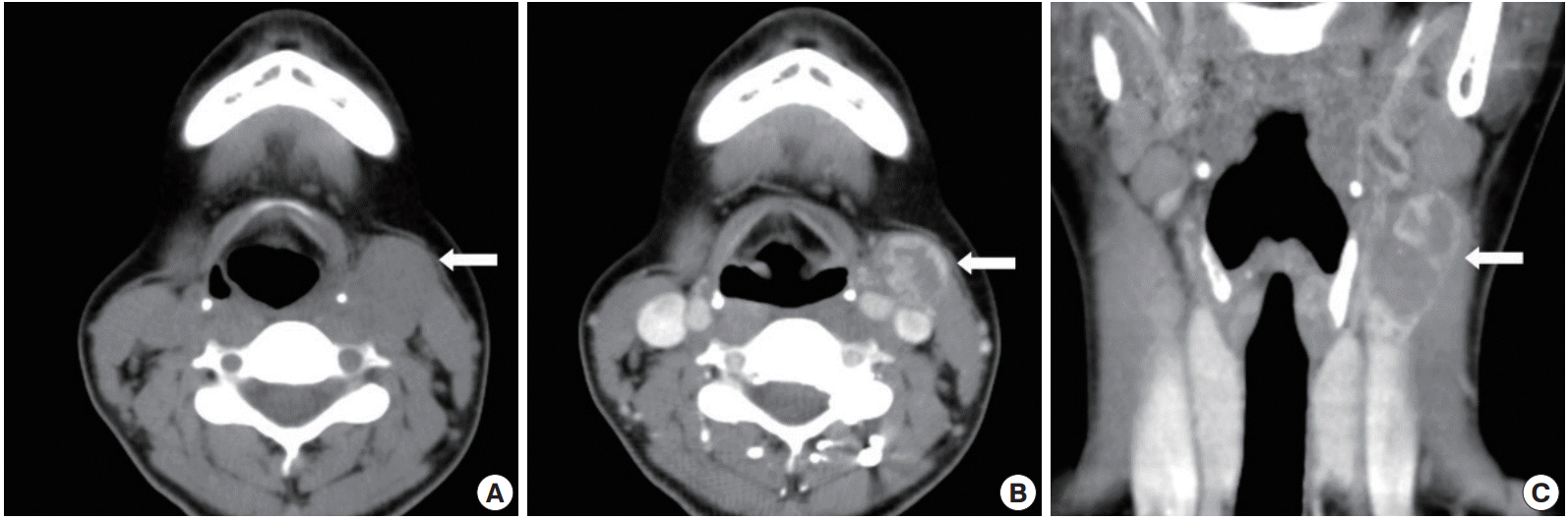
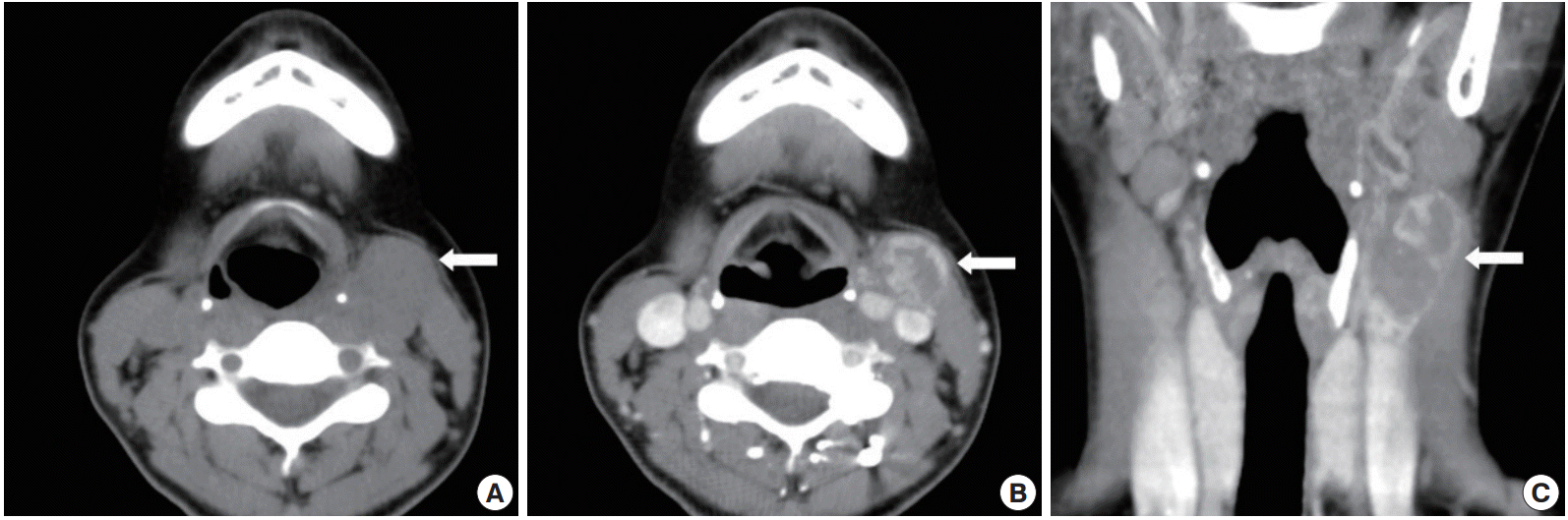
- A previously healthy 27-year-old woman presented to the out-patient department of our institution in February 2012 with a painless mass on the left side of the neck, which had increased in size over the course of one year. She had no constitutional symptoms such as weight loss, fever, or decreased appetite, and the rest of her medical history was also unremarkable. Physical examination revealed a 4×3-cm mass occupying the left side of the neck at the level III lymph node region. On palpation, the lesion was non-tender, hard, movable, and showed well-defined and smooth borders. The overlying skin was normal. Cervical lymph node enlargement was not observed, and the remainder of the otolaryngopharyngeal examination was unremarkable. Ultrasound-guided fine-needle aspiration was performed, and the cytology results were negative for malignancy. Computed tomography of the neck showed a well-defined heterogeneous mass with prominent ring enhancement occupying the left side of the neck at the level III lymph node region (Fig. 1). Surgical excision revealed a well-encapsulated and hypervascular tumor within the left anterior neck triangle.
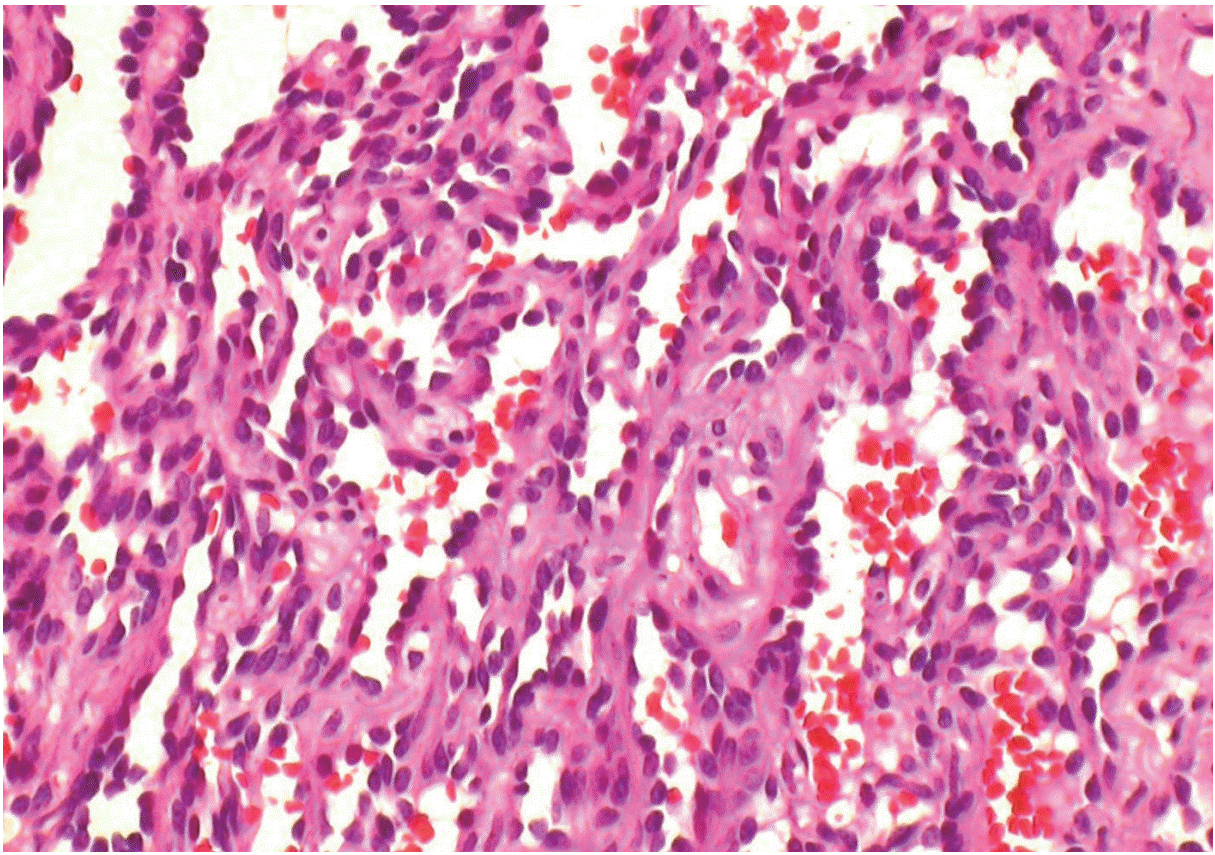
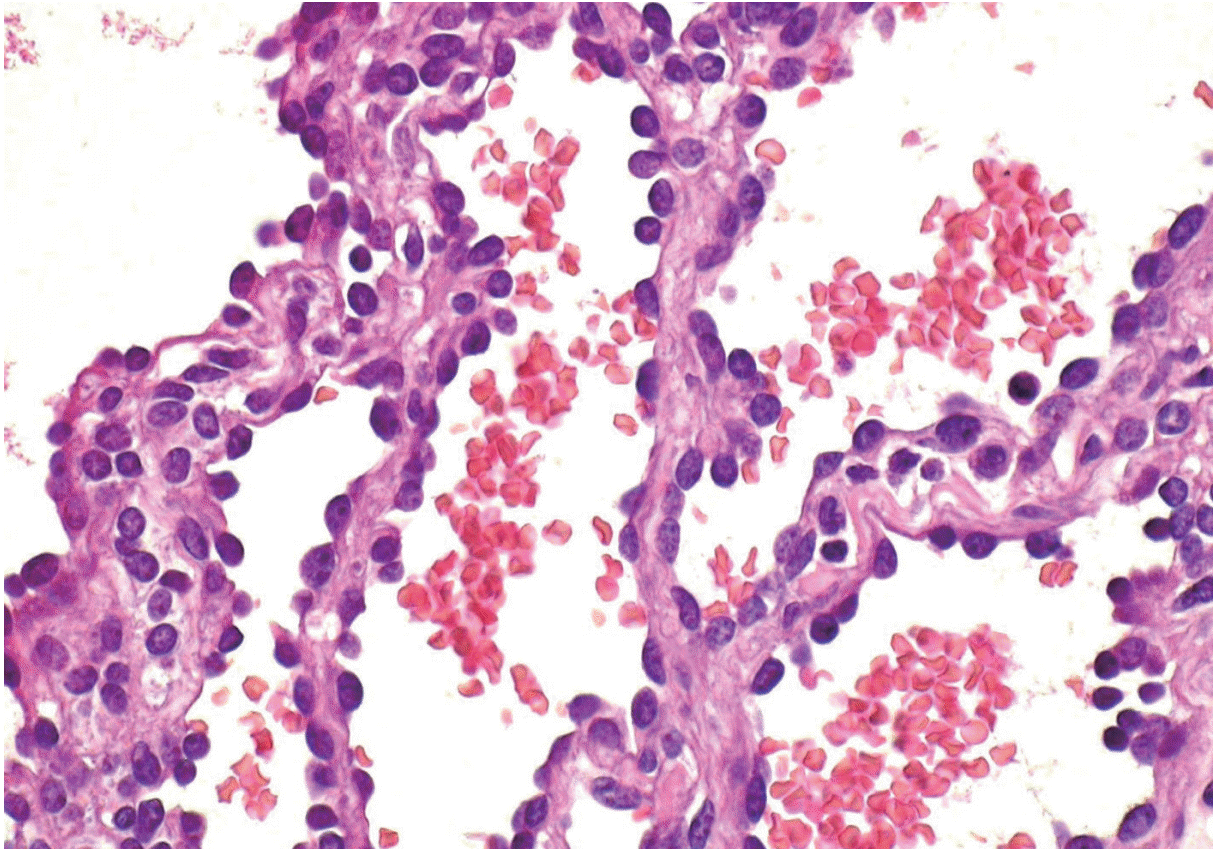
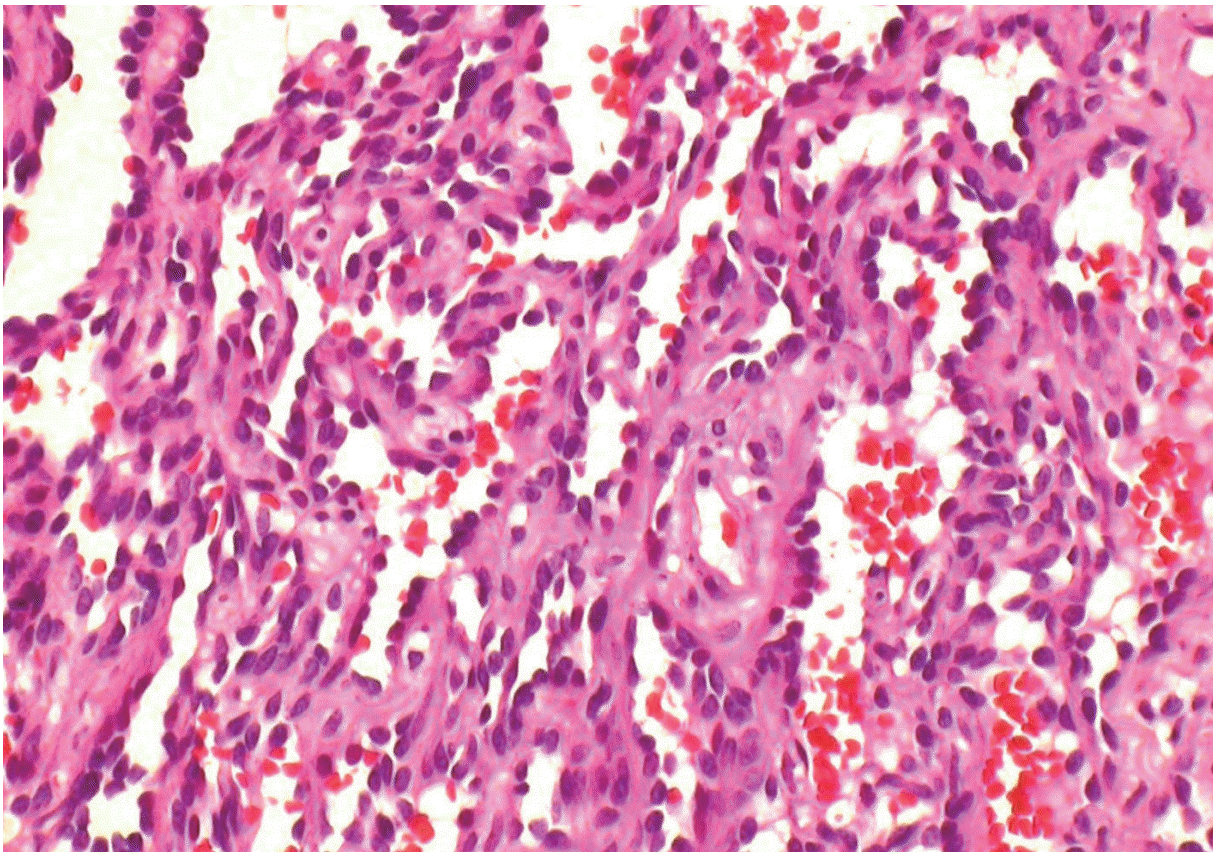
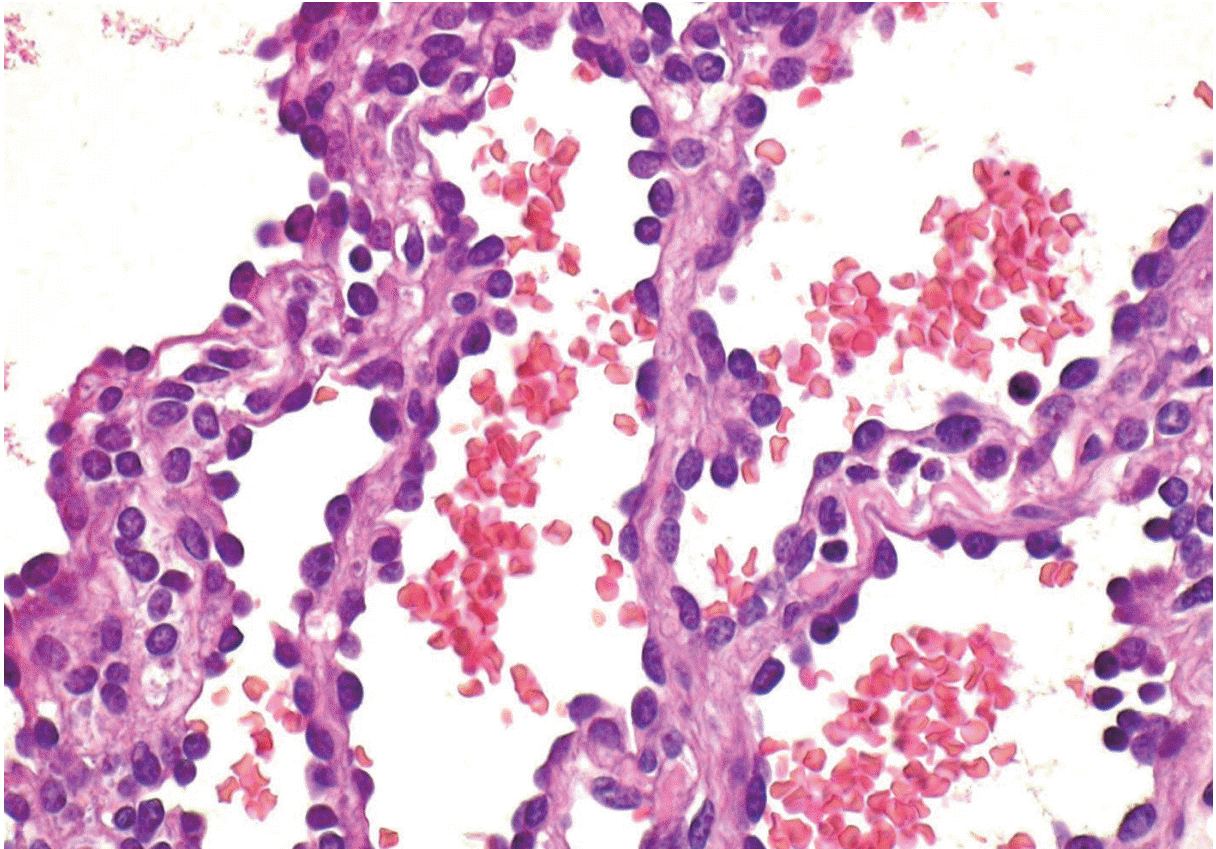
- Histologic examination revealed a 3.8×2×2-cm solid tumor with clear surgical margins. The tumor was composed of vascular structures containing elongated and narrow arborizing vascular channels simulating the structure of a rete testis (Fig. 2). The vascular channels were lined with hobnail endothelial cells (Fig. 3), which demonstrated rare mitosis. The tumor cells were immune-reactive for CD31 and friend leukemia virus integration 1 (Fli-1), but not for smooth muscle actin. Based on these histopathologic and immunohistochemical features, a diagnosis of RH was reached.
- After discussion with the head and neck oncology multidisciplinary team, a regular follow-up program was scheduled for the patient without postoperative radiation or chemotherapy, because of the clear surgical margins and rare mitosis observed, and to avoid the potential life-long complications associated with radiation therapy. No recurrence was evident at 1-year follow-up.
CASE REPORT
- To date, only 32 cases of RH have been reported in the English literature [4]. The etiology of this disease remains unclear; although cases related to previous epidermal malignant neoplasms, radiotherapy, human herpes virus type 8 and lymphedema have been reported, the exact associations have not been clearly established [2,4,5]. Disease duration from the time of diagnosis ranges from 2 months to several years [2,3,6]. Most patients present between the second to fourth decades of life (mean age, 36 years) [2]. There is a female predominance, with a female-to-male ratio of 2:1 [2,3,6]. RH can develop as either exophytic or plaque-like lesions with size ranging from 1 to 30 cm [3,6]. Most cases appear in the extremities, with a higher incidence in the lower limbs. Occasionally, this disease occurs in the trunk, scalp, pinna, and penis [2-5]. An RH presenting as a neck mass, as seen in this case, is unusual and has not been previously reported. The differential diagnoses generally include angiosarcoma, papillary intralymphatic angioendothelioma (PILA), hobnail hemangioma and Dabska-RH [3-5,7,8].
- The most important differential diagnosis of RH is angiosarcoma because of the therapeutic and prognostic considerations. Angiosarcoma is an aggressive neoplasm with a high mortality rate and a very high incidence of local recurrence and metastasis [6]. RH also exhibits frequent local recurrence, but rarely metastasizes [2,3], and no tumor-related deaths have been reported [6]. The vessels of angiosarcoma usually do not have a retiform appearance. Angiosarcoma is characterized by more nuclear atypia, conspicuous mitotic activity, tumor infiltration between individual collagen bundles, and multi-layering of endothelial cells without hobnail morphology [3,6]. PILA is another important differential diagnosis of RH. Although the histologic features of RH overlap with those of PILA, PILA lacks a retiform growth pattern, and is characterized by cavernous lymphangioma-like vascular spaces. Unlike RH, which typically occurs in the extremities, PILA shows no preference for a specific anatomic site [7]. Hobnail hemangioma can be confused with RH due to its similar hobnail endothelial cells. Hobnail hemangioma, however, lacks the complex retiform vessels and is a circumscribed tumor. Furthermore, the hobnail endothelial cells of hobnail hemangioma are seen in only the most superficial vessels. Collagen dissection is usually present, and an inflammatory infiltrate is not usually prominent [3]. Dabska-retiform hemangioendothelioma and RH are closely related tumors characterized by the presence of “hobnail”-type endothelial cells. Although RH typically appears in young to middle-age adults in the lower limbs and trunk, Dabska tumors have no particular pattern in terms of age or anatomical site. In addition, RH is considered to be a vascular tumor, while Dabska-RHs have a lymphatic endothelial phenotype [8].
- Cytologic or pathological examination reports may provide surgeons with appropriate guidelines for the treatment of patients, and the most compelling evidence in the differential diagnosis of RH is provided by immunohistochemistry, in which tumor cells react with endothelial markers (e.g., CD31, Fli-1, and factor VIII-related antigen) [2,5,9,10]. CD31 is generally regarded as the single best marker of endothelial cell differentiation because it is expressed in 90% of endothelial cell tumors, but very rarely in carcinomas, lymphomas, and mesotheliomas [9]. Factor VIII-related antigen has low sensitivity, and frequent presence of significant “background” due to staining of circulating antigen greatly limits the use of this marker [10]. Hence, the expression of this marker was not determined in this patient.
- There are differences between the present case and those in previous reports of RH in terms of disease location (neck vs mainly extremities), morphology (encapsulated mass vs mainly exophytic lesion), and histology (deep soft tissue vs mainly dermal involvement). These differences may expand the understanding of RH. In summary, there can be diagnostic pitfalls in general practice when managing neck masses, particularly an uncommon tumor in an unusual site. Awareness of this unique RH with uncertain malignant behavior is essential. In addition, as the prognosis and adjuvant therapy for RH and angiosarcoma are very different, it is important to distinguish them histopathologically.
DISCUSSION
Acknowledgments
- 1. Chu YC, Choi SJ, Park IS, Kim L, Han JY, Kim JM. Composite hemangioendothelioma: a case report. Korean J Pathol 2006; 40: 142-7.
- 2. Calonje E, Fletcher CD, Wilson-Jones E, Rosai J. Retiform hemangioendothelioma: a distinctive form of low-grade angiosarcoma delineated in a series of 15 cases. Am J Surg Pathol 1994; 18: 115-25. PubMed
- 3. Tan D, Kraybill W, Cheney RT, Khoury T. Retiform hemangioendothelioma: a case report and review of the literature. J Cutan Pathol 2005; 32: 634-7. ArticlePubMed
- 4. O’Duffy F, Timon C, Toner M. A rare angiosarcoma: retiform haemangioendothelioma. J Laryngol Otol 2012; 126: 200-2. ArticlePubMed
- 5. Ioannidou D, Panayiotides J, Krasagakis K, Stefanidou M, Manios A, Tosca A. Retiform hemangioendothelioma presenting as bruise-like plaque in an adult woman. Int J Dermatol 2006; 45: 53-5. ArticlePubMed
- 6. Hirsh AZ, Yan W, Wei L, Wernicke AG, Parashar B. Unresectable retiform hemangioendothelioma treated with external beam radiation therapy and chemotherapy: a case report and review of the literature. Sarcoma 2010; 2010: 756246.ArticlePubMedPMCPDF
- 7. Fanburg-Smith JC, Michal M, Partanen TA, Alitalo K, Miettinen M. Papillary intralymphatic angioendothelioma (PILA): a report of twelve cases of a distinctive vascular tumor with phenotypic features of lymphatic vessels. Am J Surg Pathol 1999; 23: 1004-10. PubMed
- 8. Yarmel D, Dormans JP, Pawel BR, Chang B. Recurrent pedal hobnail (Dabska-retiform) hemangioendothelioma with forefoot reconstructive surgery using a digital fillet flap. J Foot Ankle Surg 2008; 47: 487-93. ArticlePubMed
- 9. Pusztaszeri MP, Seelentag W, Bosman FT. Immunohistochemical expression of endothelial markers CD31, CD34, von Willebrand factor, and Fli-1 in normal human tissues. J Histochem Cytochem 2006; 54: 385-95. ArticlePubMedPDF
- 10. Billings SD, Folpe AL, Weiss SW. Epithelioid sarcoma-like hemangioendothelioma. Am J Surg Pathol 2003; 27: 48-57. ArticlePubMed
REFERENCES
Figure & Data
References
Citations

- Retiform hemangioendothelioma of the vulva: a case report
Hai-Jiao Wang, Zhi-Chen Ding, Xu Yan, Ying Yue
Clinical and Experimental Obstetrics & Gynecology.2021;[Epub] CrossRef - Atypical retiform hemangioendothelioma arising in a patient with Milroy disease: a case report and review of the literature
Aileen Grace P. Arriola, Laura A. Taylor, Eseosa Asemota, Markus D. Boos, David E. Elder, Kristy L. Weber, Robert G. Micheletti, Paul J. Zhang
Journal of Cutaneous Pathology.2017; 44(1): 98. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure

 E-submission
E-submission