Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 49(2); 2015 > Article
-
Original Article
The Diagnostic Usefulness of HMGA2, Survivin, CEACAM6, and SFN/14-3-3 δ in Follicular Thyroid Carcinoma - Min Hye Jang1,2, Kyeong Cheon Jung2, Hye Sook Min3
-
Journal of Pathology and Translational Medicine 2015;49(2):112-117.
DOI: https://doi.org/10.4132/jptm.2015.01.31
Published online: March 12, 2015
1Department of Pathology, Seoul National University Bundang Hospital, Seongnam, Korea
2Department of Pathology, Seoul National University College of Medicine, Seoul, Korea
3Department of Epidemiology and Preventive Medicine, Graduate School of Public Health, Seoul National University, Seoul, Korea
- Corresponding Author: Hye Sook Min, M.D. Department of Epidemiology and Preventive Medicine, Graduate School of Public Health, Seoul National University, 1 Gwanak-ro, Gwanak-gu, Seoul 151-742, Korea Tel: +82-2-880-2743 Fax: +82-2-762-9105 E-mail: lilloa@snu.ac.kr
© 2015 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background:
- Follicular thyroid carcinoma (FTC) is the second most common thyroid malignancy and its differential diagnosis includes follicular adenoma (FA) and adenomatous goiter (AG). Several ancillary markers have been suggested to aid in the diagnosis of FTC, but the successful use of these methods still needs to be validated.
-
Methods:
- In the present study, we verified the immunoexpression of HMGA2, CEACAM6, survivin, and SFN/14-3-3 δ in lesions including 41 AGs, 72 FAs, and 79 FTCs. We evaluated their diagnostic usefulness, combined with galectin 3, Hector Battifora mesothelial 1 (HBME1), cytokeratin 19, and cyclin D1, in diagnosing FTC.
-
Results:
- The expressions of HBME1 (65.8%) and HMGA2 (55.7%) were significantly higher in FTCs than in FAs and AGs (p<.001 and p=.005, respectively). HBME1 was the only marker that was more frequently expressed in FTCs than in FAs (p=.021) and it was more frequently expressed in follicular neoplasms than in AGs (p<.001). Among the novel markers, the combination of HMGA2 and HBME1 showed the highest sensitivity (72.2%) and specificity (76.1%) for diagnosing FTC. CEACAM6, survivin, and SFN/14-3-3 δ were barely expressed in most cases.
-
Conclusions:
- Our present results show that only HMGA2 can be beneficial in differentiating FTC using the novel markers.
- Tissue specimens and microarray construction
- Formalin-fixed, paraffin-embedded thyroid tissue blocks were retrieved from the archive maintained at the Department of Pathology, Seoul National University Hospital, from 2001 to 2013. A total of 192 cases of thyroid lesions consisting of 41 AGs, 72 FAs, and 79 FTCs were identified, and representative tissue blocks were obtained for all lesions. All cases were surgically resected. The hematoxylin and eosin–stained slides were reviewed in each case to confirm the original diagnosis using the strict criteria of capsular invasion and vascular invasion defined by two pathologists (M.H.J. and H.S.M.). AG cases were selected from the archives from 2001 to 2007, with identification of the follow-up records for confirming their benign nature.
- Tissue microarrays were constructed for immunohistochemical staining. A single, large tissue core (4.0 mm in diameter) was obtained from the most representative area of individual cases. Additionally, 74 cores of normal thyroid tissue from each matched thyroid lesion were included for negative controls. This study was approved by the Institutional Review Board of Seoul National University Hospital (E-1302-023-462).
- Immunohistochemical analyses
- Immunohistochemistry was performed on 4-µm-thick sections of tissue microarray blocks that included 192 surgicallyremoved samples. Tissue sections were deparaffinized and rehydrated following standard procedures. Heat-induced antigen retrieval was carried out and sections were incubated with primary antibodies for 32 minutes at 37°C at a dilution of 1:50 for HBME1 and cyclin D1, 1:100 for Gal-3 and SFN/14-3-3 δ, 1:200 for CK19, HMGA2 and CEACAM6, and 1:600 for survivin. Monoclonal antibodies were used for Gal-3 (clone 9C4, Novocastra, Newcastle, United Kingdom), HBME1 (clone HBME-1, Dako, Carpinteria, CA, USA), CK19 (clone RCK108, Dako), cyclin D1 (clone SP4, Thermo Fisher Scientific, Waltham, MA, USA), CEACAM6 (clone 9A6, Abcam, Cambridge, MA, USA) and SFN/14-3-3 δ (clone 5D7, Santacruz, Dalla, TX, USA). Polyclonal antibodies were used for HMGA2 (Biocheck, Foster city, CA, USA) and survivin (Novus Biologicals, Littleton, CO, USA). All immunohistochemical staining was carried out in a BenchMark XT autostainer (Ventana Medical Systems, Tucson, AZ, USA) using the DAB detection kit (Ventana Medical Systems).
- Immunohistochemical interpretation
- The immunohistochemical staining of tissue microarrays was evaluated by two pathologists (M.H.J. and H.S.M.). The immunoreactivity was scored for Gal-3, HBME1, CK19, cyclin D1, HMGA2, CEACAM6, survivin, and SFN/14-3-3 δ by categorizing methods based on the percentage of positive cells: 0 (less than 10%), 1 (10%–25%), 2 (26%–50%), and 3 (more than 50%). In Gal-3, CK19, survivin, and SFN/14-3-3 δ, cytoplasmic staining was considered as positive immunoreactivity. Membranous staining was regarded as positive for HBME1 and CEACAM6, and nuclear expression was regarded as positive for cyclin D1 and HMGA2.
- Statistical analysis
- The data was analyzed using SPSS ver. 21.0.0 for Windows (SPSS Inc., Chicago, IL, USA). The χ2 test or Fisher exact test was used to compare the expression of markers between different diagnostic subgroups. A p-value of <.05 was considered statistically significant. Sensitivity, specificity, and diagnostic accuracy were calculated using standard formulae for each marker individually, using histological diagnosis as the gold standard.
MATEIRALS AND METHODS
- Clinicopathologic features
- The whole series of samples was obtained from 36 males and 156 females, with a median age of 46 years (range, 9 to 76 years). The mean size of FAs and FTCs was 1.83 cm (range, 1.4 to 4.7 cm) and 3.7 cm (range, 0.8 to 7.3 cm), respectively. The FTC series included samples from 17 males and 61 females, with a median age of 42 years (range, 9 to 76 years). There were 19 cases with vascular invasion. There was only one case that metastasized to lung, and one case recurred in neck soft tissue 15 months after a total thyroidectomy. Unfortunately, we could not obtain the clinicopathologic information of the primary tumor of the metastatic FTC. The clinicopathologic characteristics of the 78 primary FTCs are summarized in Table 1.
- Immunohistochemical expressions of markers in AGs, FAs, and FTCs
- Most markers, including Gal-3, HBME1, cyclin D1, HMGA2, CEACAM6, survivin, and SNF/14-3-3 δ, were not expressed in the 74 normal thyroid tissues. On the contrary, CK19 was expressed as grade 1 (10%–25%) or 2 (26%–50%) in 31.1% of the normal thyroid tissues.
- In the AG group (n=41), most cases showed negative or limited immunoreactivity (less than grade 1, ≤25%) for all eight markers. Gal-3 was expressed in one case, but it showed relatively diffuse expression (grade 2, 26%–50%). HBME1, CK19, and cyclin D1 were expressed in only a few cases, showing grade 2 or 3 (grade 2: HBME1, 2/41; CK19, 3/41; cyclin D1, 3/41; grade 3: HBME1, 2/41; CK19, 0/41; cyclin D1, 0/41). However, HMGA2 was expressed in more cases, showing grade 2 (6/41) and grade 3 (4/41). All cases with expression of HBME1 (3/41) and HMGA2 (6/41) exhibited a characteristic feature of microfollicular proliferation of various extents, reminiscent of FN, and showed HBME1 and HMGA2 positivity in this area.
- In the FA group (n=72), HBME1, cyclin D1, and HMGA2 were expressed in more than 25% of tumor cells (grade 2: HBME1, 12/72; cyclin D1, 24/72; HMGA2, 12/72; grade 3: HBME1, 22/72; cyclin D1, 14/72; HMGA2, 18/72). There was no diffuse staining of CEACAM6, survivin, and SNF/14-3-3 δ. CK19 was expressed only in four cases, either as grade 2 (3/72) or grade 3 (1/72).
- The expression of HBME1 and HMGA2 was the highest in the FTC group, followed by that of cyclin D1, which was expressed at a similar frequency with the FA group. However, HBME1 and HMGA2 expression were not significantly different between FTCs without vascular invasion and FTCs with vascular invasion (p=.382 and p=.418, respectively). The frequency of CK19 expression was higher in the FTC group than in the FA group, but the difference was not statistically significant (p=.133). There was no diffuse staining of CEACAM6, survivin, or SNF/14-3-3 δ (Table 2).
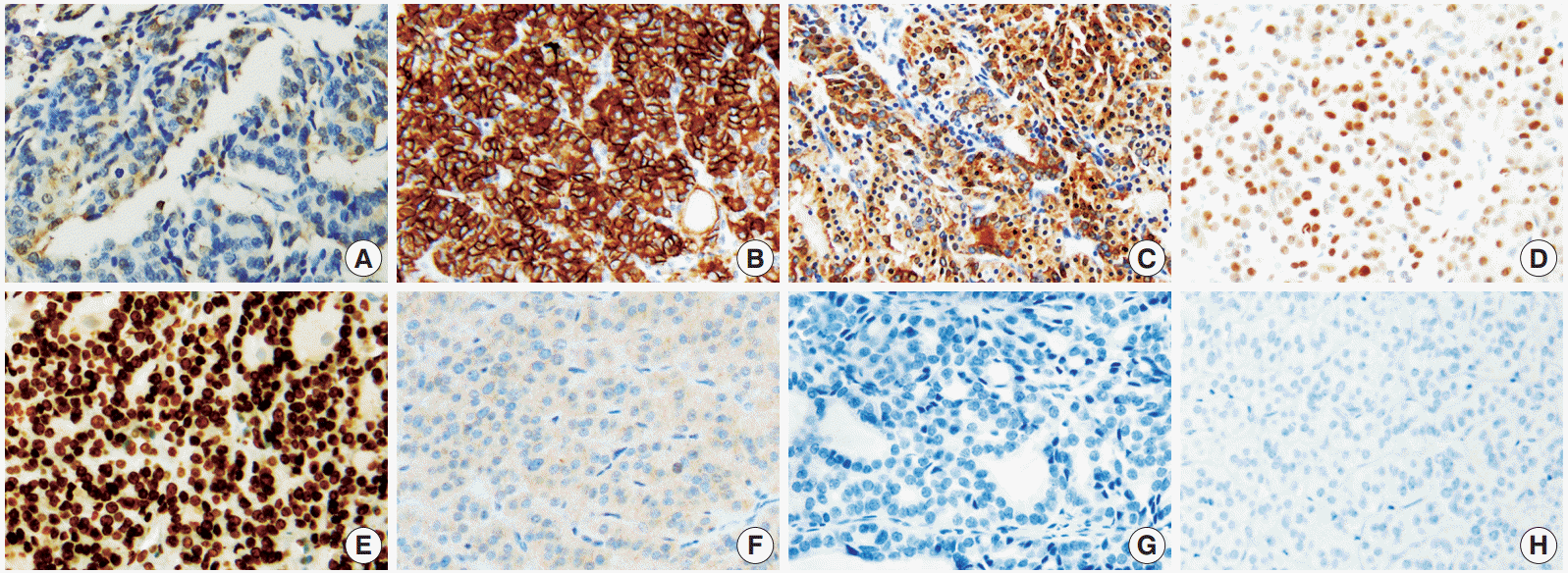
- Overall, among the novel markers, SFN/14-3-3 δ and CEACAM6 were not expressed in any of the subgroups, and survivin was only expressed in a small percentage of lesions (grade 0, less than 10%) in 14/97 FTCs. Therefore, these three markers were not helpful in distinguishing the diagnostic subgroups. In contrast, HMGA1 was significantly expressed in FTC and FA cases. The represented immunohistochemical expression of all markers is shown in Fig. 1.
- Diagnostic utilities of markers in differentiating FTC and FN
- Next, we compared the expression of each marker between the diagnostic subgroups. As survivin, CEACAM6 and SNF/14- 3-3 δ were expressed in only a few cases or were not expressed at all, they were excluded from the statistical analysis for the evaluation of diagnostic utilities.
- HBME1 was the only marker that showed differential expression frequency between FTC and FA (p=.021) (Table 2). However, it was only expressed in 52 of 79 FTCs (sensitivity, 65.8%) and its specificity remained as 52.8%. When comparing malignant and benign lesions (FTC vs FA and AG), the expression of HBME1 (p<.001) and HMGA2 (p=.005) was significantly different. In the diagnosis of malignancy, HBME1 showed a slightly better sensitivity (65.8%) and specificity (66.4%) than HMGA2 (sensitivity, 55.7%; specificity, 64.6%).
- Additionally, we calculated the sensitivity, specificity, and diagnostic accuracy of the combination of HBME1 and HMGA2 (Table 3). The combined expression of HBME1 or HMGA2 reached the highest sensitivity (72.2%), but the specificity (54.9%) and the diagnostic accuracy (62.0%) were similar or only slightly higher than those of the single markers. The simultaneous expression of HBME1 and HMGA2 increased the specificity up to 76.1%, but its sensitivity (49.4%) was poor.
- There were no differences when comparing the diagnostic utility of the combination of HBME1, cyclin D1 and HMGA2 with those of each single marker in neoplastic lesions (FN including FTC and FA vs AG) (Table 4). As a single marker, HBME1 showed the highest sensitivity (57.0%), and both HBME1 and cyclin D1 showed the highest specificity (90.2%). When more than one marker was expressed among HBME1, cyclin D1, and HMGA2, the sensitivity reached 80.9%, but the specificity decreased.
RESULTS
- Until now, the entire histologic examination of the fibrous capsule and vasculature after surgery has been the only way to precisely diagnose FTC. Thus, a preoperative diagnosis of FTC is needed in making an accurate preoperative plan and avoiding unnecessary surgery. In this study, we validated the diagnostic utility of HMGA2, CEACAM6, survivin, and SNF/14-3-3 δ with several known markers for distinguishing FTCs, expecting to find out a powerful diagnostic panel.
- HMGA2, CEACAM6, and SFN/14-3-3 δ were identified as promising molecular markers that were differentially expressed between benign and malignant thyroid tumors in a previous report by Prasad et al. [14]. In the immunohistochemical study, HMGA2 and SFN/14-3-3 δ were highly expressed in malignant tumors (HMGA2, p<.001, area under the curve [AUC]= 0.84; SFN/14-3-3 δ, p<.001, AUC=0.83). However, CEACAM6 did not show significantly different immunoreactivity [8]. Belge et al. [12] suggested that quantifying HMGA2 expression by reverse transcription polymerase chain reaction had a high potential to improve the diagnosis of FNs with a sensitivity of 95.9% and a specificity of 93.9%. In our study, the sensitivity and specificity of HMGA2 for FTC were 55.7% and 64.6%, respectively. However, CEACAM6 was only expressed in infiltrated inflammatory cells (Fig. 1). SFN/14-3-3 δ was only validated in 14 FTC cases in a previous study and thus, the results needed to be verified. Interestingly, its expression was specific for PTC [8,21]. Nevertheless, our study suggested that SFN/ 14-3-3 δ was not an applicable marker for FTC and FN.
- Haghpanah et al. [11] reported that the cytoplasmic expression of survivin was significantly higher in FTCs than in FAs (p<.005), with a high odds ratio (odds ratio, 21.4), but the number of cases was limited (11 FTC cases, 23 FA cases). Recently, Kim et al. [22] observed the immunoexpression of survivin in 13/57 FTCs but also in 21/58 FA cases. However, survivin was only expressed in a small proportion (<10%) of FTCs (14/ 97) in our study (Fig. 1).
- Among the well-known markers for PTC that we tested in this study (Gal-3, CK19, and HBME1), HBME1 was the only one expressed at significantly higher levels in the FTC group compared to other groups. The use of HBME1 as a marker of FTC is controversial, and its low specificity has not allowed for the differential diagnosis of FTCs in previous reports [23,24]. Our study yielded a similar result, showing that although the expression of HBME1 was significantly higher in the FTC group, it was expressed in almost half of FAs as well. However, it could differentiate FNs from AGs (p<.001), showing positivity in only 4/41 AG cases.
- Lastly, our results suggest that the combination use of HBME1 and HMGA2 can be beneficial in the differential diagnosis of FTC. Both markers showed significantly increased expression in FTCs when used alone. When either HBME1 or HMGA2 alone, or both HBME1 and HMGA2 were expressed in lesions, the sensitivity for detecting FTC reached 72.7%. When both markers were simultaneously positive, the specificity reached 76.1%. Therefore, the concurrent use of HBME1 and HMGA2 may be more beneficial than the single use, but it requires a more sophisticated interpretation for FTC diagnosis.
- In summary, among all the novel immunohistochemical markers that we tested, HMGA2 was expressed at a higher level in FTCs than in FAs or AGs, but its overall sensitivity was slightly lower than that of HBME1. However, the combination of HMGA2 and HBME1 may be beneficial in differentiating FTCs, as it increased the sensitivity and the specificity for FTCs. Although survivin, CEACAM6, and SFN/14-3-3 δ were initially promising in differentiating malignancy, our results showed that only HMGA2 could help in the diagnosis of FTC.
DISCUSSION
- 1. Kapur U, Wojcik EM. Follicular neoplasm of the thyroid: vanishing cytologic diagnosis? Diagn Cytopathol 2007; 35: 525-8. ArticlePubMed
- 2. Wiseman SM, Baliski C, Irvine R, et al. Hemithyroidectomy: the optimal initial surgical approach for individuals undergoing surgery for a cytological diagnosis of follicular neoplasm. Ann Surg Oncol 2006; 13: 425-32. ArticlePubMedPDF
- 3. McHenry CR, Phitayakorn R. Follicular adenoma and carcinoma of the thyroid gland. Oncologist 2011; 16: 585-93. ArticlePubMedPMCPDF
- 4. LiVolsi VA, Baloch ZW. Follicular-patterned tumors of the thyroid: the battle of benign vs. malignant vs. so-called uncertain. Endocr Pathol 2011; 22: 184-9. ArticlePubMedPDF
- 5. Sobrinho-Simões M, Eloy C, Magalhães J, Lobo C, Amaro T. Follicular thyroid carcinoma. Mod Pathol 2011; 24 Suppl 2: S10-8.
- 6. Mazzaferri EL. Management of a solitary thyroid nodule. N Engl J Med 1993; 328: 553-9. ArticlePubMed
- 7. Baloch ZW, LiVolsi VA. Our approach to follicular-patterned lesions of the thyroid. J Clin Pathol 2007; 60: 244-50. ArticlePubMedPMC
- 8. Prasad NB, Kowalski J, Tsai HL, et al. Three-gene molecular diagnostic model for thyroid cancer. Thyroid 2012; 22: 275-84. ArticlePubMedPMC
- 9. Cerutti JM, Delcelo R, Amadei MJ, et al. A preoperative diagnostic test that distinguishes benign from malignant thyroid carcinoma based on gene expression. J Clin Invest 2004; 113: 1234-42. ArticlePubMedPMC
- 10. Cerutti JM, Latini FR, Nakabashi C, et al. Diagnosis of suspicious thyroid nodules using four protein biomarkers. Clin Cancer Res 2006; 12(11 Pt 1): 3311-8. ArticlePubMedPDF
- 11. Haghpanah V, Shooshtarizadeh P, Heshmat R, Larijani B, Tavangar SM. Immunohistochemical analysis of survivin expression in thyroid follicular adenoma and carcinoma. Appl Immunohistochem Mol Morphol 2006; 14: 422-5. ArticlePubMed
- 12. Belge G, Meyer A, Klemke M, et al. Upregulation of HMGA2 in thyroid carcinomas: a novel molecular marker to distinguish between benign and malignant follicular neoplasias. Genes Chromosomes Cancer 2008; 47: 56-63. ArticlePubMed
- 13. Chiappetta G, Ferraro A, Vuttariello E, et al. HMGA2 mRNA expression correlates with the malignant phenotype in human thyroid neoplasias. Eur J Cancer 2008; 44: 1015-21. ArticlePubMed
- 14. Prasad NB, Somervell H, Tufano RP, et al. Identification of genes differentially expressed in benign versus malignant thyroid tumors. Clin Cancer Res 2008; 14: 3327-37. ArticlePubMedPMCPDF
- 15. Paunovic I, Isic T, Havelka M, Tatic S, Cvejic D, Savin S. Combined immunohistochemistry for thyroid peroxidase, galectin-3, CK19 and HBME-1 in differential diagnosis of thyroid tumors. APMIS 2012; 120: 368-79. ArticlePubMed
- 16. Bryson PC, Shores CG, Hart C, et al. Immunohistochemical distinction of follicular thyroid adenomas and follicular carcinomas. Arch Otolaryngol Head Neck Surg 2008; 134: 581-6. ArticlePubMed
- 17. Sigstad E, Paus E, Bjøro T, et al. The new molecular markers DDIT3, STT3A, ARG2 and FAM129A are not useful in diagnosing thyroid follicular tumors. Mod Pathol 2012; 25: 537-47. ArticlePubMedPDF
- 18. de Matos PS, Ferreira AP, de Oliveira Facuri F, Assumpção LV, Metze K, Ward LS. Usefulness of HBME-1, cytokeratin 19 and galectin-3 immunostaining in the diagnosis of thyroid malignancy. Histopathology 2005; 47: 391-401. ArticlePubMed
- 19. Prasad ML, Pellegata NS, Huang Y, Nagaraja HN, de la Chapelle A, Kloos RT. Galectin-3, fibronectin-1, CITED-1, HBME1 and cytokeratin-19 immunohistochemistry is useful for the differential diagnosis of thyroid tumors. Mod Pathol 2005; 18: 48-57. ArticlePubMedPDF
- 20. Papale F, Cafiero G, Grimaldi A, et al. Galectin-3 expression in thyroid fine needle cytology (t-FNAC) uncertain cases: validation of molecular markers and technology innovation. J Cell Physiol 2013; 228: 968-74. ArticlePubMed
- 21. Lal G, Padmanabha L, Provenzano M, Fitzgerald M, Weydert J, Domann FE. Regulation of 14-3-3sigma expression in human thyroid carcinoma is epigenetically regulated by aberrant cytosine methylation. Cancer Lett 2008; 267: 165-74. PubMedPMC
- 22. Kim YA, Chang M, Park YJ, Kim JE. Detection of survivin and COX- 2 in thyroid carcinoma: anaplastic carcinoma shows overexpression of nuclear survivin and low COX-2 expression. Korean J Pathol 2012; 46: 55-60. ArticlePubMedPMC
- 23. Park YJ, Kwak SH, Kim DC, et al. Diagnostic value of galectin-3, HBME-1, cytokeratin 19, high molecular weight cytokeratin, cyclin D1 and p27(kip1) in the differential diagnosis of thyroid nodules. J Korean Med Sci 2007; 22: 621-8. ArticlePubMedPMC
- 24. Saleh HA, Jin B, Barnwell J, Alzohaili O. Utility of immunohistochemical markers in differentiating benign from malignant follicular-derived thyroid nodules. Diagn Pathol 2010; 5: 9.ArticlePubMedPMCPDF
REFERENCES
Figure & Data
References
Citations

- High mobility group protein 2 (HMGA2) is highly expressed in a broad range of benign and malignant tumors
Viktoria Chirico, Fatih Baybars Ergüven, Katharina Möller, Florian Lutz, Florian Viehweger, Martina Kluth, Claudia Hube-Magg, Christian Bernreuther, Guido Sauter, Andreas H. Marx, Ronald Simon, Frank Jacobsen, Patrick Lebok, Till S. Clauditz, Waldemar Wil
Virchows Archiv.2025; 487(1): 183. CrossRef - Assessing the potential of high-mobility group AT-hook 2 immunohistochemical staining as a prognostic marker of metastatic recurrence in follicular thyroid cancer: a retrospective cohort study
Yuka Ito, Junko Sakumoto, Hideki Hirabayashi, Shinichi Haruna, Wataru Konno, Itsuo Nakajima, Kazuyuki Ishida, Yasuo Haruyama, Toshimi Sairenchi, Eijun Nishihara, Shuji Fukata, Akira Hishinuma, Takahiko Kogai
Endocrine Journal.2025; 72(5): 535. CrossRef - HMGA2 promotes nasopharyngeal carcinoma progression and is associated with tumor resistance and poor prognosis
Xinting Ouyang, Kangxin Li, Jiaqi Wang, Weijian Zhu, Qiang Yi, Jinghua Zhong
Frontiers in Oncology.2024;[Epub] CrossRef - miR‐98‐5p promotes apoptosis and inhibits migration and cell growth in papillary thyroid carcinoma through Bax/Caspase‐3 by HMGA2
Kai Qiu, QingJi Xie, Shan Jiang, Ting Lin
Journal of Clinical Laboratory Analysis.2020;[Epub] CrossRef - High mobility group A protein-2 as a tumor cancer diagnostic and prognostic marker: a systematic review and meta-analysis
Yen Thi-Hai Pham, Ovie Utuama, Claire E. Thomas, Jong A. Park, Carlo La Vecchia, Harvey A. Risch, Chi Thi-Du Tran, Thanh V. Le, Paolo Boffetta, Leon Raskin, Hung N. Luu
European Journal of Cancer Prevention.2020; 29(6): 565. CrossRef - Diagnostic performance of HMGA2 gene expression for differentiation of malignant thyroid nodules: A systematic review and meta‐analysis
Bo Hyun Kim, Seong Jang Kim, Mijin Kim, Sang‐Woo Lee, Shin Young Jeong, Kyoungjune Pak, Keunyoung Kim, In Joo Kim
Clinical Endocrinology.2018; 89(6): 856. CrossRef - Thyroid follicular adenomas and carcinomas: molecular profiling provides evidence for a continuous evolution
Geneviève Dom, Sandra Frank, Sebastien Floor, Pashalina Kehagias, Frederick Libert, Catherine Hoang, Guy Andry, Alex Spinette, Ligia Craciun, Nicolas de Saint Aubin, Christophe Tresallet, Frederique Tissier, Frederique Savagner, Samira Majjaj, Ilse Gutier
Oncotarget.2018; 9(12): 10343. CrossRef - APLP2, RRM2, and PRC1: New Putative Markers for the Differential Diagnosis of Thyroid Follicular Lesions
Esmeralda Castelblanco, Carles Zafon, Javier Maravall, Pilar Gallel, Montserrat Martinez, Ismael Capel, Maria Rosa Bella, Irene Halperin, Jordi Temprana, Carmela Iglesias, Manel Puig-Domingo, Mercedes Robledo, Xavier Matias-Guiu, Didac Mauricio
Thyroid.2017; 27(1): 59. CrossRef - Prognostic implication of histological features associated with EHD2 expression in papillary thyroid carcinoma
Yourha Kim, Min-Hee Kim, Sora Jeon, Jeeyoon Kim, Chankyung Kim, Ja Seong Bae, Chan Kwon Jung, Yves St-Pierre
PLOS ONE.2017; 12(3): e0174737. CrossRef - Survivin DEx3 as a biomarker of thyroid cancers: A study at the mRNA and protein level
Joanna Waligórska-Stachura, Nadia Sawicka-Gutaj, Maciej Zabel, Mirosław Andrusiewicz, Paweł Gut, Agata Czarnywojtek, Marek Ruchała
Oncology Letters.2017; 13(4): 2437. CrossRef - High-Frequency Ultrasound-Guided Injection for the Generation of a Novel Orthotopic Mouse Model of Human Thyroid Carcinoma
Adelaide Greco, Sandra Albanese, Luigi Auletta, Peppino Mirabelli, Antonella Zannetti, Crescenzo D'Alterio, Gennaro Di Maro, Francesca Maria Orlandella, Giuliana Salvatore, Andrea Soricelli, Marco Salvatore
Thyroid.2016; 26(4): 552. CrossRef - The study of galectin-3, Ki-67, ubiquitin, HMGA-2 by polymerase chain reaction in real time (RT-PCR) in the puncture specimens of nodular goiter
Irina S. Berjozkina, Tat'jana V. Saprina, Anastasija P. Zima, Anna V. Isaeva, Venera N. Latipova, Marat R. Muhamedov, Leonid R. Bazilevich, Oleg S. Popov, Dar'ja A. Skuratovskaja, Kristina A. Jurova, Larisa C. Litvinova
Clinical and experimental thyroidology.2016; 12(2): 19. CrossRef - High-mobility group A2 overexpression is an unfavorable prognostic biomarker for nasopharyngeal carcinoma patients
Zhuoxing Liu, Kunpeng Wu, Zhixiong Yang, Aibing Wu
Molecular and Cellular Biochemistry.2015; 409(1-2): 155. CrossRef - Defining the value of CD56, CK19, Galectin 3 and HBME-1 in diagnosis of follicular cell derived lesions of thyroid with systematic review of literature
Duško Dunđerović, Jasmina Marković Lipkovski, Ivan Boričic, Ivan Soldatović, Vesna Božic, Dubravka Cvejić, Svetislav Tatić
Diagnostic Pathology.2015;[Epub] CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
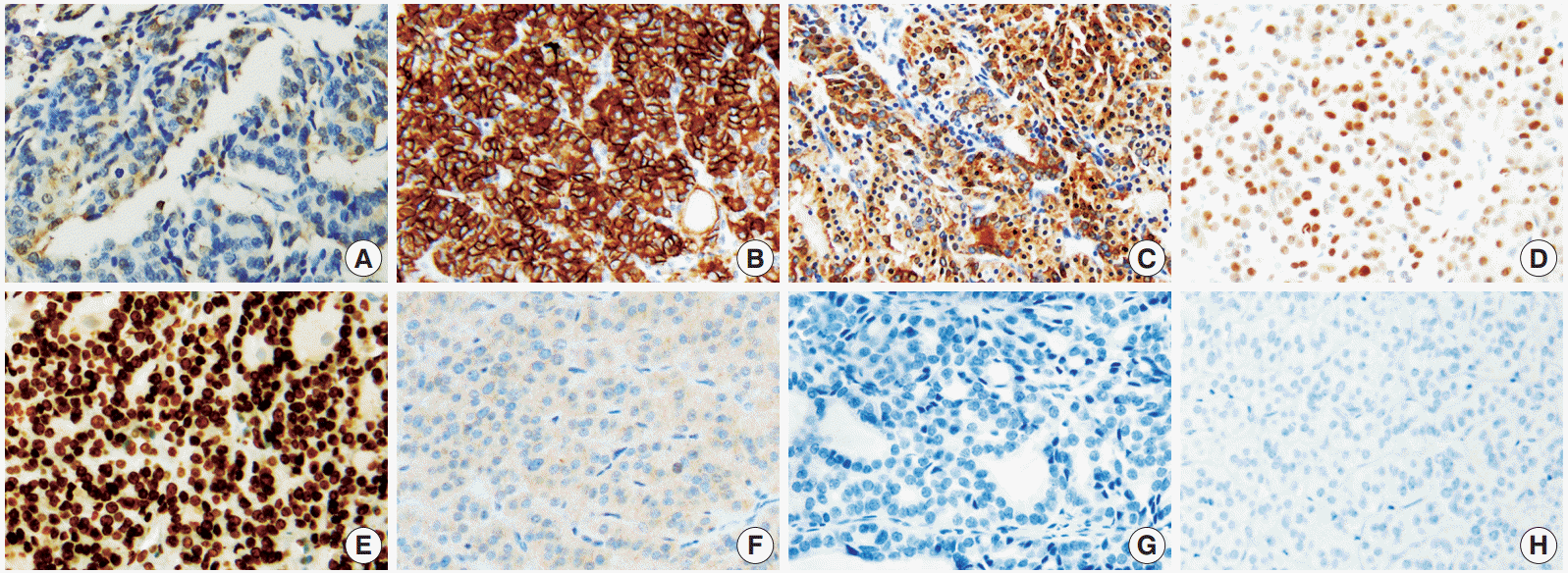
Fig. 1.
| Characteristic | Value (n = 78) |
|---|---|
| Age (yr) | |
| Median (range) | 42 (9–76) |
| Sex | |
| Female | 61 |
| Male | 17 |
| Location | |
| Right lobe | 37 |
| Left lobe | 38 |
| Isthmus | 1 |
| Both lobe | 2 |
| T category | |
| T1a | 2 |
| T1b | 5 |
| T2 | 37 |
| T3 | 33 |
| T4 | 0 |
| N category | |
| NX | 38 |
| N0 | 37 |
| N1a | 1 |
| N1b | 2 |
| Tumor size (cm) | |
| Mean (range) | 3.7 (0.8–7.3) |
| Capsular invasion | |
| Minimal | 67 |
| Widely | 11 |
| Vascular invasion | |
| Absent | 59 |
| Present | 19 |
| Follow-up duration (yr) | |
| Mean (range) | 3.33 (0.91–8.98) |
| Distant metastasis | |
| Absent | 77 |
| Present | 1 |
| Local recurrence | |
| Absent | 77 |
| Present | 1 |
| Diagnosis | Gal-3 | HBME1 | CK19 | Cyclin D1 | HMGA2 |
|---|---|---|---|---|---|
| AG (n = 41) | 1 (2.4) | 4 (9.8) | 3 (7.3) | 4 (9.8) | 10 (24.4) |
| FA (n = 72) | 4 (5.6) | 34 (47.2) | 4 (5.6) | 38 (52.8) | 30 (41.7) |
| FTC (n = 79) | 6 (7.6) | 52 (65.8) | 10 (12.7) | 35 (44.3) | 44 (55.7) |
| p-value (FTC vs FA and AG) | .364 | < .001 | .121 | .321 | .005 |
| p-value (FTC vs FA) | .748 | .021 | .133 | .298 | .085 |
| p-value (FTC and FA vs AG) | .462 | < .001 | 1.000 | < .001 | .005 |
| Antibody | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | Diagnostic accuracy (%) |
|---|---|---|---|---|---|
| Single marker | |||||
| HBME1 | 65.8 | 66.4 | 57.8 | 73.5 | 66.1 |
| HMGA2 | 55.7 | 64.6 | 52.4 | 67.6 | 60.9 |
| Double markers | |||||
| HBME1 and HMGA2 | 49.4 | 76.1 | 52.8 | 73.8 | 65.1 |
| HBME1 or HMGA2 | 72.2 | 54.9 | 59.1 | 68.3 | 62.0 |
| Antibody | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | Diagnostic accuracy (%) |
|---|---|---|---|---|---|
| Single marker | |||||
| HBME1 | 57.0 | 90.2 | 96.0 | 36.3 | 64.1 |
| Cyclin D1 | 48.3 | 90.2 | 94.8 | 32.2 | 57.3 |
| HMGA2 | 49.0 | 75.6 | 88.1 | 28.7 | 54.7 |
| Co-expression among 3 markers | |||||
| ≥ 1 among HBME1, cyclin D1, HMGA2 | 80.8 | 75.6 | 92.4 | 51.7 | 79.7 |
| ≥ 2 among HBME1, cyclin D1, HMGA2 | 53.6 | 85.4 | 93.1 | 33.3 | 60.4 |
FTC, follicular thyroid carcinoma.
Numbers in parentheses indicate percentage of positive cases. p-values are calculated using the χ2 test or Fisher exact test. AG, adenomatous goiter; FA, follicular adenoma; FTC, follicular thyroid carcinoma; Gal-3, galectin-3; HBME1, Hector Battifora mesothelial 1; CK19, cytokeratin 19.
HBME1, Hector Battifora mesothelial 1; FTC, follicular thyroid carcinoma; FA, follicular adenoma; AG, adenomatous goiter; PPV, positive predictive value; NPV, negative predictive value.
FN, follicular neoplasm; FTC, follicular thyroid carcinoma; FA, follicular adenoma; AG, adenomatous goiter; PPV, positive predictive value; NPV, negative predictive value; HBME1, Hector Battifora mesothelial 1.

 E-submission
E-submission



