Search
- Page Path
- HOME > Search
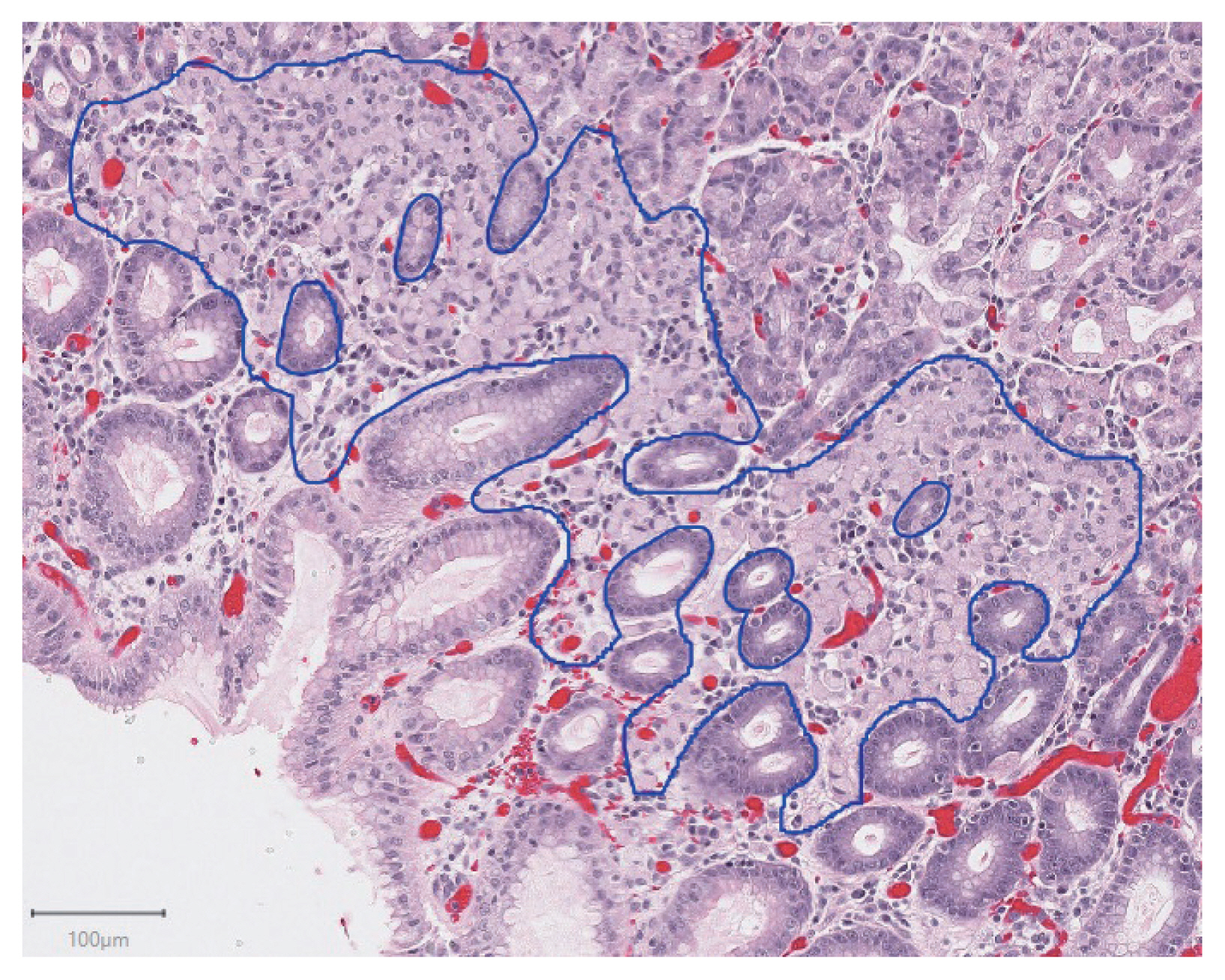
- Deep learning for computer-assisted diagnosis of hereditary diffuse gastric cancer
- Sean A. Rasmussen, Thomas Arnason, Weei-Yuarn Huang
- J Pathol Transl Med. 2021;55(2):118-124. Published online January 22, 2021
- DOI: https://doi.org/10.4132/jptm.2020.12.22
- 5,843 View
- 135 Download
- 9 Web of Science
- 9 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
Patients with hereditary diffuse gastric cancer often undergo prophylactic gastrectomy to minimize cancer risk. Because intramucosal poorly cohesive carcinomas in this setting are typically not grossly visible, many pathologists assess the entire gastrectomy specimen microscopically. With 150 or more slides per case, this is a major time burden for pathologists. This study utilizes deep learning methods to analyze digitized slides and detect regions of carcinoma.
Methods
Prophylactic gastrectomy specimens from seven patients with germline CDH1 mutations were analyzed (five for training/validation and two for testing, with a total of 133 tumor foci). All hematoxylin and eosin slides containing cancer foci were digitally scanned, and patches of size 256×256 pixels were randomly extracted from regions of cancer as well as from regions of normal background tissue, resulting in 15,851 images for training/validation and 970 images for testing. A model with DenseNet-169 architecture was trained for 150 epochs, then evaluated on images from the test set. External validation was conducted on 814 images scanned at an outside institution.
Results
On individual patches, the trained model achieved a receiver operating characteristic (ROC) area under the curve (AUC) of 0.9986. This enabled it to maintain a sensitivity of 90% with a false-positive rate of less than 0.1%. On the external validation dataset, the model achieved a similar ROC AUC of 0.9984. On whole slide images, the network detected 100% of tumor foci and correctly eliminated an average of 99.9% of the non-cancer slide area from consideration.
Conclusions
Overall, our model shows encouraging progress towards computer-assisted diagnosis of hereditary diffuse gastric cancer. -
Citations
Citations to this article as recorded by- Now and future of artificial intelligence-based signet ring cell diagnosis: A survey
Zhu Meng, Junhao Dong, Limei Guo, Fei Su, Jiaxuan Liu, Guangxi Wang, Zhicheng Zhao
Expert Systems with Applications.2026; 296: 129188. CrossRef - Development and application of deep learning-based diagnostics for pathologic diagnosis of gastric endoscopic submucosal dissection specimens
Soomin Ahn, Yiyu Hong, Sujin Park, Yunjoo Cho, Inwoo Hwang, Ji Min Na, Hyuk Lee, Byung-Hoon Min, Jun Haeng Lee, Jae J. Kim, Kyoung-Mee Kim
Gastric Cancer.2025; 28(4): 609. CrossRef - A Comprehensive Literature Review of the CDH1 Mutation and Its Role in Gastric Cancer
Malik Samardali, Jehad Samardaly, Ibrahim Shanti
Cureus.2025;[Epub] CrossRef - Deep learning for multiclass tumor cell detection in histopathology slides of hereditary diffuse gastric cancer
Robin Lomans, Valentina Angerilli, Joey Spronck, Liudmila L. Kodach, Irene Gullo, Fátima Carneiro, Rachel S. van der Post, Francesco Ciompi
iScience.2025; 28(8): 113064. CrossRef - Non-endoscopic Applications of Machine Learning in Gastric Cancer: A Systematic Review
Marianne Linley L. Sy-Janairo, Jose Isagani B. Janairo
Journal of Gastrointestinal Cancer.2024; 55(1): 47. CrossRef - Artificial intelligence applicated in gastric cancer: A bibliometric and visual analysis via CiteSpace
Guoyang Zhang, Jingjing Song, Zongfeng Feng, Wentao Zhao, Pan Huang, Li Liu, Yang Zhang, Xufeng Su, Yukang Wu, Yi Cao, Zhengrong Li, Zhigang Jie
Frontiers in Oncology.2023;[Epub] CrossRef - Deep learning of endoscopic features for the assessment of neoadjuvant therapy response in locally advanced rectal cancer
Anqi Wang, Jieli Zhou, Gang Wang, Beibei Zhang, Hongyi Xin, Haiyang Zhou
Asian Journal of Surgery.2023; 46(9): 3568. CrossRef - Preparing Data for Artificial Intelligence in Pathology with Clinical-Grade Performance
Yuanqing Yang, Kai Sun, Yanhua Gao, Kuansong Wang, Gang Yu
Diagnostics.2023; 13(19): 3115. CrossRef - Using Deep Learning to Predict Final HER2 Status in Invasive Breast Cancers That are Equivocal (2+) by Immunohistochemistry
Sean A. Rasmussen, Valerie J. Taylor, Alexi P. Surette, Penny J. Barnes, Gillian C. Bethune
Applied Immunohistochemistry & Molecular Morphology.2022; 30(10): 668. CrossRef
- Now and future of artificial intelligence-based signet ring cell diagnosis: A survey
- Introduction to digital pathology and computer-aided pathology
- Soojeong Nam, Yosep Chong, Chan Kwon Jung, Tae-Yeong Kwak, Ji Youl Lee, Jihwan Park, Mi Jung Rho, Heounjeong Go
- J Pathol Transl Med. 2020;54(2):125-134. Published online February 13, 2020
- DOI: https://doi.org/10.4132/jptm.2019.12.31
- 22,426 View
- 657 Download
- 87 Web of Science
- 91 Crossref
-
 Abstract
Abstract
 PDF
PDF - Digital pathology (DP) is no longer an unfamiliar term for pathologists, but it is still difficult for many pathologists to understand the engineering and mathematics concepts involved in DP. Computer-aided pathology (CAP) aids pathologists in diagnosis. However, some consider CAP a threat to the existence of pathologists and are skeptical of its clinical utility. Implementation of DP is very burdensome for pathologists because technical factors, impact on workflow, and information technology infrastructure must be considered. In this paper, various terms related to DP and computer-aided pathologic diagnosis are defined, current applications of DP are discussed, and various issues related to implementation of DP are outlined. The development of computer-aided pathologic diagnostic tools and their limitations are also discussed.
-
Citations
Citations to this article as recorded by- Artificial Intelligence and Digital Pathology: Toward Next‐Generation Diagnostic Hematology
Valeriia Tsekhovska, Pier Paolo Piccaluga, Nikhat Kaura
International Journal of Clinical Practice.2026;[Epub] CrossRef - Deep learning model to diagnose cardiac amyloidosis from haematoxylin/eosin-stained myocardial tissue
Takeshi Tohyama, Takeshi Iwasaki, Masataka Ikeda, Masato Katsuki, Tatsuya Watanabe, Kayo Misumi, Keisuke Shinohara, Takeo Fujino, Toru Hashimoto, Shouji Matsushima, Tomomi Ide, Junji Kishimoto, Koji Todaka, Yoshinao Oda, Kohtaro Abe
European Heart Journal - Imaging Methods and Practice.2025;[Epub] CrossRef - The current landscape of artificial intelligence in oral and maxillofacial surgery– a narrative review
Rushil Rajiv Dang, Balram Kadaikal, Sam El Abbadi, Branden R. Brar, Amit Sethi, Radhika Chigurupati
Oral and Maxillofacial Surgery.2025;[Epub] CrossRef - Assessing the quality of whole slide images in cytology from nuclei features
Paul Barthe, Romain Brixtel, Yann Caillot, Benoît Lemoine, Arnaud Renouf, Vianney Thurotte, Ouarda Beniken, Sébastien Bougleux, Olivier Lézoray
Journal of Pathology Informatics.2025; 17: 100420. CrossRef - An update on applications of digital pathology: primary diagnosis; telepathology, education and research
Shamail Zia, Isil Z. Yildiz-Aktas, Fazail Zia, Anil V. Parwani
Diagnostic Pathology.2025;[Epub] CrossRef - Artificial intelligence–driven digital pathology in urological cancers: current trends and future directions
Inyoung Paik, Geongyu Lee, Joonho Lee, Tae-Yeong Kwak, Hong Koo Ha
Prostate International.2025; 13(4): 181. CrossRef - Label-free optical microscopy with artificial intelligence: a new paradigm in pathology
Chiho Yoon, Eunwoo Park, Donggyu Kim, Byullee Park, Chulhong Kim
Biophotonics Discovery.2025;[Epub] CrossRef - EPIIC: Edge-Preserving Method Increasing Nuclei Clarity for Compression Artifacts Removal in Whole-Slide Histopathological Images
Julia Merta, Michal Marczyk
Applied Sciences.2025; 15(8): 4450. CrossRef - Comparative analysis of a 5G campus network and existing networks for real-time consultation in remote pathology
Ilgar I. Guseinov, Arnab Bhowmik, Somaia AbuBaker, Anna E. Schmaus-Klughammer, Thomas Spittler
Journal of Pathology Informatics.2025; 18: 100444. CrossRef - The Evolution of Digital Pathology in Infrastructure, Artificial Intelligence and Clinical Impact
Chan Kwon Jung
International Journal of Thyroidology.2025; 18(1): 6. CrossRef - Role of Telepathology, Artificial Intelligence, and Emerging Technologies in Enhancing Diagnostic Accuracy
Yugeshwari R. Tiwade, Obaid Noman, Pratibha Dawande, Nandkishor J Bankar, Sweta Bahadure, Praful Patil
Journal of Nature and Science of Medicine.2025; 8(2): 115. CrossRef - Analysis of system and scanner downtime in a digital pathology–predominant institution: A 6-year experience
Ryan Reagans, Lokman Cevik, Himani Kumar, David Kellough, Abberly Lott Limbach, Giovanni Lujan, Anil Parwani, Hamza N Gokozan
American Journal of Clinical Pathology.2025; 164(4): 634. CrossRef - Integration of Digital Cytology in Quality Assurance Programs for Cytopathology
Yosep Chong, Maria Jesús Fernández Aceñero, Zaibo Li, Andrey Bychkov
Acta Cytologica.2025; 70(1): 126. CrossRef - Telepathology for Consultation in the Military Health System: An Evaluation of Pathologists’ Impressions of Facilitators and Barriers Prior to Implementation
Victoria Mahar, Zachary Colburn, Joshua Sakai
Laboratory Investigation.2025; 105(11): 104236. CrossRef - Online histostereometric analysis in digital forensic pathology: a technical report
Vladimir G. Nedugov, Anna V. Zhukova, German V. Nedugov
Russian Journal of Forensic Medicine.2025; 11(2): 145. CrossRef - Latent representation of H&E images retains biological information in a breast cancer cohort
Chloé Benmussa, Esther Sanfeliu, Anabel Martínez-Romero, Blanca González-Farré, Tomás Pascual, Joaquín Gavilá, Alona Levy-Jurgenson, Ariel Shamir, Fara Brasó-Maristany, Aleix Prat, Zohar Yakhini, Amgad Muneer
PLOS One.2025; 20(9): e0329221. CrossRef - Modernizing Colorectal Cancer Care With Artificial Intelligence: Real-Time Detection, Radiomics, and Digital Pathology
Elmoatazbellah Nasr, Zaid Al-Hamid, Mina H Younan, Mohamed Omran, Maan Sarsam, Mohamed Abdellatif
Cureus.2025;[Epub] CrossRef - A multi-task learning model for evaluating non-tumor gastric diseases indicators in whole slide images
Mingxi Fu, Liming Liu, Fanglei Fu, Jingli Ouyang, Xueying Shi, Song Duan, Tian Guan, Yonghong He, Zhiqiang Cheng, Lianghui Zhu
Scientific Reports.2025;[Epub] CrossRef - Performance Evaluation of MambaVision in Breast Cancer Detection from Histopathology Images
Hasan Zan
Dicle Üniversitesi Mühendislik Fakültesi Mühendislik Dergisi.2025; 16(4): 879. CrossRef - Artificial intelligence for automatic detection of basal cell carcinoma from frozen tissue tangential biopsies
Dennis H Murphree, Yong-hun Kim, Kirk A Sidey, Nneka I Comfere, Nahid Y Vidal
Clinical and Experimental Dermatology.2024; 49(7): 719. CrossRef - Performance of externally validated machine learning models based on histopathology images for the diagnosis, classification, prognosis, or treatment outcome prediction in female breast cancer: A systematic review
Ricardo Gonzalez, Peyman Nejat, Ashirbani Saha, Clinton J.V. Campbell, Andrew P. Norgan, Cynthia Lokker
Journal of Pathology Informatics.2024; 15: 100348. CrossRef - Invisible for a few but essential for many: the role of Histotechnologists in the establishment of digital pathology
Gisela Magalhães, Rita Calisto, Catarina Freire, Regina Silva, Diana Montezuma, Sule Canberk, Fernando Schmitt
Journal of Histotechnology.2024; 47(1): 39. CrossRef - Using digital pathology to analyze the murine cerebrovasculature
Dana M Niedowicz, Jenna L Gollihue, Erica M Weekman, Panhavuth Phe, Donna M Wilcock, Christopher M Norris, Peter T Nelson
Journal of Cerebral Blood Flow & Metabolism.2024; 44(4): 595. CrossRef - PATrans: Pixel-Adaptive Transformer for edge segmentation of cervical nuclei on small-scale datasets
Hexuan Hu, Jianyu Zhang, Tianjin Yang, Qiang Hu, Yufeng Yu, Qian Huang
Computers in Biology and Medicine.2024; 168: 107823. CrossRef - CNAC-Seg: Effective segmentation for cervical nuclei in adherent cells and clusters via exploring gaps of receptive fields
Hexuan Hu, Jianyu Zhang, Tianjin Yang, Qiang Hu, Yufeng Yu, Qian Huang
Biomedical Signal Processing and Control.2024; 90: 105833. CrossRef - Artificial Intelligence-Enabled Prostate Cancer Diagnosis and Prognosis: Current State and Future Implications
Swati Satturwar, Anil V. Parwani
Advances in Anatomic Pathology.2024; 31(2): 136. CrossRef - Ensemble Deep Learning Model to Predict Lymphovascular Invasion in Gastric Cancer
Jonghyun Lee, Seunghyun Cha, Jiwon Kim, Jung Joo Kim, Namkug Kim, Seong Gyu Jae Gal, Ju Han Kim, Jeong Hoon Lee, Yoo-Duk Choi, Sae-Ryung Kang, Ga-Young Song, Deok-Hwan Yang, Jae-Hyuk Lee, Kyung-Hwa Lee, Sangjeong Ahn, Kyoung Min Moon, Myung-Giun Noh
Cancers.2024; 16(2): 430. CrossRef - Artificial intelligence’s impact on breast cancer pathology: a literature review
Amr Soliman, Zaibo Li, Anil V. Parwani
Diagnostic Pathology.2024;[Epub] CrossRef - Automated Analysis of Nuclear Parameters in Oral Exfoliative Cytology Using Machine Learning
Shubhangi Mhaske, Karthikeyan Ramalingam, Preeti Nair, Shubham Patel, Arathi Menon P, Nida Malik, Sumedh Mhaske
Cureus.2024;[Epub] CrossRef - Enhancing AI Research for Breast Cancer: A Comprehensive Review of Tumor-Infiltrating Lymphocyte Datasets
Alessio Fiorin, Carlos López Pablo, Marylène Lejeune, Ameer Hamza Siraj, Vincenzo Della Mea
Journal of Imaging Informatics in Medicine.2024; 37(6): 2996. CrossRef - Current Developments in Diagnosis of Salivary Gland Tumors: From Structure to Artificial Intelligence
Alexandra Corina Faur, Roxana Buzaș, Adrian Emil Lăzărescu, Laura Andreea Ghenciu
Life.2024; 14(6): 727. CrossRef - Comparative analysis of chronic progressive nephropathy (CPN) diagnosis in rat kidneys using an artificial intelligence deep learning model
Yeji Bae, Jongsu Byun, Hangyu Lee, Beomseok Han
Toxicological Research.2024; 40(4): 551. CrossRef - A Pan-Cancer Patient-Derived Xenograft Histology Image Repository with Genomic and Pathologic Annotations Enables Deep Learning Analysis
Brian S. White, Xing Yi Woo, Soner Koc, Todd Sheridan, Steven B. Neuhauser, Shidan Wang, Yvonne A. Evrard, Li Chen, Ali Foroughi pour, John D. Landua, R. Jay Mashl, Sherri R. Davies, Bingliang Fang, Maria Gabriela Rosa, Kurt W. Evans, Matthew H. Bailey, Y
Cancer Research.2024; 84(13): 2060. CrossRef - Non-contrasted computed tomography (NCCT) based chronic thromboembolic pulmonary hypertension (CTEPH) automatic diagnosis using cascaded network with multiple instance learning
Mayang Zhao, Liming Song, Jiarui Zhu, Ta Zhou, Yuanpeng Zhang, Shu-Cheng Chen, Haojiang Li, Di Cao, Yi-Quan Jiang, Waiyin Ho, Jing Cai, Ge Ren
Physics in Medicine & Biology.2024; 69(18): 185011. CrossRef - MR_NET: A Method for Breast Cancer Detection and Localization from Histological Images Through Explainable Convolutional Neural Networks
Rachele Catalano, Myriam Giusy Tibaldi, Lucia Lombardi, Antonella Santone, Mario Cesarelli, Francesco Mercaldo
Sensors.2024; 24(21): 7022. CrossRef - Advances in AI-Enhanced Biomedical Imaging for Cancer Immunology
Willa Wen-You Yim, Felicia Wee, Zheng Yi Ho, Xinyun Feng, Marcia Zhang, Samuel Lee, Inti Zlobec, Joe Yeong, Mai Chan Lau
World Scientific Annual Review of Cancer Immunology.2024;[Epub] CrossRef - Blockchain: A safe digital technology to share cancer diagnostic results in pandemic times—Challenges and legacy for the future
Bruno Natan Santana Lima, Lucas Alves da Mota Santana, Rani Iani Costa Gonçalo, Carla Samily de Oliveira Costa, Daniel Pitanga de Sousa Nogueira, Cleverson Luciano Trento, Wilton Mitsunari Takeshita
Oral Surgery.2023; 16(3): 300. CrossRef - Pathologists’ acceptance of telepathology in the Ministry of National Guard Health Affairs Hospitals in Saudi Arabia: A survey study
Raneem Alawashiz, Sharifah Abdullah AlDossary
DIGITAL HEALTH.2023;[Epub] CrossRef - An Atrous Convolved Hybrid Seg-Net Model with residual and attention mechanism for gland detection and segmentation in histopathological images
Manju Dabass, Jyoti Dabass
Computers in Biology and Medicine.2023; 155: 106690. CrossRef - Validation of a Machine Learning Expert Supporting System, ImmunoGenius, Using Immunohistochemistry Results of 3000 Patients with Lymphoid Neoplasms
Jamshid Abdul-Ghafar, Kyung Jin Seo, Hye-Ra Jung, Gyeongsin Park, Seung-Sook Lee, Yosep Chong
Diagnostics.2023; 13(7): 1308. CrossRef - Diagnosing Infectious Diseases in Poultry Requires a Holistic Approach: A Review
Dieter Liebhart, Ivana Bilic, Beatrice Grafl, Claudia Hess, Michael Hess
Poultry.2023; 2(2): 252. CrossRef - Recent application of artificial intelligence on histopathologic image-based prediction of gene mutation in solid cancers
Mohammad Rizwan Alam, Kyung Jin Seo, Jamshid Abdul-Ghafar, Kwangil Yim, Sung Hak Lee, Hyun-Jong Jang, Chan Kwon Jung, Yosep Chong
Briefings in Bioinformatics.2023;[Epub] CrossRef - Canine Mammary Tumor Histopathological Image Classification via Computer-Aided Pathology: An Available Dataset for Imaging Analysis
Giovanni P. Burrai, Andrea Gabrieli, Marta Polinas, Claudio Murgia, Maria Paola Becchere, Pierfranco Demontis, Elisabetta Antuofermo
Animals.2023; 13(9): 1563. CrossRef - Rapid digital pathology of H&E-stained fresh human brain specimens as an alternative to frozen biopsy
Bhaskar Jyoti Borah, Yao-Chen Tseng, Kuo-Chuan Wang, Huan-Chih Wang, Hsin-Yi Huang, Koping Chang, Jhih Rong Lin, Yi-Hua Liao, Chi-Kuang Sun
Communications Medicine.2023;[Epub] CrossRef - Applied machine learning in hematopathology
Taher Dehkharghanian, Youqing Mu, Hamid R. Tizhoosh, Clinton J. V. Campbell
International Journal of Laboratory Hematology.2023; 45(S2): 87. CrossRef - Automated diagnosis of 7 canine skin tumors using machine learning on H&E-stained whole slide images
Marco Fragoso-Garcia, Frauke Wilm, Christof A. Bertram, Sophie Merz, Anja Schmidt, Taryn Donovan, Andrea Fuchs-Baumgartinger, Alexander Bartel, Christian Marzahl, Laura Diehl, Chloe Puget, Andreas Maier, Marc Aubreville, Katharina Breininger, Robert Klopf
Veterinary Pathology.2023; 60(6): 865. CrossRef - Artificial Intelligence in the Pathology of Gastric Cancer
Sangjoon Choi, Seokhwi Kim
Journal of Gastric Cancer.2023; 23(3): 410. CrossRef - Efficient Convolution Network to Assist Breast Cancer Diagnosis and Target Therapy
Ching-Wei Wang, Kai-Lin Chu, Hikam Muzakky, Yi-Jia Lin, Tai-Kuang Chao
Cancers.2023; 15(15): 3991. CrossRef - Multi-Configuration Analysis of DenseNet Architecture for Whole Slide Image Scoring of ER-IHC
Wan Siti Halimatul Munirah Wan Ahmad, Mohammad Faizal Ahmad Fauzi, Md Jahid Hasan, Zaka Ur Rehman, Jenny Tung Hiong Lee, See Yee Khor, Lai-Meng Looi, Fazly Salleh Abas, Afzan Adam, Elaine Wan Ling Chan, Sei-Ichiro Kamata
IEEE Access.2023; 11: 79911. CrossRef - Digitization of Pathology Labs: A Review of Lessons Learned
Lars Ole Schwen, Tim-Rasmus Kiehl, Rita Carvalho, Norman Zerbe, André Homeyer
Laboratory Investigation.2023; 103(11): 100244. CrossRef - Artificial Intelligence in Endoscopic Ultrasonography-Guided Fine-Needle Aspiration/Biopsy (EUS-FNA/B) for Solid Pancreatic Lesions: Opportunities and Challenges
Xianzheng Qin, Taojing Ran, Yifei Chen, Yao Zhang, Dong Wang, Chunhua Zhou, Duowu Zou
Diagnostics.2023; 13(19): 3054. CrossRef - Deep Learning for the Pathologic Diagnosis of Hepatocellular Carcinoma, Cholangiocarcinoma, and Metastatic Colorectal Cancer
Hyun-Jong Jang, Jai-Hyang Go, Younghoon Kim, Sung Hak Lee
Cancers.2023; 15(22): 5389. CrossRef - AIR-UNet++: a deep learning framework for histopathology image segmentation and detection
Anusree Kanadath, J. Angel Arul Jothi, Siddhaling Urolagin
Multimedia Tools and Applications.2023; 83(19): 57449. CrossRef - Deep Learning-Based Dermatological Condition Detection: A Systematic Review With Recent Methods, Datasets, Challenges, and Future Directions
Stephanie S. Noronha, Mayuri A. Mehta, Dweepna Garg, Ketan Kotecha, Ajith Abraham
IEEE Access.2023; 11: 140348. CrossRef - Digital pathology and artificial intelligence in translational medicine and clinical practice
Vipul Baxi, Robin Edwards, Michael Montalto, Saurabh Saha
Modern Pathology.2022; 35(1): 23. CrossRef - Artificial Intelligence in Toxicological Pathology: Quantitative Evaluation of Compound-Induced Follicular Cell Hypertrophy in Rat Thyroid Gland Using Deep Learning Models
Valeria Bertani, Olivier Blanck, Davy Guignard, Frederic Schorsch, Hannah Pischon
Toxicologic Pathology.2022; 50(1): 23. CrossRef - Investigating the genealogy of the literature on digital pathology: a two-dimensional bibliometric approach
Dayu Hu, Chengyuan Wang, Song Zheng, Xiaoyu Cui
Scientometrics.2022; 127(2): 785. CrossRef - Digital Dermatopathology and Its Application to Mohs Micrographic Surgery
Yeongjoo Oh, Hye Min Kim, Soon Won Hong, Eunah Shin, Jihee Kim, Yoon Jung Choi
Yonsei Medical Journal.2022; 63(Suppl): S112. CrossRef - Assessment of parathyroid gland cellularity by digital slide analysis
Rotem Sagiv, Bertha Delgado, Oleg Lavon, Vladislav Osipov, Re'em Sade, Sagi Shashar, Ksenia M. Yegodayev, Moshe Elkabets, Ben-Zion Joshua
Annals of Diagnostic Pathology.2022; 58: 151907. CrossRef - PancreaSys: An Automated Cloud-Based Pancreatic Cancer Grading System
Muhammad Nurmahir Mohamad Sehmi, Mohammad Faizal Ahmad Fauzi, Wan Siti Halimatul Munirah Wan Ahmad, Elaine Wan Ling Chan
Frontiers in Signal Processing.2022;[Epub] CrossRef - Classification of Mouse Lung Metastatic Tumor with Deep Learning
Ha Neul Lee, Hong-Deok Seo, Eui-Myoung Kim, Beom Seok Han, Jin Seok Kang
Biomolecules & Therapeutics.2022; 30(2): 179. CrossRef - Techniques for digital histological morphometry of the pineal gland
Bogdan-Alexandru Gheban, Horaţiu Alexandru Colosi, Ioana-Andreea Gheban-Roșca, Carmen Georgiu, Dan Gheban, Doiniţa Crişan, Maria Crişan
Acta Histochemica.2022; 124(4): 151897. CrossRef - Current Trend of Artificial Intelligence Patents in Digital Pathology: A Systematic Evaluation of the Patent Landscape
Muhammad Joan Ailia, Nishant Thakur, Jamshid Abdul-Ghafar, Chan Kwon Jung, Kwangil Yim, Yosep Chong
Cancers.2022; 14(10): 2400. CrossRef - Recent Applications of Artificial Intelligence from Histopathologic Image-Based Prediction of Microsatellite Instability in Solid Cancers: A Systematic Review
Mohammad Rizwan Alam, Jamshid Abdul-Ghafar, Kwangil Yim, Nishant Thakur, Sung Hak Lee, Hyun-Jong Jang, Chan Kwon Jung, Yosep Chong
Cancers.2022; 14(11): 2590. CrossRef - Development of a prognostic prediction support system for cervical intraepithelial neoplasia using artificial intelligence-based diagnosis
Takayuki Takahashi, Hikaru Matsuoka, Rieko Sakurai, Jun Akatsuka, Yusuke Kobayashi, Masaru Nakamura, Takashi Iwata, Kouji Banno, Motomichi Matsuzaki, Jun Takayama, Daisuke Aoki, Yoichiro Yamamoto, Gen Tamiya
Journal of Gynecologic Oncology.2022;[Epub] CrossRef - Digital Pathology and Artificial Intelligence Applications in Pathology
Heounjeong Go
Brain Tumor Research and Treatment.2022; 10(2): 76. CrossRef - Mass spectrometry imaging to explore molecular heterogeneity in cell culture
Tanja Bien, Krischan Koerfer, Jan Schwenzfeier, Klaus Dreisewerd, Jens Soltwisch
Proceedings of the National Academy of Sciences.2022;[Epub] CrossRef - Integrating artificial intelligence in pathology: a qualitative interview study of users' experiences and expectations
Jojanneke Drogt, Megan Milota, Shoko Vos, Annelien Bredenoord, Karin Jongsma
Modern Pathology.2022; 35(11): 1540. CrossRef - Deep Learning on Basal Cell Carcinoma In Vivo Reflectance Confocal Microscopy Data
Veronika Shavlokhova, Michael Vollmer, Patrick Gholam, Babak Saravi, Andreas Vollmer, Jürgen Hoffmann, Michael Engel, Christian Freudlsperger
Journal of Personalized Medicine.2022; 12(9): 1471. CrossRef - Deep Learning-Based Classification of Uterine Cervical and Endometrial Cancer Subtypes from Whole-Slide Histopathology Images
JaeYen Song, Soyoung Im, Sung Hak Lee, Hyun-Jong Jang
Diagnostics.2022; 12(11): 2623. CrossRef - A self-supervised contrastive learning approach for whole slide image representation in digital pathology
Parsa Ashrafi Fashi, Sobhan Hemati, Morteza Babaie, Ricardo Gonzalez, H.R. Tizhoosh
Journal of Pathology Informatics.2022; 13: 100133. CrossRef - A Matched-Pair Analysis of Nuclear Morphologic Features Between Core Needle Biopsy and Surgical Specimen in Thyroid Tumors Using a Deep Learning Model
Faridul Haq, Andrey Bychkov, Chan Kwon Jung
Endocrine Pathology.2022; 33(4): 472. CrossRef - Development of quality assurance program for digital pathology by the Korean Society of Pathologists
Yosep Chong, Jeong Mo Bae, Dong Wook Kang, Gwangil Kim, Hye Seung Han
Journal of Pathology and Translational Medicine.2022; 56(6): 370. CrossRef - Machine learning in renal pathology
Matthew Nicholas Basso, Moumita Barua, Julien Meyer, Rohan John, April Khademi
Frontiers in Nephrology.2022;[Epub] CrossRef - Whole Slide Image Quality in Digital Pathology: Review and Perspectives
Romain Brixtel, Sebastien Bougleux, Olivier Lezoray, Yann Caillot, Benoit Lemoine, Mathieu Fontaine, Dalal Nebati, Arnaud Renouf
IEEE Access.2022; 10: 131005. CrossRef - Generalizability of Deep Learning System for the Pathologic Diagnosis of Various Cancers
Hyun-Jong Jang, In Hye Song, Sung Hak Lee
Applied Sciences.2021; 11(2): 808. CrossRef - Recent advances in the use of stimulated Raman scattering in histopathology
Martin Lee, C. Simon Herrington, Manasa Ravindra, Kristel Sepp, Amy Davies, Alison N. Hulme, Valerie G. Brunton
The Analyst.2021; 146(3): 789. CrossRef - Preference and Demand for Digital Pathology and Computer-Aided Diagnosis among Korean Pathologists: A Survey Study Focused on Prostate Needle Biopsy
Soo Jeong Nam, Yosep Chong, Chan Kwon Jung, Tae-Yeong Kwak, Ji Youl Lee, Jihwan Park, Mi Jung Rho, Heounjeong Go
Applied Sciences.2021; 11(16): 7380. CrossRef - An SVM approach towards breast cancer classification from H&E-stained histopathology images based on integrated features
M. A. Aswathy, M. Jagannath
Medical & Biological Engineering & Computing.2021; 59(9): 1773. CrossRef - Diagnosis prediction of tumours of unknown origin using ImmunoGenius, a machine learning-based expert system for immunohistochemistry profile interpretation
Yosep Chong, Nishant Thakur, Ji Young Lee, Gyoyeon Hwang, Myungjin Choi, Yejin Kim, Hwanjo Yu, Mee Yon Cho
Diagnostic Pathology.2021;[Epub] CrossRef - Deep Learning for Automatic Subclassification of Gastric Carcinoma Using Whole-Slide Histopathology Images
Hyun-Jong Jang, In-Hye Song, Sung-Hak Lee
Cancers.2021; 13(15): 3811. CrossRef - A novel evaluation method for Ki-67 immunostaining in paraffin-embedded tissues
Eliane Pedra Dias, Nathália Silva Carlos Oliveira, Amanda Oliveira Serra-Campos, Anna Karoline Fausto da Silva, Licínio Esmeraldo da Silva, Karin Soares Cunha
Virchows Archiv.2021; 479(1): 121. CrossRef - Assessment of Digital Pathology Imaging Biomarkers Associated with Breast Cancer Histologic Grade
Andrew Lagree, Audrey Shiner, Marie Angeli Alera, Lauren Fleshner, Ethan Law, Brianna Law, Fang-I Lu, David Dodington, Sonal Gandhi, Elzbieta A. Slodkowska, Alex Shenfield, Katarzyna J. Jerzak, Ali Sadeghi-Naini, William T. Tran
Current Oncology.2021; 28(6): 4298. CrossRef - Prediction of genetic alterations from gastric cancer histopathology images using a fully automated deep learning approach
Hyun-Jong Jang, Ahwon Lee, Jun Kang, In Hye Song, Sung Hak Lee
World Journal of Gastroenterology.2021; 27(44): 7687. CrossRef - Clustered nuclei splitting based on recurrent distance transform in digital pathology images
Lukasz Roszkowiak, Anna Korzynska, Dorota Pijanowska, Ramon Bosch, Marylene Lejeune, Carlos Lopez
EURASIP Journal on Image and Video Processing.2020;[Epub] CrossRef - Current Trends of Artificial Intelligence for Colorectal Cancer Pathology Image Analysis: A Systematic Review
Nishant Thakur, Hongjun Yoon, Yosep Chong
Cancers.2020; 12(7): 1884. CrossRef - A bird’s-eye view of deep learning in bioimage analysis
Erik Meijering
Computational and Structural Biotechnology Journal.2020; 18: 2312. CrossRef - Pathomics in urology
Victor M. Schuettfort, Benjamin Pradere, Michael Rink, Eva Comperat, Shahrokh F. Shariat
Current Opinion in Urology.2020; 30(6): 823. CrossRef - Model Fooling Attacks Against Medical Imaging: A Short Survey
Tuomo Sipola, Samir Puuska, Tero Kokkonen
Information & Security: An International Journal.2020; 46(2): 215. CrossRef - Recommendations for pathologic practice using digital pathology: consensus report of the Korean Society of Pathologists
Yosep Chong, Dae Cheol Kim, Chan Kwon Jung, Dong-chul Kim, Sang Yong Song, Hee Jae Joo, Sang-Yeop Yi
Journal of Pathology and Translational Medicine.2020; 54(6): 437. CrossRef - A machine-learning expert-supporting system for diagnosis prediction of lymphoid neoplasms using a probabilistic decision-tree algorithm and immunohistochemistry profile database
Yosep Chong, Ji Young Lee, Yejin Kim, Jingyun Choi, Hwanjo Yu, Gyeongsin Park, Mee Yon Cho, Nishant Thakur
Journal of Pathology and Translational Medicine.2020; 54(6): 462. CrossRef
- Artificial Intelligence and Digital Pathology: Toward Next‐Generation Diagnostic Hematology
- Difference of the Nuclear Green Light Intensity between Papillary Carcinoma Cells Showing Clear Nuclei and Non-neoplastic Follicular Epithelia in Papillary Thyroid Carcinoma
- Hyekyung Lee, Tae Hwa Baek, Meeja Park, Seung Yun Lee, Hyun Jin Son, Dong Wook Kang, Joo Heon Kim, Soo Young Kim
- J Pathol Transl Med. 2016;50(5):355-360. Published online August 22, 2016
- DOI: https://doi.org/10.4132/jptm.2016.05.19
- 8,797 View
- 90 Download
-
 Abstract
Abstract
 PDF
PDF - Background
There is subjective disagreement regarding nuclear clearing in papillary thyroid carcinoma. In this study, using digital instruments, we were able to quantify many ambiguous pathologic features and use numeric data to express our findings.
Methods
We examined 30 papillary thyroid carcinomas. For each case, we selected representative cancer cells showing clear nuclei and surrounding non-neoplastic follicular epithelial cells and evaluated objective values of green light intensity (GLI) for quantitative analysis of nuclear clearing in papillary thyroid carcinoma.
Results
From 16,274 GLI values from 600 cancer cell nuclei and 13,752 GLI values from 596 non-neoplastic follicular epithelial nuclei, we found a high correlation of 94.9% between GLI and clear nuclei. GLI between the cancer group showing clear nuclei and non-neoplastic follicular epithelia was statistically significant. The overall average level of GLI in the cancer group was over two times higher than the non-neoplastic group despite a wide range of GLI. On a polygonal line graph, there was a fluctuating unique difference between both the cancer and non-neoplastic groups in each patient, which was comparable to the microscopic findings.
Conclusions
Nuclear GLI could be a useful factor for discriminating between carcinoma cells showing clear nuclei and non-neoplastic follicular epithelia in papillary thyroid carcinoma.
- Nuclear Image Analysis Study of Neuroendocrine Tumors
- Meeja Park, Taehwa Baek, Jongho Baek, Hyunjin Son, Dongwook Kang, Jooheon Kim, Hyekyung Lee
- Korean J Pathol. 2012;46(1):38-41. Published online February 23, 2012
- DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.1.38
- 8,705 View
- 41 Download
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF Background There is a subjective disagreement about nuclear chromatin in the field of pathology. Objective values of red, green, and blue (RGB) light intensities for nuclear chromatin can be obtained through a quantitative analysis using digital images.
Methods We examined 10 cases of well differentiated neuroendocrine tumors of the rectum, small cell lung carcinomas, and moderately differentiated squamous cell lung carcinomas respectively. For each case, we selected 30 representative cells and captured typical microscopic findings. Using an image analyzer, we determined the longest nuclear line profiles and obtained graph files and Excel data on RGB light intensities. We assessed the meaningful differences in graph files and Excel data among the three different tumors.
Results The nucleus of hematoxylin and eosin-stained tumor cells was expressed as a combination of RGB light sources. The highest intensity was from blue, whereas the lowest intensity was from green. According to the graph files, green showed the most noticeable change in the light intensity, which is consistent with the difference in standard deviations.
Conclusions The change in the light intensity for green has an important implication for differentiating between tumors. Specific features of the nucleus can be expressed in specific values of RGB light intensities.
-
Citations
Citations to this article as recorded by- Difference of the Nuclear Green Light Intensity between Papillary Carcinoma Cells Showing Clear Nuclei and Non-neoplastic Follicular Epithelia in Papillary Thyroid Carcinoma
Hyekyung Lee, Tae Hwa Baek, Meeja Park, Seung Yun Lee, Hyun Jin Son, Dong Wook Kang, Joo Heon Kim, Soo Young Kim
Journal of Pathology and Translational Medicine.2016; 50(5): 355. CrossRef - Comparison of diagnostic accuracy between CellprepPlus® and ThinPrep® liquid‐based preparations in effusion cytology
Yong‐Moon Lee, Ji‐Yong Hwang, Seung‐Myoung Son, Song‐Yi Choi, Ho‐Chang Lee, Eun‐Joong Kim, Hye‐Suk Han, Jin young An, Joung‐Ho Han, Ok‐Jun Lee
Diagnostic Cytopathology.2014; 42(5): 384. CrossRef
- Difference of the Nuclear Green Light Intensity between Papillary Carcinoma Cells Showing Clear Nuclei and Non-neoplastic Follicular Epithelia in Papillary Thyroid Carcinoma
- Computerization of Surgical Pathology Reporting and Data Storage by Automatic Coding System using Personal Computer.
- Woo Ho Kim, Jeong Wook Seo, Yong Il Kim
- Korean J Pathol. 1989;23(4):410-415.
- 1,964 View
- 14 Download
-
 Abstract
Abstract
 PDF
PDF - The authors developed a computer program for use in report printing as well as data storage and retrieval system at the surgical pathology and its efficacy was evaluated at the Department of Pathology, Seoul National University Hospital. This program used IBM PC XT and was written in DBASE III plus language. The main features of the program included an automatic coding and decoding of the diagnosis, automatic searching of the previous biopsy during gross dictation, powerful word processing function and flexibility of the program. The data storage was carried out during the typewriting of the report, so that the typist's workload became markedly reduced. Two kinds of data files wer stored in the hard disk ; the temporary file contained full informations and the permanent file contained the core data only. Searching of a specific case was performed by pathology accession number, chart number, patient's name or by SNOMED code within a second. All the cases were arranged by copied to the diskette during the daily service automatically, with which data were easily restored in case of hard disk failure. The advantages of this program using a persosnal computer were discussed with comparison to those of larger computer system. Based on the experience of 8 months usage in Seoul National University, we assume that this program gives a sufficient solution to the surgical pathology service of many institutes where a large computer system with well designed software is not available yet.
- Image Analysis of Glomerular Changes in Patients with Post-transplant IgA Nephropathy.
- Kye Won Kwon, Hyeon Joo Jeong
- Korean J Pathol. 2001;35(3):206-211.
- 2,131 View
- 18 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
IgA nephropathy after renal transplantation (post-transplant IgAN) may recapitulate the IgAN of native kidneys, however, little has been reported about the histologic characteristics. The aim of this study is to apply glomerular morphometry using an image analyser to examine the histologic characteristics of post-transplant IgAN.
METHODS
The outer margin of the glomerulus (Bowman's area, BA) and glomerular tuft area (GA) were traced manually. The measured area were automatically calculated by KS300 image analysis system (Kontron, Munchen, Germany). The mesangial area (MA) was calculated with a summing each manually traced mesangial area. The total number of glomerular (GC) and mesangial cells (MC) were counted. Eight cases of renal section obtained by nephrectomy due to renal cell carcinoma (normal control: N-CTRL) and nineteen cases of renal section obtained from post-transplantation patients without IgAN (transplantation control: Tx-CTRL) served as controls.
RESULTS
A total of 35 biopsies were finally selected for measurement. BA and GA of post-transplant IgAN were 1.6 and 1.4 times larger than the N-CTRL, respectively, and were not significantly different from Tx-CTRL. MA was 1.4 times significantly larger than that of the Tx-CTRL. As compared to that of the N-CTRL, it was 1.2 times larger, but this difference was not statistically significant. The GC and MC of post-transplant IgAN and the Tx-CTRL were significantly lower than the N-CTRL. There were no significant correlations between glomerular hypertrophy and duration after renal transplantation, mesangial changes, segmental sclerosis, or degree of renal cortical interstitial fibrosis in post-transplant IgAN.
CONCLUSIONS
Prominent glomerular hypertrophy and mesangial expansion suggest a hyperfiltration injury in post-transplant IgAN and a possible way to glomerulosclerosis.
- Development of the Pathology Laboratory Teaching Material Composed of Digital Pictures and Presentation over the Internet.
- Sang Yeop Yi
- Korean J Pathol. 2001;35(4):338-343.
- 1,972 View
- 18 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
To maximize the efficiency of the pathology laboratory class, it has been realized that students should be given an environment with repeated learning situations. For this purpose, this study was designed to present the significance that pathologists need in order to publish teaching materials over the Internet.
METHODS
The pictures were captured as different magnified digital data from teaching glass slides. To modify the images, a graphics program was used, and these were transferred to the Microsoft PowerPoint software and developed as final teaching material. The final teaching material was then published on the Internet. This material can be browsed by searching through windows and by the indices of diagnosis. RESULTS: The comments from all users of the teaching material used in this study showed that it was user-friendly and appropriate for searching and reviewing. The users could assess information easily before and after the laboratory sessions. The quality of the images in this material was appropriate for printing. All users from our university were satisfied with the fact that all pictures were captured from their own teaching slides.
Conclusions
: Therefore, the teaching material used in this study is helpful for medical students studying pathology. Furthermore, this trial may induce others to develop pathology teaching materials over the Internet.
- Computerization of Microscope Slide Labelling.
- Dong Sug Kim, Dae Hong Suh
- Korean J Pathol. 1997;31(7):628-634.
- 1,758 View
- 12 Download
-
 Abstract
Abstract
 PDF
PDF - The automation in surgical pathology, particularly in the reporting and encoding system using personal computers has been greatly improved in recent years, but the computerization of microscope slide labelling has not been improved. The authors have developed database program for slide labelling using FoxPro 2.5 and FoxBASE SCOUNIX 2.1.2: For I year during trial an effect has been put forward to simplify and organize the labelling work in routine surgical pathology. The program is now become easily applicable to the labelling work without disturbing the normal flow in a pathology laboratory. It is possible to get information concerning how many paraffin blocks and H&E slides have been made, as well as what kind of special stains have been requested for each case. The authors think that the computerization of labelling work in routine surgical pathology is a fairly easy task, and this should simplify the labelling work at a lower cost, diminish the workload of a typist or technician, and indirect information concerning the workload in a pathology laboratory can be calculated.
- Computerization of 100,000 cases of Surgical Pathology Data at SNUH by Automatic Coding System using Personal Computer.
- Woo Ho Kim, Ghee Young Choe, Jeong Wook Seo, Yong Il Kim
- Korean J Pathol. 1990;24(4):509-512.
- 1,967 View
- 21 Download
-
 Abstract
Abstract
 PDF
PDF - A computer program using automatic coding of the diagnosis has been used for report printing as well as data storage and retrieval system at the Department of Pathology, Seoul National University Hospital. Previous surgical pathology files were also computerized by the automatic coding system using personal computer, and 100,000 cases of surgical pathology data during 7 years were computerized at present. The cpmputerized surgical files were counted and listed according to topograph and morphologic diagnosis. It is available to print out the list of a specific diagnosis or to copy the records to a floppy diskette. Collection of cases in surgical pathology files using cpmputerized automatic coding system becomes much convenient and accurate than using stored file cards or log books. In addition, previous biopsy records of the patient are automatically searched during the routine work so that understanding of a patient as a whole is possible through the informations about previous pathologic diagnosis. We confirm that automatic coding methods is the most practical and economic method for computerization of the surgical pathology records.
- Korea University Laboratory Data Management and Communication System III-Pathology (KULAB III).
- Kap N Lee, Seung Y Paik
- Korean J Pathol. 1987;21(3):168-175.
- 1,799 View
- 11 Download
-
 Abstract
Abstract
 PDF
PDF - This pathology laboratory data management system is developed at the department of pathology and laboratory medicine, College of Medicine, Korea University, as an extension of Korea University laboratory Data managment and communication system. This is built in hospital computer for business, with the connection to the hospital business information system by software. The pathology department is only using terminals and printers, which enables us to maintain the lowest cost to maintain and operate the system. It has been written in COBO1 and utilized TOTAL Data Base and SNOMED (microglossary for surgical pathology). The input keys include patient hospital registration number, laboratory test code and SNOMED code or direct English written diagnostic words through dual masters of SNOMED.
- Development of a CD-ROM Titled 'Atlas of Pathology' for Medical Students.
- Dong Sug Kim, Yong Jin Kim, Sun Woo Park
- Korean J Pathol. 2000;34(5):374-380.
- 1,870 View
- 11 Download
-
 Abstract
Abstract
 PDF
PDF - The authors have made a CD-ROM titled 'Atlas of Pathology (AP)' for medical students to understand histopathologic findings with ease. We used a 35 mm film scanner to convert an existing file into digital images. A pathologist and two professional programmers collaborated to create the program 'AtlasMaster 1.0' based on (IBM) PC for organization of previously captured digital images. Minimum system requirement for the 'AP' was Pentium II 166 MHz, 32 MB RAM, Windows 95 or 98, 800 600 resolution, 16-bit color, 20 speed CD-ROM drive. The 'AP' was composed of a execution file (Pathology_Atlas.exe), a DB file (pathology.mdb), and reference files (*.bmp, *.jpg, *.txt). The DB file contained fields for chapter, section, disease entity, and information for location of reference files. About 1,000 color images for various kinds of gross and microscopic pictures were stored in the CD-ROM and those were classified according to the chapters, sections, and disease entities. The 'AP' was easy to manipulate, and had advantages of self-learning for students. It could be applied to other fields in which many images were dealt with, such as histology, radiology, endoscopy, dermatology, and plastic surgery. The 'AP' was handy and very useful for medical students to study pathology and it would be a powerful self-learning tool.
- Image Standardization and Determination of Gray Level Threshold in the Assessment of the Myocardial Fibrosis by the Computerized Image Analysis.
- Nam Young Lee, Young Sik Park, Jin Haeng Chung, Jeong Wook Seo
- Korean J Pathol. 1998;32(7):494-503.
- 2,075 View
- 10 Download
-
 Abstract
Abstract
- The computerized image analysis is a useful tool for the quantitative assessment of histopathologic findings. In contrast to the usual microscopic examination by pathologists, the computerization should be accompanied with the standardization process of the image. We developed an algorithm to standardize images and to determine the optimal gray level threshold, using a myocardial fibrosis model. Sirius red staining was more convenient for the image analysis than Masson's trichrome staining because of a better contrast with the surrounding structures. To get an optimal measurement, light intensity was standardized at each of the fibrosis, myocardium and background. In this study, the most promising method to determine the degree of fibrosis was that of revising the background without tissue to a gray level of 200, obtaining a green component of the color image, revising the myocardial fiber to 163, and defining a partial ratio as fibrosis index when the gray level threshold was 120. These threshold levels and parameters were determined after drawing the binarization index curves according to the change of the gray level threshold and by the morphological examination of the actual binarization figures overlaid to the original color image. Through these processes we could get a consistent result on the myocardial fibrosis and we expect a similar principle applies when we analyze color images in the histopathologic quantitation by computerized image analysis.
- Pathologic Diagnoses Management System "Pathology Slide Bank" Using IBM-PC Microcomputer.
- Yong Koo Park, Moon Ho Yang, Won Tae Lee, Joo Young Park
- Korean J Pathol. 1988;22(3):285-288.
- 1,929 View
- 12 Download
-
 Abstract
Abstract
 PDF
PDF - A simple and convenient management system in coding and storage of the pathologic diagnoses was developed by generating the dBASE III with IBM-PC microcomputer. The program named "Pathology Slide Bank" can store about 3,600 data according to the SNOP coding in a standard 51/4 inch floppy disk and retrieve the data within two minutes.
- Computerization of Surgical Pathology Reporting by Personal Computer.
- Dong Sug Kim, Young Ran Shim, Mee Jin Kim, Hae Joo Nam, Won Hee Choi, Tae Sook Lee
- Korean J Pathol. 1992;26(2):146-153.
- 1,971 View
- 14 Download
-
 Abstract
Abstract
 PDF
PDF - The authors have been developed a menu-driven FoxBASE system for surgical pathology reporting and automatic encoding in Systematized Nomenclature of Medicine. The system requires no prior knowledge of FoxBASE and is readily installed on any IBM or it's compatible personal computer. Working sheet generation is automatically accompanied by data from previous cases on the same patient. Important data which include patient name, age, sex, surgical number, hospital unit number and encoded diagnoses, are stored on the hard disk permanently; complete reports are saved on floppy diskettes. Cases can be retrieved by patient name, surgical number, hospital unit number and SNOMED codes within 0.1 second. Daily work lists and listings of incomplete cases are easily obtained. This FoxBASE system has been in use for 1 year and 6 months and resulted in increased efficiency of retrieval and gathering of basic information for specific study, cost effectiveness, markedly diminished workload of typist and very short wasting time during complete restoration of data file for hard disk failure.

 E-submission
E-submission


 First
First Prev
Prev



