Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 48(5); 2014 > Article
-
Brief Case Report
One Case of a BRCA1 Germ Line Mutation Ovarian Carcinoma Patient Based on Abnormal Immunohistochemistry Finding - Hyun Joo Kim, Jung Min Park, Hyoun Wook Lee1, Eun Hee Lee1, Min Kyu Kim2
-
Korean Journal of Pathology 2014;48(5):379-381.
DOI: https://doi.org/10.4132/KoreanJPathol.2014.48.5.379
Published online: October 27, 2014
Department of Obstetrics and Gynecology, Samsung Medical Center, Seoul, Korea
1Department of Pathology, Samsung Changwon Hospital, Sungkyunkwan University School of Medicine, Changwon, Korea
2Division of Gynecologic Oncology, Department of Obstetrics and Gynecology, Samsung Changwon Hospital, Sungkyunkwan University School of Medicine, Changwon, Korea
- Corresponding Author: Min Kyu Kim, M.D. Division of Gynecologic Oncology, Department of Obstetrics and Gynecology, Samsung Changwon Hospital, Sungkyunkwan University School of Medicine, 158 Paryong-ro, Masanhoewon-gu, Changwon 630-723, Korea Tel: +82-55-290-6040, Fax: +82-2-6442-9285, E-mail: minkyukim@skku.edu
• Received: July 31, 2013 • Revised: October 2, 2013 • Accepted: October 17, 2013
© 2014 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 8,470 Views
- 33 Download
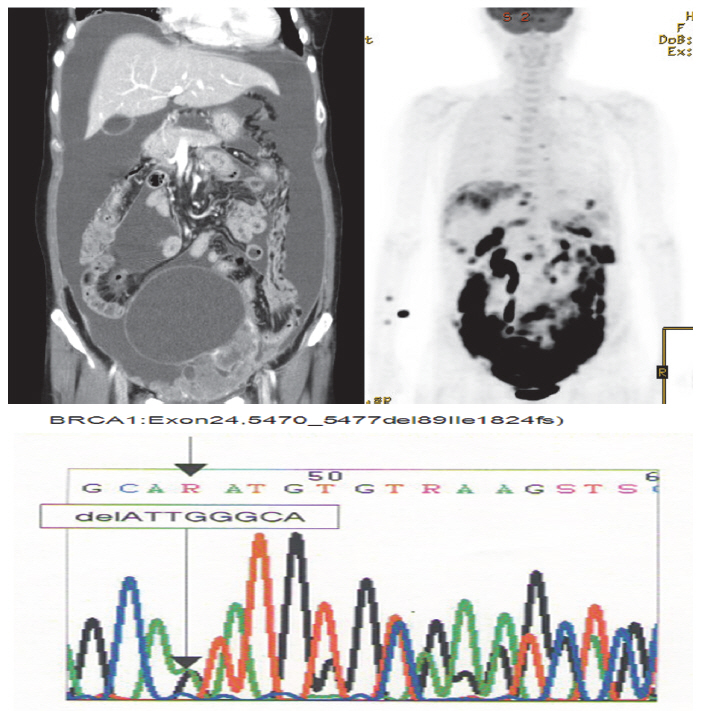
- A 52-year-old woman complaining of constipation and abdominal distension was referred to our hospital from a local clinic. Computerized axial tomography scans showed a 12-cm solid and cystic ovarian mass with a large amount of ascites and right pleural effusion (Fig. 1). Her cancer antigen-125 level was 432.4 U/mL. After other laboratory tests were performed, we conducted a staging exploratory laparotomy. Pathology showed grade 3, stage IV serous papillary adenocarcinoma from the ovary invading the pelvic cavity and right pleura. The patient underwent adjuvant taxane and carboplatin combination chemotherapy via six cycles of paclitaxel (175 mg/m2) plus carboplatin of the area under the curve of 5 mg given on day one of a 21-day cycle.
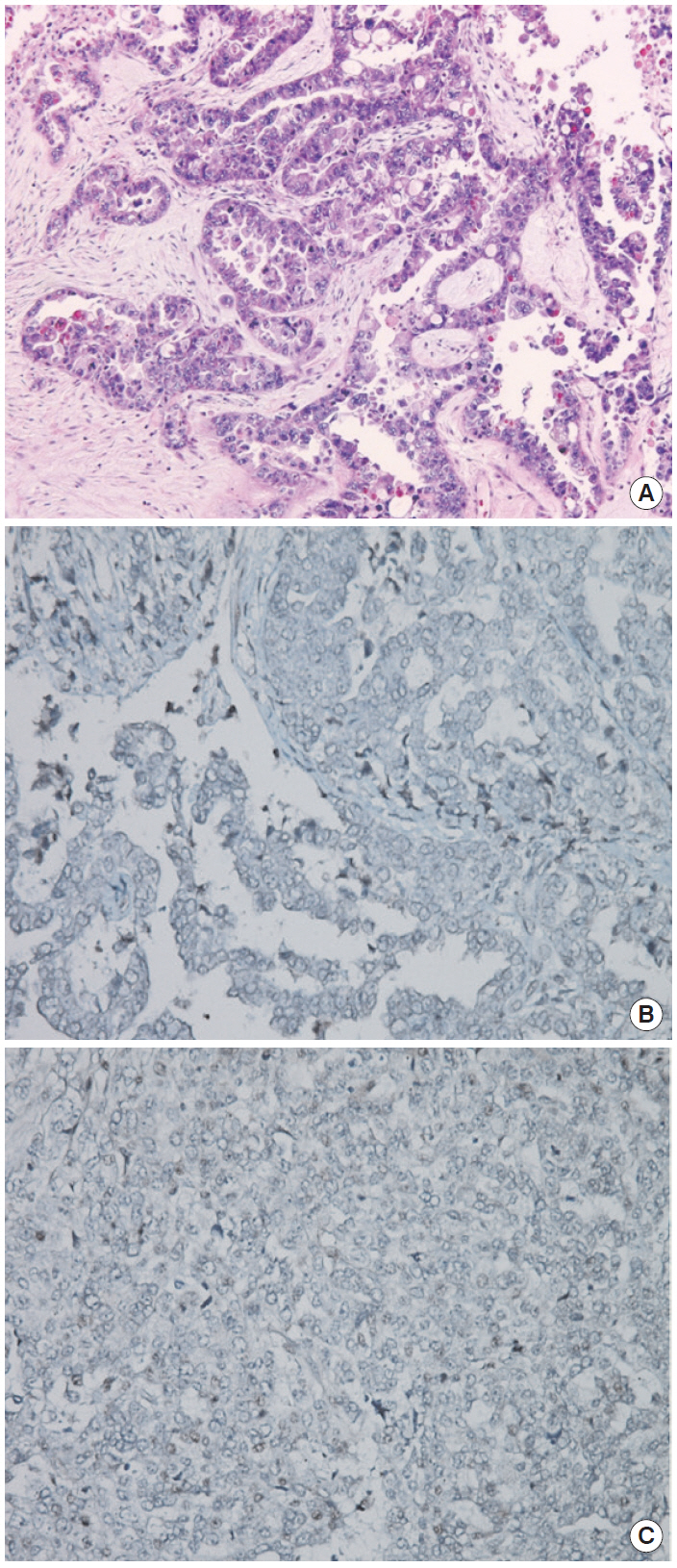
- The patient declined to discuss her family history and her family members were not contacted for further information. However, she approved genetic testing after counseling. Immunohistochemistry (IHC) screening of the mismatch repair genes BRCA1 and BRCA2 was performed, showing abnormal immunoreactivity for BRCA1 (negative staining) and focal positive reaction for BRCA2 (Fig. 2). Gene sequencing for BRCA1 and BRCA2 was conducted after counseling and a deletion of ATTGGGCA at codon 1824 in exon 24 was detected, with the stop codon (TGA) appearing in 1826 [5470_ 5477del8 (p.Ile1824-AspfsX3)]. This frame shift mutation produced a truncated protein (Fig. 1).
CASE REPORT
- This case demonstrates that IHC screening can be helpful for detecting BRCA mutations in sporadic ovarian cancer patients with unknown family histories. We retrospectively confirmed a germ line mutation after observing abnormal findings on IHC for BRCA. Unfortunately, we could not persuade the patient’s family members to undergo genetic testing for the mutation.
- The frequency of BRCA1 or BRCA2 mutations in general population is estimated to be 1 in 300 to 1 in 800[7]. There are many types of BRCA mutations with differences in penetration. Such mutations have previously been reported in Korean breast cancer patients, but not in patients with ovarian carcinoma[8].
- Although progress in molecular diagnosis has assisted in cancer diagnosis[9], the use of vague guidelines for selecting patients for genetic testing, high cost, and lack of insurance coverage make it difficult to detect hereditary cancer patients.
- After diagnosis, three methods are commonly applied to manage BRCA mutation carriers: screening, prophylactic surgery, and chemoprevention. Our patient and her family were offered genetic counseling by gynecologic oncologist and breast cancer surgeon. Her breast mammogram and sonography showed negative results.
- The average lifetime risk of developing breast and ovarian cancer in women with a BRCA1 mutation are 65% and 39%, respectively[5]. The penetrance of ovarian cancer in BRCA1 mutation carriers is known to be 11% to 23%[10]. The patient was offered counseling on breast cancer risks and prevention.
- As our abilities to recognize genetic risks improve, patients with an inherited risk of ovarian carcinoma can be identified before cancer onset, rendering effective prevention of ovarian carcinoma associated with inherited risks. IHC screening can help diagnose BRCA mutations, and clinicians should recommend genetic sequencing in patients with unknown family histories of disease.
DISCUSSION
Fig. 2.(A) Hematoxylin and eosin staining of sample. (B) BRCA1 immunohistochemistry (IHC) (negative staining). (C) BRCA2 IHC (focal positive staining). Agent: BRCA1 (Abcam ab16870), BRCA2 (Abcam ab110967). Evaluation: Nuclear staining of the tumor is scored as either 2 (strong, normal), 1 (weak), or 0 (negative, abnormal) compared with the corresponding internal control.

- 1. Lynch HT, Casey MJ, Snyder CL, et al. Hereditary ovarian carcinoma: heterogeneity, molecular genetics, pathology, and management. Mol Oncol 2009; 3: 97-137. ArticlePubMedPMCPDF
- 2. Miki Y, Swensen J, Shattuck-Eidens D, et al. A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science 1994; 266: 66-71. PubMed
- 3. Wooster R, Bignell G, Lancaster J, et al. Identification of the breast cancer susceptibility gene BRCA2. Nature 1995; 378: 789-92. ArticlePubMedPDF
- 4. Kang YK, Kim WH. Microsatellite instability and mismatch repair protein (hMLH1, hMSH2) expression in intrahepatic cholangiocarcinoma. Korean J Pathol 2005; 39: 9-14.
- 5. Antoniou A, Pharoah PD, Narod S, et al. Average risks of breast and ovarian cancer associated with BRCA1 or BRCA2 mutations detected in case series unselected for family history: a combined analysis of 22 studies. Am J Hum Genet 2003; 72: 1117-30. ArticlePubMedPMC
- 6. Kauff ND, Satagopan JM, Robson ME, et al. Risk-reducing salpingo-oophorectomy in women with a BRCA1 or BRCA2 mutation. N Engl J Med 2002; 346: 1609-15. ArticlePubMed
- 7. Whittemore AS, Gong G, Itnyre J. Prevalence and contribution of BRCA1 mutations in breast cancer and ovarian cancer: results from three U.S. population-based case-control studies of ovarian cancer. Am J Hum Genet 1997; 60: 496-504. PubMedPMC
- 8. Kim H, Cho DY, Choi DH, et al. Characteristics and spectrum of BRCA1 and BRCA2 mutations in 3,922 Korean patients with breast and ovarian cancer. Breast Cancer Res Treat 2012; 134: 1315-26. ArticlePubMedPDF
- 9. Hwang TS. Molecular biologic techniques in cytopathologic diagnosis. Korean J Pathol 2009; 43: 387-92. Article
- 10. Eisen A, Lubinski J, Klijn J, et al. Breast cancer risk following bilateral oophorectomy in BRCA1 and BRCA2 mutation carriers: an international case-control study. J Clin Oncol 2005; 23: 7491-6. ArticlePubMed
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
One Case of a BRCA1 Germ Line Mutation Ovarian Carcinoma Patient Based on Abnormal Immunohistochemistry Finding
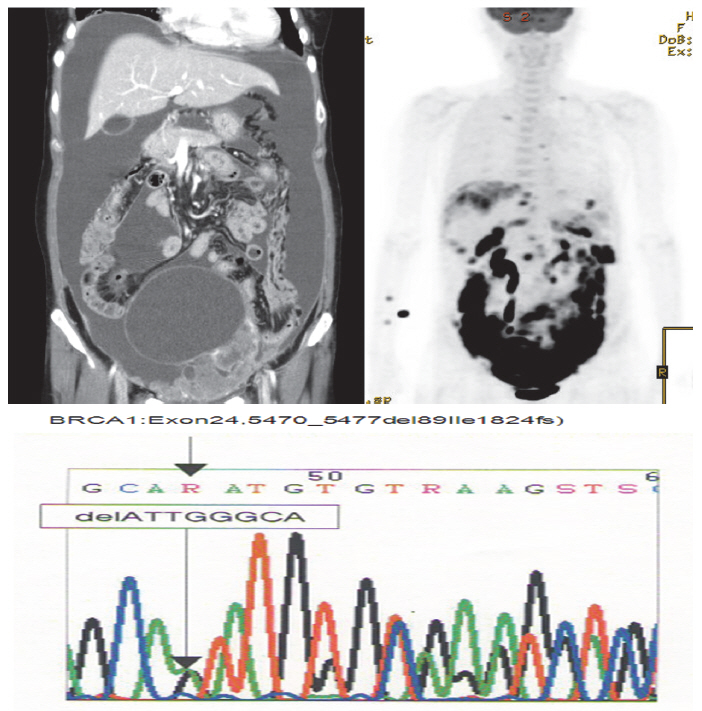
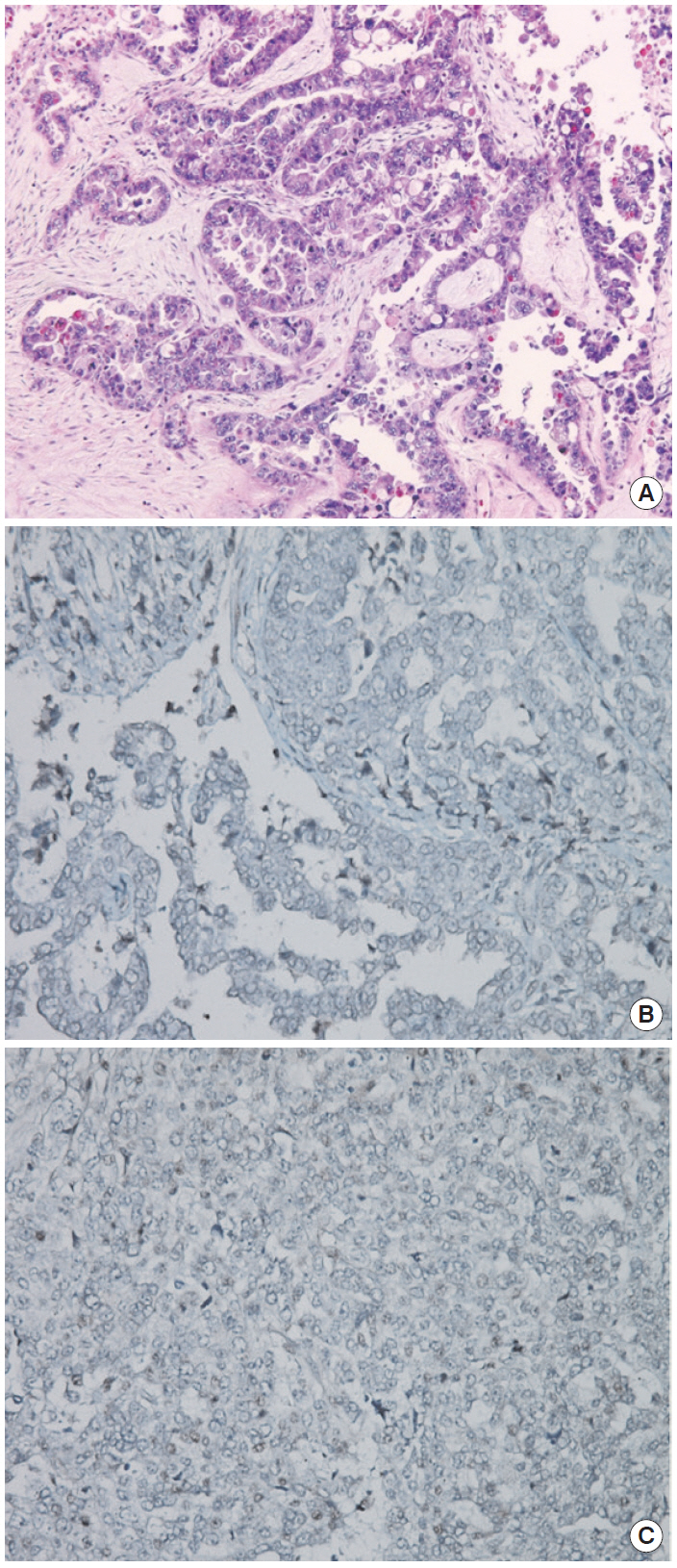
Fig. 1. Computerized axial tomography scan and gene sequencing results.
Fig. 2. (A) Hematoxylin and eosin staining of sample. (B) BRCA1 immunohistochemistry (IHC) (negative staining). (C) BRCA2 IHC (focal positive staining). Agent: BRCA1 (Abcam ab16870), BRCA2 (Abcam ab110967). Evaluation: Nuclear staining of the tumor is scored as either 2 (strong, normal), 1 (weak), or 0 (negative, abnormal) compared with the corresponding internal control.
Fig. 1.
Fig. 2.
One Case of a BRCA1 Germ Line Mutation Ovarian Carcinoma Patient Based on Abnormal Immunohistochemistry Finding

 E-submission
E-submission