Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(6); 2013 > Article
-
Case Study
Diffuse Large B-Cell Lymphoma Arising in Warthin's Tumor: Case Study and Review of the Literature - Güliz Özkök, Funda Taşlı, Nazan Özsan1, Rafet Öztürk, Hakan Postacı
-
Korean Journal of Pathology 2013;47(6):579-582.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.6.579
Published online: December 24, 2013
Department of Pathology, Izmir Bozyaka Training and Research Hospital, Izmir, Turkey.
1Department of Pathology, Ege University School of Medicine, Izmir, Turkey.
- Corresponding Author: Funda Taşlı, M.D. Department of Pathology, Izmir Bozyaka Training and Research Hospital, Saim Çıkrıkçı Cad. No. 59 Bozyaka, İzmir, Turkey. Tel: +90-232-2505050, Fax: +90-232-2614444, fundadr@gmail.com
• Received: June 1, 2012 • Revised: September 15, 2012 • Accepted: September 17, 2012
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- Warthin's tumor is the second most common type of salivary gland tumor. Microscopically, Warthin's tumor displays a proliferative epithelial component and lymphoid stroma. Carcinomas arising from the epithelial component are well known, but malignant transformations of the lymphoid stroma are rare. When they do occur, they are most commonly B-cell type non-Hodgkin lymphomas. A 60-year-old male patient underwent surgical resection of a parotid mass. After superficial parotidectomy, microscopic examination indicated that the tumor was of epithelial components with basaloid and oncocytic columns of cells neighboring lymphoid components. In addition to the lymphoid follicles with distinct germinal centers, there were large, bizarre and extremely atypical neoplastic cells seen in the lymphoid component. Large neoplastic cells were diffusely CD20 and CD30 positive. The patient was diagnosed with "Warthin's tumor and diffuse large B-cell lymphoma with expression of CD30." The histopathologic and clinical features are discussed along with a review of the literature.
- A 60-year-old male patient was referred to an otorhinolaryngology clinic due to a lump on the left side of his jaw, which had grown in 2 months. Ultrasound sonography test examination revealed a cystic mass that was 24×17 mm in size with smooth contours. Multiple echogenic and reactive lymph nodes with partially visible hila were visualized in the neighboring upper jugular chain, with the largest being 16×10 mm in size. Following a neck magnetic resonance imaging and a preliminary diagnosis of WT, a left superficial parotidectomy was conducted. The patient had a history of smoking, hypertension, and coronary artery disease.
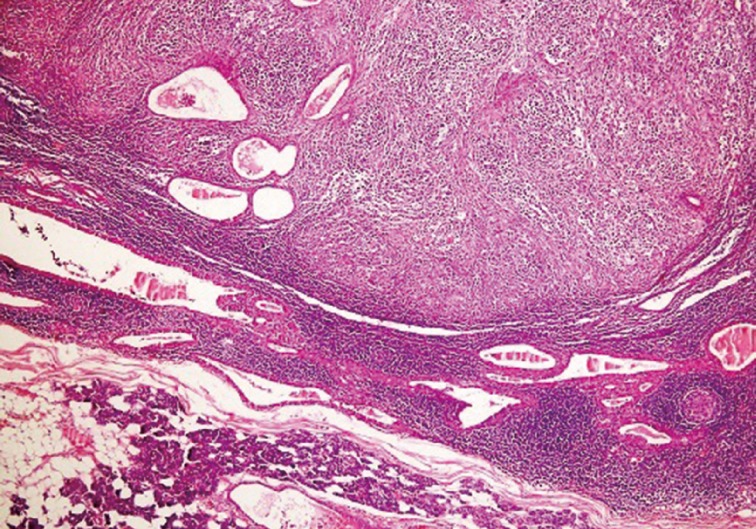
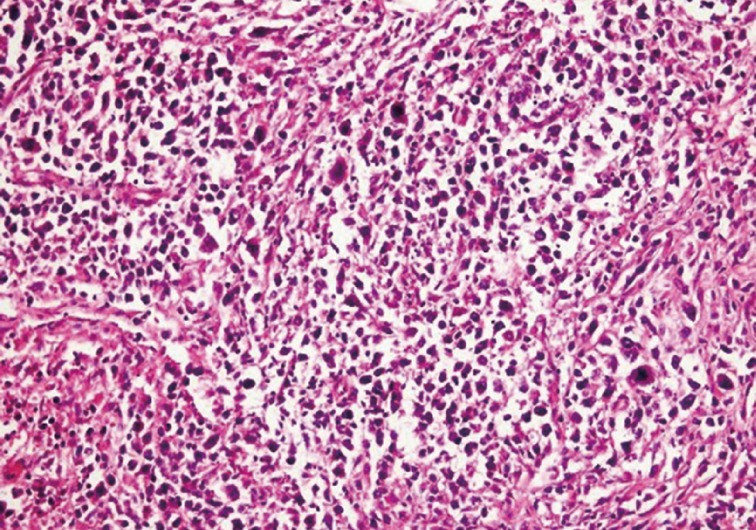
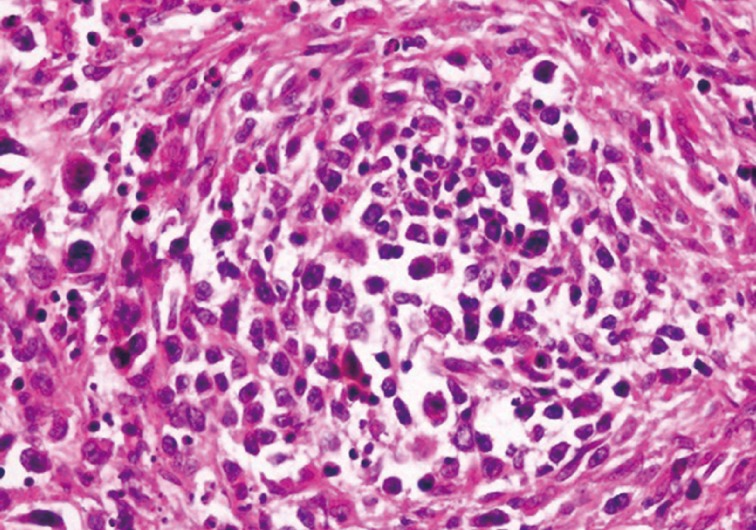
- Left superficial parotidectomy materials were sent for pathologic examination in two pieces, which were 5×3.2×2 cm and 4.5×3×1.2 cm in size. Cross section analysis showed an off white-yellowish, well-contoured nodular tumor with a bleeding center of 4×2.5×2.2 cm. Microscopic examination indicated that the tumor had epithelial components with basaloid and oncocytic columns of cells neighboring lymphoid components (Fig. 1). In addition to the lymphoid follicles with distinct germinal centers, infiltration of large neoplastic cells with bizarre and extremely atypical morphology was seen in the lymphoid component (Figs. 2, 3). The large neoplastic lymphoid cells constituted more than 50% percent of the lymphoid component.
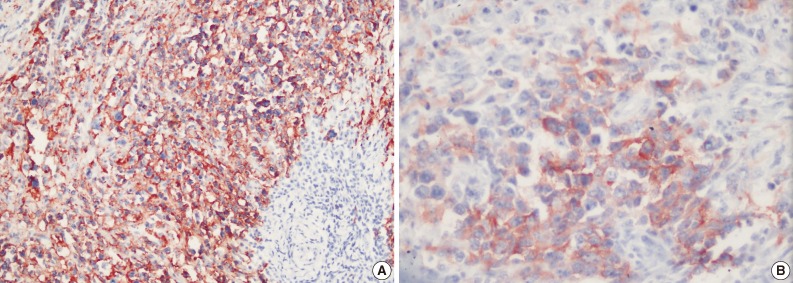
- Immunohistochemical examination showed that neoplastic cells expressed strong positivity for CD20 (Fig. 4A), CD79a, CD30 (Fig. 4B), leukocyte common antigen, IgG, CD138, MUM1, and focal positivity for kappa. Staining for lambda, IgM, IgA, CD3, CD5, CD10, CD15, CD56, epithelial membrane antigen, Bcl2, Bcl6, cyclinD1, S100, pancytokeratin, cytokeratin 20, human melanoma black 45, actin, and desmin were negative. Latent Epstein-Barr virus (EBV) was shown to be negative in tumor cells by using EBV-encoded RNA chromogenic in situ hybridization. Due to these findings, the patient was diagnosed with "WT and CD30 positive diffuse large B-cell lymphoma in the parotid gland."
- Following the lymphoma diagnosis, a full body screen was performed. Results indicated lymphadenopathies of a pathologic size in the inguinal and iliac regions. In addition to these findings, the left suprarenal gland showed two nodular mass lesions, which were assessed as likely adenomas; however, this preliminary diagnosis was not confirmed by histopathology. Bone marrow biopsy revealed a normocellular bone marrow with no lymphoma involvement.
- The patient was stage 3A and received six courses of rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) therapy. During 6-month follow-up, the patient was free of disease.
CASE REPORT
- WT is the second most common type of salivary gland tumor. In 10-15% of cases, it is bilateral, and it accounts for 70% of all bilateral salivary gland tumors.2 The male/female ratio is 1.6/1, and it typically develops in the 6th and 7th decades. Smoking increases the risk of developing WT.5 Microscopically the tumors are typically composed of proliferative epithelial components accompanied by lymphoid stroma with lymphoid follicles that have distinct germinal centers. Histogenesis of the lymphoid stroma in WT has been a topic of discussion for many years.
- Lymphoid stroma can arise as a cell response to epithelial neoplasms or as a normal lymph node due to residue held by the epithelial neoplasm.5,6 The most widely accepted hypothesis suggests that WT is a neoplasm that develops in the heterotopic salivary gland ductus within or around the parotid lymph nodes.7 Transformation to carcinoma in WT is a well-known phenomenon; however, the development of lymphomas from WTs is very rare.4,8 Although some cases contain a normal residual lymphoid component, in others cases the lymphoid component contains entirely neoplastic lymphoid cells.4 In the present case, non-neoplastic lymphoid tissue was also present in the neighboring areas.
- The pathogenesis of malignant transformation of WT remains unclear; however, exposure to radiation is of particular interest, as the relationship between previous radiotherapy and lymphomas arising from WTs has been determined by some authors.4,5,9
- Chronic immune sialadenitis is thought to play an important role, independent of the presence of Sjögren syndrome symptoms.4,7,10 In this case, there was no history of radiotherapy or sialadenitis, but a history of smoking may have provoked the development of WT.
- Saxena et al.1 state that because the lymphoid stroma of WT is part of the systemic lymphoid tissue, in patients with lymphomatous spread of WT, disseminated disease is present during the staging either at the time of the diagnosis or after.
- In the present case, bone marrow biopsy showed no disease involvement. With screening techniques, lymphadenopathies of a pathologic size were found in the inguinal and iliac regions. This indicates that disseminated disease may have been present synchronously.
- Some researchers suggested that although the relationship between WT and lymphoma could be coincidental, it might also be of a pathogenic nature. According to the latter statement, a single agent can affect different tissues or one tumor could trigger the formation of another. From this point of view, the epithelial component is a continuous antigenic stimulator for the lymphoid component, which provides the stimulus for the development of lymphoma.1,6,8 According to this theory, the frequently observed reactive follicular hyperplasia in WT may be histological evidence of chronic antigen stimulation.1
- It has been suggested that the lymphomas seen with WT are typically non-Hodgkin lymphomas; however, there are a few cases reporting Hodgkin's lymphomas.11,12 The majority of non-Hodgkin's lymphomas in WT are follicular lymphomas. DLBCL, small lymphocytic lymphoma, extranodal marginal zone lymphoma of mucosa associated lymphoid tissue, and mantle cell lymphoma have also been reported.4,6,8,9 A small number of T-cell lymphomas such as peripheric T-cell lymphoma and T-cell lymphoblastic lymphoma have also been described in WT.4,8,13
- In summary, malignant lymphomas in WT are very rare. The presented case is a diffuse large B-cell lymphoma expressing CD30 positivity. To the best of our knowledge this is the first case in literature describing DLBCL with expression of CD30 in WT.
DISCUSSION
- 1. Saxena A, Memauri B, Hasegawa W. Initial diagnosis of small lymphocytic lymphoma in parotidectomy for Warthin tumour, a rare collision tumour. J Clin Pathol 2005; 58: 331-333. ArticlePubMedPMC
- 2. Rosai J. Rosai and Ackerman's surgical pathology. 10th ed. Edinburgh: Elsevier, 2011; 826-828.
- 3. Cheuk W, Chan JK. Salivary gland tumors. In: Fletcher CD, ed. Diagnostic histopathology of tumors. 3rd ed. Philadelphia: Churchill Livingstone Elsevier, 2007; 266-269.
- 4. Gorai S, Numata T, Kawada S, Nakano M, Tamaru J, Kobayashi T. Malignant lymphoma arising from heterotopic Warthin's tumor in the neck: case report and review of the literature. Tohoku J Exp Med 2007; 212: 199-205. ArticlePubMed
- 5. Simpson RH, Eveson JW. Warthin tumour. In: Barnes L, Eveson JW, Reichart P, Sidransky D, eds. Pathology and genetics head and neck tumours: World Health Organization classification of tumours. Lyon: IARC Press, 2005; 263-265.
- 6. Park CK, Manning JT Jr, Battifora H, Medeiros LJ. Follicle center lymphoma and Warthin tumor involving the same anatomic site: report of two cases and review of the literature. Am J Clin Pathol 2000; 113: 113-119. PubMed
- 7. Ellis GL. Lymphoid lesions of salivary glands: malignant and benign. Med Oral Patol Oral Cir Bucal 2007; 12: E479-E485. PubMed
- 8. Giaslakiotis K, Androulaki A, Panagoulias G, et al. T cell lymphoblastic lymphoma in parotidectomy for Warthin's tumor: case report and review of the literature. Int J Hematol 2009; 89: 359-364. ArticlePubMedPDF
- 9. Song K, Cotelingam JD, Lowery-Nordberg M, Sun W. Characterization of the lymphoid stroma in Warthin's tumor of salivary gland by immunohistochemistry, heavy chain gene and Bcl-2 gene rearrangement. Am J Transl Res 2009; 1: 352-357. PubMedPMC
- 10. Alvarez-Buylla Blanco M, Martínez Moran A, Vázquez Barro JC, Vidal JM. Primary non-Hodgkin lymphoma of the parotid gland: revision of 8 cases. Acta Otorrinolaringol Esp 2010; 61: 371-374. ArticlePubMed
- 11. Melato M, Falconieri G, Fanin R, Baccarani M. Hodgkin's disease occurring in a Warthin's tumor: first case report. Pathol Res Pract 1986; 181: 615-620. ArticlePubMed
- 12. Badve S, Evans G, Mady S, Coppen M, Sloane J. A case of Warthin's tumour with coexistent Hodgkin's disease. Histopathology 1993; 22: 280-281. ArticlePubMed
- 13. Pescarmona E, Perez M, Faraggiana T, Granati L, Baroni CD. Nodal peripheral T-cell lymphoma associated with Warthin's tumour. Histopathology 2005; 47: 221-222. ArticlePubMed
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- From the archives of MD Anderson Cancer Center: Small lymphocytic lymphoma/chronic lymphocytic leukemia diagnosed in Warthin tumor
Jing Zhou, L. Jeffrey Medeiros
Annals of Diagnostic Pathology.2025; 79: 152533. CrossRef - Warthin’s Tumor Involved by Monoclonal B-Cell Lymphocytosis: A Case Report
Alexie Kindy, Ziang Wang, Robin Gautam, Robert Bennett, Zijian Wang
Cureus.2025;[Epub] CrossRef - Warthin tumor concomitant with mantle cell lymphoma: a case report and review of literature
Hai-Chao Tong, Shuang Ma, Lan Chen, Xiangyun Meng, Ying-Chun Li, Le-Yao Li, Lingyun Dong, Wan-Lin Zhang, Tyler Wildes, Lian-He Yang, Endi Wang
Diagnostic Pathology.2024;[Epub] CrossRef - A Rare Diagnosis of Parotid Gland Follicular Lymphoma Arising in Warthin Tumor: Case Report and Literature Review
Ido Vaknin, Irit Allon, Shirley Zafrir-Haver, Alex Abramson
Medicina.2024; 60(12): 2086. CrossRef - Warthin’s Tumor of the Parotid Gland With Degeneration to Diffuse Large B-cell Lymphoma: A Case Report and Review of Literature
Mauricio Gutierrez-Alvarez, Cynthia Martinez, Ana Priscila Campollo Lopez, Kevin Fuentes, Jorge Alberto Robles Aviña
Cureus.2023;[Epub] CrossRef - Pathogenesis Analysis of Salivary Gland Tumors Through the Expression of Programmed Death-Ligand 1 (PD-L1)
Aisyah Izzatul Muna, Maria Evata Krismawati Surya, Meiske Margaretha, Jane Kosasih, Mei Syafriadi
Indian Journal of Otolaryngology and Head & Neck Surgery.2023; 75(4): 3098. CrossRef - Role of cytotoxic T-lymphocyte antigen 4 (CTLA-4) expression in the pathogenesis of Warthin’s tumor growth
Alvionika Nadyah Qotrunnada, Tecky Indriana, Jane Kosasih, Meiske Margaretha, Mei Syafriadi
Dental Journal (Majalah Kedokteran Gigi).2022; 55(4): 194. CrossRef - Lymphoepithelial Carcinoma of Salivary Glands
Lester D.R. Thompson, Rumeal D. Whaley
Surgical Pathology Clinics.2021; 14(1): 75. CrossRef - Follicular Lymphoma Diagnosed in Warthin Tumor: A Case Report and Review of the Literature
Fnu Alnoor, Jatin S. Gandhi, Matthew K. Stein, Joel F. Gradowski
Head and Neck Pathology.2020; 14(2): 386. CrossRef - Prevalence of Lymphoid Neoplasia in a Retrospective Analysis of Warthin Tumor: A Single Institution Experience
F. N. U. Alnoor, Jatin S. Gandhi, Matthew K. Stein, Jorge Solares, Joel F. Gradowski
Head and Neck Pathology.2020; 14(4): 944. CrossRef - A Case of Diffuse Large B Cell Lymphoma in Submandibular Gland
Sang Hoo Park, Min Gyoung Pak, Dong Kun Lee
Journal of Clinical Otolaryngology Head and Neck Surgery.2020; 31(1): 111. CrossRef - Analysis of the Clinical Relevance of Histological Classification of Benign Epithelial Salivary Gland Tumours
Henrik Hellquist, António Paiva-Correia, Vincent Vander Poorten, Miquel Quer, Juan C. Hernandez-Prera, Simon Andreasen, Peter Zbären, Alena Skalova, Alessandra Rinaldo, Alfio Ferlito
Advances in Therapy.2019; 36(8): 1950. CrossRef - Diffuse large B-cell lymphoma arising from follicular lymphoma with warthin’s tumor of the parotid gland - immunophenotypic and genetic features: A case report
Chang-Song Wang, Xia Chu, Di Yang, Lei Ren, Nian-Long Meng, Xue-Xia Lv, Tian Yun, Yan-Sha Cao
World Journal of Clinical Cases.2019; 7(22): 3895. CrossRef - Presentation of Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma in a Warthin Tumor: Case Report and Literature Review
Hadeel Jawad, Peter McCarthy, Gerard O’Leary, Cynthia C. Heffron
International Journal of Surgical Pathology.2018; 26(3): 256. CrossRef - Bilateral Warthin tumor in psoriatic patients in therapy with multiple immunosuppressive therapy
M Burlando, E Cozzani, C Chinazzo, M larosa, M Boggio, A Parodi
International Journal of Immunopathology and Pharmacology.2015; 28(1): 138. CrossRef - Nodular Lymphocyte-Predominant Hodgkin Lymphoma in a Warthin Tumor of the Parotid Gland
Arianna Di Napoli, Giuseppe Mallel, Armando Bartolazzi, Elena Cavalieri, Roberto Becelli, Claudia Cippitelli, Luigi Ruco
International Journal of Surgical Pathology.2015; 23(5): 419. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
Diffuse Large B-Cell Lymphoma Arising in Warthin's Tumor: Case Study and Review of the Literature
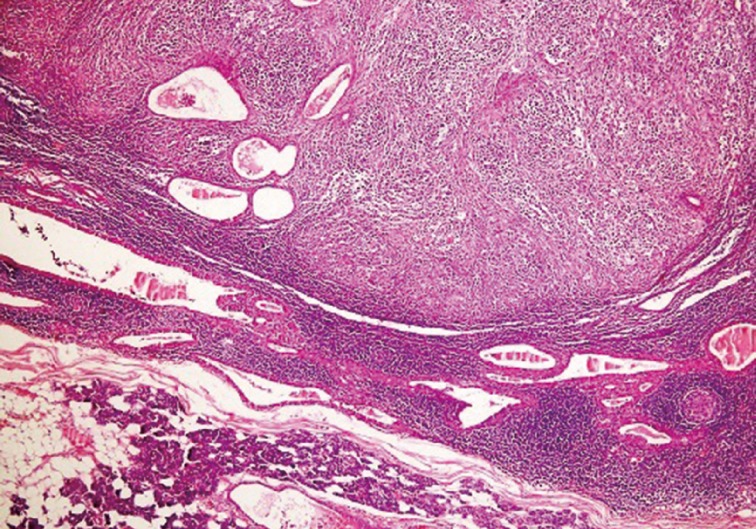
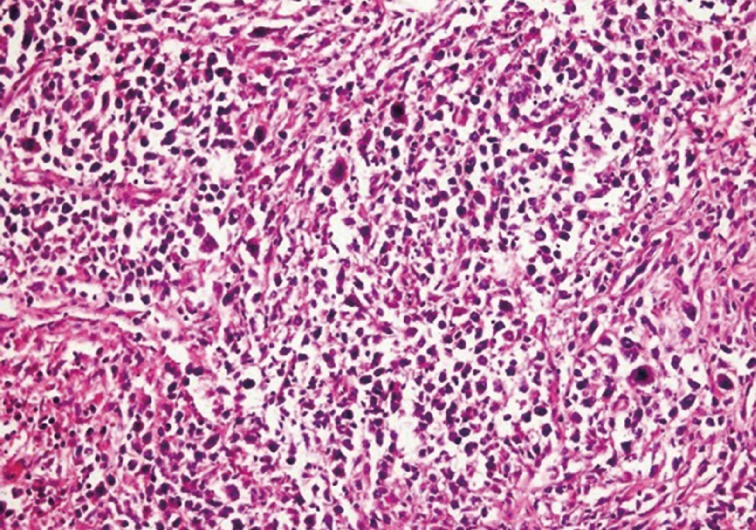
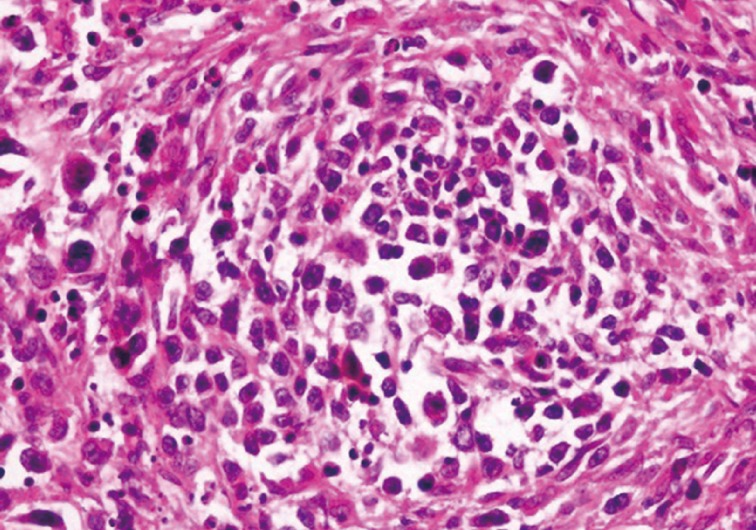
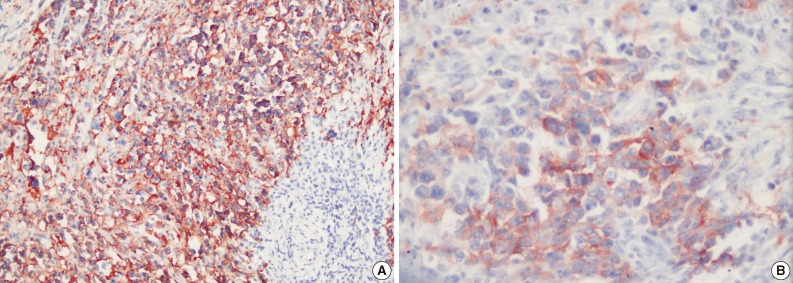
Fig. 1 Cystic Warthin's tumor epithelium surrounded by lymphoma next to normal salivary gland tissue.
Fig. 2 Diffuse infiltration of the stroma by large atypical lymphoid cells with irregular size.
Fig. 3 Neoplastic lymphoid cells display scanty cytoplasm, large nucleolus, and prominent nucleoli. Atypical mitotic figures are also seen.
Fig. 4 CD20 positivity in tumor cells (A) and CD30 positivity in tumor cells (B).
Fig. 1
Fig. 2
Fig. 3
Fig. 4
Diffuse Large B-Cell Lymphoma Arising in Warthin's Tumor: Case Study and Review of the Literature

 E-submission
E-submission