Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(6); 2013 > Article
-
Case Study
Development of Six Tumors in a Sebaceus Nevus of Jadassohn: Report of a Case - Serap Gozel1, Melahat Donmez1, Noyan Can Akdur1, Hulya Yikilkan2,3
-
Korean Journal of Pathology 2013;47(6):569-574.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.6.569
Published online: December 24, 2013
1Department of Pathology, Ankara Training and Research Hospital, Ankara, Turkey.
2Department of Family Medicine, Diskapi Yildirim Beyazit Training and Research Hospital, Ankara, Turkey.
3Department of Family Medicine, University of Antwerp, Antwerp, Belgium.
- Corresponding Author: Hulya Yikilkan, M.D. Department of Family Medicine, Diskapi Yildirim Beyazit Training and Research Hospital, Kasalar, 06010 Ankara, Turkey. Tel: +90-312-3260010 (ext. 1477), Fax: +90-312-3230632, hulyayikilkan@hotmail.com
• Received: October 15, 2011 • Revised: February 2, 2012 • Accepted: February 6, 2012
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- Nevus sebaceus of Jadassohn is a congenital cutaneous hamartoma comprised of multiple skin structures. It has the potential to develop into variety of neoplasms of various epidermal adnexal origins. While multiple tumors may occasionally arise, it is unusual for more than four tumors to arise simultaneously within a single sebaceus nevus. Here in, we report a case of a 70-year-old woman with six neoplastic proliferations including a syringocystadenoma papilliferum, pigmented trichoblastoma, tubular apocrine adenoma, sebaceoma, tumors of follicular infundibulum and superficial epithelioma with sebaceus differentiation arising in a long standing nevus sebaceus on the scalp. Our case is extraordinary because a single nevus sebaceus contained six neoplastic proliferations with differentiation toward the folliculosebaceous-apocrine unit.
- A 70-year-old woman presented to our outpatient clinic with a verrucous growth on her scalp measuring 2.5×1.8 cm. The lesion presented at birth. While she was unaware of any specific changes in the lesion for decades, she noticed a rapid progression in the last two years. She was generally healthy and had no accompanying symptoms. The patient had no previous history of cutaneous or internal malignancies and a complete systems review was negative. There was no family history of malignancy.
- The lesion of NS was completely excised with a free margin of 0.2 cm and submitted for histopathological evaluation. Macroscopic examination of the lesion revealed a 2.5×1.8 cm, yellowish, verrucous plaque devoid of hair. One large, vegetative plaque with a hemorrhagic crust lay within the yellowish plaque.
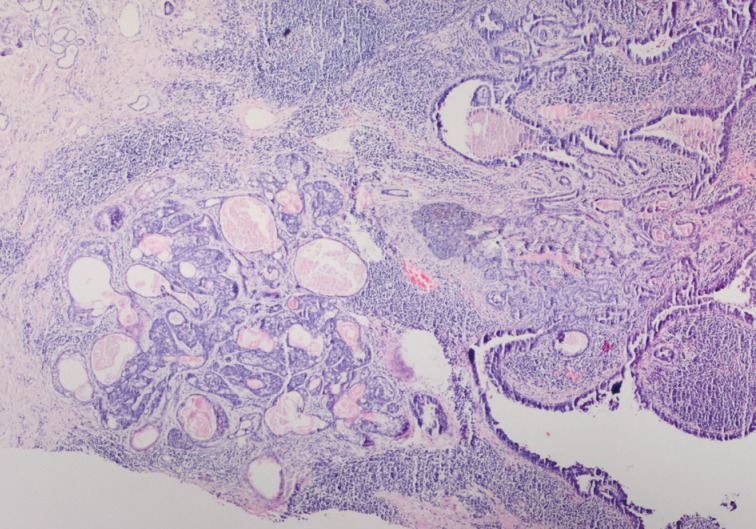
- Histopathological findings of the lesion by hematoxylin and eosin staining showed acanthosis, papillomatosis, sebaceus gland hyperplasia, and immature hair follicles in the dermis in the peripheral parts of the lesion. These findings were consistent with a diagnosis of NS. In addition, there were 6 adnexal tumors in the lesion including SCAP, PT, TAA, sebaceoma, TFI, and superficial epithelioma with sebaceus differentiation (SESD). The majority of the lesion was composed of SCAP, PT, and TAA. The tumors were separated from each other by loose fibrillary connective tissue stroma. These benign tumors were partly intertwined (Figs. 1, 2A).
- Cytomorphologically, the SCAP was composed of irregular papillary projections covered by double layers of epithelium and stratified squamous epithelium with stroma containing lymphocytes and large numbers of plasma cells (Fig. 2B, C). The deeper portion of the SCAP consisted of tubular structures with two layers of epithelial cells. While the luminal cells of the tubules were columnar and showed decapitation secretion, the outer layer was composed of flattened cells. These tubules were surrounded by a fibrous stroma. TAA was diagnosed from these findings (Fig. 1).
- The PT was characterized by a well-circumscribed large basaloid dermal tumor without an epidermal connection. There were dispersedly brown pigmentation fields in the center of large tumor islands (Fig. 2B, D).
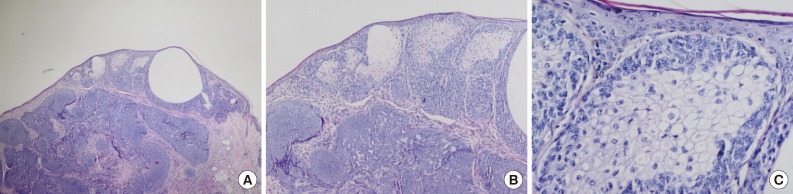
- In our case, small foci of follicular infundibulum, sebaceoma and SESD were observed. The TFI was composed of a plate like growth of basaloid keratinocytes with several points of attachment to the epidermis (Fig. 3). A collection of benign basaloid sebaceus cells suggestive of a sebaceoma was present and the sebaceoma existed as two types: superficial and deep in the dermis. There were cysts containing debris from holocrine degeneration (Fig. 4). The SESD was characterized by a sharply defined and well-circumscribed plate-like epidermal growth. Merging with the superficial keratinocytes was a cytologically bland basaloid cell population in which admixed mature sebocytes were present in clusters (Fig. 5).
CASE REPORT
- While multiple neoplasms may occasionally arise within the same lesion of NS, it is rare for four or more neoplasms to occur simultaneously.9 There are only three articles in the English medical literature describing the development and progression of 5 neoplasms in a solitary lesion. Our case is the second involving 6 neoplasms in a solitary lesion.2,5,10 This is not surprising considering that the skin adnexal structures arise from pluripotent stem cells.
- The 6 benign tumors arising within the nevus sebaceus of our patient included SCAP, TB, TAA, TFI, sebaceoma, and SESD. SCAP and TB are the most common neoplasms associated with nevus sebaceus.4
- TBs are fibroepithelial tumors composed of benign basaloid epithelial cells with cellular fibrous stroma. There are histopathologic overlaps between TB and BCC. The main distinction between pigmented TB and pigmented BCC is the stroma of TB shows a tight association with basaloid epithelial cells. In addition, no clefts or retractions are seen between epithelium and stroma as is characteristic of BCC. Mitoses and apoptotic bodies can occur in TB but are not as common as in BCC.5,11
- TAAs are benign tumors with glandular differentiation. They tend to be centered in the dermis and are well-circumscribed. The important issue is to distinguish adenomas from well differentiated adenocarcinomas. In the majority of cases, this distinction is straight forward because many carcinomas are large, infiltrative and show significant cytologic atypia.1
- The fourth tumor in our patient was TFI. The tumor cells had basaloid features and resembled a superficial BCC and superficial portion of an infundibulocystic BCC. In contrast with BCC, TFI lacks hyperchromatic nuclei, myxoid stroma and peripheral clefting.1
- The fifth tumor in our patient was sebaceoma. Sebaceoma, as presented in our case, should be differentiated from mantleoma and sebaceus carcinoma. In our case there was no atypia observed in the sebaceus carcinoma. The mantleoma is a tumor of the sebaceus mantle. Histopathologically, early neoplasms consist only of cords and columns of undifferentiated epithelial cells that radiate from an infundibulum. In late stages, these cords interweave in a retiform pattern that contains sebocytes of varying degrees of vacuolization and sebaceus ductal structures. In our case, there were multiple nests of basaloid cells with a random admixture of sebaceus cells. In addition, cysts and duct like structures containing debris from holocrine degeneration are suggestive of sebaceoma.1
- The sixth tumor in our patient was SESD. SESD is very closely related to reticulated acanthoma with sebaceus differentiation but is a distinct tumor. Reticulated acanthoma with sebaceous differentiation (RASD) is mainly characterized by proliferation of spinous cells, aggregates of mature sebocytes in the bottom of the strand of epithelium, bridging of the bases of the epithelium by those mature sebocytes and lack of pseudocysts and melanin granules.12,13 Utilizing features like sebaceous aggregates located only at the basal aspects without a reticulated pattern, our case was differentiated from RASD.
- Seborrheic keratosis with sebaceous differentiation was also included in the differential diagnosis. However, this lesion showed features of an acanthotic variant with only small numbers of mature sebaceous cells scattered randomly throughout the epithelium. There were no pseudohorn cysts and no melanin pigments.
- SESD should also be distinguished from follicular infundibulum tumor. Tumor of follicular infundibulum is characterized by a plate-like epithelial proliferation suspended from the epidermis. However, tumor trabeculae are characteristically thin, vacuolated and often surrounded by a thickened eosinophilic basement membrane.14 In terms of the differential diagnosis, it is important that our case did not have a reticular pattern.
- In our case, there was no direct connection of sebaceus cells with the superficial epithelium as they were located only in the basal aspects of the epithelium. In addition, our lesion contained not only sebaceus cells but also basaloid cells.
- As a result, many publications have discussed extensively whether SESD is a completely different entity. From our perspective, we believe SESD is unique as although it may have some similar features, it does not fit exactly into any of the other diagnoses. These kinds of cases are difficult to interpret since they are rare, are not published in a wide range of classical books and are not defined by exact diagnostic criteria. Therefore, it is important to create awareness on this issue and share information through the literature.
- There have been recent reports of benign and malignant complex neoplasms arising within NS. Manonukul et al.10 reported TB, trichilemmoma, sebaceus adenoma, TFI, SCAP, and mucoepidermoid carcinoma arising within 2 persistent lesions of NS. Kantrow et al.2 reported a coexistence of tumor of the follicular infundibulum, basaloid proliferations resembling superficial TB, a focus reminiscent of syringofibroadenoma, a trichilemmoma, a sebaceus lesion consistent with sebaceoma/sebaceus epithelioma and an apocrine adenocarcinoma in a long-standing NS. Miller et al.5 also reported five tumors in a NS (sebaceus carcinoma, BCC, trichoadenoma, TB, and SCAP). The multiplicity of tumors was considered not to be coincidental and was attributed to histogenetic mechanisms of NS.9 Tumors complicating NS may arise from the pluripotential primary epithelial germ as a result of either dedifferentiation of NS cells or from embryonic cells that lie dormant until the onset of neoplasia.3
- In their study, Xin et al.11 concluded that the genetic defect at 9q22.3 might possibly also contribute to the development of other appendageal tumors in NS such as SCAP, primitive follicular germs and TBs. They also suggested that further studies needed to address whether patched (PTCH) deletion in NS is associated with progression to BCC and/or other appendageal tumors.11
- Sebaceus tumor existence is important in NS because sebaceus lesions are associated with two syndromes: linear sebaceus nevus syndrome and Muir-Torre syndrome.1 Muir-Torre syndrome is characterized by the development of usually multiple sebaceus tumors in association with visceral neoplasms, usually gastrointestinal carcinomas and occasionally the genitourinary system. A single skin tumor with sebaceous differentiation could be a sign of Muir-Torre syndrome.1 Fortunately, in this case, careful examinations for internal malignancies were all negative.
- Currently, the necessity of prophylactic excision or topical photodynamic therapy in NS has been discussed in many publications.2,5,9,10,15,16 Clinical features are not sufficient to make an exact diagnosis of benign or malignant secondary tumors.17 In our opinion, owing to the possibility of malignant tumor outgrowth from a NS and the increasing frequency of occurrence with age, it is necessary to conduct prophylactic excision especially in adulthood. If excision is not conducted, at a minimum, changes in the lesion should be monitored by close clinical follow-up.
DISCUSSION
- 1. Weedon D. Skin pathology. 3rd ed. London: Churchill Livingstone, 2010.
- 2. Kantrow SM, Ivan D, Williams MD, Prieto VG, Lazar AJ. Metastasizing adenocarcinoma and multiple neoplastic proliferations arising in a nevus sebaceus. Am J Dermatopathol 2007; 29: 462-466. ArticlePubMed
- 3. Elder DE, Elenitsas R, Johnson BL, Murphy GF. Lever's histopathology of the skin. 9th ed. Philadelphia: Lippincott Williams & Wilkins, 2005.
- 4. Jaqueti G, Requena L, Sánchez Yus E. Trichoblastoma is the most common neoplasm developed in nevus sebaceus of Jadassohn: a clinicopathologic study of a series of 155 cases. Am J Dermatopathol 2000; 22: 108-118. PubMed
- 5. Miller CJ, Ioffreda MD, Billingsley EM. Sebaceous carcinoma, basal cell carcinoma, trichoadenoma, trichoblastoma, and syringocystadenoma papilliferum arising within a nevus sebaceus. Dermatol Surg 2004; 30(12 Pt 2): 1546-1549. ArticlePubMed
- 6. Gonzalez Guemes M, Gonzalez Hermosa R, Calderon Gutierrez MJ, Gonzalez-Perez R, Saracibar Oyon N, Soloeta Arechavala R. Development of multiple tumours arising in a nevus sebaceus of Jadassohn. J Eur Acad Dermatol Venereol 2005; 19: 658-659. ArticlePubMed
- 7. Mehregan AH, Pinkus H. Life history of organoid nevi: special reference to nevus sebaceus of Jadassohn. Arch Dermatol 1965; 91: 574-588. ArticlePubMed
- 8. Jones EW, Heyl T. Naevus sebaceus: a report of 140 cases with special regard to the development of secondary malignant tumours. Br J Dermatol 1970; 82: 99-117. ArticlePubMed
- 9. Stavrianeas NG, Katoulis AC, Stratigeas NP, Karagianni IN, Patertou-Stavrianea M, Varelzidis AG. Development of multiple tumors in a sebaceous nevus of Jadassohn. Dermatology 1997; 195: 155-158. ArticlePubMedPDF
- 10. Manonukul J, Omeapinyan P, Vongjirad A. Mucoepidermoid (adenosquamous) carcinoma, trichoblastoma, trichilemmoma, sebaceous adenoma, tumor of follicular infundibulum and syringocystadenoma papilliferum arising within 2 persistent lesions of nevus sebaceous: report of a case. Am J Dermatopathol 2009; 31: 658-663. ArticlePubMed
- 11. Xin H, Matt D, Qin JZ, Burg G, Böni R. The sebaceous nevus: a nevus with deletions of the PTCH gene. Cancer Res 1999; 59: 1834-1836. PubMed
- 12. Fukai K, Sowa J, Ishii M. Reticulated acanthoma with sebaceous differentiation. Am J Dermatopathol 2006; 28: 158-161. ArticlePubMed
- 13. Steffen CG, Ackerman AB. Neoplasms with sebaceus differentiation. Philadelphia: Lea & Febiger, 1994; 449-467.
- 14. McKee PH, Calonje E, Granter SR. Pathology of the skin. 3rd ed. Philadelphia: Elsevier Mosby, 2005.
- 15. Premalata CS, Kumar RV, Malathi M, Shenoy AM, Nanjundappa N. Cutaneous leiomyosarcoma, trichoblastoma, and syringocystadenoma papilliferum arising from nevus sebaceus. Int J Dermatol 2007; 46: 306-308. ArticlePubMed
- 16. In SI, Lee JY, Kim YC. Topical photodynamic therapy for nevus sebaceous on the face. Eur J Dermatol 2010; 20: 590-592. PubMed
- 17. Baykal C, Buyukbabani N, Yazganoglu KD, Saglik E. Tumors associated with nevus sebaceous. J Dtsch Dermatol Ges 2006; 4: 28-31. ArticlePubMed
REFERENCES
Fig. 1Syringocystadenoma papilliferum in the superficial part of the lesion. Tubular apocrine adenoma and cystic dilate glands in the deeper part.
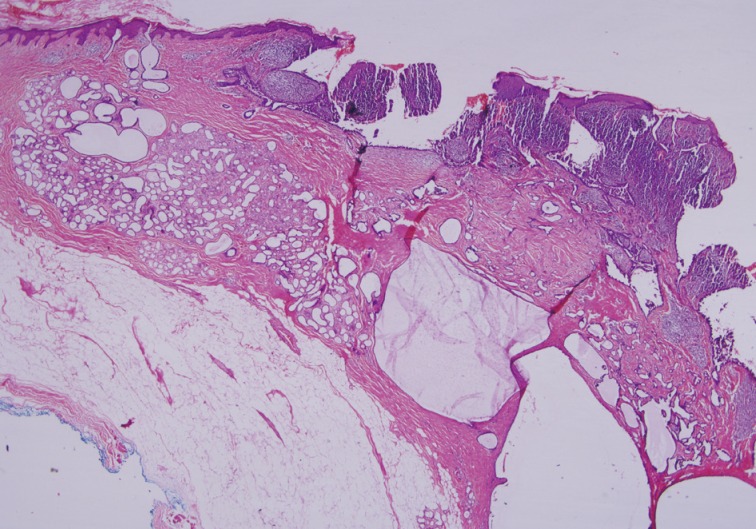

Fig. 2Syringocystadenoma papilliferum in the superficial part of the lesion. (A) Pigmented trichoblastoma and cystic dilate glands in the deeper part. (B) Pigmented trichoblastoma in the deeper part. (C) Typical features of syringocystadenoma papilliferum. (D) Pigmented trichoblastoma; lobules of basaloid aggregates with peripheral palisading.
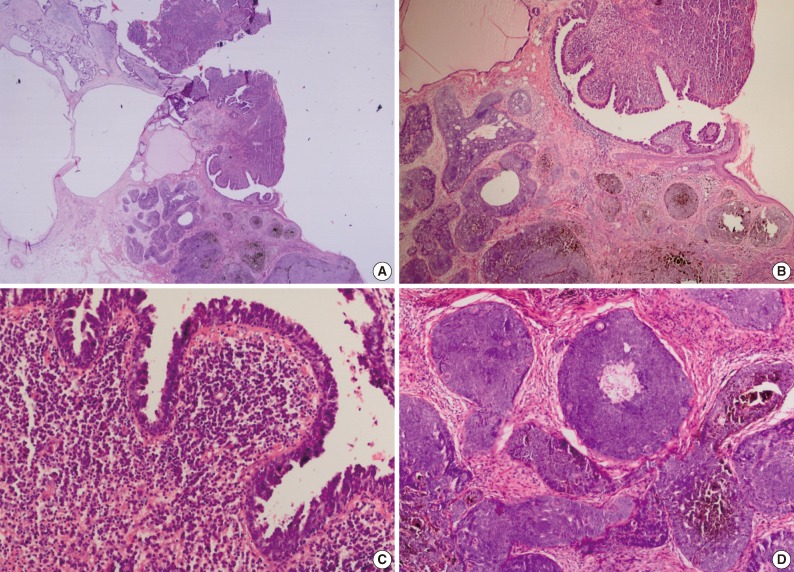

Fig. 3Tumor of follicular infundibulum. Aggregates of small keratinocytes show multiple connections to the epidermis and form a superficial dermal plaque.
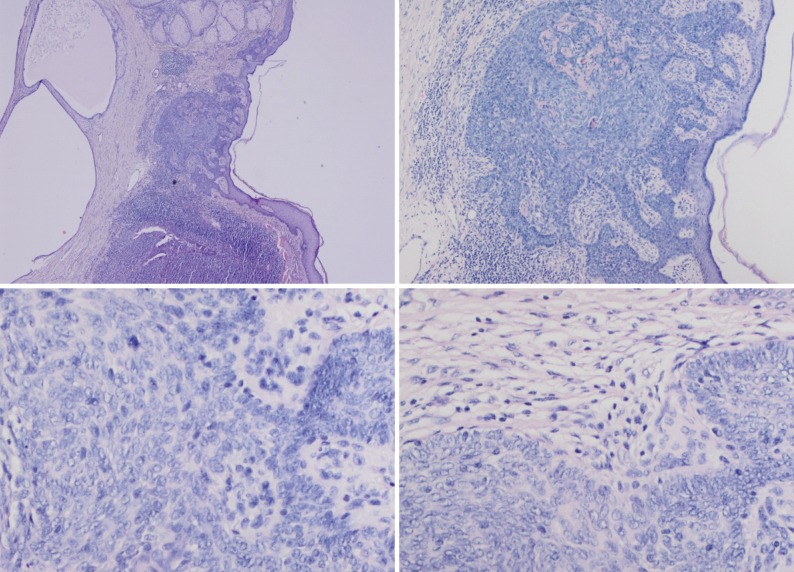

Figure & Data
References
Citations
Citations to this article as recorded by 

- Melanotrichoblastoma Arising on Nevus Sebaceous: A Rare Occurence
Apaopa J. Thekho, Deepika Uikey, Shanta Passi, V. Ramesh
Indian Journal of Dermatology.2025; 70(2): 105. CrossRef - Co-occurrence of Tubular Apocrine Adenoma and Syringocystadenoma Papilliferum over the Hypogastrium: A Rare Case Report
R Raghunatha Reddy, Mukunda Ranga Swaroop, Yogesh Devaraj, Greeshma Jagadish, Namratha Govindaraju
Clinical Dermatology Review.2025; 9(1): 69. CrossRef - Tumor of follicular infundibulum – reappraisal in a series of 28 patients with critical review of the literature
Michael Wilk, Bettina G. Zelger, Bernhard Zelger
JDDG: Journal der Deutschen Dermatologischen Gesellschaft.2024; 22(2): 223. CrossRef - Tumor des follikulären Infundibulums – Neubewertung in einer Serie von 28 Patienten mit kritischer Analyse der Literatur
Michael Wilk, Bettina G. Zelger, Bernhard Zelger
JDDG: Journal der Deutschen Dermatologischen Gesellschaft.2024; 22(2): 223. CrossRef - Adnexal neoplasms of the eye
Roman Drozdowski, Jane M. Grant-Kels, Madina Falcone, Campbell L. Stewart
Clinics in Dermatology.2024; 42(4): 321. CrossRef - Melanotrichoblastoma: sixth case report in the literature
Juliana Polizel Ocanha-Xavier, José Cândido Caldeira Xavier-Júnior
Anais Brasileiros de Dermatologia.2023; 98(6): 871. CrossRef - Multiple secondary neoplasms in nevus sebaceus excision
Travis S. Dowdle, David A. Mehegran, Dylan Maldonado, Cort D. McCaughey
Baylor University Medical Center Proceedings.2022; 35(2): 241. CrossRef - Congenital tumors arising from nevus sebaceous in 2 neonates
Lynette Wei Yi Wee, Bori Born, Sharon Mun Yee Wong, Hui-Ling Chia, Sithach Mey, Suresh Chandran, Mark Jean Aan Koh
JAAD Case Reports.2022; 21: 70. CrossRef - Development of seven secondary neoplasms in a nevus sebaceous: a case report and literature review
Yi-Wen Kuo, Jung-Chia Lin, Wei-Hsuan Tsai
Archives of Craniofacial Surgery.2022; 23(2): 83. CrossRef - Multiple rare neoplasms arising from the nevus sebaceous of the scalp: A case report
Deepthi Shetty, Anilkumar Desai, Niranjan Kumar, Dinesh U.S., Aditya Agnihotri, Saurav Bhaduri
Gulhane Medical Journal.2022; 64(2): 197. CrossRef - Syringocystadenoma Papilliferum and Basal Cell Carcinoma Arising in Nevus Sebaceous
Jingjing Jiang, Yujuan Chen, Qi He, Jiao Yang, Zhengzhong Zhang, Hao Yang, Huan Zhang, Chuan Yang
Clinical, Cosmetic and Investigational Dermatology.2022; Volume 15: 2021. CrossRef - Eyelid trichoblastoma – A case series
Gunja Chowdhury, Meghana Tanwar, Usha Kim, Shanthi R. Krishnan
Journal of Clinical Ophthalmology and Research.2021; 9(3): 123. CrossRef - Trilogy Revisited
Anand Bardia, Debajyoti Chatterjee, Keshavamurthy Vinay
Indian Dermatology Online Journal.2021; 12(4): 577. CrossRef - Trichilemmoma coexisting with sebaceous nevus
AngooriG Rao, VangaliS Reddy, M Tejal, M Divya
Indian Dermatology Online Journal.2020; 11(2): 253. CrossRef - Syndromic sebaceous nevus: current findings
Oumama El Ezzi, Anthony S. de Buys Roessingh, Michèle Bigorre, Guillaume Captier
International Journal of Dermatology.2018; 57(5): 599. CrossRef - Syringocystadenoma papilliferum and trichoblastoma arising in the nevus sebaceous
Feifei Wang, Yatong Wu, Zhancai Zheng, Yanping Bai
Indian Journal of Pathology and Microbiology.2018; 61(1): 106. CrossRef - Dermoscopic Analysis of Nevus Sebaceus of Jadassohn: A Study of 13 Cases
Awatef Kelati, Hanane Baybay, Salim Gallouj, Fatima Zahra Mernissi
Skin Appendage Disorders.2017; 3(2): 83. CrossRef - Secondary neoplasms arising from nevus sebaceus: A retrospective study of 450 cases in Taiwan
Ming‐Chun Hsu, Jau‐Yu Liau, Jin‐Liern Hong, Yin Cheng, Yi‐Hua Liao, Jau‐Shiuh Chen, Yi‐Shuan Sheen, Jin‐Bon Hong
The Journal of Dermatology.2016; 43(2): 175. CrossRef - A Histological Snapshot of Hypothetical Multistep Progression From Nevus Sebaceus to Invasive Syringocystadenocarcinoma Papilliferum
Vishwas Parekh, Cesar E. Guerrero, Charles F. Knapp, Craig A. Elmets, Kristopher M. McKay
The American Journal of Dermatopathology.2016; 38(1): 56. CrossRef - Trichoblastoma, syringocystadenoma papilliferum, desmoplastic trichilemmoma and tumor of the follicular infundibulum with signet‐ring cells, all arising in nevus sebaceus
Emilie Dore, Megan H. Noe, Brian L. Swick
Journal of Cutaneous Pathology.2015; 42(9): 645. CrossRef - Ceruminous adenoma (ceruminoma) arising in a nevus sebaceus of Jadassohn within the external auditory canal of a 3 year-old boy – A case report
Elżbieta Niemczyk, Kazimierz Niemczyk, Jadwiga Małdyk, Lidia Zawadzka-Głos
International Journal of Pediatric Otorhinolaryngology.2015; 79(11): 1932. CrossRef - Fehlbildungen und Nävi des behaarten Kopfes
V. Behle, H. Hamm
Der Hautarzt.2014; 65(12): 1022. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
Development of Six Tumors in a Sebaceus Nevus of Jadassohn: Report of a Case
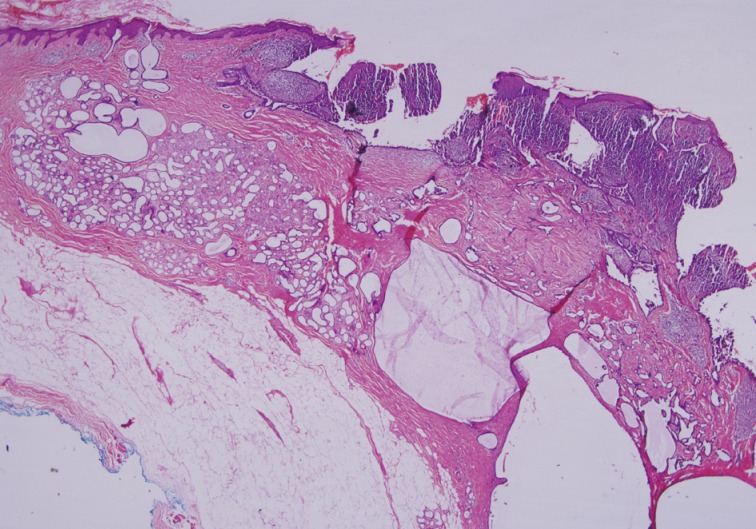
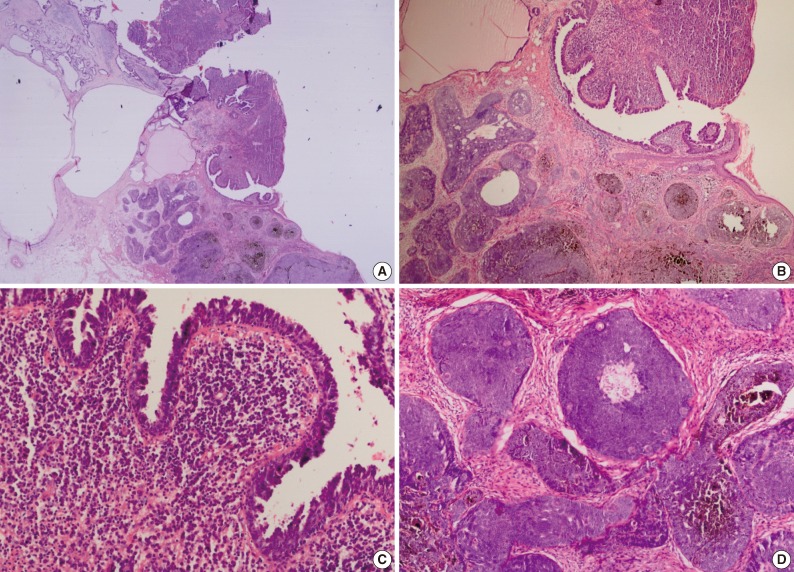
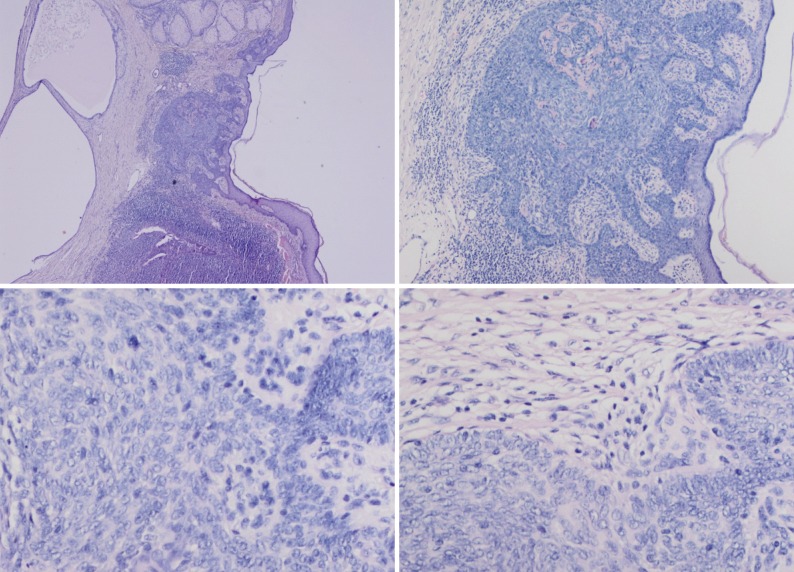
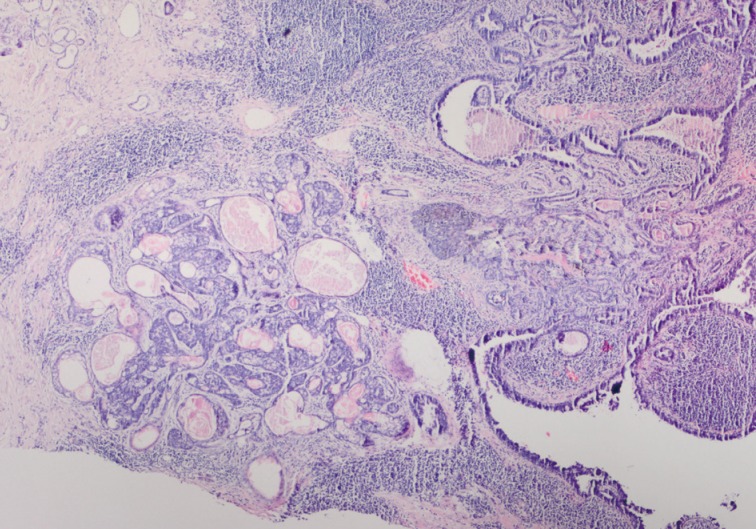
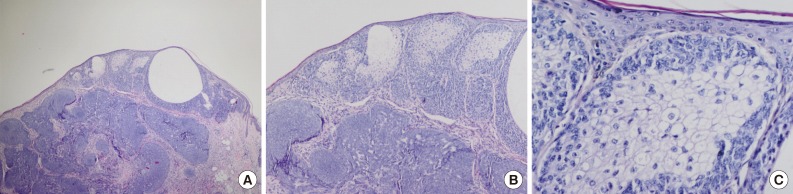
Fig. 1 Syringocystadenoma papilliferum in the superficial part of the lesion. Tubular apocrine adenoma and cystic dilate glands in the deeper part.
Fig. 2 Syringocystadenoma papilliferum in the superficial part of the lesion. (A) Pigmented trichoblastoma and cystic dilate glands in the deeper part. (B) Pigmented trichoblastoma in the deeper part. (C) Typical features of syringocystadenoma papilliferum. (D) Pigmented trichoblastoma; lobules of basaloid aggregates with peripheral palisading.
Fig. 3 Tumor of follicular infundibulum. Aggregates of small keratinocytes show multiple connections to the epidermis and form a superficial dermal plaque.
Fig. 4 Syringocystadenoma papilliferum in the superficial part, pigmented trichoblastoma and sebaceoma in the deeper part.
Fig. 5 (A) Superficial epithelioma with sebaceus differentiation above the trichoblastoma. (B) Superficial epithelioma with sebaceous differentiation. (C) Sebaceous differentiation in the superficial epithelioma.
Fig. 1
Fig. 2
Fig. 3
Fig. 4
Fig. 5
Development of Six Tumors in a Sebaceus Nevus of Jadassohn: Report of a Case

 E-submission
E-submission