Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(6); 2013 > Article
-
Original Article
Immunohistochemical Classification of Primary and Secondary Glioblastomas - Kyu Sang Lee, Gheeyoung Choe1, Kyung Han Nam1, An Na Seo1, Sumi Yun, Kyung Ju Kim, Hwa Jin Cho, Sung Hye Park
-
Korean Journal of Pathology 2013;47(6):541-548.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.6.541
Published online: December 24, 2013
Department of Pathology, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea.
1Department of Pathology, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seongnam, Korea.
- Corresponding Author: Gheeyoung Choe, M.D. Department of Pathology, Seoul National University Bundang Hospital, Seoul National University College of Medicine, 82 Gumi-ro 173beon-gil, Bundang-gu, Seongnam 463-707, Korea. Tel: +82-31-787-7711, Fax: +82-31-787-4012, gychoe@snu.ac.kr
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Glioblastomas may develop de novo (primary glioblastomas, P-GBLs) or through progression from lower-grade astrocytomas (secondary glioblastomas, S-GBLs). The aim of this study was to compare the immunohistochemical classification of glioblastomas with clinically determined P-GBLs and S-GBLs to identify the best combination of antibodies for immunohistochemical classification.
-
Methods
- We evaluated the immunohistochemical expression of epidermal growth factor receptor (EGFR), p53, and isocitrate dehydrogenase 1 (IDH-1) in 150 glioblastoma cases.
-
Results
- According to clinical history, the glioblastomas analyzed in this study consisted of 146 P-GBLs and 4 S-GBLs. Immunohistochemical expression of EGFR, p53, and IDH-1 was observed in 62.6%, 49.3%, and 11.1%, respectively. Immunohistochemical profiles of EGFR(+)/p53(-), IDH-1(-)/EGFR(+)/p53(-), and EGFR(-)/p53(+) were noted in 41.3%, 40.2%, and 28.7%, respectively. Expression of IDH-1 and EGFR(-)/p53(+) was positively correlated with young age. The typical immunohistochemical features of S-GBLs comprised IDH-1(+)/EGFR(-)/p53(+), and were noted in 3.6% of clinically P-GBLs. The combination of IDH-1(-) or EGFR(+) was the best set of immunohistochemical stains for identifying P-GBLs, whereas the combination of IDH-1(+) and EGFR(-) was best for identifying S-GBLs.
-
Conclusions
- We recommend a combination of IDH-1 and EGFR for immunohistochemical classification of glioblastomas. We expect our results to be useful for determining treatment strategies for glioblastoma patients.
- Patients and specimens
- We collected formalin-fixed paraffin-embedded tissues from 150 glioblastoma patients. More than one available pathologic examination was available for 10 patients due to tumor recurrence, and for these cases the initial paraffin blocks were used for our study. A total of 118 undergoing craniotomy or stereotactic biopsy in Seoul National University Bundang Hospital from May 2003 to September 2011, and 32 patients received craniotomy or stereotactic biopsy in Seoul National University Hospital from May 2011 to September 2011.
- The mean age of 150 glioblastoma patients was 58.8 years with range of 19 to 85 years. The patient group consisted of equal numbers of men and women, 75 patients each. Patients with history of an evolution from diffuse or anaplastic astrocytoma were diagnosed as having clinically secondary glioblastoma, and de novo cases were regarded as clinically primary glioblastomas. They consisted of 146 cases of clinically primary glioblastomas and 4 cases of clinically secondary glioblastomas. The mean age of clinically primary and secondary glioblastomas was 59.2 years and 44.0 years, respectively.
- Immunohistochemical staining
- Immunohistochemical staining for p53 and EGFR was performed in 150 cases using formalin-fixed paraffin-embedded tumor blocks. IDH-1 immunohistochemical stains were performed in 144 cases of glioblastoma for which paraffin blocks were available.
- Briefly, 4-µm-thick tissue sections were deparaffinized in xylene and hydrated by immersing in a series of graded ethanol. Antigen retrieval was performed in a microwave by placing the sections in epitope retrieval solution (0.01 M citrate buffer, pH 6.0) for 20 minutes; endogenous peroxidase was inhibited by immersing the sections in 0.3% hydrogen peroxide for 10 minutes.15 Sections were then incubated with combinations of EGFR (1:150, Dako, Camarillo, CA, USA), p53 (1:1,000, Dako, Glostrup, Denmark), and/or IDH-1 (1:100, DIANOVA, Hamburg, Germany) antibodies.
- Immunohistochemical stains for EGFR were graded as follows: 0 (no cell stained), 1+ (<5% tumor cells stained), 2+ (5-50% cells stained), and 3+ (>50% cells stained). For statistical analysis, a score of 0 and 1 was considered negative and a score of 2 or 3 was considered positive. Nuclear staining of p53 was scored semi-quantitatively in the most prominently stained area of the tissue slides. The percentage of positive cells was counted as follows: cases with ≥10% cells were considered positive (overexpression of p53), and <10% cells were considered negative. Cytoplasmic immunoreactivity to the IDH-1 antibody was considered as positive immunostaining.
- Histopathological evaluation and radiological review
- The hematoxylin and eosin stained slides were reviewed and the diagnosis was confirmed according to the WHO classification of tumors of the nervous system. In cases where the results of immunohistochemical studies were consistent with those of secondary glioblastomas in clinically primary glioblastoma patients with no history of previous diffuse or anaplastic astrocytoma, we searched for histological evidence of an evolution from a diffuse or anaplastic astrocytoma in the background. In addition, we radiologically re-evaluated brain tumor images to detect any lower grade components in the background.
- Statistical analysis
- All statistical analyses were performed using SPSS ver. 19.0 (SPSS Inc., Chicago, IL, USA). Chi-squared tests were used to analyze correlations between immunohistochemical results. A p-value of less than 0.05 was accepted as statistically significant.
MATERIALS AND METHODS
- Immunohistochemical expression of EGFR and p53 in glioblastomas
- Among 150 cases, immunohistochemical expression of EGFR was noted in 94 cases (62.6%) and p53 was overexpressed in 74 cases (49.3%) (Fig. 1A, B, D, E). The typical immunohistochemical feature of primary glioblastoma, EGFR(+)/p53(-), was noted in 62 cases (41.3%) while the typical immunohistochemical feature of secondary glioblastoma, EGFR(-)/p53(+), was noted in 43 cases (28.7%). Immunohistochemical expression of EGFR(+)/p53(+) was noted in 31 cases (20.7%) and EGFR(-)/p53(-) in 14 cases (9.3%). The inverse correlation between EGFR and p53 was statistically significant (independent samples t-test, p<.001).
- The mean age of EGFR(+)/p53(-) immunohistochemically primary glioblastoma patients was 65.4 years and the mean age of EGFR(-)/p53(+) immunohistochemically secondary glioblastoma patients was 53.8 years. The mean age of EGFR(+)/p53(-) immunohistochemically primary glioblastoma patients was 11.6 years older than that of EGFR(-)/p53(+) immunohistochemically secondary glioblastoma patients, the difference of which was statistically significant (p<.001) (Table 1).
- Immunohistochemical expression of IDH-1 in glioblastomas
- Immunohistochemical expression of IDH-1 was noted in 16 (11.1%) of 144 total cases (Fig. 1C, F). The mean age of IDH-1(+) glioblastoma patients was 45.2 years, whereas that of IDH-1(-) glioblastoma patients was 60.0 years. Positive IDH-1 status was significantly correlated with young age (p<.001). Among 16 cases of IDH-1(+) glioblastomas, 14 cases showed co-expression of p53 (87.5%) and two cases (12.5%) revealed no overexpression of p53 (p=.01). Among 128 cases of IDH-1(-) glioblastomas, immunohistochemical expression of EGFR was noted in 82 cases (64.1%) (p=.97). We also observed a positive correlation between IDH-1 and p53, but no correlation between IDH-1 and EGFR (Table 2).
- Immunohistochemical subtypes in clinically primary and secondary glioblastomas
- Clinically secondary glioblastomas were observed in only 4 cases (2.8%) of the 144 cases in our study, which had previously been diagnosed as having diffuse astrocytoma or anaplastic astrocytoma. The proportion of clinically secondary glioblastomas was lower than those of previous studies.1 The mean age of patients with secondary glioblastomas was 44.0 years. Immunohistochemical profiles of clinically primary and secondary glioblastomas are summarized in Table 3.
- All of the clinically secondary glioblastomas expressed IDH-1 according to immunohistochemical staining while none expressed EGFR. The secondary glioblastomas consisted of three cases with an immunohistochemical profile typical of secondary glioblastomas, IDH-1(+)/EGFR(-)/p53(+), and one case of IDH-1(+)/EGFR(-)/p53(-).
- Among 140 cases of clinically primary glioblastoma, immunohistochemical profiles typical of primary glioblastomas, IDH-1(-)/EGFR(+)/p53(-), were noted in 58 cases (41.4%). Interestingly, the immunohistochemical profile associated with secondary glioblastomas, IDH-1(+)/EGFR(-)/p53(+), was detected in five cases (3.6%) among the 140 clinically primary glioblastomas. The five patients with IDH-1(+)/EGFR(-)/p53(+) immunohistochemical profiles had no history of lower grade astrocytic tumors; however, they were young with a mean age of 44.6 years. Based on these findings, we requested a neuroradiologist to review the brain magnetic resonance imaging (MRI) images of these five cases. Nonenhancing, extensively infiltrative components without necrosis were detected at least focally in the peripheral areas of the main tumor, which provided radiological evidence of an evolution from a less malignant precursor lesion. In addition, one case presented with multiple lesions as identified by MRI. Based on these findings, the five cases of immunohistochemically secondary glioblastomas were combined with the four cases of clinically secondary glioblastomas. Following this adjustment, the proportion of clinically and immunohistochemically secondary glioblastomas (6.25%) was similar to those of previous studies.1 Clinicopathologic profiles of clinically and immunohistochemically secondary glioblastoma patients are summarized in Table 4.
- Optimal combination of antibodies for immunohistochemical classification of primary and secondary glioblastomas
- To determine the best combination of antibodies for immunohistochemical classification of primary and secondary glioblastomas, we analyzed various combinations of antibodies including expression of single individual antibodies, expression profiles of a combination of two antibodies, and expression profiles of all the three antibodies. Specifically, we calculated the statistical measures of the performance of a binary classification tests such as the sensitivity, specificity, accuracy, positive predictive value, and negative predictive value for every antibody combination for identifying immunohistochemically primary glioblastomas (Table 5) and immunohistochemically secondary glioblastomas (Table 6).
- With respect to immunostaining with a single antibody, expression of p53 exhibited relatively lower sensitivity, specificity, accuracy, positive predictive value, and negative predictive value compared with expression of IDH-1 or EGFR. Contrary to our expectations, the combination of all three antibodies (IDH-1, p53, and EGFR) was not the best way for immunohistochemical classification of primary and secondary glioblastomas. Rather, combination of two antibodies for IDH-1 and EGFR produced the best results in distinguishing primary and secondary glioblastomas. Specifically, combination of IDH-1(-) or EGFR(+) was the best way to identify primary glioblastomas (Table 5), whereas the combination of IDH-1(+) and EGFR(-) was best way to identify secondary glioblastomas (Table 6).
RESULTS
- The present study consisted of 146 clinically primary and 4 clinically secondary glioblastomas, which had immunohistochemical expression of EGFR, p53, and IDH-1 in 62.6%, 49.3%, and 11.1% of cases, respectively. Immunohistochemical profiles of EGFR(+)/p53(-), IDH-1(-)/EGFR(+)/p53(-), and EGFR(-)/p53(+) were noted in 41.3%, 40.2%, and 28.7% of cases, respectively. In addition, expression of IDH-1 and EGFR(-)/p53(+) was positively correlated with young age. Our study also showed a positive correlation between IDH-1 and p53, but no correlation between IDH-1 and EGFR. Interestingly, 3.6% of clinically primary glioblastomas exhibited the IDH-1(+)/EGFR(-)/p53(+) immunohistochemical profile which is typical of secondary glioblastomas.
- Targeted molecular therapies and personalized medicine are becoming increasingly important for the treatment of glioblastoma patients as well as other malignant tumors. Primary glioblastomas are remarkably different from secondary glioblastomas in many aspects, including their therapeutic responses to EGFR tyrosine kinase inhibitors,16-18 MMP activation,19 cell signaling pathway,20 patterns of promoter methylation, and expression profiles at the RNA and protein levels.1 Because molecular subtyping of glioblastomas is very important for personalized medicine,21 distinction of primary and secondary glioblastomas can serve as an initial step for determining the treatment strategy of glioblastoma patients.
- Amplification/overexpression of EGFR is a key pathogenesis in the development of primary glioblastomas. EGFR amplification and mRNA overexpression are strongly associated with an increased level of the EGFR protein.22,23 Increased level of the EGFR protein can be demonstrated as overexpression of EGFR by immunohistochemistry. In previous studies, overexpression of EGFR has been observed in more than 60% of glioblastomas. Likewise in our study, overexpression of EGFR was detected in 62.7% of cases.
- The TP53 pathway plays a critical role in the development of low grade gliomas. TP53 mutations are present in more than 70% of diffuse astrocytomas, anaplastic astrocytomas, and secondary glioblastomas. In addition, TP53 mutations are noted in lower than 30% of primary glioblastomas.24 In the presence of TP53 mutations, positive staining for p53 can be expected; however, a few studies concerning TP53 in glioblastomas have reported a higher rate of immunoreactivity than expected based on the actual gene mutation. In fact, immunohistochemistry does not seem to reflect TP53 mutations perfectly. Along these lines, several studies have suggested that various mechanisms of TP53 alteration can result in accumulation of p53 protein.25,26 Similarly, immunohistochemical overexpression of p53 was observed in 49.3% of glioblastomas in our study.
- Overexpression of EGFR and p53 mutations are known to be mutually exclusive in the evolution of primary and secondary glioblastomas.7 Our study revealed an inverse correlation between EGFR and p53 that was statistically significant (p<0.001); however, 30% of glioblastomas exhibited ambiguous immunoprofiles in our study. The typical immunohistochemical feature of primary glioblastomas, EGFR(+)/p53(-), was noted in 41.3% of cases while the typical immunoprofile of secondary glioblastomas, EGFR(-)/p53(+), was noted in 28.7% of cases. Unexpected immunohistochemical expression of EGFR(+)/p53(+) and EGFR(-)/p53(-) was noted in 20.7% and 9.3% of cases, respectively. Thus, our attempt to classify glioblastomas by the immunohistochemical combination of EGFR and p53 revealed certain limitations.
- Because of the inability of EGFR and p53 staining to fully distinguish between clinically primary and secondary glioblastomas, we included immunohistochemical staining for IDH-1, which is highly related to secondary glioblastoma.8 We then analyzed the expression profiles of triple markers (IDH-1, EGFR, and p53) with respect to classification of glioblastomas (primary vs secondary). Three out of four cases of clinically secondary glioblastomas exhibited the typical immunohistochemical profile consisting of IDH-1(+)/EGFR(-)/p53(+), while the typical immunoprofile of primary glioblastoma, IDH-1(-)/EGFR(+)/p53(-), was noted in 41.4% cases. Interestingly, the typical immunoprofile of secondary glioblastoma, IDH-1(+)/EGFR(-)/p53(+), was detected in five cases (3.6%) among 140 clinically primary glioblastomas. The five patients with IDH-1(+)/EGFR(-)/p53(+) immunoprofile had no history of lower grade astrocytic tumors; however, they were observed in younger patients with a mean age of 44.6 years and presented with diffuse or multiple lesions on MRI images. Therefore, these cases were reclassified as immunohistochemically secondary glioblastomas. We suspect that these cases consisted of secondary glioblastomas with subclinical diffuse or anaplastic astrocytomas and very rapid progression from precursor lower grade lesions.
- The proportion of clinically secondary glioblastomas (2.8%) observed in our study was lower previously observed.1 However, the proportion of clinically and immunohistochemically secondary glioblastomas (6.25%) was similar to those of previous studies. The mean age of secondary glioblastomas was 44.3 years (44.0 years in clinically secondary glioblastomas and 44.6 years in immunohistochemically secondary glioblastomas). Conversely, the mean age of clinically primary and immunohistochemically primary glioblastomas was 59.2 years and 59.3 years, respectively.
- To determine the best combination of antibodies for immunohistochemical classification of glioblastomas, we evaluated the statistical performance of a binary classification test using every combination of antibodies for immunohistochemically primary and secondary glioblastomas. Contrary to our expectations, the combination of all three antibodies (IDH-1, p53, and EGFR) was not the best way to distinguish primary and secondary glioblastomas. Rather, combinations of IDH-1 and/or EGFR performed better for distinguishing primary and secondary glioblastomas. Specifically, the combination of IDH-1(+) and EGFR(-) was the best way to identify secondary glioblastomas, which exhibited perfect statistical significance for sensitivity, specificity, accuracy, positive predictive value, and negative predictive value. Likewise, the immunohistochemical combination of IDH-1(-) or EGFR(+) was the best method for identifying primary glioblastomas. In other words, an IDH-1(+)/EGFR(-) immunoprofile accurately identified secondary glioblastomas, while IDH-1(-)/EGFR(+), IDH-1(+)/EGFR(+), and IDH-1(-)/EGFR(-) immunoprofiles identified primary glioblastomas. Based on these results, the combination of IDH-1 and EGFR immunohistochemistry, excluding p53, was determined to be a good method for subtyping glioblastomas. This result was attributed primarily to one case of the four clinically secondary glioblastomas that did not express p53, which affected the statistical significance of the data considerably. Moreover, overexpression of p53 was frequently observed in primary glioblastomas. The sensitivity of p53(-) was too low (53.3%) in primary glioblastomas, and thus the immunohistochemical overexpression of p53 was not statistically useful for distinguishing primary and secondary glioblastomas.
- In summary, immunohistochemical expression of EGFR, p53, and IDH-1 in glioblastomas was observed in 62.6%, 49.3%, and 11.1%, respectively. The immunohistochemical profiles of EGFR(+)/p53(-), IDH-1(-)/EGFR(+)/p53(-), and EGFR(-)/p53(+) were noted in 41.3%, 40.2%, and 28.7% of cases, respectively. In addition, expression of IDH-1 and EGFR(-)/p53(+) was positively correlated with young age. Our study demonstrated a positive correlation between IDH-1 and p53, but not between IDH-1 and EGFR. The typical immunoprofile of secondary glioblastomas, IDH-1(+)/EGFR(-)/p53(+), was detected in 3.6% of clinically primary glioblastomas. The combination of IDH-1(-) or EGFR(+) was the best method for identifying primary glioblastomas, whereas the combination of IDH-1(+) and EGFR(-) was the best method for identifying secondary glioblastomas.
- In conclusion, we recommend the combination of IDH-1 and EGFR for immunohistochemical classification of glioblastomas. We expect that our results will be useful for determining treatment strategies for patients with glioblastoma.
DISCUSSION
- 1. Ohgaki H, Kleihues P. Genetic pathways to primary and secondary glioblastoma. Am J Pathol 2007; 170: 1445-1453. ArticlePubMedPMC
- 2. Kim YZ, Song YJ, Kim KU, Kim DC. MGMT gene promoter methylation analysis by pyrosequencing of brain tumour. Korean J Pathol 2011; 45: 455-462. Article
- 3. Peiffer J, Kleihues P. Hans-Joachim Scherer (1906-1945), pioneer in glioma research. Brain Pathol 1999; 9: 241-245. ArticlePubMed
- 4. Ohgaki H, Dessen P, Jourde B, et al. Genetic pathways to glioblastoma: a population-based study. Cancer Res 2004; 64: 6892-6899. PubMed
- 5. Cancer Genome Atlas Research Network. Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature 2008; 455: 1061-1068. ArticlePubMedPMCPDF
- 6. von Deimling A, von Ammon K, Schoenfeld D, Wiestler OD, Seizinger BR, Louis DN. Subsets of glioblastoma multiforme defined by molecular genetic analysis. Brain Pathol 1993; 3: 19-26. ArticlePubMed
- 7. Watanabe K, Tachibana O, Sata K, Yonekawa Y, Kleihues P, Ohgaki H. Overexpression of the EGF receptor and p53 mutations are mutually exclusive in the evolution of primary and secondary glioblastomas. Brain Pathol 1996; 6: 217-223. ArticlePubMed
- 8. Yan H, Parsons DW, Jin G, et al. IDH1 and IDH2 mutations in gliomas. N Engl J Med 2009; 360: 765-773. PubMedPMC
- 9. van den Bent MJ, Dubbink HJ, Marie Y, et al. IDH1 and IDH2 mutations are prognostic but not predictive for outcome in anaplastic oligodendroglial tumors: a report of the European Organization for Research and Treatment of Cancer Brain Tumor Group. Clin Cancer Res 2010; 16: 1597-1604. ArticlePubMedPDF
- 10. Sonoda Y, Kumabe T, Nakamura T, et al. Analysis of IDH1 and IDH2 mutations in Japanese glioma patients. Cancer Sci 2009; 100: 1996-1998. ArticlePubMedPMC
- 11. Balss J, Meyer J, Mueller W, Korshunov A, Hartmann C, von Deimling A. Analysis of the IDH1 codon 132 mutation in brain tumors. Acta Neuropathol 2008; 116: 597-602. ArticlePubMedPDF
- 12. Takano S, Tian W, Matsuda M, et al. Detection of IDH1 mutation in human gliomas: comparison of immunohistochemistry and sequencing. Brain Tumor Pathol 2011; 28: 115-123. ArticlePubMedPDF
- 13. Mellai M, Piazzi A, Caldera V, et al. IDH1 and IDH2 mutations, immunohistochemistry and associations in a series of brain tumors. J Neurooncol 2011; 105: 345-357. ArticlePubMedPDF
- 14. Lee HJ, Xu X, Kim H, et al. Comparison of direct sequencing, PNA clamping-real time polymerase chain reaction, and pyrosequencing methods for the detection of EGFR mutations in non-small cell lung carcinoma and the correlation with clinical responses to EGFR tyrosine kinase inhibitor treatment. Korean J Pathol 2013; 47: 52-60. ArticlePubMedPMC
- 15. Hwangbo W, Lee JH, Ahn S, et al. EGFR gene amplification and protein expression in invasive ductal carcinoma of the breast. Korean J Pathol 2013; 47: 107-115. ArticlePubMedPMC
- 16. Das P, Puri T, Jha P, et al. A clinicopathological and molecular analysis of glioblastoma multiforme with long-term survival. J Clin Neurosci 2011; 18: 66-70. ArticlePubMed
- 17. Mellinghoff IK, Wang MY, Vivanco I, et al. Molecular determinants of the response of glioblastomas to EGFR kinase inhibitors. N Engl J Med 2005; 353: 2012-2024. PubMed
- 18. Haas-Kogan DA, Prados MD, Tihan T, et al. Epidermal growth factor receptor, protein kinase B/Akt, and glioma response to erlotinib. J Natl Cancer Inst 2005; 97: 880-887. ArticlePubMed
- 19. Choe G, Park JK, Jouben-Steele L, et al. Active matrix metalloproteinase 9 expression is associated with primary glioblastoma subtype. Clin Cancer Res 2002; 8: 2894-2901. PubMed
- 20. Choe G, Horvath S, Cloughesy TF, et al. Analysis of the phosphatidylinositol 3'-kinase signaling pathway in glioblastoma patients in vivo. Cancer Res 2003; 63: 2742-2746. PubMed
- 21. Mischel PS, Cloughesy TF, Nelson SF. DNA-microarray analysis of brain cancer: molecular classification for therapy. Nat Rev Neurosci 2004; 5: 782-792. ArticlePubMedPDF
- 22. Rong Y, Belozerov VE, Tucker-Burden C, et al. Epidermal growth factor receptor and PTEN modulate tissue factor expression in glioblastoma through JunD/activator protein-1 transcriptional activity. Cancer Res 2009; 69: 2540-2549. ArticlePubMedPMCPDF
- 23. Benito R, Gil-Benso R, Quilis V, et al. Primary glioblastomas with and without EGFR amplification: relationship to genetic alterations and clinicopathological features. Neuropathology 2010; 30: 392-400. ArticlePubMed
- 24. Momota H, Narita Y, Matsushita Y, Miyakita Y, Shibui S. p53 abnormality and tumor invasion in patients with malignant astrocytoma. Brain Tumor Pathol 2010; 27: 95-101. ArticlePubMedPDF
- 25. Srividya MR, Thota B, Arivazhagan A, et al. Age-dependent prognostic effects of EGFR/p53 alterations in glioblastoma: study on a prospective cohort of 140 uniformly treated adult patients. J Clin Pathol 2010; 63: 687-691. ArticlePubMed
- 26. Stoczynska-Fidelus E, Witusik-Perkowska M, Banaszczyk M, et al. Glioblastoma specimens with TP53 mutations do not show EGFRvIII amplification. Cancer Genet 2011; 204: 282-283. ArticlePubMed
REFERENCES
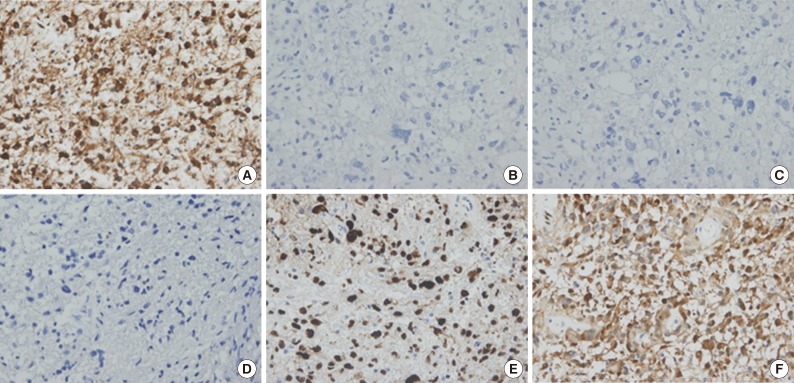
Figure & Data
References
Citations

- The Co-Expression and Cellular Location of HER Family Members, EGFRvIII, Putative Cancer Stem Cell Biomarkers CD44 and CD109 in Patients with Glioblastoma, and Their Impacts on Prognosis
Ermira Mulliqi, Said Khelwatty, Izhar Bagwan, Ahmad Kamaludin, Anna Morgan, Natalie Long, Keyoumars Ashkan, Helmout Modjtahedi
Cancers.2025; 17(7): 1221. CrossRef - Predicting p53 Status in IDH‐Mutant Gliomas Using MRI‐Based Radiomic Model
Jiamin Li, Zhihong Lan, Xiao Zhang, Xiaoyun Liang, Hanwei Chen, Xiangrong Yu
Cancer Medicine.2025;[Epub] CrossRef - Classification of Glioblastoma Based on Immunohistochemical Expression of IDH-1, p53, and ATRX: A Study from a Tertiary Care Center in South India
Hiba Thankayathil, Aparna Govindan, Supriya Nilambur Kovilakam, Rajeev Mandaka Parambil
Indian Journal of Neurosurgery.2025; 14(S 01): S48. CrossRef - Endothelial transdifferentiation of glioma stem cells: a literature review
Andrei Buruiana, Stefan Ioan Florian, Alexandru Ioan Florian, Olga Soritau, Sergiu Susman
Acta Neuropathologica Communications.2025;[Epub] CrossRef - Cutaneous Melanoma and Glioblastoma Multiforme Association—Case Presentation and Literature Review
Olguța Anca Orzan, Călin Giurcăneanu, Bogdan Dima, Monica Beatrice Dima, Ana Ion, Beatrice Bălăceanu, Cornelia Nițipir, Irina Tudose, Cătălina Andreea Nicolae, Alexandra Maria Dorobanțu
Diagnostics.2023; 13(6): 1046. CrossRef - Primary Extra-axial Glioblastoma: Case Report and Literature Review
Baraa Dabboucy, Philippe Younes, Abdallah Rahbani, Elie Fahed, Gérard Abadjian
Arquivos Brasileiros de Neurocirurgia: Brazilian Neurosurgery.2021; 40(04): e368. CrossRef - TERT Promoter Mutation in Adult Glioblastomas: It's Correlation with Other Relevant Molecular Markers
Mukesh Barange, Sridhar Epari, Mamta Gurav, Omshree Shetty, Ayushi Sahay, Prakash Shetty, Jayantsastri Goda, Aliasagar Moyiadi, Tejpal Gupta, Rakesh Jalali
Neurology India.2021; 69(1): 126. CrossRef - Immunohistochemical characterisation and histopathology of astrocytic neoplasms at a tertiary Nigerian hospital
Michael Nweke, Gabriel Ogun, Amos Adeleye, Clement A. Okolo, Adekunle Adesina
International Journal of Clinical Practice.2021;[Epub] CrossRef - Cytotoxic Effects of Blue Scorpion Venom (Rhopalurus junceus) in a Glioblastoma Cell Line Model
Laura A. Lozano-Trujillo, Diana K. Garzón-Perdomo, Andrea C.R. Vargas, Lina M. de los Reyes, Marco F. Avila-Rodriguez, Olivia T.G. Gay, Liliana F. Turner
Current Pharmaceutical Biotechnology.2021; 22(5): 636. CrossRef - Molecular Subgroups of Glioblastoma– an Assessment by Immunohistochemical Markers
Ádám Nagy, Ferenc Garzuly, Gergely Padányi, Iván Szűcs, Ádám Feldmann, Balázs Murnyák, Tibor Hortobágyi, Bernadette Kálmán
Pathology & Oncology Research.2019; 25(1): 21. CrossRef - Proteomic Advances in Glial Tumors through Mass Spectrometry Approaches
Radu Pirlog, Sergiu Susman, Cristina Adela Iuga, Stefan Ioan Florian
Medicina.2019; 55(8): 412. CrossRef - Isocitrate dehydrogenase 1 mutant glioblastomas demonstrate a decreased rate of pseudoprogression: a multi-institutional experience
Homan Mohammadi, Kevin Shiue, G Daniel Grass, Vivek Verma, Kay Engellandt, Dirk Daubner, Gabriele Schackert, Mercia J Gondim, Dibson Gondim, Alexander O Vortmeyer, Aaron P Kamer, William Jin, Timothy J Robinson, Gordon Watson, Hsiang-Hsuan M Yu, Tim Laute
Neuro-Oncology Practice.2019;[Epub] CrossRef - Calvarium mass as the first presentation of glioblastoma multiforme: A very rare manifestation of high-grade glioma
S. Taghipour Zahir, M. Mortaz, M. Baghi Yazdi, N. Sefidrokh Sharahjin, M. Shabani
Neurochirurgie.2018; 64(1): 76. CrossRef - Malignant Gliomas as Second Neoplasms in Pediatric Cancer Survivors: Neuropathological Study
Ewa Izycka-Swieszewska, Ewa Bien, Joanna Stefanowicz, Edyta Szurowska, Ewa Szutowicz-Zielinska, Magdalena Koczkowska, Dawid Sigorski, Wojciech Kloc, Wojciech Rogowski, Elzbieta Adamkiewicz-Drozynska
BioMed Research International.2018; 2018: 1. CrossRef - Prognostic significance of mutant IDH1, CD133, and β-catenin immunohistochemical expression in glioblastoma multiforme
Azza Abdel-Aziz, Mie A. Mohamed, Dina Abdallah, Fatma M.F. Akl, Ghada E. Eladawy, Ahmed N. Taha, Hossam Shata
Egyptian Journal of Pathology.2018; 38(1): 27. CrossRef - On glioblastoma and the search for a cure: where do we stand?
John Bianco, Chiara Bastiancich, Aleksander Jankovski, Anne des Rieux, Véronique Préat, Fabienne Danhier
Cellular and Molecular Life Sciences.2017; 74(13): 2451. CrossRef - Expression of p53 & epidermal growth factor receptor in glioblastoma
Sameera Karnam, Radhika Kottu, Amit Kumar Chowhan, Prasad Chandramouleswara Bodepati
Indian Journal of Medical Research.2017; 146(6): 738. CrossRef - Development of Glioblastoma after Treatment of Brain Abscess
Hiroaki Matsumoto, Hiroaki Minami, Shogo Tominaga, Yasuhisa Yoshida
World Neurosurgery.2016; 88: 686.e19. CrossRef - Clinical, immunohistochemical, and molecular genetic prognostic factors in adult patients with glioblastoma
N. V. Lobanova, L. V. Shishkina, M. V. Ryzhova, G. L. Kobyakov, R. V. Sycheva, S. A. Burov, A. V. Lukyanov, Zh. R. Omarova
Arkhiv patologii.2016; 78(4): 10. CrossRef - Concordance analysis and diagnostic test accuracy review of IDH1 immunohistochemistry in glioblastoma
Jung-Soo Pyo, Nae Yu Kim, Roy Hyun Jai Kim, Guhyun Kang
Brain Tumor Pathology.2016; 33(4): 248. CrossRef - Methyl Guanine Methyl Transferase Methylation Status and Epidermal Growth Factor Receptor expression in a cohort of Egyptian glioblastoma patients
Soheir M. Hamam, Bassma M. El Sabaa, Iman M. Talaat, Rasha A. Nassra, Doaa A. Abdelmonsif
Egyptian Journal of Pathology.2016; 36(2): 282. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
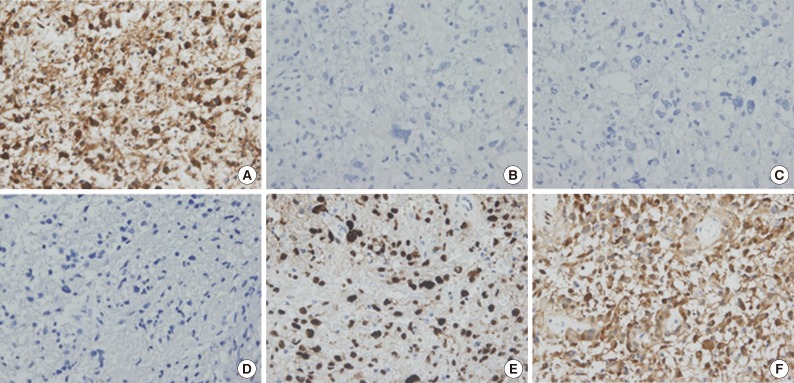
Fig. 1
| IHC | No. of cases (%) | Age (yr) | p-value |
|---|---|---|---|
| EGFR(+) | 93/150 (62.7) | 61.2 | .016 |
| p53(+) | 74/150 (49.3) | 53.5 | < .001 |
| EGFR(+)/p53(–) | 62/150 (41.3) | 65.4 | < .001 |
| EGFR(–)/p53(+) | 43/150 (28.7) | 53.8 | .019 |
| EGFR(+)/p53(+) | 31/150 (20.7) | 53.09 | .126 |
| EGFR(–)/p53(–) | 14/150 (9.3) | 57.9 | .075 |
| IDH-1 positive | IDH-1 negative | p-value | |
|---|---|---|---|
| No. of cases | 16 (11.1) | 128 (88.9) | |
| Age (yr) | 45.2 | 60.0 | < .001 |
| Male/Female ratio | 1.33 | 0.56 | .136 |
| Clinically secondary GBM | 4/4 | 0/4 | .006 |
| EGFR(+) | 7/16 (43.8) | 82/128 (64.1) | .97 |
| p53(+) | 14/16 (87.5) | 57/128 (44.5) | .01 |
| IHC | Clinically primary GBM | Clinically secondary GBM |
|---|---|---|
| IDH-1(–)/EGFR(+)/p53(–) | 58 (40.2) | 0 (0) |
| IDH-1(–)/EGFR(+)/p53(+) | 24 (16.6) | 0 (0) |
| IDH-1(–)/EGFR(–)/p53(+) | 33 (22.9) | 0 (0) |
| IDH-1(–)/EGFR(–)/p53(–) | 13 (9.0) | 0 (0) |
| IDH-1(+)/EGFR(+)/p53(–) | 1 (0.6) | 0 (0) |
| IDH-1(+)/EGFR(+)/p53(+) | 6 (4.1) | 0 (0) |
| IDH-1(+)/EGFR(–)/p53(–) | 0 (0) | 1 (0.6) |
| IDH-1(+)/EGFR(–)/p53(+) | 5 (3.4) | 3 (2.0) |
| Case No. | Age | Sex | Previous Diagnosis | EGFR | p53 | IDH-1 |
|---|---|---|---|---|---|---|
| 1 | 36 | F | Diffuse astrocytoma | – | – | + |
| 2 | 39 | M | Anaplastic astrocytoma | – | + | + |
| 3 | 45 | M | Diffuse astrocytoma | – | + | + |
| 4 | 56 | F | Anaplastic astrocytoma | – | + | + |
| 5 | 57 | F | None | – | + | + |
| 6 | 38 | M | None | – | + | + |
| 7 | 69 | F | None | – | + | + |
| 8 | 19 | F | None | – | + | + |
| 9 | 40 | M | None | – | + | + |
| Sensitivity | Specificity | Accuracy | PPV | NPV | |
|---|---|---|---|---|---|
| EGFR(+) | 65.93 | 100.00 | 68.06 | 100.00 | 16.36 |
| p53(–) | 53.33 | 88.89 | 55.56 | 98.63 | 11.27 |
| IDH-1(–) | 94.07 | 100.00 | 94.44 | 100.00 | 52.94 |
| EGFR(+) and p53(–) | 43.70 | 100.00 | 47.22 | 100.00 | 10.59 |
| EGFR(+) or p53(–) | 90.37 | 11.11 | 85.42 | 93.85 | 7.14 |
| IDH-1(–) and EGFR(+) | 60.74 | 100.00 | 63.19 | 100.00 | 14.52 |
| IDH-1(–) or EGFR(+) | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 |
| IDH-1(–) and p53(–) | 52.59 | 100.00 | 55.56 | 100.00 | 12.33 |
| IDH-1(–) or p53(–) | 95.56 | 88.89 | 95.14 | 99.23 | 57.14 |
| EGFR(+) and p53(–) and IDH-1(–) | 42.96 | 100.00 | 46.53 | 100.00 | 10.47 |
| Sensitivity | Specificity | Accuracy | PPV | NPV | |
|---|---|---|---|---|---|
| EGFR(–) | 100.00 | 65.93 | 68.06 | 16.36 | 100.00 |
| p53(+) | 88.89 | 53.33 | 55.56 | 11.27 | 98.63 |
| IDH-1(+) | 100.00 | 94.81 | 95.14 | 56.20 | 100.00 |
| EGFR(–) and p53(+) | 88.89 | 75.56 | 76.39 | 19.51 | 99.03 |
| EGFR(–) or p53(+) | 100.00 | 43.70 | 47.22 | 10.59 | 100.00 |
| IDH-1(+) and EGFR(–) | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 |
| IDH-1(+) or EGFR(–) | 100.00 | 60.74 | 63.19 | 14.52 | 100.00 |
| IDH-1(+) and p53(+) | 88.89 | 95.56 | 95.14 | 57.14 | 99.23 |
| IDH-1(+) or p53(+) | 100.00 | 53.33 | 56.25 | 12.50 | 100.00 |
| EGFR(–) and p53(+) and IDH-1(+) | 88.89 | 100.00 | 99.31 | 100.00 | 99.26 |
EGFR, epidermal growth factor receptor; IHC, immunohistochemistry.
Values are presented as number (%). IDH-1, isocitrate dehydrogenase 1; GBM, glioblastoma; EGFR, epidermal growth factor receptor.
Values are presented as number (%). IDH-1, isocitrate dehydrogenase 1; EGFR, epidermal growth factor receptor; IHC, immunohistochemistry; GBM, glioblastoma.
EGFR, epidermal growth factor receptor; IDH-1, isocitrate dehydrogenase 1; F, female; M, male.
Values are presented as percentage. PPV, positive predictive value; NPV, negative predictive value; EGFR, epidermal growth factor receptor; IDH-1, isocitrate dehydrogenase 1.
Values are presented as percentage. PPV, positive predictive value; NPV, negative predictive value; EGFR, epidermal growth factor receptor; IDH-1, isocitrate dehydrogenase 1.

 E-submission
E-submission



