Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(6); 2013 > Article
-
Original Article
Prognostic Significance of Absolute Lymphocyte Count/Absolute Monocyte Count Ratio at Diagnosis in Patients with Multiple Myeloma - Su-Jin Shin, Jin Roh, Misung Kim, Min Jung Jung, Young Wha Koh, Chan-Sik Park, Dok Hyun Yoon1, Cheolwon Suh1, Chan-Jeong Park2, Hyun Sook Chi2, Jooryung Huh
-
Korean Journal of Pathology 2013;47(6):526-533.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.6.526
Published online: December 24, 2013
Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
1Department of Oncology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
2Department of Laboratory Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- Corresponding Author: Jooryung Huh, M.D. Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, 88 Olympic-ro 43-gil, Songpa-gu, Seoul 138-736, Korea. Tel: +82-2-3010-4553, Fax: +82-2-472-7898, jrhuh@amc.seoul.kr
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Absolute lymphocyte count (ALC) in peripheral blood has recently been reported to be an independent prognostic factor in multiple myeloma (MM). Previous studies indicated that the absolute monocyte count (AMC) in peripheral blood reflects the state of the tumor microenvironment in lymphomas. Neither the utility of the AMC nor its relationship with ALC has been studied in MM.
-
Methods
- The prognostic value of ALC, AMC, and the ALC/AMC ratio at the time of diagnosis was retrospectively examined in 189 patients with MM.
-
Results
- On univariate analysis, low ALC (<1,400 cells/µL), high AMC (≥490 cells/µL), and low ALC/AMC ratio (<2.9) were correlated with worse overall survival (OS) (p=.002, p=.038, and p=.001, respectively). On multivariate analysis, the ALC/AMC ratio was an independent prognostic factor (p=.047), whereas ALC and AMC were no longer statistical significant. Low ALC, high AMC, and low ALC/AMC ratio were associated with poor prognostic factors such as high International Staging System stage, plasmablastic morphology, hypoalbuminemia, and high β2-microglobulin.
-
Conclusions
- Univariate analysis demonstrated that changes in ALC, AMC, and the ALC/AMC ratio are associated with patient survival in MM. Multivariate analysis showed that, of these factors, the ALC/AMC ratio was an independent prognostic factor for OS.
- Patients
- This study included 189 patients who were diagnosed with MM between 2001 and 2011 at the Asan Medical Center, Seoul, Korea. Disease was considered to be plasmablastic when >30% of myeloma cells in the BM biopsy exhibited plasmablastic morphology. All patients met the following criteria: BM involvement; no previous treatment; no previous history of other malignancies, transplantation or immunosuppression; no anti-human immunodeficiency virus antibodies; and availability of laboratory and radiologic data and follow-up information.
- AMC and ALC were obtained from routine complete blood count (CBC) with a four-part differential (lymphocytes, monocytes, eosinophils, and neutrophils) using a Sysmex automated hematology analyzer (model E-4000, SE-9000 or XE-2100, Sysmex Co., Kobe, Japan), which was performed at the time of the diagnosis.19
- Statistical analysis
- Overall survival (OS) was defined as the time between the date of diagnosis and the date of death from any cause. For living patients, OS was defined as the time between diagnosis and the last follow-up date. OS was analyzed using the Kaplan-Meier method and log-rank testing was used to compare groups. Median follow-up with a 95% confidence interval was calculated using the reverse Kaplan-Meier method.20 Multivariate analyses of demographic and clinical characteristics prognostic for OS were performed using the Cox proportional hazards regression model.
- The maximal chi-square method was adapted to evaluate the cutoff points in the dataset that best segregated patients into poor and good prognosis subgroups (based on the likelihood of surviving), with the log-rank test as statistic used to measure the strength of the grouping. Maxstat, a maximal chi-square package in R 2.15.2. (R Development Core Team, Vienna, Austria; http://www.R-project.org), was used to identify the optimal cut-off points for ALC, AMC and the ALC/AMC ratio.21,22 Correlations between ALC, AMC, and the ALC/AMC ratio with categorical variables were analyzed using Pearson's chi-square test or Fisher's exact test, and the Mann-Whitney U test was used to evaluate associations with continuous variables. All statistical analyses were performed using the SPSS ver. 18.0 (SPSS Inc., Chicago, IL, USA) or R 2.15.2. A p-value of ≤.05 was considered to be statistically significant.
- Ethical permission
- The Institutional Review Board (IRB) of Asan Medical Center (Seoul, Korea) approved this study protocol and provided all necessary ethical permissions.
MATERIALS AND METHODS
- Patient characteristics
- The mean age of the 189 patients with MM in the study was 60 years (range, 29 to 84 years), and the male to female ratio was 1.077:1. Paraprotein type was determined by immunoelectrophoresis; IgG was the most common (n=95, 50.3%), followed by light chain (n=48, 25.4%), IgA (n=34, 18%), IgD (n=7, 3.7%), and IgM (n=5, 2.6%). Of the 189 patients, 57 (30.2%) exhibited plasmablastic disease morphology. Punched-out, osteolytic bone lesions (>3 lesions) were identified in 96 patients (50.8%). Following diagnosis, patients were treated with chemotherapy (47.1%), radiotherapy (6.3%), chemotherapy followed by ASCT (35.4%), or conservative treatment (11.1%). At the time of analysis, 146 patients (77.2%) had died of MM, and the estimated 5-year OS was 26%.
- At the time of diagnosis, the median AMC was 372 cells/µL (range, 41 to 2,040 cells/µL), the median ALC was 1,581 cells/µL (range, 319 to 5,742 cells/µL) and the median ALC/AMC ratio was 4 (range, 0.83 to 33).
- Correlation of ALC, AMC, and the ALC/AMC ratio with clinicopathological variables
- The patients were divided into two groups according to ALC, AMC, and the ALC/AMC ratio at diagnosis. Cut-off points for the division of groups, chosen according to the results of maximal chi-square analysis to best segregate patients, were as follows: ALC 1,400 cells/µL, AMC 490 cells/µL, and an ALC/AMC ratio of 2.9. One hundred and fifteen patients (60.8%) belonged to the high ALC (≥1,400 cells/µL) group, 57 patients (30.2%) to the high AMC (≥490 cells/µL) group and 135 patients (71.4%) to the high ALC/AMC ratio (≥2.9) group.
- The low ALC, high AMC, and low ALC/AMC ratio groups were associated with poor prognostic factors such as high International Staging System (ISS) stage, plasmablastic morphology, hypoalbuminemia and high β2-microglobulin (β2m). Patients with a low ALC at diagnosis were more likely to present with a high ISS stage (p=.004), plasmablastic morphology (p=.027), hypoalbuminemia (p=.004), anemia (p=.005), low platelets (p=.001), and leukopenia (p=.001). Patients with a high AMC at diagnosis were more likely to be male (p<.0001) and present with leukocytosis (p<.0001) and high β2m (p=.015), serum creatinine (p=.014) and platelets (p=.028). Patients with a low ALC/AMC ratio at diagnosis were more likely to present with plasmablastic morphology (p=.008) and leukocytosis (p=.003), and be male (p=.011) (Table 1).
- Prognostic significance of ALC, AMC, and the ALC/AMC ratio
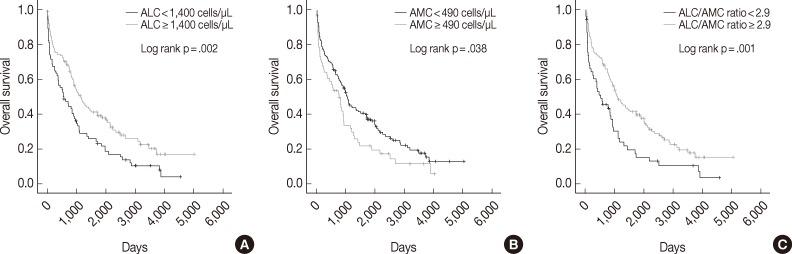
- The median follow-up period was 938 days (range, 2 to 5,011 days). Univariate analysis showed that low ALC, high AMC, and low ALC/AMC ratio were correlated with poor OS (p=.002, p=.038, and p=.001, respectively) (Fig. 1). The 5-year OS rate was also shorter in the low ALC (17% vs 33% in the high ALC group), high AMC (18% vs 30% in the low AMC group), and low ALC/AMC ratio groups (13% vs 31% in the high ALC/AMC ratio group).
- Univariate analyses also showed that other factors related to poor OS included plasmablastic morphology (p=.016), older age (≥65 years; p=.001), high ISS stage disease (p<.0001), high Durie-Salmon stage (p<.0001), high β2m (p<.0001), hypoalbuminemia (p=.009) and treatment without ASCT (p<.0001) (Table 2).
- Multivariate analysis including ALC, AMC, morphology, age, ISS stage, Durie-Salmon stage, and treatment type showed that only treatment type (ASCT vs no ASCT) was an independent prognostic factor for OS (p=.001); ALC and AMC were not statistically significant (p=.106 and p=.107, respectively). Multivariate analysis including the ALC/AMC ratio, morphology, age, ISS stage, Durie-Salmon stage, and treatment type showed that the ALC/AMC ratio and treatment type were both independent prognostic markers (p=.047 and p=.001, respectively) (Table 3).
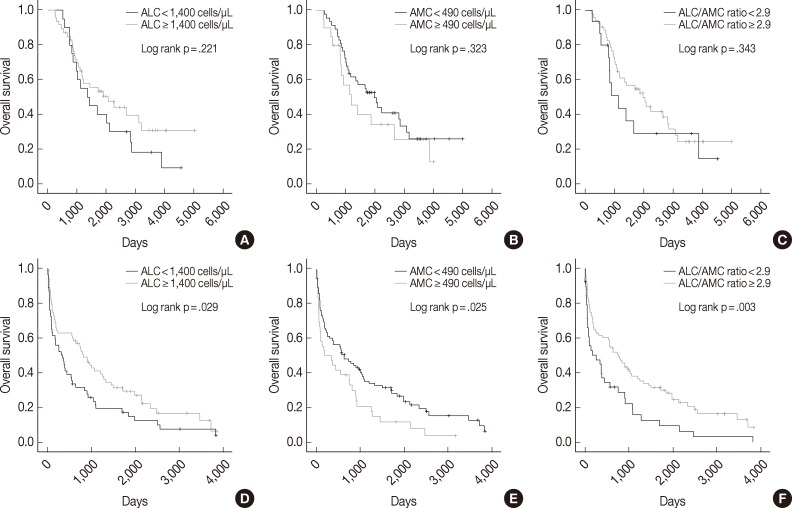
- We also assessed ALC, AMC and the ALC/AMC ratio in patients subdivided for factors that can affect prognosis and treatment selection (such as ASCT vs no ASCT, age, sex, and ISS) to determine whether they could predict OS. Among patients who did not undergo ASCT, those with low ALC, high AMC, and low ALC/AMC ratio had significantly shorter OS (p=.029, p=.025, and p=.003, respectively), but patients who received ASCT did not show significant differences in survival (p=.221, p=.323, and p=.343, respectively) (Fig. 2). When we stratified patients according to age, sex, and ISS, low ALC was associated with poor OS in patients who were <65 years-old (p=.009), male (p=.001), or had ISS stage II disease (p=.031). High AMC was associated with poor survival in the ISS stage I group (p<.0001). Low ALC/AMC ratio was associated with poor survival in patients who were <65 years-old (p=.008), male (p=.018), or had ISS stage I (p=.017) or stage III disease (p=.010) (Table 4).
RESULTS
- The aim of the present study was to investigate the role of AMC as a prognostic biomarker for MM and to determine its relationship with ALC, an established prognostic factor for MM. To our knowledge, this is the first study to demonstrate the prognostic value of AMC and the ALC/AMC ratio in newly diagnosed MM.
- Low ALC (<1,400 cells/µL), high AMC (≥490 cells/µL), and low ALC/AMC ratio (<2.9) were associated with poor OS and poor prognostic factors such as high ISS stage, plasmablastic morphology, hypoalbuminemia, and high β2m. However, on multivariate analysis, only low ALC/AMC ratio was an independent prognostic factor for MM.
- Although ALC did not have statistical significance in multivariate analysis due to the relatively small sample size, our results are in agreement with previous reports showing that a high ALC is associated with better prognosis in newly diagnosed MM (cut-off point, 1,400 cells/µL; the same as that used our study),3 relapsed MM after Vel-Dex therapy (cut-off point, 1,100 cells/µL)23 and other lymphomas.24
- We also found that shorter OS in MM patients was associated with high AMC and low ALC/AMC ratio, which is similar to the findings of previous studies of malignant lymphomas. AMC has been shown to be a reliable prognostic marker for DLBCL (cut-off point, 630 cells/µL),16 FL (cut-off point, 570 cells/µL),17 and HL (cut-off point, 900 cells/µL),18 and in classical HL, the ALC/AMC ratio at diagnosis is an independent prognostic factor for survival (cut-off point, 1.1).18
- Recently it has been reported that TAMs are a prognostic marker for the survival of patients with MM.13 TAMs, which constitute a significant proportion of tumor-infiltrating inflammatory cells, have been linked to the growth, angiogenesis and metastasis of a variety of cancers.7 In MM, macrophages are an abundant and important component of BM stromal cells and contribute to tumor angiogenesis.25,26 TAMs, which are continually being recruited and activated both in an autocrine manner and by cytokines secreted by myeloma cells, adapt functionally, phenotypically and morphologically to collaborate with endothelial cells in vessel formation.27 Moreover, BM macrophages protect myeloma cells from apoptosis.28 TAMs therefore promote tumor growth not only by supporting angiogenesis, but also by protecting tumor cells from apoptosis, via the induction of immunosuppression.29 The important role TAMs play in the biology of MM, thereby affecting patient outcome, makes them a potential target for anti-vascular therapy in MM.26
- As TAMs are derived from circulating monocytes and are recruited to the tumor site by tumor-derived chemotactic factors,14,15,30 obtaining an AMC from a CBC at diagnosis is simple and could be more easily applied in clinical practice than counting TAMs. In this study, the ALC/AMC ratio was an independent prognostic factor in MM. Moreover, the ALC/AMC ratio, which may be a reflection of both the tumor microenvironment and host immune status, could provide prognostic information independently.
- We found that the patient group with high AMC and low ALC/AMC ratio trended toward worse OS whether or not they received ASCT, although statistical significance was not reached in the ASCT group because of the small sample size. A larger prospective cohort study of uniformly treated patients is required to validate these findings.
- In conclusion, the results of the present study suggest that AMC, and, in particular, ALC/AMC ratio, which was found to be an independent prognostic factor for patient survival, could be used to determine the prognosis of MM.
DISCUSSION
- 1. Porrata LF, Ristow K, Habermann TM, Witzig TE, Inwards DJ, Markovic SN. Absolute lymphocyte count at the time of first relapse predicts survival in patients with diffuse large B-cell lymphoma. Am J Hematol 2009; 84: 93-97. ArticlePubMed
- 2. Kim H, Sohn HJ, Kim S, Lee JS, Kim WK, Suh C. Early lymphocyte recovery predicts longer survival after autologous peripheral blood stem cell transplantation in multiple myeloma. Bone Marrow Transplant 2006; 37: 1037-1042. ArticlePubMedPDF
- 3. Ege H, Gertz MA, Markovic SN, et al. Prediction of survival using absolute lymphocyte count for newly diagnosed patients with multiple myeloma: a retrospective study. Br J Haematol 2008; 141: 792-798. ArticlePubMed
- 4. Podar K, Chauhan D, Anderson KC. Bone marrow microenvironment and the identification of new targets for myeloma therapy. Leukemia 2009; 23: 10-24. ArticlePubMedPDF
- 5. Mitsiades CS, Mitsiades NS, Richardson PG, Munshi NC, Anderson KC. Multiple myeloma: a prototypic disease model for the characterization and therapeutic targeting of interactions between tumor cells and their local microenvironment. J Cell Biochem 2007; 101: 950-968. ArticlePubMed
- 6. Coussens LM, Werb Z. Inflammation and cancer. Nature 2002; 420: 860-867. ArticlePubMedPMCPDF
- 7. Pollard JW. Tumour-educated macrophages promote tumour progression and metastasis. Nat Rev Cancer 2004; 4: 71-78. ArticlePubMedPDF
- 8. Lamagna C, Aurrand-Lions M, Imhof BA. Dual role of macrophages in tumor growth and angiogenesis. J Leukoc Biol 2006; 80: 705-713. ArticlePubMedPDF
- 9. Alvaro-Naranjo T, Lejeune M, Salvadó-Usach MT, et al. Tumor-infiltrating cells as a prognostic factor in Hodgkin's lymphoma: a quantitative tissue microarray study in a large retrospective cohort of 267 patients. Leuk Lymphoma 2005; 46: 1581-1591. ArticlePubMed
- 10. Steidl C, Lee T, Shah SP, et al. Tumor-associated macrophages and survival in classic Hodgkin's lymphoma. N Engl J Med 2010; 362: 875-885. ArticlePubMedPMC
- 11. Pusztaszeri MP, Seelentag W, Bosman FT. Immunohistochemical expression of endothelial markers CD31, CD34, von Willebrand factor, and Fli-1 in normal human tissues. J Histochem Cytochem 2006; 54: 385-395. ArticlePubMedPDF
- 12. Farinha P, Masoudi H, Skinnider BF, et al. Analysis of multiple biomarkers shows that lymphoma-associated macrophage (LAM) content is an independent predictor of survival in follicular lymphoma (FL). Blood 2005; 106: 2169-2174. ArticlePubMedPDF
- 13. Suyanı E, Sucak GT, Akyürek N, et al. Tumor-associated macrophages as a prognostic parameter in multiple myeloma. Ann Hematol 2013; 92: 669-677. ArticlePubMedPDF
- 14. Ribatti D, Nico B, Crivellato E, Vacca A. Macrophages and tumor angiogenesis. Leukemia 2007; 21: 2085-2089. ArticlePubMedPDF
- 15. Green CE, Liu T, Montel V, et al. Chemoattractant signaling between tumor cells and macrophages regulates cancer cell migration, metastasis and neovascularization. PLoS One 2009; 4: e6713.ArticlePubMedPMC
- 16. Porrata LF, Ristow K, Habermann TM, et al. Absolute monocyte/lymphocyte count prognostic score is independent of immunohistochemically determined cell of origin in predicting survival in diffuse large B-cell lymphoma. Leuk Lymphoma 2012; 53: 2159-2165. ArticlePubMed
- 17. Wilcox RA, Ristow K, Habermann TM, et al. The absolute monocyte count is associated with overall survival in patients newly diagnosed with follicular lymphoma. Leuk Lymphoma 2012; 53: 575-580. ArticlePubMed
- 18. Porrata LF, Ristow K, Colgan JP, et al. Peripheral blood lymphocyte/monocyte ratio at diagnosis and survival in classical Hodgkin's lymphoma. Haematologica 2012; 97: 262-269. ArticlePubMedPMC
- 19. Inoue H. Overview of automated hematology analyzer XE-2100TM. Sysmex J Int 1999; 9: 58-64.
- 20. Clark TG, Bradburn MJ, Love SB, Altman DG. Survival analysis part I: basic concepts and first analyses. Br J Cancer 2003; 89: 232-238. ArticlePubMedPMCPDF
- 21. Hothorn T, Lausen B. On the exact distribution of maximally selected rank statistics. Comput Stat Data Anal 2003; 43: 121-137. Article
- 22. Boulesteix AL. Maximally selected chi-square statistics for ordinal variables. Biom J 2006; 48: 451-462. ArticlePubMed
- 23. Song MK, Chung JS, Joo YD, et al. Clinical value of absolute lymphocyte counts before bortezomib-dexamethasone therapy in relapsed multiple myeloma patients. Acta Haematol 2010; 124: 34-39. ArticlePubMedPDF
- 24. Le Jeune C, Bertoli S, Elhamri M, et al. Initial absolute lymphocyte count as a prognostic factor for outcome in acute myeloid leukemia. Leuk Lymphoma 2013 8 05 [Epub]. http://dx.doi.org/10.3109/10428194.2013.813504.
- 25. Scavelli C, Nico B, Cirulli T, et al. Vasculogenic mimicry by bone marrow macrophages in patients with multiple myeloma. Oncogene 2008; 27: 663-674. ArticlePubMedPDF
- 26. Berardi S, Ria R, Reale A, et al. Multiple myeloma macrophages: pivotal players in the tumor microenvironment. J Oncol 2013; 2013: 183602.ArticlePubMedPMCPDF
- 27. Vacca A, Ribatti D, Presta M, et al. Bone marrow neovascularization, plasma cell angiogenic potential, and matrix metalloproteinase-2 secretion parallel progression of human multiple myeloma. Blood 1999; 93: 3064-3073. ArticlePubMedPDF
- 28. Zheng Y, Cai Z, Wang S, et al. Macrophages are an abundant component of myeloma microenvironment and protect myeloma cells from chemotherapy drug-induced apoptosis. Blood 2009; 114: 3625-3628. ArticlePubMedPMCPDF
- 29. Ben-Baruch A. Inflammation-associated immune suppression in cancer: the roles played by cytokines, chemokines and additional mediators. Semin Cancer Biol 2006; 16: 38-52. ArticlePubMed
- 30. Dirkx AE, Oude Egbrink MG, Wagstaff J, Griffioen AW. Monocyte/macrophage infiltration in tumors: modulators of angiogenesis. J Leukoc Biol 2006; 80: 1183-1196. ArticlePubMedPDF
REFERENCES
| Characteristic | ALC < 1,400 cells/µL | ALC ≥ 1,400 cells/µL | p-value | AMC < 490 cells/µL | AMC ≥ 490 cells/µL | p-value | ALC/AMC < 2.9 | ALC/AMC ≥ 2.9 | p-value |
|---|---|---|---|---|---|---|---|---|---|
| Age | 60 (31-81) | 60 (29-84) | .939a | 60 (29-81) | 60 (31-84) | .934a | 62 (31-84) | 59 (29-81) | .130a |
| Sex | .459b | < .0001b | .011b | ||||||
| Male | 41 (41.8) | 57 (58.2) | 55 (56.1) | 43 (43.9) | 36 (36.7) | 62 (63.3) | |||
| Female | 33 (36.3) | 58 (63.7) | 77 (84.6) | 14 (15.4) | 18 (19.8) | 73 (80.2) | |||
| International Staging System (ISS) | .004c | .195c | .142c | ||||||
| I | 8 (22.9) | 27 (77.1) | 27 (77.1) | 8 (22.9) | 7 (20.0) | 28 (80.0) | |||
| II | 29 (33.3) | 58 (66.7) | 62 (71.3) | 25 (28.7) | 23 (26.4) | 64 (73.6) | |||
| III | 33 (54.1) | 28 (45.9) | 37 (60.7) | 24 (39.3) | 23 (37.7) | 38 (62.3) | |||
| Durie-Salmon stage | .745c | .641c | .263c | ||||||
| I | 6 (28.6) | 15 (71.4) | 15 (71.4) | 6 (28.6) | 8 (38.1) | 13 (61.9) | |||
| II | 22 (42.3) | 30 (57.7) | 37 (71.2) | 15 (28.8) | 12 (23.1) | 40 (76.9) | |||
| IIIA | 35 (39.3) | 54 (60.7) | 64 (71.9) | 25 (28.1) | 23 (25.8) | 66 (74.2) | |||
| IIIB | 11 (40.7) | 16 (59.3) | 16 (59.3) | 11 (40.7) | 11 (40.7) | 16 (59.3) | |||
| Morphology | .027b | .112b | .008b | ||||||
| Plasmablastic | 29 (50.9) | 28 (49.1) | 33 (57.9) | 24 (42.1) | 26 (45.6) | 31 (54.4) | |||
| Plasmacytic | 32 (32.3) | 67 (67.7) | 71 (71.7) | 28 (28.3) | 24 (24.2) | 75 (75.8) | |||
| Bone lesion | .233b | 1.000b | .873b | ||||||
| ≥ 3 lytic bone lesions | 42 (43.8) | 54 (56.3) | 67 (69.8) | 29 (30.2) | 28 (29.2) | 68 (70.8) | |||
| < 3 lytic bone lesions | 32 (34.4) | 61 (65.6) | 65 (69.9) | 28 (30.1) | 26 (28.0) | 67 (72.0) | |||
| β2m (mg/L) | 8.98 (0.89-82.50) | 7.80 (1.40-65.80) | .122a | 6.47 (0.89-48.40) | 12.20 (1.60-82.50) | .015a | 10.96 (0.89-82.50) | 7.14 (1.00-63.80) | .087a |
| Albumin (g/dL) | 3.00 (0.90-10.70) | 3.22 (1.10-4.60) | .004a | 3.12 (0.90-10.70) | 3.17 (0.90-4.60) | .316a | 2.99 (0.90-4.50) | 3.19 (0.90-10.70) | .265a |
| Cr (mg/dL) | 1.70 (0.50-14.40) | 1.75 (0.39-18.70) | .976a | 1.50 (0.50-14.40) | 2.27 (0.39-18.70) | .014a | 2.22 (0.39-18.70) | 1.53 (0.50-8.50) | .282a |
| Hb (g/dL) | 9.81 (6.40-14.80) | 10.68 (5.50-15.40) | .005a | 10.17 (5.50-15.40) | 10.74 (6.30-15.00) | .085a | 10.48 (6.30-14.80) | 10.29 (5.50-15.40) | .510a |
| Ca (mg/dL) | 9.13 (4.00-14.60) | 9.35 (6.70-15.10) | .131a | 9.14 (4.00-14.10) | 9.56 (7.10–15.10) | .098a | 9.57 (7.10-15.10) | 9.14 (4.00-13.80) | .928a |
| Serum protein (g/dL) | 8.22 (3.90-17.00) | 8.17 (4.70-13.60) | .453a | 8.30 (3.90-17.00) | 7.93 (4.70-12.50) | .280a | 7.90 (4.60-12.50) | 8.30 (3.90-17.00) | .373a |
| 24 hr urine protein (mg/day) | 2,092 (11-13,947) | 2,123 (9-14,286) | .912a | 2,049 (9-14,286) | 2,261 (11-13,947) | .793a | 1,749 (11-13,947) | 2,250 (9-14,286) | .238a |
| WBC count (× 109/L) | 5.72 (2.40-12.70) | 6.86 (2.90-17.40) | .001a | 5.55 (2.40-16.00) | 8.40 (4.40-17.40) | < .0001a | 7.44 (2.40-17.00) | 5.99 (2.40-17.40) | .003a |
| Platelets (× 109/L) | 176 (23-461) | 225 (39-663) | .001a | 194 (31-523) | 231 (23-663) | .028a | 199 (23-663) | 208 (38-584) | .448a |
Figure & Data
References
Citations

- Association between systemic immune-inflammatory markers and monoclonal gammopathy of undetermined significance in the elderly population: findings from the National health and nutrition examination survey
Chengpeng Zhang, Kangyan Hou, Dongjun Lin, Cong Xu
Clinical and Experimental Medicine.2026;[Epub] CrossRef - Low peripheral blood B lymphocyte count predicts poor outcome in patients with multiple myeloma
Yuqi Wang, Zhongxin Zheng, Qiaoxi Kang, Linjing Cai, Shanshan Zhang, Huan Chen, Youhai Yuan, Hanzhen Zhang, Xiaolei Wei, Ru Feng, Yongqiang Wei
Immunobiology.2025; 230(4): 153096. CrossRef - Inflammatory markers from routine blood tests predict survival in multiple myeloma: a Systematic Review and meta-analysis
Mengjiao Luo, Ling Qin, Yujie Li, Qianru Mei, Qiaoping Wu, Xudong Feng
Frontiers in Immunology.2025;[Epub] CrossRef - Variation of peripheral blood-based biomarkers for response of anti-PD-1 immunotherapy in non-small-cell lung cancer
Xiaoming Wang, Dayu Chen, Yuyan Ma, Dongping Mo, Feng Yan
Clinical and Translational Oncology.2024; 26(8): 1934. CrossRef - Descriptive analysis and prognostic factors in cats with myeloma-related disorders: A multicenter retrospective study of 50 cases
Lorris Lecot, Isabelle Desmas-Bazelle, Sarah Benjamin, Pauline De Fornel, Frédérique Ponce, Matthew Kornya, Loïc Desquilbet, Claire Beaudu-Lange, Catherine Ibisch, David Sayag, Ghita Benchekroun, Jérémy Béguin
Journal of Veterinary Internal Medicine.2024; 38(3): 1693. CrossRef - Clinical features and treatment of newly diagnosed multiple myeloma with secondary myelofibrosis: a retrospective study
Han Xu, Yujie Xu, Mengying Wang, Chunxia Mao, Junxia Huang, Tianlan Li, Yan Gao, Shanshan Liu, Jingjing Zhou, Yi Zhang, Xianqi Feng
Therapeutic Advances in Hematology.2024;[Epub] CrossRef - Definers and drivers of functional high-risk multiple myeloma: insights from genomic, transcriptomic, and immune profiling
Rahul Banerjee, Kara I. Cicero, Sarah S. Lee, Andrew J. Cowan
Frontiers in Oncology.2023;[Epub] CrossRef - Normal Absolute Monocyte Count in Combination with Normal/High Absolute Lymphocyte Count at the Time of Relapse is Associated with Improved Survival in Patients with Early Relapsed Acute Myeloid Leukemia
Yu Zhang, Kanchun Dai, Qianying Zhang, Yisha Huang, Yiyun Feng, Deeksha Bhardwaj, Kang Yu, Jianhua Feng
Cancer Investigation.2021; 39(6-7): 550. CrossRef - Real World Experience of Daratumumab: Evaluating Lymphopenia and Adverse Events in Multiple Myeloma Patients
Francesca Cottini, Ying Huang, Nita Williams, Naresh Bumma, Abdullah M. Khan, Maria Chaudhry, Srinivas Devarakonda, Yvonne A. Efebera, Don M. Benson, Ashley E. Rosko
Frontiers in Oncology.2021;[Epub] CrossRef - Are the Derived Indexes of Peripheral Whole Blood Cell Counts (NLR, PLR, LMR/MLR) Clinically Significant Prognostic Biomarkers in Multiple Myeloma? A Systematic Review And Meta-Analysis
Xinwen Zhang, Jialin Duan, Zhenyu Wen, Hao Xiong, Xiaomin Chen, Yang Liu, Kunyu Liao, Chunlan Huang
Frontiers in Oncology.2021;[Epub] CrossRef - Combined immune score of lymphocyte to monocyte ratio and immunoglobulin levels predicts treatment-free survival of multiple myeloma patients after autologous stem cell transplant
Karen Sweiss, Jonathan Lee, Nadim Mahmud, Gregory S. Calip, Youngmin Park, Dolores Mahmud, Damiano Rondelli, Pritesh R. Patel
Bone Marrow Transplantation.2020; 55(1): 199. CrossRef - Low absolute CD4+ T cell counts in peripheral blood predict poor prognosis in patients with newly diagnosed multiple myeloma
Yan Gu, Yuanyuan Jin, Jie Ding, Wu Yujie, Qinglin Shi, Xiaoyan Qu, Sishu Zhao, Jianyong Li, Chen Lijuan
Leukemia & Lymphoma.2020; 61(8): 1869. CrossRef Normal Absolute Monocyte Count at the Time of Relapse is Associated with Improved Survival After First Salvage Therapy in Adult Patients with Early Relapsed B-Lineage Acute Lymphoblastic Leukemia
Yi-fen Shi, Na Wang, Zi-yang Huang, Rong-rong Chen, Yi-sha Huang, Yi-yi Zhu, Chong-yun Xing, Bin Liang, Kang Yu, Jian-hua Feng
Cancer Management and Research.2020; Volume 12: 7097. CrossRef- Peripheral blood biomarkers of early immune reconstitution in newly diagnosed multiple myeloma
Moritz Binder, S. Vincent Rajkumar, Martha Q. Lacy, Morie A. Gertz, Francis K. Buadi, Angela Dispenzieri, Yi L. Hwa, Amie Fonder, Miriam Hobbs, Suzanne R. Hayman, Steven R. Zeldenrust, John A. Lust, Stephen J. Russell, Nelson Leung, Prashant Kapoor, Ronal
American Journal of Hematology.2019; 94(3): 306. CrossRef - Effect of absolute monocyte count post-transplant on the outcome of patients with acute myeloid leukemia undergoing myeloablative allogeneic hematopoietic stem cell transplant with busulfan and cyclophosphamide conditioning
Liyuan Tang, Na Wang, Chongyun Xing, Qiang Zhuang, Bin Liang, Lan Sun, Yi Chen, Yan Qian, Zhijian Shen, Songfu Jiang, Kang Yu, Jianhua Feng
Leukemia Research.2018; 69: 60. CrossRef - A lower ALC/AMC ratio is associated with poor prognosis of peripheral T-cell lymphoma-not otherwise specified
Qian Li, Shuang Gao, Jing Ma, Su Liu, Yuanfang Yue, Lin Chen, Han Li, Xue Wang, Dongying Li, Zeng Cao, Zhigang Zhao, Xiaofang Wang, Yong Yu, Yizhuo Zhang, Yafei Wang
Leukemia Research.2018; 73: 5. CrossRef - Peripheral Blood Lymphocyte-to-Monocyte Ratio as a Useful Prognostic Factor in Newly Diagnosed Multiple Myeloma
Ying Tian, Yue Zhang, Wan-Qiu Zhu, Xiao-Lei Chen, He-Bing Zhou, Wen-Ming Chen
BioMed Research International.2018; 2018: 1. CrossRef - Significance of the absolute lymphocyte/monocyte ratio as a prognostic immune biomarker in newly diagnosed multiple myeloma
T Dosani, F Covut, R Beck, J J Driscoll, M de Lima, E Malek
Blood Cancer Journal.2017; 7(6): e579. CrossRef - Lymphocyte-to-monocyte ratio can predict mortality in pancreatic adenocarcinoma
Gurshawn Singh, Ammar Nassri, David Kim, Hong Zhu, Zeeshan Ramzan
World Journal of Gastrointestinal Pharmacology and Therapeutics.2017; 8(1): 60. CrossRef - Bone marrow microenvironmental CD4 + and CD8 + lymphocyte infiltration patterns define overall- and progression free survival in standard risk multiple myeloma – an analysis from the Austrian Myeloma Registry
Wolfgang Willenbacher, Ella Willenbacher, Claudia Zelle-Rieser, Rainer Biedermann, Roman Weger, Karin Jöhrer, Andrea Brunner
Leukemia & Lymphoma.2016; 57(6): 1478. CrossRef - Absolute lymphocyte count as a prognostic marker in newly diagnosed multiple myeloma patients
C. Suriu, L. Akria, D. Azoulay, E. Shaoul, M. Barhoum, A. Braester
International Journal of Laboratory Hematology.2016;[Epub] CrossRef - The Peripheral Blood Mononuclear Cell Count Is Associated With Bone Health in Elderly Men
Xianfeng Lin, Hejun Yu, Chenchen Zhao, Yu Qian, Dun Hong, Kangmao Huang, Jian Mo, An Qin, Xiangqian Fang, Shunwu Fan
Medicine.2016; 95(15): e3357. CrossRef - Prognostic role of lymphocyte to monocyte ratio for patients with cancer: evidence from a systematic review and meta-analysis
Liangyou Gu, Hongzhao Li, Luyao Chen, Xin Ma, Xintao Li, Yu Gao, Yu Zhang, Yongpeng Xie, Xu Zhang
Oncotarget.2016; 7(22): 31926. CrossRef - Do lymphocytes count in myeloma? Are we absolutely sure?
Tamar Tadmor
Leukemia & Lymphoma.2015; 56(5): 1193. CrossRef - Absolute lymphocyte count is unrelated to overall survival in newly diagnosed elderly patients with multiple myeloma treated with immunomodulatory drugs
Mariasanta Napolitano, Giorgia Saccullo, Roberto Bono, Antonio Branca, Clotilde Cangialosi, Salvatrice Mancuso, Simona Raso, Gerlando Quintini, Maria Grazia Lipari, Francesco Fabbiano, Giorgina Specchia, Alberto Dolce, Francesco Di Raimondo, Sergio Siragu
Leukemia & Lymphoma.2015; 56(5): 1507. CrossRef - Distinct Transcriptional and Anti-Mycobacterial Profiles of Peripheral Blood Monocytes Dependent on the Ratio of Monocytes: Lymphocytes
Vivek Naranbhai, Helen A. Fletcher, Rachel Tanner, Matthew K. O'Shea, Helen McShane, Benjamin P. Fairfax, Julian C. Knight, Adrian V.S. Hill
EBioMedicine.2015; 2(11): 1619. CrossRef - Prognostic value of absolute monocyte count in chronic lymphocytic leukaemia
László Szerafin, János Jakó, Ferenc Riskó
Orvosi Hetilap.2015; 156(15): 592. CrossRef - The lymphocyte to monocyte ratio in peripheral blood represents a novel prognostic marker in patients with pancreatic cancer
Michael Stotz, Joanna Szkandera, Tatjana Stojakovic, Julia Seidel, Hellmut Samonigg, Peter Kornprat, Renate Schaberl-Moser, Fridericke Seggewies, Gerald Hoefler, Armin Gerger, Martin Pichler
Clinical Chemistry and Laboratory Medicine (CCLM).2015;[Epub] CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
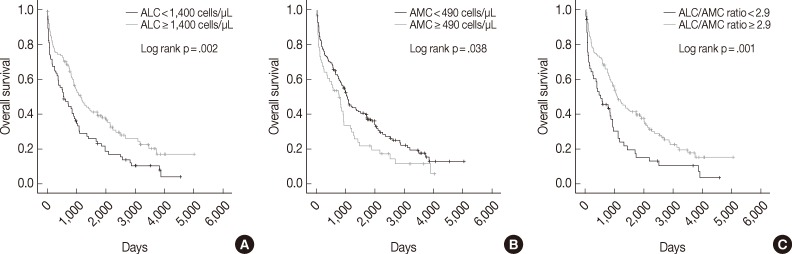
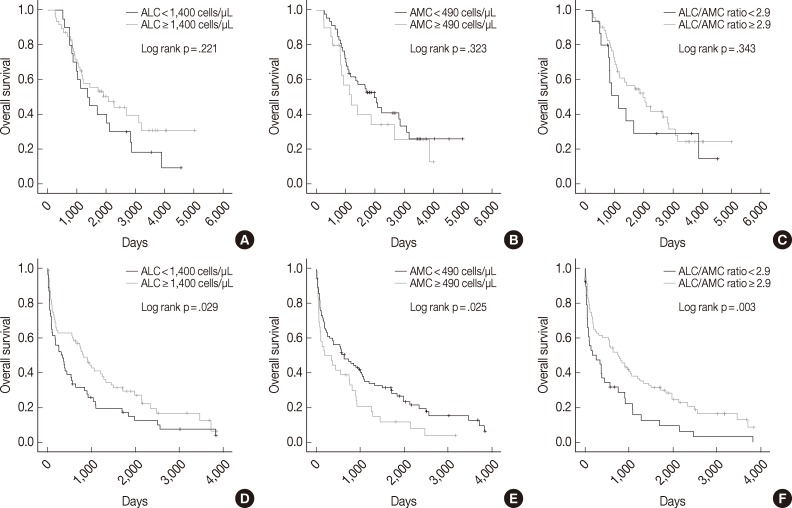
Fig. 1
Fig. 2
| Characteristic | ALC < 1,400 cells/µL | ALC ≥ 1,400 cells/µL | p-value | AMC < 490 cells/µL | AMC ≥ 490 cells/µL | p-value | ALC/AMC < 2.9 | ALC/AMC ≥ 2.9 | p-value |
|---|---|---|---|---|---|---|---|---|---|
| Age | 60 (31-81) | 60 (29-84) | .939 |
60 (29-81) | 60 (31-84) | .934 |
62 (31-84) | 59 (29-81) | .130 |
| Sex | .459 |
< .0001 |
.011 |
||||||
| Male | 41 (41.8) | 57 (58.2) | 55 (56.1) | 43 (43.9) | 36 (36.7) | 62 (63.3) | |||
| Female | 33 (36.3) | 58 (63.7) | 77 (84.6) | 14 (15.4) | 18 (19.8) | 73 (80.2) | |||
| International Staging System (ISS) | .004 |
.195 |
.142 |
||||||
| I | 8 (22.9) | 27 (77.1) | 27 (77.1) | 8 (22.9) | 7 (20.0) | 28 (80.0) | |||
| II | 29 (33.3) | 58 (66.7) | 62 (71.3) | 25 (28.7) | 23 (26.4) | 64 (73.6) | |||
| III | 33 (54.1) | 28 (45.9) | 37 (60.7) | 24 (39.3) | 23 (37.7) | 38 (62.3) | |||
| Durie-Salmon stage | .745 |
.641 |
.263 |
||||||
| I | 6 (28.6) | 15 (71.4) | 15 (71.4) | 6 (28.6) | 8 (38.1) | 13 (61.9) | |||
| II | 22 (42.3) | 30 (57.7) | 37 (71.2) | 15 (28.8) | 12 (23.1) | 40 (76.9) | |||
| IIIA | 35 (39.3) | 54 (60.7) | 64 (71.9) | 25 (28.1) | 23 (25.8) | 66 (74.2) | |||
| IIIB | 11 (40.7) | 16 (59.3) | 16 (59.3) | 11 (40.7) | 11 (40.7) | 16 (59.3) | |||
| Morphology | .027 |
.112 |
.008 |
||||||
| Plasmablastic | 29 (50.9) | 28 (49.1) | 33 (57.9) | 24 (42.1) | 26 (45.6) | 31 (54.4) | |||
| Plasmacytic | 32 (32.3) | 67 (67.7) | 71 (71.7) | 28 (28.3) | 24 (24.2) | 75 (75.8) | |||
| Bone lesion | .233 |
1.000 |
.873 |
||||||
| ≥ 3 lytic bone lesions | 42 (43.8) | 54 (56.3) | 67 (69.8) | 29 (30.2) | 28 (29.2) | 68 (70.8) | |||
| < 3 lytic bone lesions | 32 (34.4) | 61 (65.6) | 65 (69.9) | 28 (30.1) | 26 (28.0) | 67 (72.0) | |||
| β2m (mg/L) | 8.98 (0.89-82.50) | 7.80 (1.40-65.80) | .122 |
6.47 (0.89-48.40) | 12.20 (1.60-82.50) | .015 |
10.96 (0.89-82.50) | 7.14 (1.00-63.80) | .087 |
| Albumin (g/dL) | 3.00 (0.90-10.70) | 3.22 (1.10-4.60) | .004 |
3.12 (0.90-10.70) | 3.17 (0.90-4.60) | .316 |
2.99 (0.90-4.50) | 3.19 (0.90-10.70) | .265 |
| Cr (mg/dL) | 1.70 (0.50-14.40) | 1.75 (0.39-18.70) | .976 |
1.50 (0.50-14.40) | 2.27 (0.39-18.70) | .014 |
2.22 (0.39-18.70) | 1.53 (0.50-8.50) | .282 |
| Hb (g/dL) | 9.81 (6.40-14.80) | 10.68 (5.50-15.40) | .005 |
10.17 (5.50-15.40) | 10.74 (6.30-15.00) | .085 |
10.48 (6.30-14.80) | 10.29 (5.50-15.40) | .510 |
| Ca (mg/dL) | 9.13 (4.00-14.60) | 9.35 (6.70-15.10) | .131 |
9.14 (4.00-14.10) | 9.56 (7.10–15.10) | .098 |
9.57 (7.10-15.10) | 9.14 (4.00-13.80) | .928 |
| Serum protein (g/dL) | 8.22 (3.90-17.00) | 8.17 (4.70-13.60) | .453 |
8.30 (3.90-17.00) | 7.93 (4.70-12.50) | .280 |
7.90 (4.60-12.50) | 8.30 (3.90-17.00) | .373 |
| 24 hr urine protein (mg/day) | 2,092 (11-13,947) | 2,123 (9-14,286) | .912 |
2,049 (9-14,286) | 2,261 (11-13,947) | .793 |
1,749 (11-13,947) | 2,250 (9-14,286) | .238 |
| WBC count (× 109/L) | 5.72 (2.40-12.70) | 6.86 (2.90-17.40) | .001 |
5.55 (2.40-16.00) | 8.40 (4.40-17.40) | < .0001 |
7.44 (2.40-17.00) | 5.99 (2.40-17.40) | .003 |
| Platelets (× 109/L) | 176 (23-461) | 225 (39-663) | .001 |
194 (31-523) | 231 (23-663) | .028 |
199 (23-663) | 208 (38-584) | .448 |
| Factor | HR | 95% CI | p-value | |
|---|---|---|---|---|
| ALC (cells/µL) | ≥ 1,400 vs < 1,400 | 1.649 | 1.189-2.286 | .002 |
| AMC (cells/µL) | < 490 vs ≥ 490 | 1.443 | 1.018-2.045 | .038 |
| ALC/AMC ratio | ≥ 2.9 vs < 2.9 | 1.772 | 1.249-2.514 | .001 |
| Morphology | Plasmacytic vs plasmablastic | 1.552 | 1.081-2.228 | .016 |
| Age (yr) | < 65 vs ≥ 65 | 1.793 | 1.279-2.513 | .001 |
| ISS | I | 1 | ||
| II | 2.148 | 1.258-3.670 | .005 | |
| III | 4.360 | 2.526-7.523 | < .0001 | |
| D-S stage | I | 1 | ||
| II | 1.982 | 1.061-3.702 | .032 | |
| IIIA | 1.403 | 0.769-2.560 | .270 | |
| IIIB | 3.368 | 1.712-6.627 | < .0001 | |
| β2-Microglobulin (mg/L) | < 3.5 vs ≥ 3.5 | 2.128 | 1.468-3.084 | < .0001 |
| Albumin (g/dL) | < 3.5 vs ≥ 3.5 | 1.573 | 1.115-2.219 | .009 |
| Bone lesion (lytic bone lesions) | < 3 vs ≥ 3 | 1.108 | 0.799-1.535 | .539 |
| Treatment type | ASCT vs no ASCT | 2.345 | 1.637-3.359 | < .0001 |
| Factors | HR | 95% CI | p-value | HR | 95% CI | p-value | |
|---|---|---|---|---|---|---|---|
| ALC (cells/µL) | ≥ 1,400 vs < 1,400 | 1.389 | 0.933-2.069 | .106 | |||
| AMC (cells/µL) | < 490 vs ≥ 490 | 1.415 | 0.928-2.157 | .107 | |||
| ALC/AMC ratio | ≥ 2.9 vs < 2.9 | 1.511 | 1.006-2.272 | .047 | |||
| Morphology | Plasmacytic vs plasmablastic | 1.396 | 0.940-2.074 | .099 | 1.366 | 0.917-2.033 | .125 |
| Age (yr) | < 65 vs ≥ 65 | 1.441 | 0.949-2.189 | .087 | 1.388 | 0.910-2.116 | .128 |
| ISS | I | 1 | 1 | ||||
| II | 1.129 | 0.543-2.348 | .745 | 1.122 | 0.540-2.331 | .757 | |
| III | 1.799 | 0.824-3.925 | .140 | 2.048 | 0.947-4.429 | .069 | |
| D-S stage | I | 1 | 1 | ||||
| II | 1.089 | 0.454-2.611 | .849 | 1.151 | 0.482-2.750 | .751 | |
| IIIA | 0.997 | 0.427-2.326 | .994 | 1.099 | 0.479-2.520 | .824 | |
| IIIB | 1.786 | 0.681-4.683 | .239 | 1.899 | 0.725-4.975 | .192 | |
| Treatment type | ASCT vs no ASCT | 2.252 | 1.405-3.610 | .001 | 2.131 | 1.337-3.396 | .001 |
| Characteristics | ALC < 1,400 cells/µL |
AMC ≥ 490 cells/µL |
ALC/AMC < 2.9 |
||||||
|---|---|---|---|---|---|---|---|---|---|
| HR | 95% CI | p-value | HR | 95% CI | p-value | HR | 95% CI | p-value | |
| Treatment | |||||||||
| ASCT (n = 67) | 1.459 | 0.794-2.679 | .221 | 1.376 | 0.728-2.600 | .323 | 1.393 | 0.700-2.769 | .343 |
| No ASCT (n = 122) | 1.542 | 1.043-2.281 | .029 | 1.613 | 1.058-2.457 | .025 | 1.850 | 1.227-2.789 | .003 |
| Age (yr) | |||||||||
| < 65 (n = 121) | 1.728 | 1.139-2.622 | .009 | 1.517 | 0.971-2.369 | .065 | 1.843 | 1.163-2.920 | .008 |
| ≥ 65 (n = 68) | 1.606 | 0.934-2.762 | .084 | 1.323 | 0.752-2.327 | .330 | 1.408 | 0.812-2.442 | .221 |
| Sex | |||||||||
| Male (n = 98) | 2.112 | 1.351-3.301 | .001 | 1.259 | 0.806-1.967 | .310 | 1.719 | 1.091-2.708 | .018 |
| Female (n = 91) | 1.245 | 0.764-2.028 | .379 | 1.433 | 0.748-2.744 | .276 | 1.559 | 0.873-2.784 | .130 |
| ISS | |||||||||
| I (n = 35) | 1.303 | 0.451-3.761 | .624 | 6.865 | 2.408-19.576 | < .0001 | 3.169 | 1.166-8.617 | .017 |
| II (n = 87) | 1.720 | 1.044-2.834 | .031 | 0.982 | 0.563-1.712 | .949 | 1.209 | 0.693-2.110 | .504 |
| III (n = 61) | 0.936 | 0.554-1.580 | .804 | 1.171 | 0.683-2.005 | .566 | 2.011 | 1.172-3.450 | .010 |
Values are presencted as mean (range) or number (%). ALC, absolute lymphocyte count; AMC, absolute monocyte count; β2m, β2-microglobulin; Cr, creatinine; Hb, hemoglobin; Ca, calcium; WBC, white blood cell. Mann-Whitney U test; Two-sided Fisher’s exact test; Two-sided Pearson’s chi-square test.
ALC, absolute lymphocyte count; AMC, absolute monocyte count; OS, overall survival; HR, hazard ratio; CI, confidence interval; ISS, International Staging System; D-S stage, Durie-Salmon stage; ASCT, autologous stem cell transplantation.
ALC, absolute lymphocyte count; AMC, absolute monocyte count; OS, overall survival; HR, hazard ratio; CI, confidence interval; ISS, International Staging System; D-S stage, Durie-Salmon stage; ASCT, autologous stem cell transplantation.
OS, overall survival; ALC, absolute lymphocyte count; AMC, absolute monocyte count; HR, hazard ratio; CI, confidence interval; ASCT, autologous stem cell transplantation; ISS, International Staging System.

 E-submission
E-submission




