Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(6); 2013 > Article
-
Original Article
Endogenous Gastric-Resident Mesenchymal Stem Cells Contribute to Formation of Cancer Stroma and Progression of Gastric Cancer - Eun-Kyung Kim1,, Hye-Jung Kim1,, Young-Il Yang1,2, Jong Tae Kim2, Min-Young Choi2, Chang Soo Choi3, Kwang-Hee Kim3, Jeong-Han Lee2,4, Won-Hee Jang2, Soon-Ho Cheong2,4
-
Korean Journal of Pathology 2013;47(6):507-518.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.6.507
Published online: December 24, 2013
1Department of Pathology, Inje University School of Medicine, Busan, Korea.
2Paik Institute for Clinical Research, Inje University School of Medicine, Busan, Korea.
3Department of Surgery, Inje University School of Medicine, Busan, Korea.
4Department of Anesthesiology, Inje University School of Medicine, Busan, Korea.
- Corresponding Author: Young-Il Yang, M.D. Department of Pathology, Paik Institute for Clinical Research, Inje University School of Medicine, 75 Bokji-ro, Busanjin-gu, Busan 614-735, Korea. Tel: +82-51-890-6630, Fax: +82-51-894-6709, pathyang@inje.ac.kr
- *Eun-Kyung Kim and Hye-Jung Kim contributed equally to this work.
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Carcinoma-associated fibroblasts (CAFs) contribute to carcinogenesis and cancer progression, although their origin and role remain unclear. We recently identified and investigated the in situ identity and implications of gastric submucosa-resident mesenchymal stem cells (GS-MSCs) in the progression of gastric carcinogenesis.
-
Methods
- We isolated GS-MSCs from gastric submucosa using hydrogel-supported organ culture and defined their identity. Isolated cells were assessed in vitro by immunophenotype and mesengenic multipotency. Reciprocal interactions between GS-MSCs and gastric cancer cells were evaluated. To determine the role of GS-MSCs, xenografts were constructed of gastric cancer cells admixed with or without GS-MSCs.
-
Results
- Isolated cells fulfilled MSCs requirements in regard to plastic adherence, stromal cell immunophenotype, and multipotency. We demonstrated a paracrine loop that gastric cancer cells enhanced the migration, proliferation, and differentiation of GS-MSCs; additionally, GS-MSCs promoted the proliferation of gastric cancer cell in vitro. Xenograft experiments showed that GS-MSCs significantly promoted cancer growth and angiogenesis. GS-MSCs that integrated into gastric cancer became not only CAFs but also rarely endothelial cells which contributed to the formation of cellular and vascular cancer stroma.
-
Conclusions
- Endogenous GS-MSCs play an important role in gastric cancer progression.
- 3D organ culture of gastric submucosa and isolation of outgrown cells
- All gastric submucosa (GS) were obtained from brain-dead patients (n=12; 35 to 56 years; mean, 51 years). The procedure and guidelines were approved by the ethical committee at Busan Paik Hospital, Inje University School of Medicine. The GS was minced into 2 to 3 mm3 fragments, embedded in fibrin hydrogel, and cultured as described previously.5 After 14 days of organ culture, outgrown cells were selectively recovered from the hydrogel and propagated in a monolayer condition. When the cells reached confluence, they were detached and seeded at a density of 2×103 cells/cm2. This initial passage was referred to as passage 1. A limited dilution assay was performed to determine the clonogenic potential of recovered cells. The colony was counted using Image J (NIH, Bethesda, MD, USA) after 11 days of culture. All cell-based assays used subcultured outgrown cells at passage 3.
- Localization and phenotype of in vitro renewed and outgrown cells
- Paraffin sections including cultured GS fragments and outgrown cells were used to determine the in situ localization and phenotype of in vitrorenewed and outgrown cells during organ culture. Immunophenotype was determined with immunohistochemical staining using an EnVision detection system (Dako, Glosrup, Denmark) and diaminobenzidine (Sigma, St. Louis, MO, USA).13 In situ localization of the renewed cells was evaluated using immunofluorescent staining and markers for endothelial cells (EC; CD31 and CD34), pericytes (CD140b and smooth muscle actin [SMA]), and MSCs (CD29 and CD105).14 The signals were visualized with fluorochrome-conjugated secondary antibodies. All antibodies used in this experiment are listed in Table 1.
- Flow cytometry analysis
- The immunophenotype of the subcultured cells was analyzed with a FACSCalibur (Becton Dickinson, San Jose, CA, USA). After the cells were incubated with primary or isotype-matched antibodies, the cells were washed with phosphate buffered saline and fixed. For unconjugated antibodies, the signal was detected after incubation with fluorochrome-conjugated secondary antibodies. Isotype-matched immunoglobulin was used as a negative control. A minimum of 10,000 events were acquired for each analysis.
- In vitro mesengenic differentiation
- Subcultured cells were induced into adipogenic and osteogenic cells as described previously.5 Adipogenic or osteogenic differentiation was determined with Oil Red O (Sigma) or Alizarin Red (Sigma) stainings, respectively. To determine the EC differentiation potential, subcultured cells were seeded on fibrin hydrogel and induced with 20 ng/mL vascular endothelial growth factor (VEGF; R&D Systems, Minneapolis, MN, USA) or conditioned media. Endothelial cell differentiation was determined by capillary-like network formation and expression of EC-specific markers. The tube length and the branching points of the capillary-like structure were measured with the photoplanimetric method using Image J (NIH). The expression of EC-specific proteins and mRNAs was determined by immunofluorescent staining and by quantitative real-time reverse transcriptase polymerase chain reaction, respectively. The primers used in this experiment are listed in Table 2.
- Conditioned media
- All gastric cancer cell lines including AGS, NCI-N87, and SNU-1 were purchased from Korean Cell Line Bank (Seoul, Korea). When the cancer cells or GS-MSCs reached 80% confluence, the cells were washed three times with Dulbecco's modified Eagle media (DMEM; Invitrogen, Carlsbad, CA, USA) and incubated with vehicle (99% DMEM and 1% calf serum) for 12 hours. Conditioned media comprised of adipose-derived stem cells (ADSCs) and HEK293 were used as positive or negative controls, respectively.
- In vitro scratch assay
- A confluent monolayer of GS-MSCs on 60-mm dishes was scraped with a p200 pipet tip to create a straight scratch line. The debris was removed by washing with vehicle media, conditioned media were added, and then the cells were cultured for 24 hours. The photoplanimetric method was used to measure the migration distance with Image J. The mitogenic effect of gastric cancer cells on GS-MSCs was determined by measuring the DNA content using PicoGreen dsDNA quantitation kit (Invitrogen) after the cells were incubated for three days with conditioned gastric cancer cell line media.
- In vitro tumor sphere formation assay
- NCI-N87 cells (1×104) were suspended in vehicle, 10% or 100% conditioned media of GS-MSCs, transferred into each well of an ultra-low attachment 12-well plate (Coring, Lowell, MA, USA), and cultured for 12 days. The sphere ratio was calculated as the percentage of the sphere with more than 100 µm of diameter to the total sphere number.
- Xenograft transplantation
- The xenograft animal experiment was approved by the Institutional Animal Review Committee of Inje University School of Medicine. Athymic nude mice (Balb/c nu/nu, 6 weeks old, female, n=20) were used for the procedure, and NCI-N87 cells (2×106) admixed with or without GS-MSCs (2×106) or 5×106 NCI-N87 cells were injected subcutaneously. Tumorigenesis was assessed daily with macroscopic observation. Three weeks after the injection, the formed tumor was dissected, and its weight was measured. To evaluate the general features and CD34+ microvascular density, hematoxylin and eosin staining and immunohistochemistry were performed. Double immunostaining against a human-specific mitochondrial antigen (hMt-Ag) and ECs and CAFs markers was performed to determine the role of the transplanted GS-MSCs. The signals were visualized with fluorochrome-conjugated secondary antibodies. Isotype-matched immunoglobulin was used as a negative control.
- Statistical analysis
- One-way analysis of variance (ANOVA) was performed using the SPSS ver. 10.0.7 (SPSS Inc., Chicago, IL, USA). All data are expressed as the mean±standard deviation (SD) of three independent experiments. A p<.05 was considered statistically significant.
MATERIALS AND METHODS
- In situ renewed and outgrown cells showed MSC-like phenotypes
- The sol-gel phase transitional hydrogel provided physical support for GS fragments and a large surface area for outgrowth of the in vitro renewed cells residing in the GS. Outgrown cells were recognized on day 1 of culture; thereafter, these cells proliferated extensively and distributed in the fibrin hydrogel and within the GS (Fig. 1A). Most of the outgrown cells that had a spindle-shape appearance expressed or incorporated proliferating cell nuclear antigen or bromodeoxyuridine, which indicated that these cells were derived from in vitro cell division (Fig. 1B). After 14 days of culture, outgrown cells were successfully recovered from the hydrogel after urokinase treatment. Recovered cells were rapidly attached and spread on the plastic dish within 30 minutes. The limited dilution assay showed that more than 15% of the recovered cells that formed colonies were composed of homogeneous fibroblastoid cells (Fig. 1C). Outgrown cells uniformly expressed a set of MSC markers including CD29, CD44, and CD105 (Fig. 2A). Moreover, these cells expressed pericyte markers including CD140b, CD146, and α-SMA, but no EC markers (CD31 and CD34) or hematopoietic cell markers (CD34 and CD45, data not shown) were observed, indicating that outgrown cells showed MSC-and pericyte-like phenotypes. Next, we determined where the cells resided in the GS. The cell density within the GS increased around the perivascular area of microvessels that did not have a mural layer after 14 days of organ culture (Fig. 2B). Immunostaining demonstrated that the in vitro renewed cells increased around CD31+ and CD34+ microvessels. These cells did not express EC markers but consistently expressed MSC and pericyte markers. Our results suggest that perivascular MSC-like cells were predominantly renewed during the in vitro culture and outgrew into the hydrogel, indicating that the origin of the outgrown cells was a specific population of microvascular pericytes residing in the GS.
- Subcultured outgrown cells exhibit typical MSC properties
- Regardless of tissue origin, MSCs are defined by stromal cell immunophenotype and multipotency which are categorized into mesodermal lineages. This study determined whether in vitro subcultured outgrown cells fulfilled these requirements. The outgrown cells rapidly adhered and showed typical fibroblastic features once seeded on cultured plastic. Subcultured cells did not show any alterations in the initial immunophenotype observed for the in situ renewed and outgrown cells. Additionally, more than 90% of the cells consistently expressed MSC markers, such as CD29, CD44, CD73, CD90, and CD105, and expressed pericyte markers at variable frequencies, indicating the MSC-and pericyte-like immunophenotypes were maintained after in vitro expansion (Table 3).
- Subcultured cells showed cytoplasmic accumulation of fat droplets and mineral crystal deposition in response to the defined culture conditions, which indicated that the cells retained adipogenic and osteogenic differentiation potential (Fig. 2C). MSCs have been considered as tissue-resident endothelial progenitor cells due to their ability to differentiate toward ECs.15 The subcultured outgrown cells induced by VEGF formed a capillary-like structure within 12 hours (Fig. 2D), which indicated angiogenic potential. Next, the capillary-like structure disassembled, and the outgrown cells aggregated and transformed into a sphere-like structure at three days. Interestingly, when the spheres formed, the outgrown cells expressed EC markers (Flk-1, CD31, and CD34), indicating EC differentiation potential. Collectively, our results indicated that the outgrown cells from the GS fulfilled the minimum requirement of MSCs in terms of plastic adherence, stromal cell immunophenotype, and multipotency.
- Gastric cancer cells enhance GS-MSC migration, proliferation, and endothelial differentiation
- Evidence indicates that the reciprocal interaction between cancer cells and BM-MSCs is important in carcinogenesis.16 However, the reciprocal interactions between gastric-resident MSCs and gastric cancer cells have not been elucidated. The in vitro migration assay revealed that conditioned gastric cancer cell line media significantly increased GS-MSC migration (p<.01) (Fig. 3A) and the proliferation of GS-MSCs (p<.05) (Fig. 3B) compared to vehicle.
- Since angiogenesis is critical for tumorigenesis, cancer cells secrete proangiogenic factors that directly or indirectly contribute to neovascularization. Therefore, we determined the effects of gastric cancer cells on GS-MSCs EC differentiation. All conditioned gastric cancer cell line media and ADSCs enabled GS-MSCs to form a capillary-like structure (data not shown). However, HEK293 vehicle or conditioned media did not form capillary-like structures. In addition, the tube length and the number of branch points of the capillary-like network were significantly higher in cells induced by conditioned gastric cancer cell line media than in conditioned HEK293 media (p<.01) (Fig. 3C).
- Next, we investigated the EC-specific mRNA expression levels in induced GS-MSCs with conditioned gastric cancer cell line media. Interestingly, GS-MSCs induced by conditioned gastric cancer cell media showed significantly higher EC-specific mRNA expression of CD31, CD34, CD144, and von Willebrand factor compared to GS-MSCs before induction or those induced by conditioned HEK293 media (Fig. 3D). However, no meaningful difference in the EC-specific mRNA expression level was observed according to gastric cancer cell type. VEGF and basic fibroblast growth factor (bFGF) play important roles in mobilization and differentiation of endothelial progenitor cells, and immunoabsorbent analysis showed that the VEGF and bFGF were detected in all conditioned gastric cancer cell line media but not in conditioned HEK293 media. This indicates that proangiogenic factors secreted by gastric cancer cells might directly induce GS-MSCs toward ECs (Fig. 3E).
- GS-MSCs promote tumor growth and angiogenesis
- Although our data indicated chemotactic and mitotic effects of gastric cancer cells on GS-MSCs, whether GS-MSCs play specific roles in gastric cancer is unclear. The tumor sphere-forming assay indicated that GS-MSC had a mitogenic effect on the growth of gastric cancer cells (Fig. 4A). Conditioned GS-MSC medium dose-dependently enhanced NCI-N87 tumor sphere formation. Next, we investigated the role of GS-MSCs in tumorigenesis and progression in vivo. All mice injected with 5×106 NCI-N87 only showed de novo tumor formation at one week, but mice injected with 2×106 NCI-N87 had late-forming tumors at two weeks. The tumor volume was related to the number of NCI-N87 injections in a dose-dependent manner at three weeks (Fig. 4B). Interestingly, tumors in mice injected with 2×106 NCI-N87 and 2×106 GS-MSCs were detected earlier, at one week. The size and weight of the tumors were significantly higher in mice injected with both NCI-N87 and GS-MSCs compared to those of mice injected with 2×106 NCI-N87 only (p<.01). However, GS-MSCs did not induce de novo tumor formation by non-tumorigenic gastric cancer cells, such as AGS, until 12 weeks of observation (data not shown). These data suggest that GS-MSCs can accelerate cancer growth but not cancer initiation.
- De novo formed tumors exhibited typical features of poorly differentiated adenocarcinoma regardless of GS-MSC injection (Fig. 4C). Interestingly, tumors formed by NCI-N87 and GS-MSC coinjections showed decreased coagulative necrosis compared to tumors formed by injection of NCI-N87 only (Fig. 4C). The CD34+ microvascular density was significantly increased in tumors injected with NCI-N87 and GS-MSCs (2.79/mm2) compared to tumors injected with 2×106 (1.87/mm2) and 5×106 (1.95/mm2) NCI-N87, respectively (p<.05). Intriguingly, the cancer stroma formed by coinjection of NCI-N87 and GS-MSCs showed increased cellularity and vascularity compared to the cancer stroma formed by injection of NCI-N87 only (Fig. 4D).
- GS-MSCs directly integrate into tumor stroma and become SMA+ CAFs
- To investigate the role of GS-MSCs in gastric cancer, we traced the injected cells using hMt-Ag. All cancer cells homogeneously expressed hMt-Ag in their cytoplasm, but hMt-Ag tracing did not detect intervening cancer stroma formed by NCI-N87-only injections. More than 20% of the cancer stromal cells formed by NCI-N87 and GS-MSC coinjections expressed hMt-Ag.
- We performed double immunostaining to determine the role of the injected GS-MSCs. hMt-Ag+ stromal cells were widely distributed along the cancer stroma and coexpressed CD105, CD140b, and SMA (Fig. 5). The analysis indicated that approximately 20% of hMt-Ag+ stromal cells coexpressed SMA. Occasionally, hMt-Ag+ stromal cells coexpressing CD34 were observed in the intraluminal side of microvessels in the cancer stroma, but less than 1% of CD34+ ECs were observed to demonstrate this coexpression. However, the cancer stroma formed by an injection of NCI-N87 alone were not localized with any hMt-Ag+ stromal cells (Fig. 6). Taken together, these results indicate that locally delivered GS-MSCs were integrated into the cancer stroma where GS-MSCs become SMA+ CAFs but rarely microvascular ECs, which contributed to the cancer stroma and gastric cancer angiogenesis.
RESULTS
- Recent studies have indicated that various cancers can attract MSCs in their microenvironment, and that MSCs become CAFs and play a role in tumorigenesis, angiogenesis, and progression. However, most of these studies described the roles of BM-MSCs.16-18 In this study, we demonstrated the existence of endogenous gastric-resident MSCs and their role in gastric cancer.
- Evidence suggests that MSCs reside in a perivascular niche, where they are in close proximity to ECs of microvessels.5 This evidence illustrates that the degree of microvascular density is closely related to the MSC density. Thus, we speculated that the GS might retain a higher density of MSCs compared to other compartments of the stomach due to their high vascularity. The results demonstrate that in situ renewed cells were apparent in perivascular locations in the GS and consequently outgrew in the hydrogel. Both in situ and subcultured outgrown cells shared typical biologic properties with BM-MSCs. Collectively, our data support not only the perivascular MSC hypothesis, but also the existence of gastric-resident MSCs residing in the GS. Additionally, our results suggest the possibility that tissue-resident MSCs are not restricted to the stomach but might be widely distributed in other areas of the intestinal tract.
- Cancer growth is determined by crosstalk between cancer cells and stromal cells.17 Our in vitro results demonstrate a paracrine loop in which gastric cancer cells exert migration and proliferation of GS-MSCs, and, in response, GS-MSCs subsequently play an important role in gastric cancer growth and angiogenesis. Proangiogenic factors secreted by gastric cancer cells, such as VEGF and bFGF, further induced GS-MSC EC differentiation in vitro. Meanwhile, GS-MSCs also facilitated not only in vitro tumor spheroid formation, but also enhanced in vivo tumor growth. Collectively, our results indicate that a reciprocal interaction between gastric cancer cells and GS-MSCs is exerted synergistically in gastric cancer.
- CAFs are thought to develop from both local and distant niches and to provide a functional and structural supportive microenvironment for cancer. A number of studies have indicated that tissue-resident MSCs integrate into cancer sites and contribute to the formation of cancer stroma in various types of cancer.7-9 Although a few studies have elucidated the existence of MSC-like cells in the stomach, the contributions of gastric-resident MSCs in gastric cancer are less well known.9,10 Our results demonstrated that tumors with admixed GS-MSCs showed hMt-Ag positive stromal cells that coexpressed with CD105, CD140b, SMA, and CD34, indicating that GS-MSCs can give rise to a set of stromal cell populations, including CAFs and ECs. Therefore, GS-MSCs constitute an important cell source for CAFs in that they promote gastric cancer growth through the formation of fibrovascular stroma.
- Promoting angiogenesis is an important function of MSCs during wound healing as well as carcinogenesis.19,20 MSCs promote angiogenesis through the expression of SDF-1 and VEGF, which mobilize ECs and/or endothelial progenitor cells.21 Moreover, MSCs can directly give rise to angiogenic cells both in vitro and in vivo.22 Thus, a specific population of MSCs has been regarded as tissue-resident endothelial progenitor cells. Our data indicate that gastric-resident MSCs also contribute to direct and indirect promotion of tumor angiogenesis since the CD34+ microvascular density was significantly increased in tumors with admixed GS-MSCs. Although our results indicate that GS-MSCs can differentiate into ECs in vitro, the frequency of CD34+/hMt-Ag+ ECs was less than 1% among the CD34+ ECs of the microvessels, indicating that the proangiogenic ability of GS-MSCs is most likely induced by an indirect mechanism.
- In summary, we showed that gastric-resident MSCs exist in the GS. These cells reciprocally interact with cancer cells to promote the growth and progression of gastric cancer. Similar to BM-MSCs, gastric-resident MSCs actively integrated, became SMA+ CAFs and CD34+ ECs, and contributed to the formation of the fibrovascular cancer stroma. These observations have implications for gastric cancer progression.
DISCUSSION
Acknowledgments
Acknowledgments
- 1. Mueller MM, Fusenig NE. Friends or foes: bipolar effects of the tumour stroma in cancer. Nat Rev Cancer 2004; 4: 839-849. ArticlePubMedPDF
- 2. Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell 2011; 144: 646-674. ArticlePubMed
- 3. Direkze NC, Hodivala-Dilke K, Jeffery R, et al. Bone marrow contribution to tumor-associated myofibroblasts and fibroblasts. Cancer Res 2004; 64: 8492-8495. ArticlePubMedPDF
- 4. Karnoub AE, Dash AB, Vo AP, et al. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature 2007; 449: 557-563. ArticlePubMedPDF
- 5. Crisan M, Yap S, Casteilla L, et al. A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell 2008; 3: 301-313. ArticlePubMed
- 6. da Silva Meirelles L, Caplan AI, Nardi NB. In search of the in vivo identity of mesenchymal stem cells. Stem Cells 2008; 26: 2287-2299. ArticlePubMedPDF
- 7. Muehlberg FL, Song YH, Krohn A, et al. Tissue-resident stem cells promote breast cancer growth and metastasis. Carcinogenesis 2009; 30: 589-597. ArticlePubMed
- 8. Lin G, Yang R, Banie L, et al. Effects of transplantation of adipose tissue-derived stem cells on prostate tumor. Prostate 2010; 70: 1066-1073. ArticlePubMedPMCPDF
- 9. Nomoto-Kojima N, Aoki S, Uchihashi K, et al. Interaction between adipose tissue stromal cells and gastric cancer cells in vitro. Cell Tissue Res 2011; 344: 287-298. ArticlePubMedPDF
- 10. Xu X, Zhang X, Wang S, et al. Isolation and comparison of mesenchymal stem-like cells from human gastric cancer and adjacent non-cancerous tissues. J Cancer Res Clin Oncol 2011; 137: 495-504. ArticlePubMedPDF
- 11. Semba S, Kodama Y, Ohnuma K, et al. Direct cancer-stromal interaction increases fibroblast proliferation and enhances invasive properties of scirrhous-type gastric carcinoma cells. Br J Cancer 2009; 101: 1365-1373. ArticlePubMedPMCPDF
- 12. Choi MY, Kim HI, Yang YI, et al. The isolation and in situ identification of MSCs residing in loose connective tissues using a niche-preserving organ culture system. Biomaterials 2012; 33: 4469-4479. ArticlePubMed
- 13. Jang TJ, Kim SW, Lee KS. The expression of pigment epithelium-derived factor in bladder transitional cell carcinoma. Korean J Pathol 2012; 46: 261-265. ArticlePubMedPMC
- 14. Jung YH, Kim J, Kim BM, et al. Neoplastic stromal cells of intracranial hemangioblastomas disclose pericyte-derived mesenchymal stromal cells-like phenotype. Korean J Pathol 2011; 45: 564-572. Article
- 15. Urbich C, Dimmeler S. Endothelial progenitor cells: characterization and role in vascular biology. Circ Res 2004; 95: 343-353. PubMed
- 16. Cuiffo BG, Karnoub AE. Mesenchymal stem cells in tumor development: emerging roles and concepts. Cell Adh Migr 2012; 6: 220-230. PubMedPMC
- 17. Keung EZ, Nelson PJ, Conrad C. Concise review: genetically engineered stem cell therapy targeting angiogenesis and tumor stroma in gastrointestinal malignancy. Stem Cells 2013; 31: 227-235. ArticlePubMedPDF
- 18. Quante M, Tu SP, Tomita H, et al. Bone marrow-derived myofibroblasts contribute to the mesenchymal stem cell niche and promote tumor growth. Cancer Cell 2011; 19: 257-272. ArticlePubMedPMC
- 19. Chamberlain G, Fox J, Ashton B, Middleton J. Concise review: mesenchymal stem cells: their phenotype, differentiation capacity, immunological features, and potential for homing. Stem Cells 2007; 25: 2739-2749. ArticlePubMedPDF
- 20. Suzuki K, Sun R, Origuchi M, et al. Mesenchymal stromal cells promote tumor growth through the enhancement of neovascularization. Mol Med 2011; 17: 579-587. ArticlePubMedPMCPDF
- 21. Foronjy RF, Majka SM. The potential for resident lung mesenchymal stem cells to promote functional tissue regeneration: understanding microenvironmental cues. Cells 2012; 1: 874.ArticlePubMedPMC
- 22. Torsney E, Xu Q. Resident vascular progenitor cells. J Mol Cell Cardiol 2011; 50: 304-311. ArticlePubMed
REFERENCES
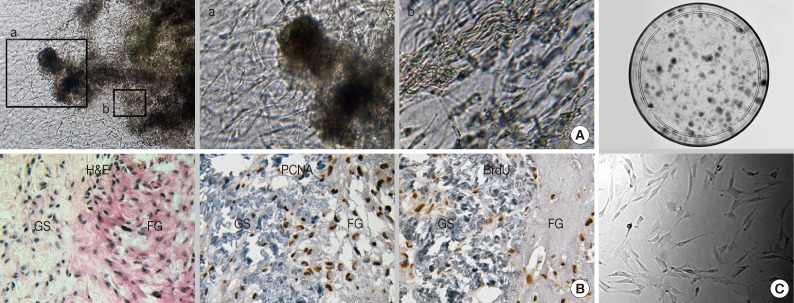
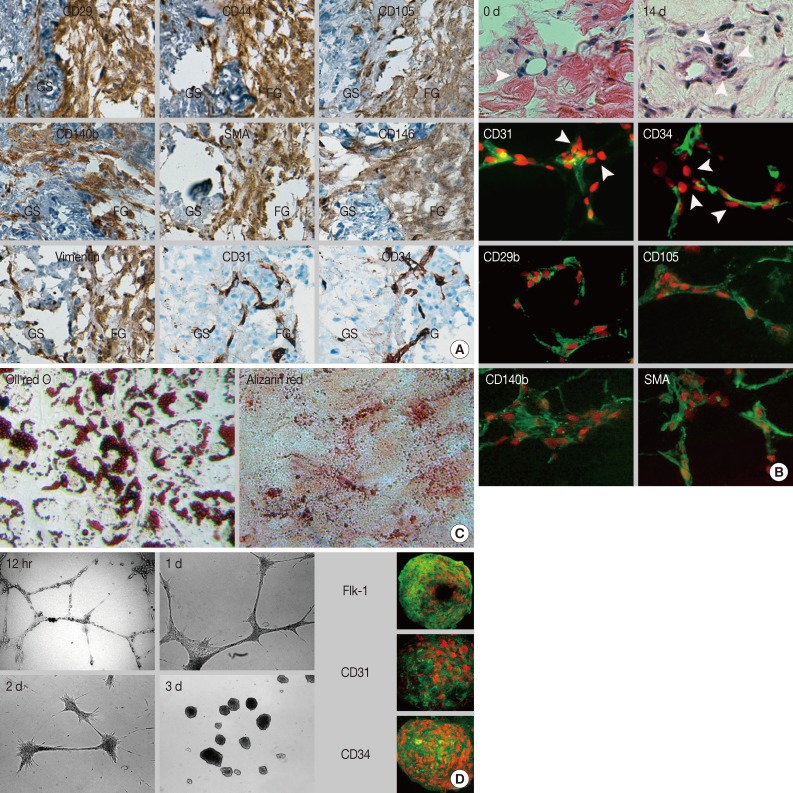
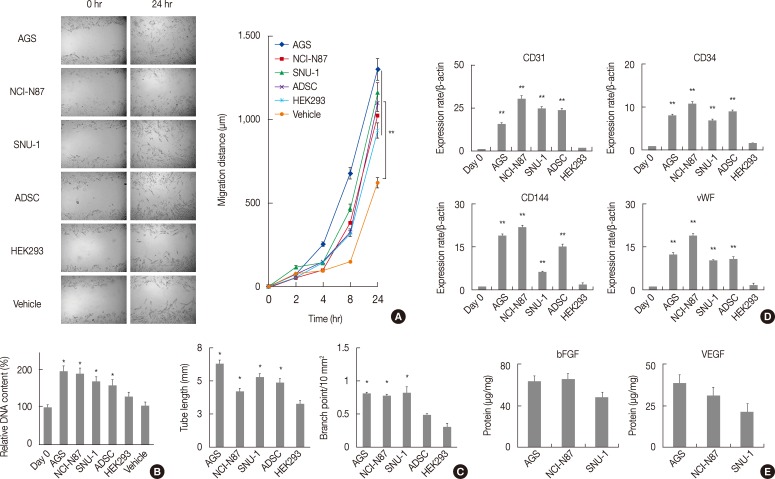
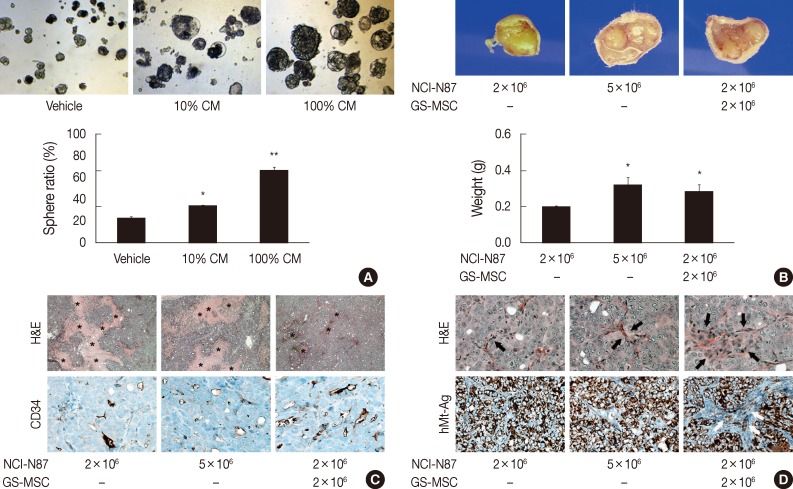
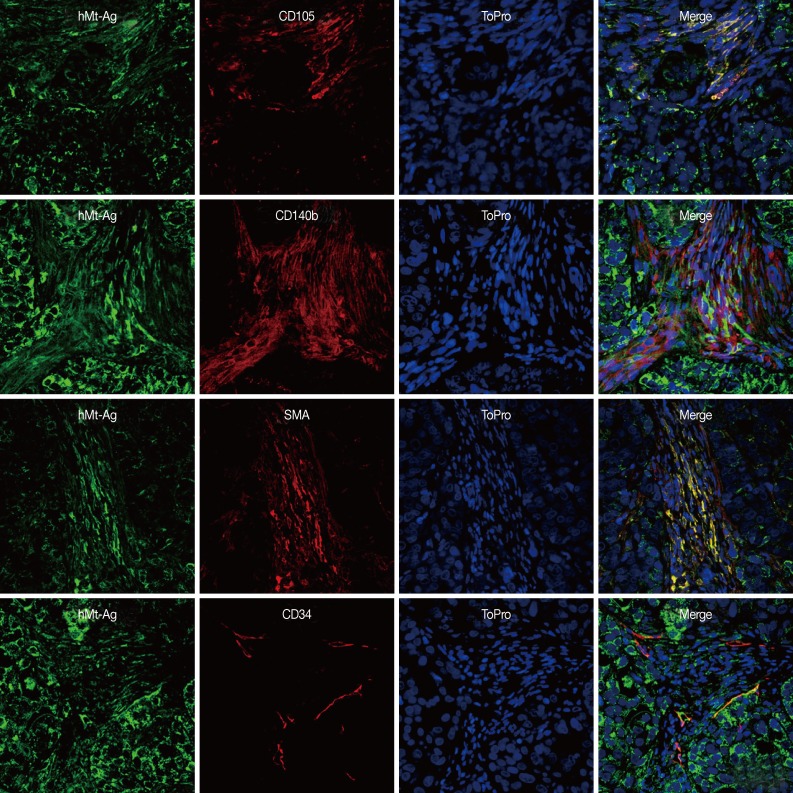
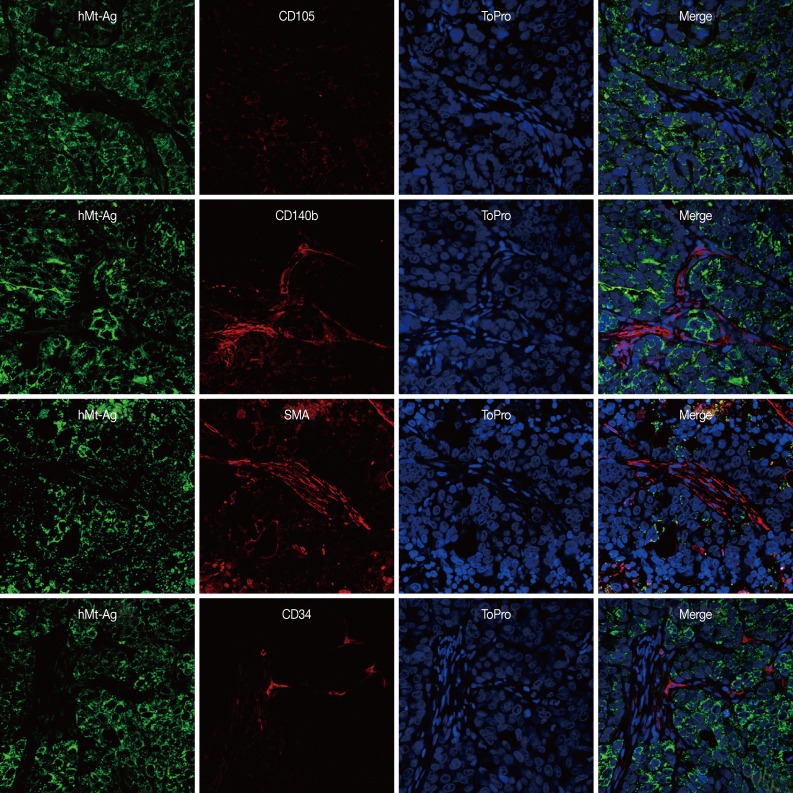
Figure & Data
References
Citations

- Tumor microenvironment dynamics in gastric cancer pathogenesis and therapeutic resistance
Zhenhua Lu, Qinnan Zhang, Jing Han, Jiafu Ji, Xiaofang Xing
Molecular Cancer.2026;[Epub] CrossRef - The therapeutic potential of mesenchymal stem cells in intestinal diseases: from mechanisms to clinical translation
Jia-Zhi Yang, Li-Yan He, Xian-Zhou Lu
Stem Cell Research & Therapy.2025;[Epub] CrossRef - Gastric cancer and mesenchymal stem cell-derived exosomes: from pro-tumorigenic effects to anti-cancer vehicles
Maryam Dolatshahi, Ahmad Reza Bahrami, Qaiser Iftikhar Sheikh, Mohsen Ghanbari, Maryam M. Matin
Archives of Pharmacal Research.2024; 47(1): 1. CrossRef - Extracellular vesicles derived from SARS-CoV-2 M-protein-induced triple negative breast cancer cells promoted the ability of tissue stem cells supporting cancer progression
Hoai-Nga Thi Nguyen, Cat-Khanh Vuong, Mizuho Fukushige, Momoko Usuda, Liora Kaho Takagi, Toshiharu Yamashita, Mana Obata-Yasuoka, Hiromi Hamada, Motoo Osaka, Toru Tsukada, Yuji Hiramatsu, Osamu Ohneda
Frontiers in Oncology.2024;[Epub] CrossRef - Revisiting the role of mesenchymal stromal cells in cancer initiation, metastasis and immunosuppression
Yanyan Zhang, Charles Wang, Jian Jian Li
Experimental Hematology & Oncology.2024;[Epub] CrossRef - Mesenchymal stem cells and their potential therapeutic benefits and challenges in the treatment and pathogenesis of gastric cancer
Asma'a H. Mohamed, Alaa Shafie, Waleed Al Abdulmonem, Hassan Swed Alzahrani, Amal Adnan Ashour, Ahmed Hjazi, Azfar Jamal, Allolo D. Aldreiwish, Mohammad Azhar Kamal, Fuzail Ahmad, Nazia Khan
Pathology - Research and Practice.2024; 260: 155422. CrossRef - Cancer-associated fibroblasts promote proliferation, angiogenesis, metastasis and immunosuppression in gastric cancer
Peiyuan Li, Huan Zhang, Tao Chen, Yajing Zhou, Jiaoyang Yang, Jin Zhou
Matrix Biology.2024; 132: 59. CrossRef - Mesenchymal Stem Cell-Derived Exosomes Modulate Angiogenesis in Gastric Cancer
Fawzy Akad, Veronica Mocanu, Sorin Nicolae Peiu, Viorel Scripcariu, Bogdan Filip, Daniel Timofte, Florin Zugun-Eloae, Magdalena Cuciureanu, Monica Hancianu, Teodor Oboroceanu, Laura Condur, Radu Florin Popa
Biomedicines.2023; 11(4): 1031. CrossRef - LRRC15 expression indicates high level of stemness regulated by TWIST1 in mesenchymal stem cells
Kensuke Toriumi, Yuta Onodera, Toshiyuki Takehara, Tatsufumi Mori, Joe Hasei, Kanae Shigi, Natsumi Iwawaki, Toshifumi Ozaki, Masao Akagi, Mahito Nakanishi, Takeshi Teramura
iScience.2023; 26(7): 106946. CrossRef - Rat Adipose-Derived Stromal Cells (ADSCs) Increases the Glioblastoma Growth and Decreases the Animal Survival
Isabele Cristiana Iser, Liziane Raquel Beckenkamp, Juliana Hofstatter Azambuja, Francine Luciano Rahmeier, Paula Andreghetto Bracco, Ana Paula Santin Bertoni, Rita de Cássia Sant’Anna Alves, Elizandra Braganhol, Léder Leal Xavier, Marilda da Cruz Fernande
Stem Cell Reviews and Reports.2022; 18(4): 1495. CrossRef - Mesenchymal/stromal stem cells: necessary factors in tumour progression
Xinyu Li, Qing Fan, Xueqiang Peng, Shuo Yang, Shibo Wei, Jingang Liu, Liang Yang, Hangyu Li
Cell Death Discovery.2022;[Epub] CrossRef - Mesenchymal stromal cells in cancer: a review of their immunomodulatory functions and dual effects on tumor progression
Sabine Galland, Ivan Stamenkovic
The Journal of Pathology.2020; 250(5): 555. CrossRef - A highly efficient non-viral process for programming mesenchymal stem cells for gene directed enzyme prodrug cancer therapy
Yoon Khei Ho, Jun Yung Woo, Geraldine Xue En Tu, Lih-Wen Deng, Heng-Phon Too
Scientific Reports.2020;[Epub] CrossRef - Fruit-Derived Polysaccharides and Terpenoids: Recent Update on the Gastroprotective Effects and Mechanisms
Mohammed Safwan Ali Khan, Syeda Umme Kulsoom Khundmiri, Syeda Rukhaiya Khundmiri, Mohammad M. Al-Sanea, Pooi Ling Mok
Frontiers in Pharmacology.2018;[Epub] CrossRef - Gastric cancer mesenchymal stem cells derived IL-8 induces PD-L1 expression in gastric cancer cells via STAT3/mTOR-c-Myc signal axis
Li Sun, Qianqian Wang, Bin Chen, Yuanyuan Zhao, Bo Shen, Hua Wang, Jing Xu, Miaolin Zhu, Xiangdong Zhao, Changgen Xu, Zhihong Chen, Mei Wang, Wenrong Xu, Wei Zhu
Cell Death & Disease.2018;[Epub] CrossRef - Mesenchymal stem cells: key players in cancer progression
Sarah M. Ridge, Francis J. Sullivan, Sharon A. Glynn
Molecular Cancer.2017;[Epub] CrossRef - Gastric cancer-derived mesenchymal stem cells prompt gastric cancer progression through secretion of interleukin-8
Wei Li, Ying Zhou, Jin Yang, Xu Zhang, Huanhuan Zhang, Ting Zhang, Shaolin Zhao, Ping Zheng, Juan Huo, Huiyi Wu
Journal of Experimental & Clinical Cancer Research.2015;[Epub] CrossRef - Mesenchymal stem cells – A new hope for radiotherapy-induced tissue damage?
Nils H. Nicolay, Ramon Lopez Perez, Juergen Debus, Peter E. Huber
Cancer Letters.2015; 366(2): 133. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
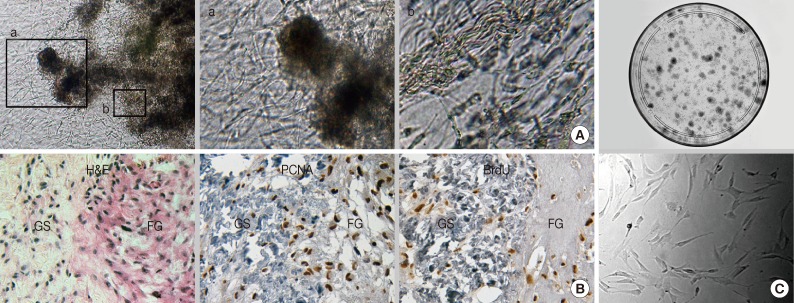
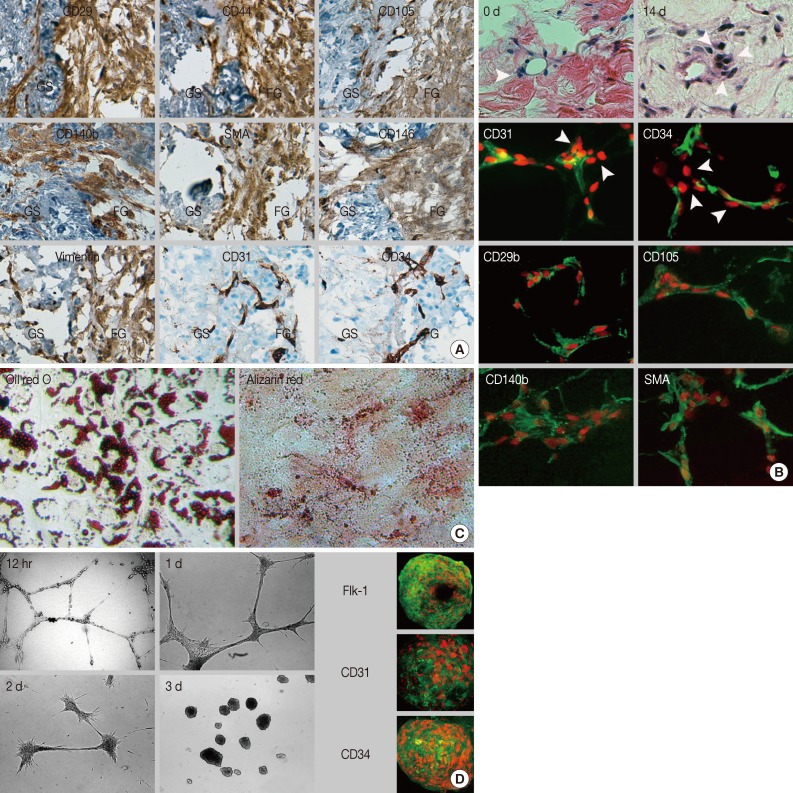
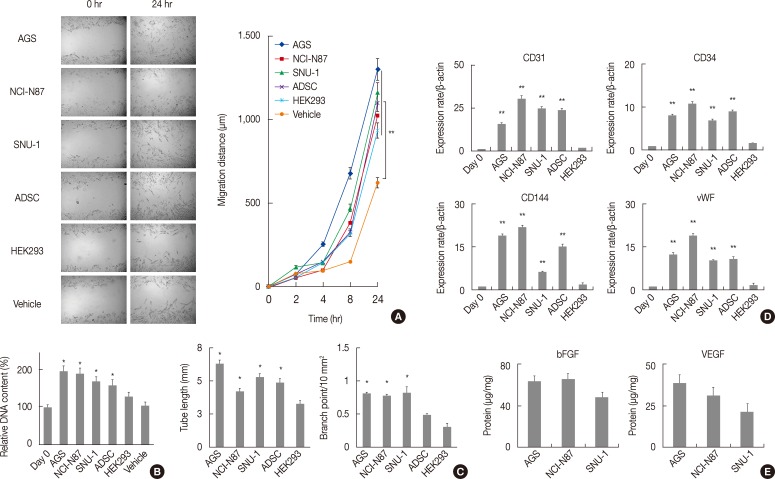
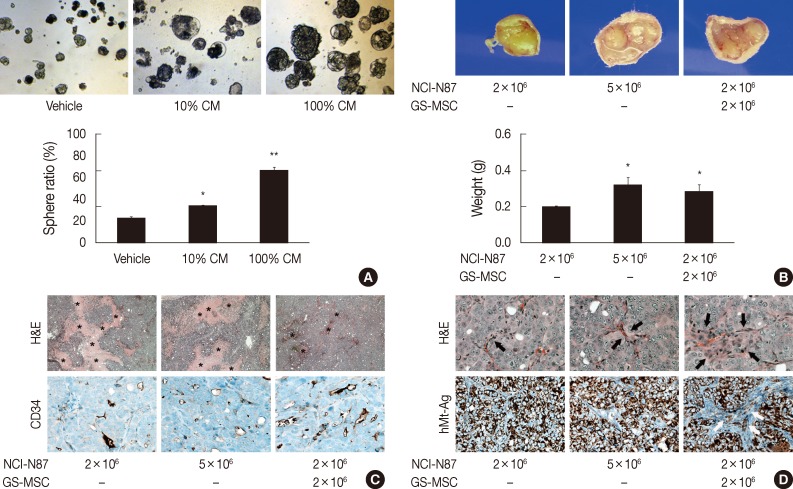
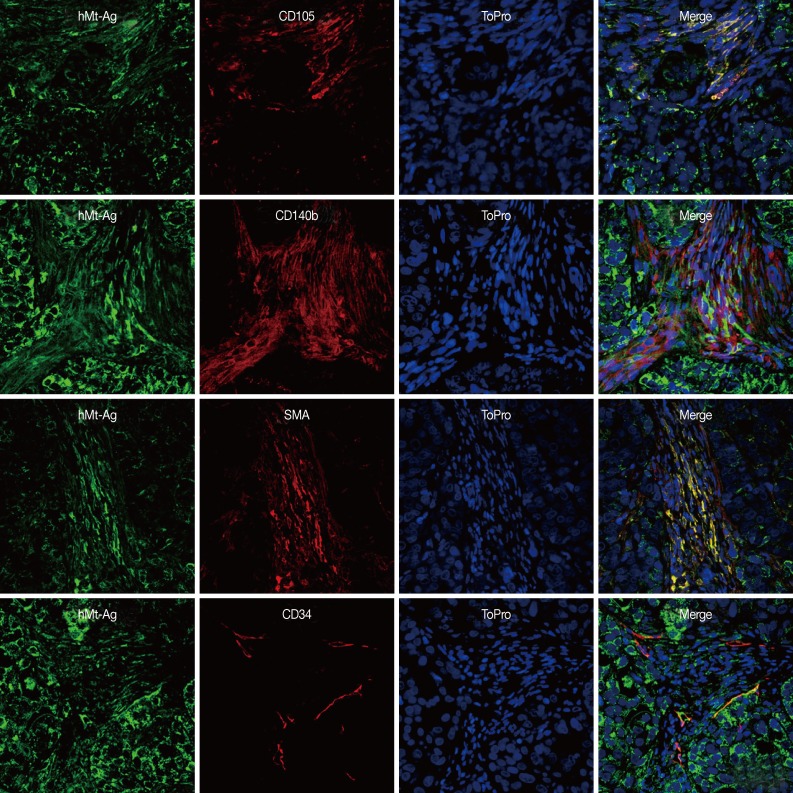
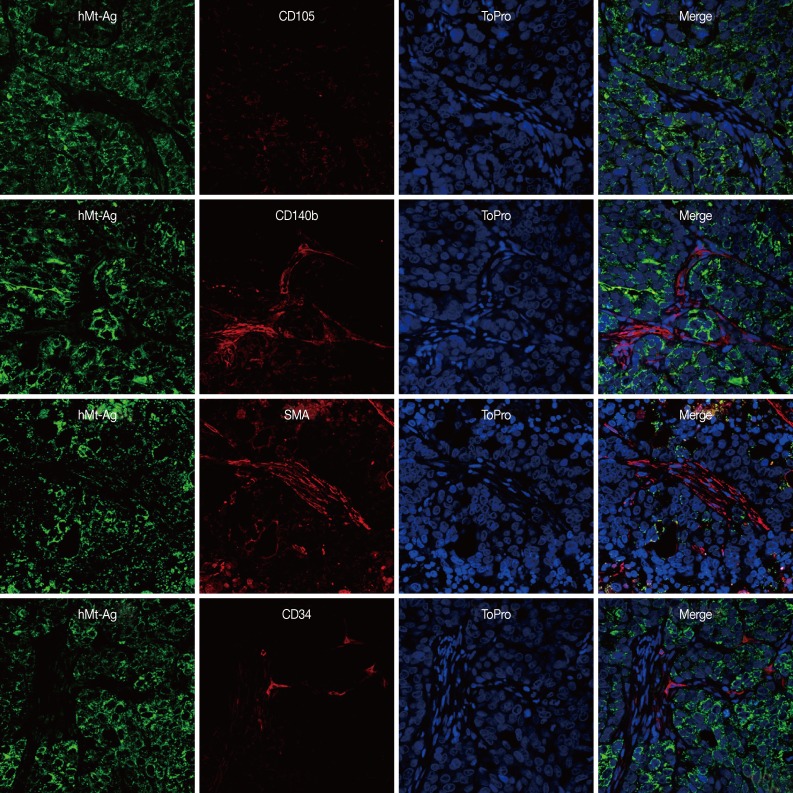
Fig. 1
Fig. 2
Fig. 3
Fig. 4
Fig. 5
Fig. 6
| Antibody | Application | Dilution | Company |
|---|---|---|---|
| CD29 | IHC, IF | 1:200 | Novus Biologicals |
| CD31 | IHC, IF | 1:40 | Dako |
| CD34 | IHC, IF | 1:800 | Neomarker |
| CD44 | IHC | 1:40 | Leica |
| CD105 | IHC, IF | 1:500 | BD Bioscicence |
| CD140b | IHC, IF | 1:400 | Abcam |
| CD146 | IHC | 1:200 | Abcam |
| SMA | IHC, IF | 1:400 | Dako |
| Vimentin | IHC | 1:200 | Zymed |
| Human-specific mitochondrial antigen | IHC, IF | 1:200 | Millipore |
| Delight 488-goat anti-mouse IgG | IF, FCM | 1:800 | Jackson Immunoresearch |
| Delight 488-goat anti-rabbit IgG | IF, FCM | 1:800 | Jackson Immunoresearch |
| Delight 594-goat anti-mouse IgG | IF, FCM | 1:800 | Jackson Immunoresearch |
| Delight 591-goat anti-rabbit IgG | IF, FCM | 1:800 | Jackson Immunoresearch |
| HRP-conjugated anti-rabbit/mouse IgG | IHC | Dako | |
| CD14 (PE conjugated) | FCM | 1:100 | BD Bioscience |
| CD29 (PE conjugated) | FCM | 1:100 | BD Bioscience |
| CD31 (PE conjugated) | FCM | 1:100 | BD Bioscience |
| CD34 (FITC conjugated) | FCM | 1:100 | BD Bioscience |
| CD44 (PE conjugated) | FCM | 1:100 | BD Bioscience |
| CD45 (PE conjugated) | FCM | 1:100 | BD Bioscience |
| CD73 (PE conjugated) | FCM | 1:100 | BD Bioscience |
| CD90 (PE conjugated) | FCM | 1:100 | BD Bioscience |
| CD105 (unconjugated) | FCM | 1:100 | R&D Systems |
| CD133 (PE conjugated) | FCM | 1:100 | Mitenyl Biotech |
| CD140b (unconjugated) | FCM | 1:100 | Abcam |
| CD146 (unconjugated) | FCM | 1:100 | Abcam |
| c-Kit (unconjugated) | FCM | 1:100 | eBioscience |
| Flk-1 (unconjugated) | FCM | 1:100 | R&D Systems |
| SMA (unconjugated) | FCM | 1:100 | Dako |
| MHC-1 (PE conjugated) | FCM | 1:100 | BD Bioscience |
| MHC-II (PE conjugated) | FCM | 1:100 | BD Bioscience |
| Gene | Primer sequence (5’-3’) |
|---|---|
| β-Actin | F: AGCAAGCAGGAGTATGACGA |
| R: TGTGAACTTTGGGGGATG | |
| CD31 | F: GTGGTGGAGTCTGGAGAGGA |
| R: TCCGATGATAACCACTGCAA | |
| CD34 | F: AAGTGAAATTGACTCAGGGC |
| R: CCAGTGCAATCAGGGTCTTT | |
| CD144 | F: GGATGACCAAGTACAGC |
| R: ACACACTTTGGGCTGGTAGG | |
| VWF | F: TGGATGAGCTTTTGCAGACC |
| R: GTGGGAGCCGTCGTGGTACT |
| Antibody | Expression rate (%) |
|---|---|
| CD29 | 97.63 ± 5.21 |
| CD44 | 97.30 ± 3.25 |
| CD73 | 94.33 ± 3.47 |
| CD90 | 95.83 ± 4.87 |
| CD105 | 93.93 ± 5.28 |
| HLA-ABC | 95.57 ± 3.14 |
| HLA-DR | 0.84 ± 0.02 |
| CD14 | 0.54 ± 0.01 |
| CD34 | 0.87 ± 0.05 |
| CD45 | 0.71 ± 0.04 |
| CD133 | 0.21 ± 0.01 |
| SMA | 81.13 ± 9.78 |
| CD140b | 94.90 ± 6.57 |
| STRO-1 | 47.90 ± 7.98 |
| c-Kit | 0.01 ± 0.01 |
| Flk-1 | 0.01 ± 0.01 |
IHC, immunohistochemical staining; IF, immunofluorescent staining; SMA, smooth muscle actin; FCM, flow cytometry; HRP, horseradish peroxidase; PE, phycoerythrin; FITC, fluorescein isothiocyanate; MHC-I, major histocompatibility complex class I; MHC-II, major histocompatibility complex class II.
F, forward primer; R, reverse primer; VWF, von Willebrand factor.
SMA, smooth muscle actin.

 E-submission
E-submission








