Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(4); 2013 > Article
-
Case Study
Spindle Cell Rhabdomyosacoma of Uterus: A Case Study - Dae Woon Kim, Jung Hwan Shin, Ho Jung Lee1, Young Ok Hong1, Jong Eun Joo1, Eun Kyung Kim1
-
Korean Journal of Pathology 2013;47(4):388-391.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.4.388
Published online: August 26, 2013
Department of Obstetrics and Gynecology, Eulji University School of Medicine, Seoul, Korea.
1Department of Pathology, Eulji University School of Medicine, Seoul, Korea.
- Corresponding Author: Eun Kyung Kim, M.D. Department of Pathology, Eulji General Hospital, 68 Hangeulbiseong-ro, Nowon-gu, Seoul 139-711, Korea. Tel: +82-2-970-8322, Fax: +82-2-970-8002, kek7402@eulji.ac.kr
• Received: October 2, 2012 • Revised: November 1, 2012 • Accepted: November 2, 2012
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- Uterine rhabdomyosarcoma (RMS) typically presents as a mixed epithelial and mesenchymal tumors. Pure RMSs of the female genital tract are uncommon. Spindle cell variant of RMS is a rare morphologic subtype of embryonal RMS and mostly occurs in the paratesticular region of children. Here, we present a case of uterine spindle cell RMS in a 76-year-old woman. The tumor, 20×15×7 cm in size, was highly necrotic and adherent to the colon and rectum. Tumor cells were mostly spindle-shaped, and isolated rhabdomyoblasts were scattered. Immunohistochemical stains for myoglobin and myo-D1 showed diffuse positivity for tumor cells. The patient died only of disease three months after diagnosis.
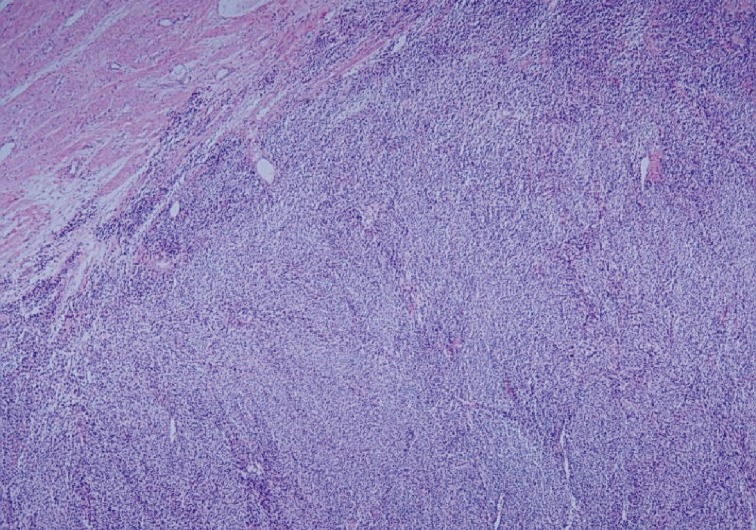
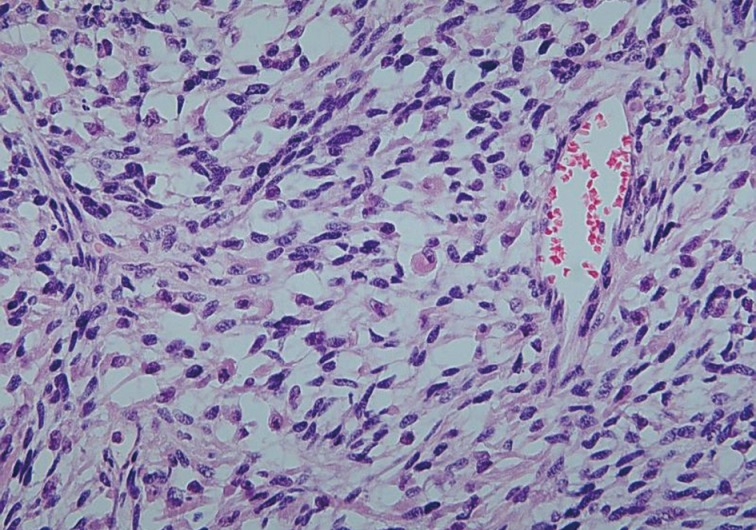
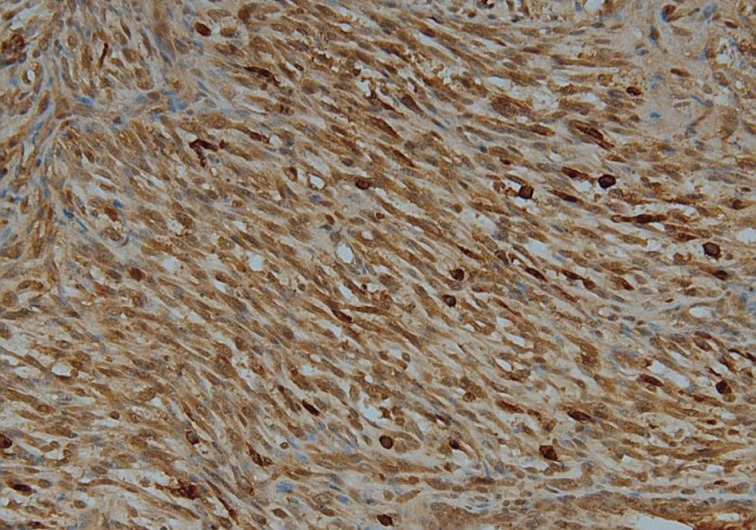
- A 76-year-old woman, gravida 3, para 2, presented with intermittent lower abdominal pain of increasing severity. The patient reported a 10 kg weight loss with associated anorexia and constipation for approximately six months. Physical examination revealed abdominal tenderness and a fixed lower abdominal mass at roughly six months in gestational size. Abdominal and pelvic ultrasonography showed a multilobulated solid and cystic mass in the pelvic cavity. A computerized tomography scan of the abdomen and pelvis demonstrated a 16×10×8 cm lobulating-contoured and heterogeneously-enhanced mass occupying the lower abdomen and pelvic cavity. Neither the uterus nor both ovaries were identified (Fig. 1). Serum levels of tumor markers, with normal values in parentheses, were as follows: cancer antigen 125, 101.2 U/mL (<35 U/mL); cancer antigen 19-9, 4.8 U/mL (<30 U/mL). An exploratory laparotomy was performed given the high-suspicion for an ovarian tumor. Intraoperatively, a yellowish, hard, friable, 20×15×7 cm mass had replaced the right lateral uterine wall and had adhered to the colon, rectum, and appendix. The right ovary was not recognized, but the left ovary and both fallopian tubes appeared normal. Tumor debulking and subtotal hysterectomy with bilateral salpingo-oophorectomy were performed. The tumor grossly occupied the entire right myometrium and was bulging into the endometrial cavity. The cut surface of the tumor had a variegated appearance, including components of greyish soft, whitish fish-flesh, and yellowish myxoid with small cystic changes (Fig. 2). Wide areas of hemorrhagic necrosis were also noted. Microscopic findings exhibited highly cellular and necrotic tumor consisting of bundles or fascicles of spindle cells in a myxoid stroma (Fig. 3). Rare rhabdomyoblasts containing eccentric nuclei and deeply eosinophilic cytoplasm were observed (Fig. 4). Pleomorphic tumor cells were mixed with spindle cells in small areas. Mitotic figures were frequently counted with more than 100 mitotic figures per 10 high power fields. Right ovarian tissue was identified in the fibrous tumor capsule without direct invasion of tumor cells. Immunohistochemical staining showed diffuse positivity of tumor cells with myoglobin (Fig. 5), myo-D1, desmin, and vimentin. No stains for smooth muscle actin, pan-cytokeratin, epithelial membrane antigen, or estrogen receptor were postive. The patient refused further treatment and died of disease three months after diagnosis.
CASE REPORT
- Uterine sarcomas are relatively uncommon with leiomyosarcoma as the most common histologic type. RMS is a common soft tissue tumor seen in children, but pure uterine sarcomas arising in the female genital tract are rare, particularly in adults.1 In the pediatric population, the most commonly involved site is the vagina, whereas the uterine cervix and corpus are the most frequent locations of RMS in adult women.2,3 In postmenopausal women, uterine RMSs appear to be entirely the pleomorphic type with extremely poor clinical outcome.4,5
- RMSs can be morphologically classified into three categories: embryonal, alveolar, and pleomorphic. The spindle cell variant is a recently characterized and rare subtype of embryonal RMS first described in 1992.6 Spindle cell RMS predominately occurs in the paratesticular region in children and young adults, and carries a relatively favorable prognosis. After Rubin et al.7 reported the first two cases of spindle cell RMS in adults, limited reports of adult spindle cell RMS have been subsequently published.8 According to the largest review performed by Nascimento and Fletcher,9 the head and neck region were the most common sites for adult spindle cell RMS followed by the retroperitoneum and lower extremity. These adult tumors seem to have a more aggressive clinical course compared to pediatric cases. Only one case of uterine spindle cell RMS has been previously reported in a 28-year-old woman who presented at International Federation of Gynecology and Obstetrics (FIGO) stage I and was well after two years of follow-up. The case here, was FIGO stage IIIA at presentation, and the patient died of disease three months after diagnosis.
- Differential diagnosis for spindle cell RMS of the uterus includes leiomyosarcoma, undifferentiated endometrial sarcoma, and rhabdomyosarcomatous components of a mullerian adenosarcoma or carcinosarcoma. Immunohistochemical staining for skeletal muscle markers can be helpful in differentiating spindle cell RMS from other spindle cell tumors. Leiomyosarcoma is the most common malignant mesenchymal tumor of the uterine corpus and has similar histologic finding to spindle cell RMS. Tumor cells of leiomyosarcoma are spindle-shaped with characteristic cigar-shaped nuclei, or large pleomorphic resembling rhabdomyoblasts. Markers such as myo-D1 and myoglobin are more specific than muscle-specific actin, smooth muscle actin, and desmin, because smooth muscle markers have limited value in differential diagnosis. Undifferentiated endometrial sarcomas, highly aggressive neoplasms, do not histologically resemble entometrial stroma and may consist of spindle or markedly atypical tumor cells. Although CD10 expression has been observed in both uterine RMS4 and undifferentiated endometrial sarcoma, immunophenotypic evidence of skeletal muscle differentiation has only been seen with RMS. Mixed epithelial and mesenchymal tumors including adenosarcoma and carcinosarcoma consist of a mixture of benign or malignant epithelial and sarcomatous components. Epithelial components are usually glandular or rarely non-glandular and can be constitute a small portion of the tumor, especially in carcinosarcoma. Sarcomatous elements may be either homologous or heterologous with heterologous components frequently exhibiting rhabdomyoblastic differentiation. When sarcomatoid overgrowth predominates, epithelial components can be obscured. Extensive tumor sampling with careful investigation for epithelial areas, and immunohistochemical confirmation may be required to differentiate RMS from mixed epithelial and mesenchymal tumors.
- In conclusion, pure RMS of the uterine corpus is uncommon, and the spindle cell variant is especially rare. Here, we present a case of spindle cell RMS of the uterine body in 76-year-old woman. Uterine RMS should be differentiated from leiomyosarcoma, undifferentiated sarcoma, and mixed epithelial and mesenchymal tumors. Extensive tumor sampling, careful microscopic examination, and immunohistochemical staining may aid in a diagnosis of spindle cell RMS.
DISCUSSION
- 1. Scully RE, Bonfiglio TA, Kurman RJ, Silverberg SG, Wilkinson EJ. Histological typing of female genital tract tumours. 2nd ed. Berlin: Springer-Verlag, 1994.
- 2. Hays DM, Shimada H, Raney RB Jr, et al. Sarcomas of the vagina and uterus: the Intergroup Rhabdomyosarcoma Study. J Pediatr Surg 1985; 20: 718-724. ArticlePubMed
- 3. Ferguson SE, Gerald W, Barakat RR, Chi DS, Soslow RA. Clinicopathologic features of rhabdomyosarcoma of gynecologic origin in adults. Am J Surg Pathol 2007; 31: 382-389. ArticlePubMed
- 4. Fadare O, Bonvicino A, Martel M, Renshaw IL, Azodi M, Parkash V. Pleomorphic rhabdomyosarcoma of the uterine corpus: a clinicopathologic study of 4 cases and a review of the literature. Int J Gynecol Pathol 2010; 29: 122-134. ArticlePubMed
- 5. McCluggage WG, Lioe TF, McClelland HR, Lamki H. Rhabdomyosarcoma of the uterus: report of two cases, including one of the spindle cell variant. Int J Gynecol Cancer 2002; 12: 128-132. ArticlePubMed
- 6. Cavazzana AO, Schmidt D, Ninfo V, et al. Spindle cell rhabdomyosarcoma. a prognostically favorable variant of rhabdomyosarcoma. Am J Surg Pathol 1992; 16: 229-235. PubMed
- 7. Rubin BP, Hasserjian RP, Singer S, Janecka I, Fletcher JA, Fletcher CD. Spindle cell rhabdomyosarcoma (so-called) in adults: report of two cases with emphasis on differential diagnosis. Am J Surg Pathol 1998; 22: 459-464. PubMed
- 8. Mentzel T, Kuhnen C. Spindle cell rhabdomyosarcoma in adults: clinicopathological and immunohistochemical analysis of seven new cases. Virchows Arch 2006; 449: 554-560. ArticlePubMedPDF
- 9. Nascimento AF, Fletcher CD. Spindle cell rhabdomyosarcoma in adults. Am J Surg Pathol 2005; 29: 1106-1113. ArticlePubMed
REFERENCES
Fig. 1Abdominal computed tomorgaphy demonstrating a huge, lobulating-contoured, lower abdominal and pelvic mass with omental and mesenteric fat infiltration.
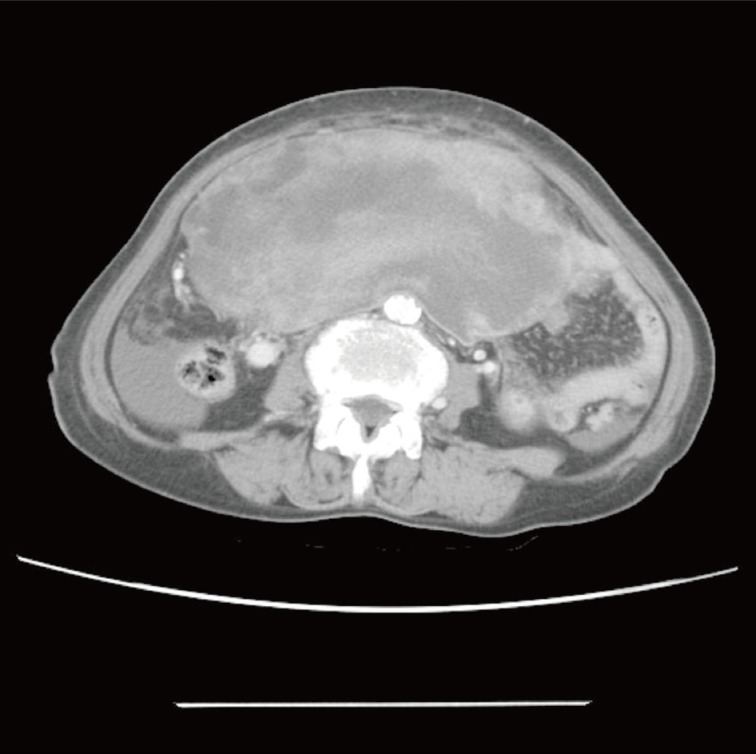

Fig. 2Gross tumor findings showing a yellowish-white cut surface with vague trabeculation and myxoid change.
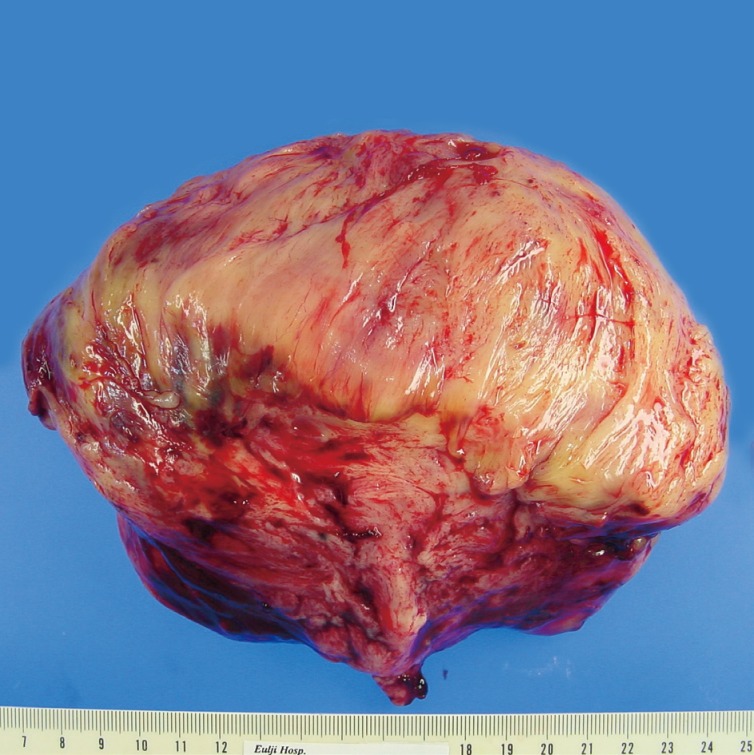

Figure & Data
References
Citations
Citations to this article as recorded by 

- A rare adult case of primary uterine rhabdomyosarcoma with mixed pattern: a clinicopathological & immunohistochemical study with literature review
Nehal K.H. Kamel, Eiman Adel Hasby
Diagnostic Pathology.2024;[Epub] CrossRef - Embryonal rhabdomyosarcoma of the uterine corpus: a clinicopathological and molecular analysis of 21 cases highlighting a frequent association with DICER1 mutations
Jennifer A. Bennett, Zehra Ordulu, Robert H. Young, Andre Pinto, Koen Van de Vijver, Eike Burandt, Pankhuri Wanjari, Rajeev Shah, Leanne de Kock, William D. Foulkes, W. Glenn McCluggage, Lauren L. Ritterhouse, Esther Oliva
Modern Pathology.2021; 34(9): 1750. CrossRef - Vaginal embryonal rhabdomyosarcoma in young woman: A case report and literature review
Lalya Issam, Laatitioui Sana, Essadi Ismail, El Omrani Abdelhamid, Khouchani Mouna
Archives of Cancer Science and Therapy.2020; 4(1): 034. CrossRef - Is fertility-preservation safe for adult non-metastatic gynecologic rhabdomyosarcoma patients? Systematic review and pooled survival analysis of 137 patients
Maha AT Elsebaie, Zeinab Elsayed
Archives of Gynecology and Obstetrics.2018; 297(3): 559. CrossRef - Spindle cell sarcoma – a rare diagnosis
SK Kathpalia, Manju Mehrotra, Pinky Jena, Archana H Deshpande
Women's Health.2018;[Epub] CrossRef - Malignant mesenchymal tumors of the uterus – time to advocate a genetic classification
Birgit Rommel, Carsten Holzmann, Jörn Bullerdiek
Expert Review of Anticancer Therapy.2016; 16(11): 1155. CrossRef - Human rhabdomyosarcoma cells express functional pituitary and gonadal sex hormone receptors: Therapeutic implications
AGATA PONIEWIERSKA-BARAN, GABRIELA SCHNEIDER, WENYUE SUN, AHMED ABDELBASET-ISMAIL, FREDERIC G. BARR, MARIUSZ Z. RATAJCZAK
International Journal of Oncology.2016; 48(5): 1815. CrossRef - Primary third ventricular tumor in an 18‐year‐old man
Tianping Yu, Mengni Zhang, Qiao Zhou, Jing Gong, Ling Nie, Xueqin Chen, Ni Chen
Neuropathology.2015; 35(6): 599. CrossRef - Rhabdomyosarcoma of vulva in a young lady
Tapesh Bhattacharyya, Firuza D. Patel, Radhika Srinivasan, Bhavana Rai, Pradeep Saha, R. Nijhawan
Journal of Cancer Research and Therapeutics.2015; 11(3): 650. CrossRef - Uterine sarcoma in a 14 year-old girl presenting with uterine rupture
Jane Özcan, Özlem Dülger, Latif Küpelioğlu, Ali İhsan Gönenç, Aynur Erşahin
Gynecologic Oncology Reports.2014; 10: 44. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
- Related articles
Spindle Cell Rhabdomyosacoma of Uterus: A Case Study


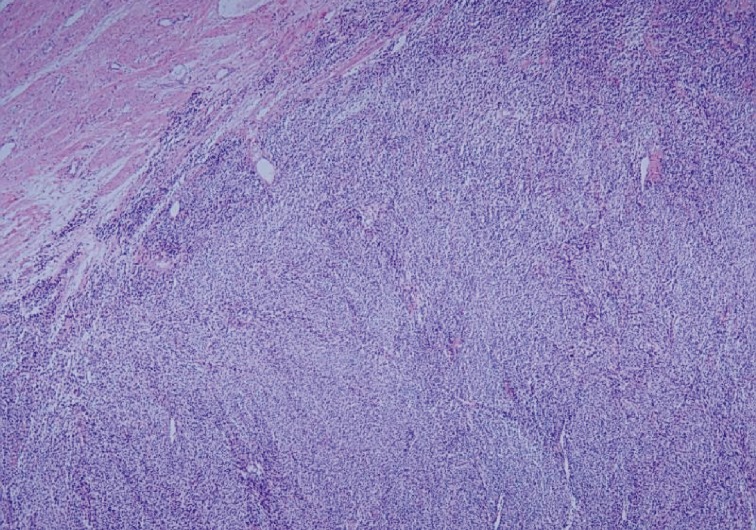
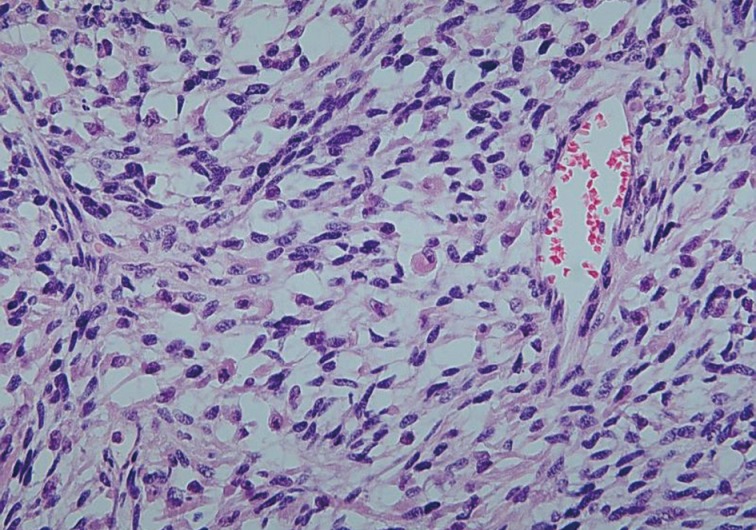

Fig. 1 Abdominal computed tomorgaphy demonstrating a huge, lobulating-contoured, lower abdominal and pelvic mass with omental and mesenteric fat infiltration.
Fig. 2 Gross tumor findings showing a yellowish-white cut surface with vague trabeculation and myxoid change.
Fig. 3 Microscopic finding exhibiting highly cellular tumor composed of interlacing bundles of spindle cells with infiltrating margin.
Fig. 4 Isolated rhabdomyoblasts admixed with spindle cells.
Fig. 5 Tumor cells strongly positive for myoglobin.
Fig. 1
Fig. 2
Fig. 3
Fig. 4
Fig. 5
Spindle Cell Rhabdomyosacoma of Uterus: A Case Study

 E-submission
E-submission