Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 47(2); 2013 > Article
-
Case Study
A Solitary Fibrous Tumor with Giant Cells in the Lacrimal Gland: A Case Study - Da Hye Son, Su Hyun Yoo, Ho-Seok Sa1, Kyung-Ja Cho
-
Korean Journal of Pathology 2013;47(2):158-162.
DOI: https://doi.org/10.4132/KoreanJPathol.2013.47.2.158
Published online: April 24, 2013
Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
1Department of Ophthalmology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- Corresponding Author: Kyung-Ja Cho, M.D. Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, 88 Olympic-ro 43-gil, Songpa-gu, Seoul 138-736, Korea. Tel: +82-2-3010-4545, Fax: +82-2-472-7898, kjc@amc.seoul.kr
• Received: May 7, 2012 • Revised: June 28, 2012 • Accepted: July 2, 2012
© 2013 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- Orbital solitary fibrous tumor (SFT) has recently been proposed as the encompassing terminology for hemangiopericytoma, giant cell angiofibroma (GCAF), and fibrous histiocytoma of the orbit. The lacrimal gland is a very rare location for both SFT and GCAF. A 39-year-old man presented with a painless left upper eyelid mass. An orbital computed tomography scan identified a 1.1 cm-sized well-defined nodule located in the left lacrimal gland. He underwent a mass excision. Histopathologic examination showed a proliferation of relatively uniform spindle cells with a patternless or focally storiform pattern. Dilated vessels were prominent, but angiectoid spaces lined with giant cells were absent. Floret-type giant cells were mostly scattered in the periphery. The tumor was immunoreactive for CD34 and CD99, but negative for smooth muscle actin and S-100 protein. This is the first Korean case of SFT of the lacrimal gland with overlapping features of GCAF, suggesting a close relationship between the two entities.
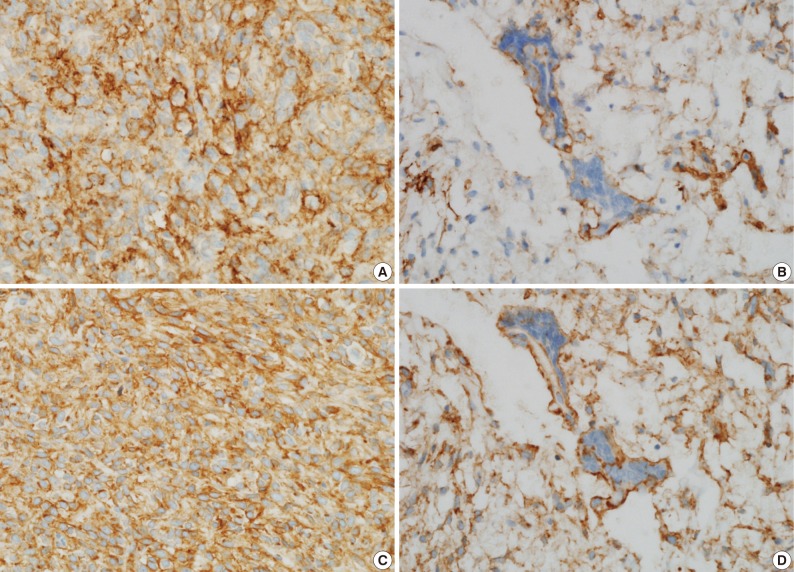
- A 39-year-old man presented with a painless left upper eyelid mass that had presented three months previous. An orbital computed tomography scan showed that a 1.1 cm-sized, well-defined nodule with an eccentric nodular enhancement, and an internal low attenuation area was present in the left lacrimal gland (Fig. 1A). An en-block excision was performed under the impression that this mass was a pleomorphic adenoma. The excised mass was well-demarcated, ovoid, and firm, with measurements of 1.5×1.3×0.7 cm. The cut surface had a whitish tan hue and was fibrotic with a multifocal hemorrhage (Fig. 1B). Histopathological examination showed a spindle cell proliferative lesion that was surrounded by the lacrimal gland parenchyma (Fig. 2A). The cellularity was variable and presented as either a patternless or a storiform pattern (Fig. 2B). Collagenized stroma and dense collagen nodules were found around dilated vessels, and perivascular hyalinization was also observed (Fig. 2C). The spindle cells had scant cytoplasm and short, oval spindle nuclei with fine chromatin and inconspicuous nucleoli. Multinucleated giant cells were either observed in the periphery of the mass giant cells or were not identified (Fig. 2D). Mitotic activity was low (only observed in two of ten high magnification fields). Immunohistochemistry demonstrated that the spindle cells were strongly positive, and the multinucleated cell cytoplasm was focally positive for CD34 and CD99 (Fig. 3). Smooth muscle actin and S-100 protein were not detected. The diagnosis of a SFT was made on the basis of the histological findings, which showed that the mass lacked the typical components and immunoprofiles of giant angiofibromas.
CASE REPORT
- Although SFTs and GCAFs are listed as separate entities in the current World Health Organization (WHO) classification, it is generally acknowledged that the features of SFTs, GCAFs, hemangiopericytomas (HPC), and lipomatous HPC are commonly shared by a subset of tumors. Traditionally, SFTs are described as having patternless spindle cell tumors that are rich in collagen, and HPCs are described as having thin-walled, branched, or staghorn blood vessels. GCAFs are distinguished from these tumors by the presence of multinucleated giant stromal cells that often line the angiectoid spaces.5,15 These distinct entities also have common immunophenotypes, including positivity for both CD34 and CD99.
- The notion that SFTs and GCAFs should be united into one category appears to be gaining popularity among oncologists. In 1999, Hasegawa et al.10 described the presence of giant cells in six of 24 extrapleural SFTs. Guillou et al.7 studied one orbital and nine extraorbital GCAFs and asserted that GCAFs are merely a giant cell-rich variant of SFTs. They observed that GCAFs and SFTs have overlapping morphologic and clinical features. Both GCAFs and SFTs are well-circumscribed, nonencapsulated lesions that occur in middle-aged adults with an equal gender distribution and which can occur either inside or outside of the orbital region, contain a varying number of multinucleated giant cells, and are immunopositive for CD34 and CD99.7,10 Piperi et al.9 also demonstrated a lingual example of a vascular SFT, where an overlapping characteristic with GCAFs included the presence of "floret" cells. Recently, Furusato et al.2 reappraised 41 orbital cases and suggested orbital SFT as an encompassing terminology for HPC, GCAFs, and fibrous histiocytomas. They observed that the majority of cases presented with a prominent vasculature with perivascular fibrosis and were composed of haphazardly disposed fibroblast-like cells with round or oval nuclei, indistinct nucleoli, and variable stromal collagen. In addition, staghorn vessels, giant cells, fat, and ropey collagen were all common features whose statuses varied among tumors and even among different portions of the same neoplasm.2 Most of the cases originated from the orbital soft tissue, and only two cases of lacrimal gland tumors were included.
- The lacrimal gland tumor presented here also exhibited a patternless spindle cell proliferation with perivascular fibrosis and giant cells but did not present prominent pseudovascular spaces. Therefore, this tumor showed features that were intermediate between those of SFTs and those of GCAFs. Therefore, this case provides another example that supports the unifying concept. There seems be a morphological spectrum of SFTs whose morphologies vary depending on the site of the tumor. The orbital cases might more often present GCAF-like features than the SFT-like features observed at other sites for unknown reasons. To our knowledge, only seven cases of lacrimal gland SFTs or GCAFs have been reported since the first one was observed in 1996 by Scott et al.14 Among the seven cases, two were Korean (a 24-year-old man and a 26-year-old woman).11 The tumors in these cases were described as having unusual adenofibromatous features without any mention of giant cells. Therefore, this is the first Korean case report of a lacrimal gland SFT with features that overlap with those of GCAF. This study further suggests the existence of a close relationship between SFTs and GCAFs.
- SFTs are mostly benign, but some cases have been associated with malignancy.16 In the case described here, the tumor was small (1.5 cm at its largest dimension) and had a low mitotic activity (only observed in two of ten high magnification fields). The tumor did not present necrosis, nuclear pleomorphism, or an invasive growth pattern. England et al.17 suggested histological parameters that indicate the malignancy of SFTs in intrathoracic regions, which include increased cellularity, nuclear pleomorphisms, observation of mitosis in more than four of ten high magnification fields, hemorrhages, and necrosis. Vallat-Decouvelaere et al.16 suggested that nuclear atypia, hypercellularity, mitosis in four of ten high magnification fields, and necrosis be considered as malignant features in extrathoracic SFTs. The morphological criteria for uncertain behavior or low-grade malignant ocular SFTs are proposed to be the presence of cytologic atypia and increased mitotic activity.4 However, the overall outcomes for malignant solitary fibrous tumors in the eyes should be further explored.
DISCUSSION
- 1. Klemperer P, Coleman BR. Primary neoplasms of the pleura: a report of five cases. Am J Ind Med 1992; 22: 1-31. ArticlePubMed
- 2. Furusato E, Valenzuela IA, Fanburg-Smith JC, et al. Orbital solitary fibrous tumor: encompassing terminology for hemangiopericytoma, giant cell angiofibroma, and fibrous histiocytoma of the orbit: reappraisal of 41 cases. Hum Pathol 2011; 42: 120-128. ArticlePubMed
- 3. Demirci H, Shields CL, Eagle RC Jr, Shields JA. Giant cell angiofibroma, a variant of solitary fibrous tumor, of the orbit in a 16-year-old girl. Ophthal Plast Reconstr Surg 2009; 25: 402-404. ArticlePubMed
- 4. Ha JK, Park BJ, Kim YH, Lim YJ. Orbital solitary fibrous tumor: a case report and diagnostic clues. J Korean Neurosurg Soc 2009; 46: 77-80. ArticlePubMedPMC
- 5. Dei Tos AP, Seregard S, Calonje E, Chan JK, Fletcher CD. Giant cell angiofibroma: a distinctive orbital tumor in adults. Am J Surg Pathol 1995; 19: 1286-1293. PubMed
- 6. de Andrade CR, Lopes MA, de Almeida OP, León JE, Mistro F, Kignel S. Giant cell angiofibroma of the oral cavity: a case report and review of the literature. Med Oral Patol Oral Cir Bucal 2008; 13: E540-E543. PubMed
- 7. Guillou L, Gebhard S, Coindre JM. Orbital and extraorbital giant cell angiofibroma: a giant cell-rich variant of solitary fibrous tumor? Clinicopathologic and immunohistochemical analysis of a series in favor of a unifying concept. Am J Surg Pathol 2000; 24: 971-979. PubMed
- 8. Thomas R, Banerjee SS, Eyden BP, et al. A study of four cases of extra-orbital giant cell angiofibroma with documentation of some unusual features. Histopathology 2001; 39: 390-396. ArticlePubMed
- 9. Piperi E, Rohrer MD, Pambuccian SE, Koutlas IG. Vascular solitary fibrous tumor with "floret" cells or giant cell angiofibroma? A lingual example highlighting the overlapping characteristics of these entities and positive immunoreaction for estrogen and progesterone receptors. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2009; 107: 685-690. ArticlePubMed
- 10. Hasegawa T, Matsuno Y, Shimoda T, Hasegawa F, Sano T, Hirohashi S. Extrathoracic solitary fibrous tumors: their histological variability and potentially aggressive behavior. Hum Pathol 1999; 30: 1464-1473. ArticlePubMed
- 11. Kim HY, Lee SY, Kang SJ, Kim HJ. Solitary fibrous tumor of the orbit: a poorly-recognized orbital lesion. Acta Ophthalmol Scand 1999; 77: 704-708. ArticlePubMedPDF
- 12. Polito E, Tosi GM, Toti P, Schürfeld K, Caporossi A. Orbital solitary fibrous tumor with aggressive behavior: three cases and review of the literature. Graefes Arch Clin Exp Ophthalmol 2002; 240: 570-574. ArticlePubMedPDF
- 13. Cho NH, Kie JH, Yang WI, Jung WH. Solitary fibrous tumour with an unusual adenofibromatous feature in the lacrimal gland. Histopathology 1998; 33: 289-290. ArticlePubMedPDF
- 14. Scott IU, Tanenbaum M, Rubin D, Lores E. Solitary fibrous tumor of the lacrimal gland fossa. Ophthalmology 1996; 103: 1613-1618. ArticlePubMed
- 15. Hayashi Y, Uchiyama N, Hayashi Y, et al. A reevaluation of the primary diagnosis of hemangiopericytoma and the clinical importance of differential diagnosis from solitary fibrous tumor of the central nervous system. Clin Neurol Neurosurg 2009; 111: 34-38. ArticlePubMed
- 16. Vallat-Decouvelaere AV, Dry SM, Fletcher CD. Atypical and malignant solitary fibrous tumors in extrathoracic locations: evidence of their comparability to intra-thoracic tumors. Am J Surg Pathol 1998; 22: 1501-1511. PubMed
- 17. England DM, Hochholzer L, McCarthy MJ. Localized benign and malignant fibrous tumors of the pleura: a clinicopathologic review of 223 cases. Am J Surg Pathol 1989; 13: 640-658. PubMed
REFERENCES
Fig. 1(A) An orbital computed tomography scan shows a 1.1 cm-sized, well-demarcated nodule with an eccentric nodular enhancement and an internal low attenuation area in the left lacrimal gland. (B) The solitary fibrous tumor specimen from the lacrimal gland is a well-circumscribed, whitish tan, firm, and fibrotic nodule with a hemorrhagic lesion.
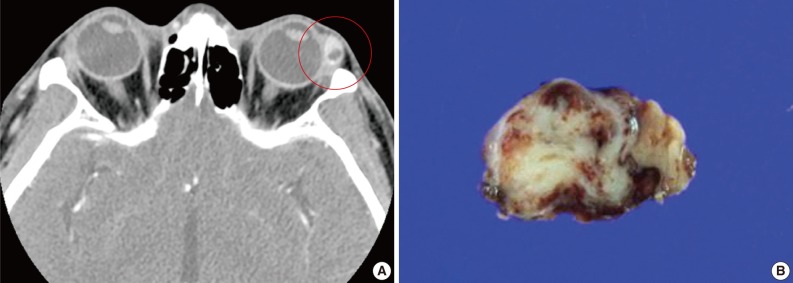

Fig. 2(A, B) Microscopic examination of the lacrimal gland lesion shows proliferation of relatively uniform spindle cells that are either patternless or have a focally storiform pattern. (C) Focal vascular dilatation with collagenized stroma, dense collagen nodules, and perivascular hyalinization is also observed. (D) A small number of floret-type giant cells are scattered at the periphery, but no angiectoid spaces lined with giant cells are found.
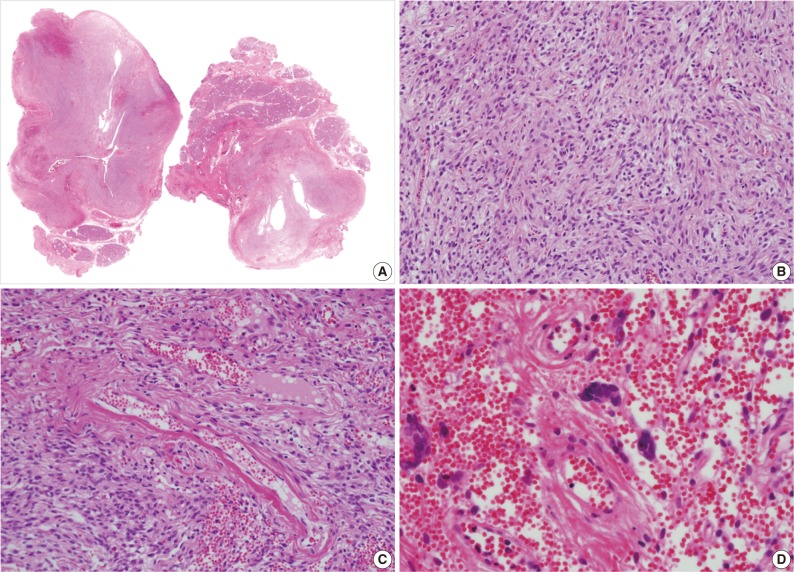

Figure & Data
References
Citations
Citations to this article as recorded by 

- Radiological features of lacrimal gland masses
Sonia Huang, Jessica Y. Tong, Valerie Juniat, Abdullah Almater, Ilse Mombaerts, Dinesh Selva
Survey of Ophthalmology.2026; 71(2): 700. CrossRef - Imaging features of lacrimal gland disease
Carmelo Caltabiano, Khizar Rana, Alexander Buckby, Sandy Patel, Dinesh Selva
Frontiers in Ophthalmology.2026;[Epub] CrossRef - A review of solitary fibrous tumours of the orbit and ocular adnexa
Cornelius René, Paolo Scollo, Dominic O’Donovan
Eye.2023; 37(5): 858. CrossRef - A giant orbital solitary fibrous tumor treated by surgical excision: a case report and literature review
Qi Zhou, Yuting Liu, Fang Wang, Yang Cao, Hongbin Lv, Xibo Zhang
Diagnostic Pathology.2023;[Epub] CrossRef - Giant cell-rich solitary fibrous tumour of the lacrimal gland with prominent angiomatoid cystic changes and an underlying NAB2ex3-STAT6ex18 fusion
Khaled A Alsaadi, Manar Alwohaib, Karen Pinto, Rola H Ali
BMJ Case Reports.2022; 15(2): e247141. CrossRef - Cystic appearance - a new feature of solid fibrous tumours in the lacrimal gland: a case report with literature review
Ancuta-Augustina Gheorghisan-Galateanu, Dana Cristina Terzea, Iulia Burcea, Roxana Dusceac, Cristina Capatina, Catalina Poiana
Diagnostic Pathology.2019;[Epub] CrossRef - Solitary Fibrous Tumor in the Lacrimal Gland Fossa: A Case Report
Jacqueline Mupas-Uy, Yoshiyuki Kitaguchi, Yasuhiro Takahashi, Emiko Takahashi, Hirohiko Kakizaki
Case Reports in Ophthalmology.2016; 7(2): 398. CrossRef - Ocular adnexal (orbital) solitary fibrous tumor: nuclear STAT6 expression and literature review
Aleksandra Petrovic, Aurélie Obéric, Alexandre Moulin, Mehrad Hamedani
Graefe's Archive for Clinical and Experimental Ophthalmology.2015; 253(9): 1609. CrossRef - Angiofibroma de células gigantes en mucosa yugal: una entidad rara en una localización infrecuente
Alejandro Rubio Fernández, María López Macías, Weimar Toro Zambrano, Mario Díaz Delgado, Alicia Hernández Amate
Revista Española de Patología.2014; 47(4): 223. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
A Solitary Fibrous Tumor with Giant Cells in the Lacrimal Gland: A Case Study
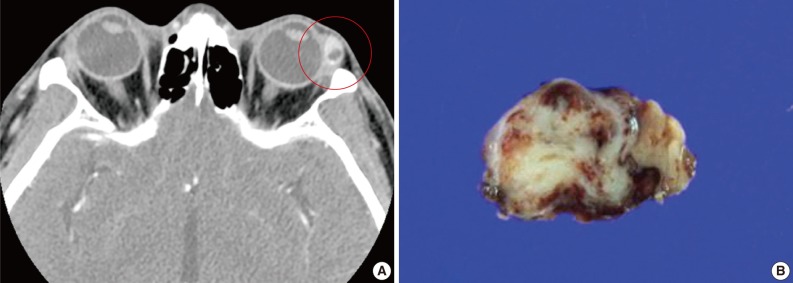
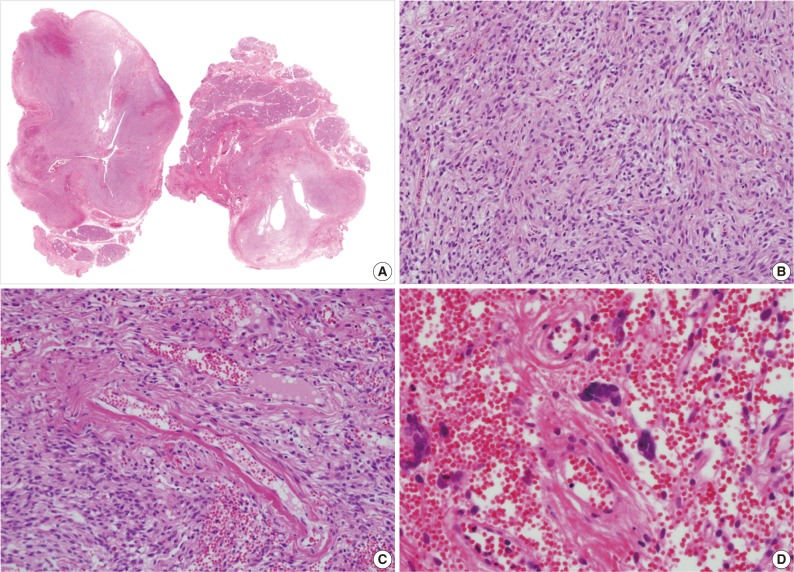
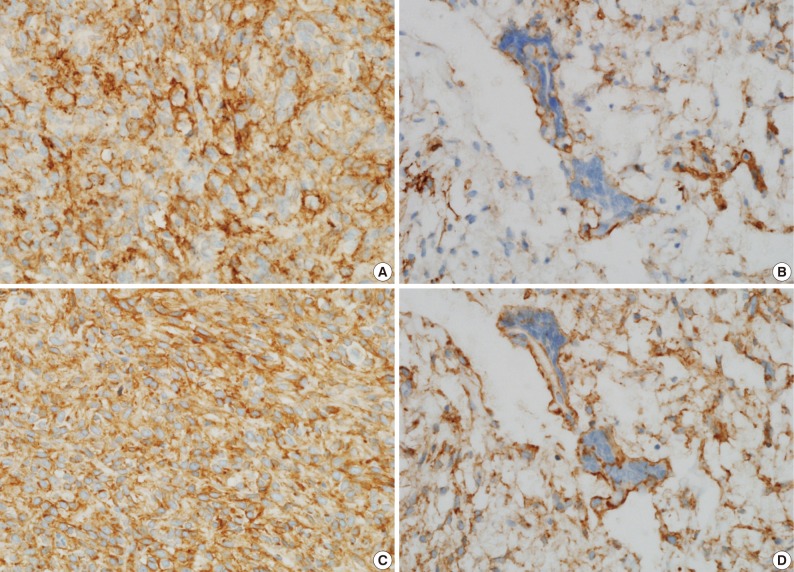
Fig. 1 (A) An orbital computed tomography scan shows a 1.1 cm-sized, well-demarcated nodule with an eccentric nodular enhancement and an internal low attenuation area in the left lacrimal gland. (B) The solitary fibrous tumor specimen from the lacrimal gland is a well-circumscribed, whitish tan, firm, and fibrotic nodule with a hemorrhagic lesion.
Fig. 2 (A, B) Microscopic examination of the lacrimal gland lesion shows proliferation of relatively uniform spindle cells that are either patternless or have a focally storiform pattern. (C) Focal vascular dilatation with collagenized stroma, dense collagen nodules, and perivascular hyalinization is also observed. (D) A small number of floret-type giant cells are scattered at the periphery, but no angiectoid spaces lined with giant cells are found.
Fig. 3 Immunohistochemical staining shows that the spindle cell cytoplasms are strongly and diffuse positive for CD34 (A) and CD99 (C), and the giant cell cytoplasms are focally positive for CD34 (B) and CD99 (D).
Fig. 1
Fig. 2
Fig. 3
A Solitary Fibrous Tumor with Giant Cells in the Lacrimal Gland: A Case Study

 E-submission
E-submission