Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 46(6); 2012 > Article
-
Original Article
Prognostic Relevance of the Expression of CA IX, GLUT-1, and VEGF in Ovarian Epithelial Cancers - Kyungbin Kim1, Won Young Park1, Jee Yeon Kim1,2, Mee Young Sol2,3, Dong Hun Shin2,3, Do Youn Park1,2, Chang Hun Lee1,2, Jeong Hee Lee3, Kyung Un Choi2,3,4
-
Korean Journal of Pathology 2012;46(6):532-540.
DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.6.532
Published online: December 26, 2012
1Department of Pathology, Pusan National University Hospital, Pusan National University School of Medicine, Yangsan, Korea.
2Department of Pathology, Pusan National University School of Medicine, Yangsan, Korea.
3Department of Pathology, Pusan National University Yangsan Hospital, Pusan National University School of Medicine, Yangsan, Korea.
4Research Institute for Convergence of Biomedical Science and Technology, Pusan National University Yangsan Hospital, Pusan National University School of Medicine, Yangsan, Korea.
- Corresponding Author: Kyung Un Choi, M.D. Department of Pathology, Pusan National University Yangsan Hospital, Pusan National University School of Medicine, 20 Geumo-ro, Yangsan 626-787, Korea. Tel: +82-55-360-1862, Fax: +82-55-360-1865, kuchoi@pusan.ac.kr
© 2012 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Tumor hypoxia is associated with malignant progression and treatment resistance. Hypoxia-related factors, such as carbonic anhydrase IX (CA IX), glucose transporter-1 (GLUT-1), and vascular endothelial growth factor (VEGF) permit tumor cell adaptation to hypoxia. We attempted to elucidate the correlation of these markers with variable clinicopathological factors and overall prognosis.
-
Methods
- Immunohistochemistry for CA IX, GLUT-1, and VEGF was performed on formalin-fixed, paraffin-embedded tissues from 125 cases of ovarian epithelial cancer (OEC).
-
Results
- CA IX expression was significantly associated with an endometrioid and mucinous histology, nuclear grade, tumor necrosis, and mitosis. GLUT-1 expression was associated with tumor necrosis and mitosis. VEGF expression was correlated only with disease recurrence. Expression of each marker was not significant in terms of overall survival in OECs; however, there was a significant correlation between poor overall survival rate and high coexpression of these markers.
-
Conclusions
- The present study suggests that it is questionable whether CA IX, GLUT-1, or VEGF can be used alone as independent prognostic factors in OECs. Using at least two markers helps to predict patient outcomes in total OECs. Moreover, the inhibition of two target gene combinations might prove to be a novel anticancer therapy.
- Patients and tissue samples
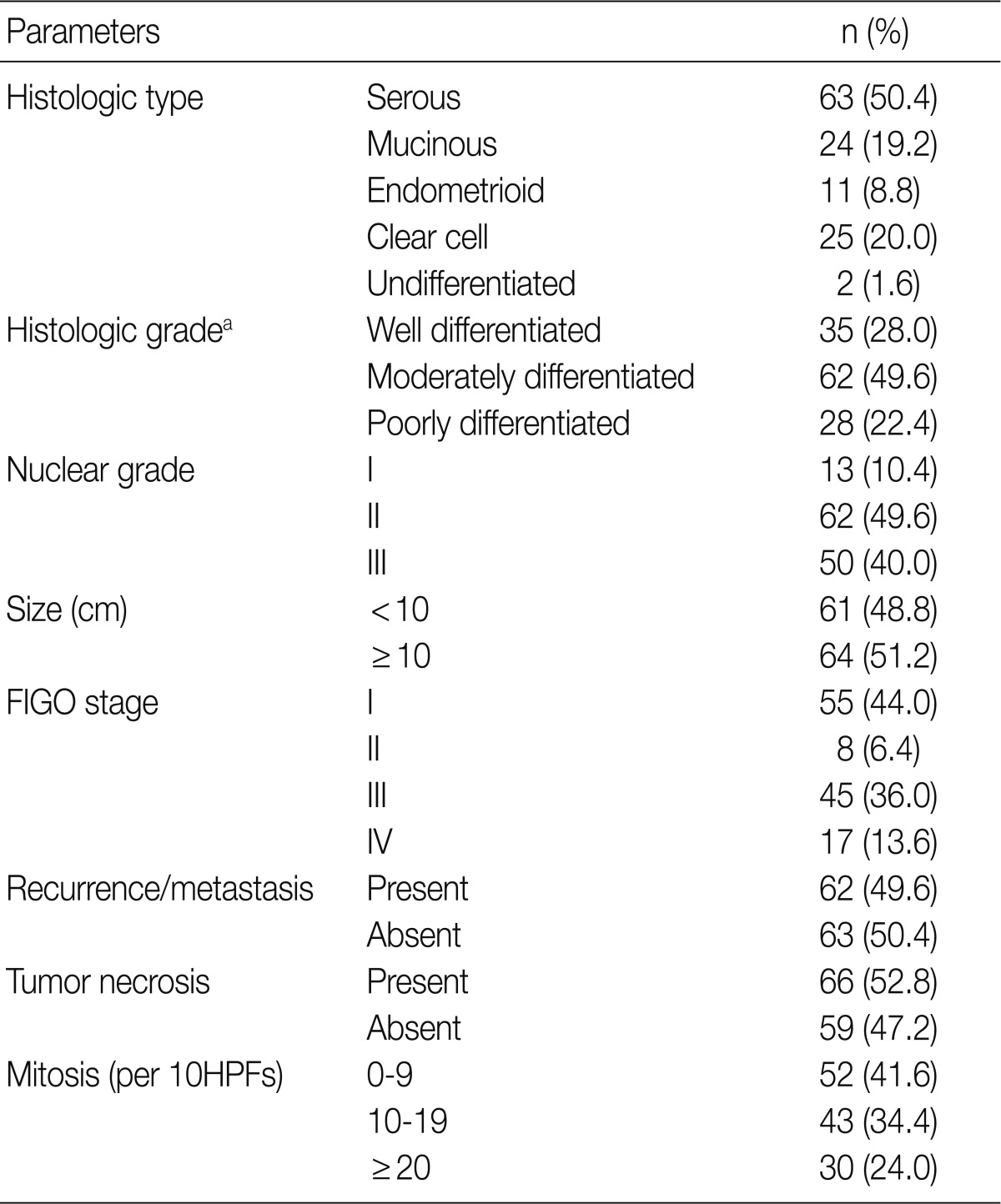
- The tissue samples of OECs were obtained during surgical operations performed at Pusan National University Hospital, Korea, from 1999 to 2008. A total of 125 patients (mean age, 49.5 years; range, 15 to 82 years) who underwent surgical resection for OECs were examined. All patients, except those with grade 1, stage IA, were given adjuvant chemotherapy of a platinum/taxol containing multiple drugs. The patients were followed up from the date of surgery until death or the last visit to the outpatient department. The follow-up period ranged from 1 to 140 months (median, 38 months). Pathological data, such as pathologic stage, tumor grading, tumor size, mitosis, and histological tumor type, were obtained from the primary pathology reports and patient medical record was reviewed retrospectively. Surgical staging was determined based on the criteria recommended by the International Federation of Gynecology and Obstetrics (FIGO). Histologic tumor type and grade were determined according to the World Health Organization criteria. Table 1 provides an overview of the main clinicopathologic features.19
- Immunohistochemistry
- Immunohistochemistry was performed on serial 4 µm-thick paraffin sections. The sections were deparaffinized in xylene and rehydrated in four decreasing grades of ethanol (100%, 95%, 80%, and then 70%) for 2 minutes each. Endogenous peroxidase activity was blocked by immersing the slides in 3% hydrogen peroxide in methanol for 15 minutes at room temperature. Heat-induced antigen retrieval was performed using a microwave. The slides were immersed in 1× citrate buffer (pH 6.3) and treated for 5 minutes and then cooled for 5 minutes. This process was performed three times. Slides were then cooled down under running tap water for 30 seconds and washed with phosphate-buffered saline (PBS) for 10 minutes. In order to reduce nonspecific absorption of antibodies to the tissues, the slides were pre-incubated with blocking serum in 1% bovine serum albumin for 30 minutes at room temperature. The slides were then left to incubate with rabbit polyclonal anti-CA IX antibody (1:1,000, Abcam, Cambridge, UK), rabbit polyclonal anti-GLUT-1 antibody (1:200, Dako, Carpinteria, CA, USA), and mouse monoclonal anti-VEGF antibody (1:200, Santa Cruz Biotechnology, Santa Cruz, CA, USA) overnight at 4℃. After incubation with primary antibodies, the slides were rinsed with PBS for 10 minutes. Thereafter, for detection of the antibody reactions, EnVision Detection System (Dako) was used at room temperature according to the manufacturer's recommended protocol. Finally, the color was developed using diaminobenzidine and the sections were then counterstained with Mayer's hematoxylin solution for 1 minute, and dehydrated with graded alcohols, dipped in 2 changes of xylene, and mounted. The following appropriate positive controls were used: renal cell carcinoma specimen for CA IX, erythrocytes for GLUT-1, and endothelial cells for VEGF. As negative controls for each antibody, adjacent sections were incubated in parallel using non-immune serum instead of primary antibodies.
- Assessment of immunohistochemical staining
- Results of immunohistochemical staining were assessed using a light microscope in a blinded fashion. Stainings were scored blinded to clinicopathologic data and the results of other stainings. For CA IX, only a membranous staining was regarded as positive. GLUT-1 and VEGF were considered positive when a membranous or cytoplasmic staining was identified. The extent of their expression was semi-quantitatively evaluated according to the following scoring system: 0, negative staining (0%); 1, weak positive (<10%); 2, moderate positive (10-50%); and 3, strong positive (>50%).16.20 Erythrocytes in each section were used as positive internal controls for GLUT-1. Finally, to perform a statistical analysis, we grouped various patterns of staining into two categories: low expression (score 0 and 1) and high expression (score 2 and 3).
- Statistical analysis
- All statistical analyses were performed using SPSS ver. 15.0 (SPSS Inc., Chicago, IL, USA). Pearson's chi-squared test and Fisher's exact test were used to study the associations between clinicopathological factors and immunohistochemical markers. Overall survival (OS) was calculated from the day of surgery until the death of the patient. Data on patients who had survived until the end of the observation period were censored at their last follow-up visit. Death from a cause other than ovarian cancer or cases lost to follow-up was considered a censoring event. The effect of immunohistochemical markers on survival was assessed using Kaplan-Meier survival curves and a log rank test. For all of the tests, a p≤0.05 was considered to be significant.
MATERIALS AND METHODS
- The expression of CA IX, GLUT-1, and VEGF
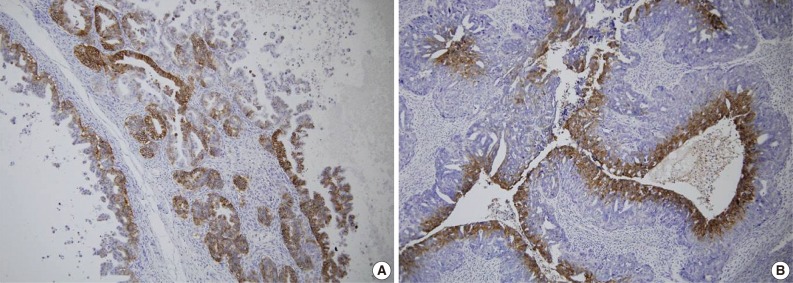
- A total of 125 OEC samples were used for the determination of CA IX, GLUT-1, and VEGF expression. CA IX expression is shown in Fig. 1. There was negative staining in 26 specimens (score 0, 20.8%). CA IX was detectable in 99 (79.2%) of the 125 specimens. CA IX expression was scored as 1 in 47 specimens (37.6%), scored as 2 in 41 specimens (32.8%), and scored as 3 in 11 specimens (8.8%). High CA IX expression was noted in 52 (41.6%) among all of the 125 cases.
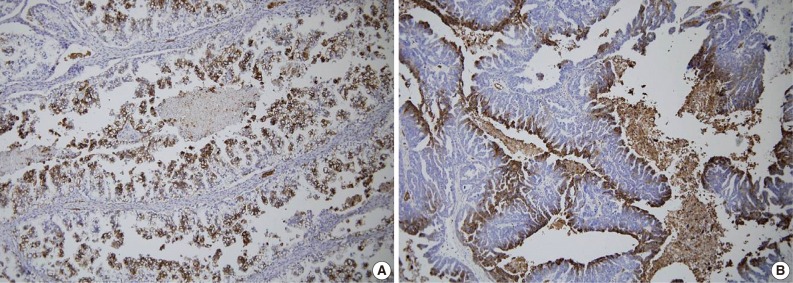
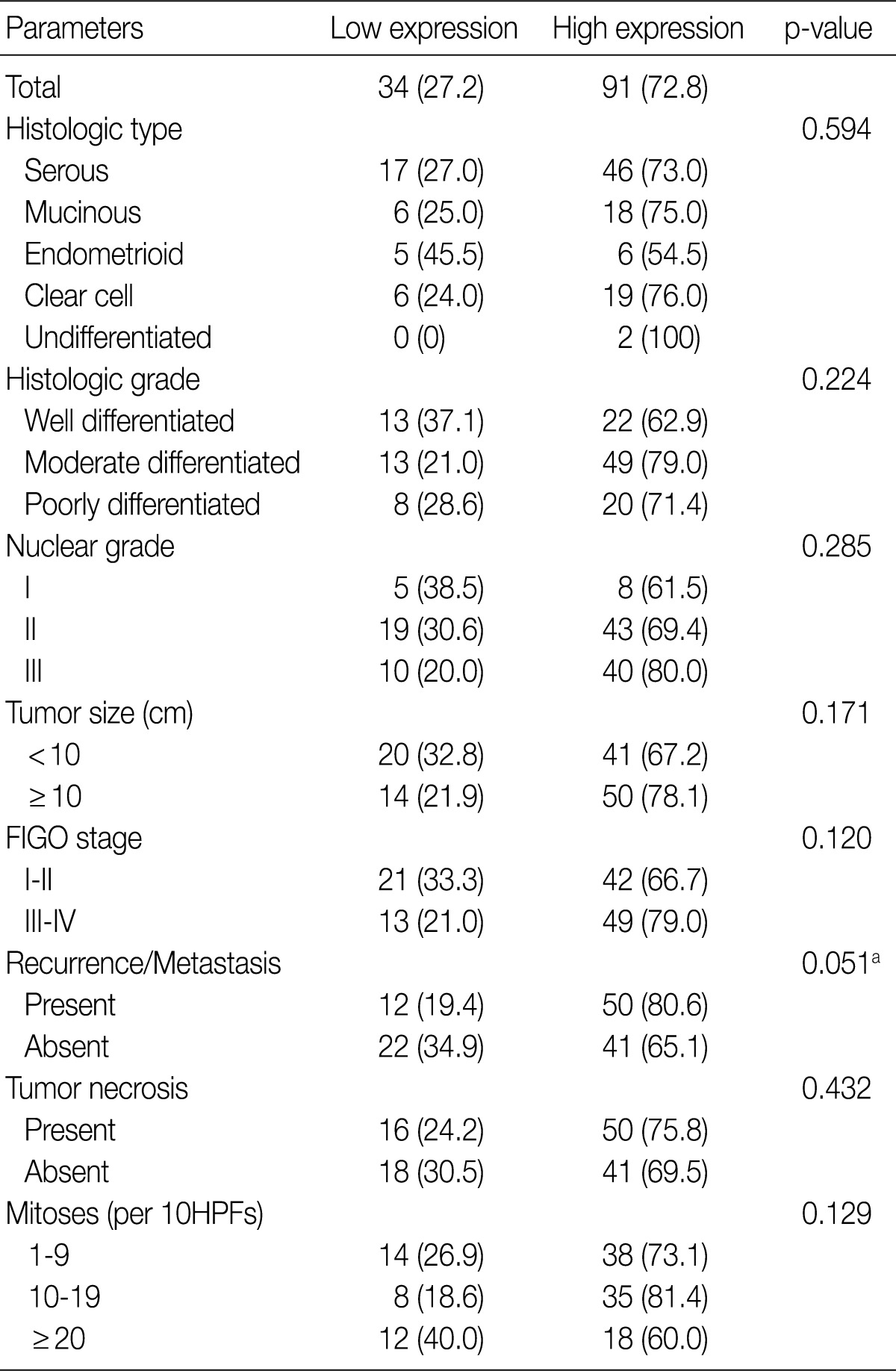
- GLUT-1 expression showed a membranous or cytoplasmic staining pattern. Of 125 OEC cases, 77 (61.6%) showed positive GLUT-1 immunostaining. Weak staining was detected in 13 cases (score 1, 10.4%), moderate staining in 46 cases (score 2, 36.8%), and strong staining in 18 cases (score 3, 14.4%) (Fig. 2). High GLUT-1 expression was noted in 64 (51.2%) of the total of 125 OEC cases, whereas, negative staining was observed in 48 cases (score 0, 38.4%).
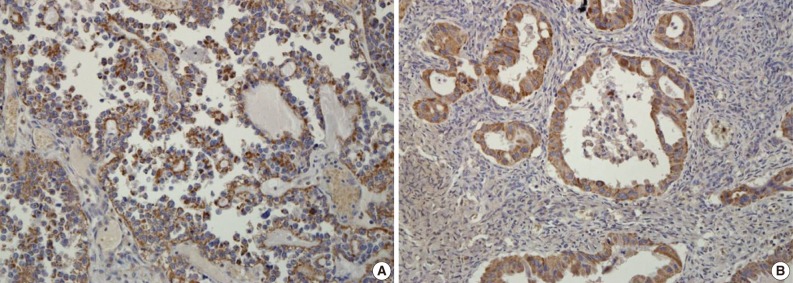
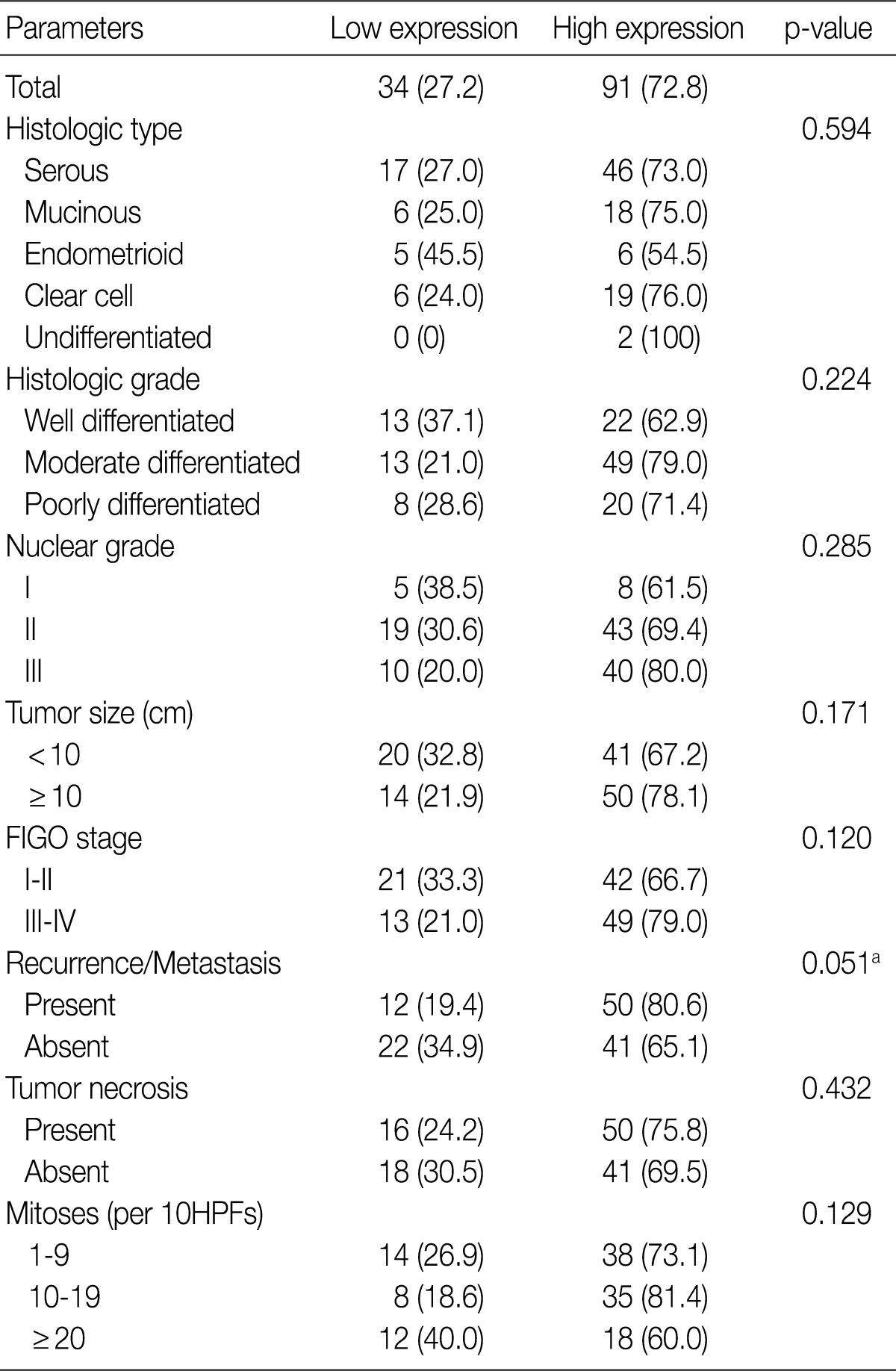
- An immunohistochemical study of VEGF expression in the 125 OEC cases revealed no VEGF expression in 10 cases (score 0, 8.0%), a score of 1 in 24 cases (19.2%), a score of 2 in 60 cases (48.0%), and a score of 3 in 31 cases (24.8%) (Fig. 3). Thus high VEGF expression was observed in 91 (72.8%) of all of the 125 OEC cases.
- Correlation between high expression of HRFs and clinicopathological characteristics in OECs
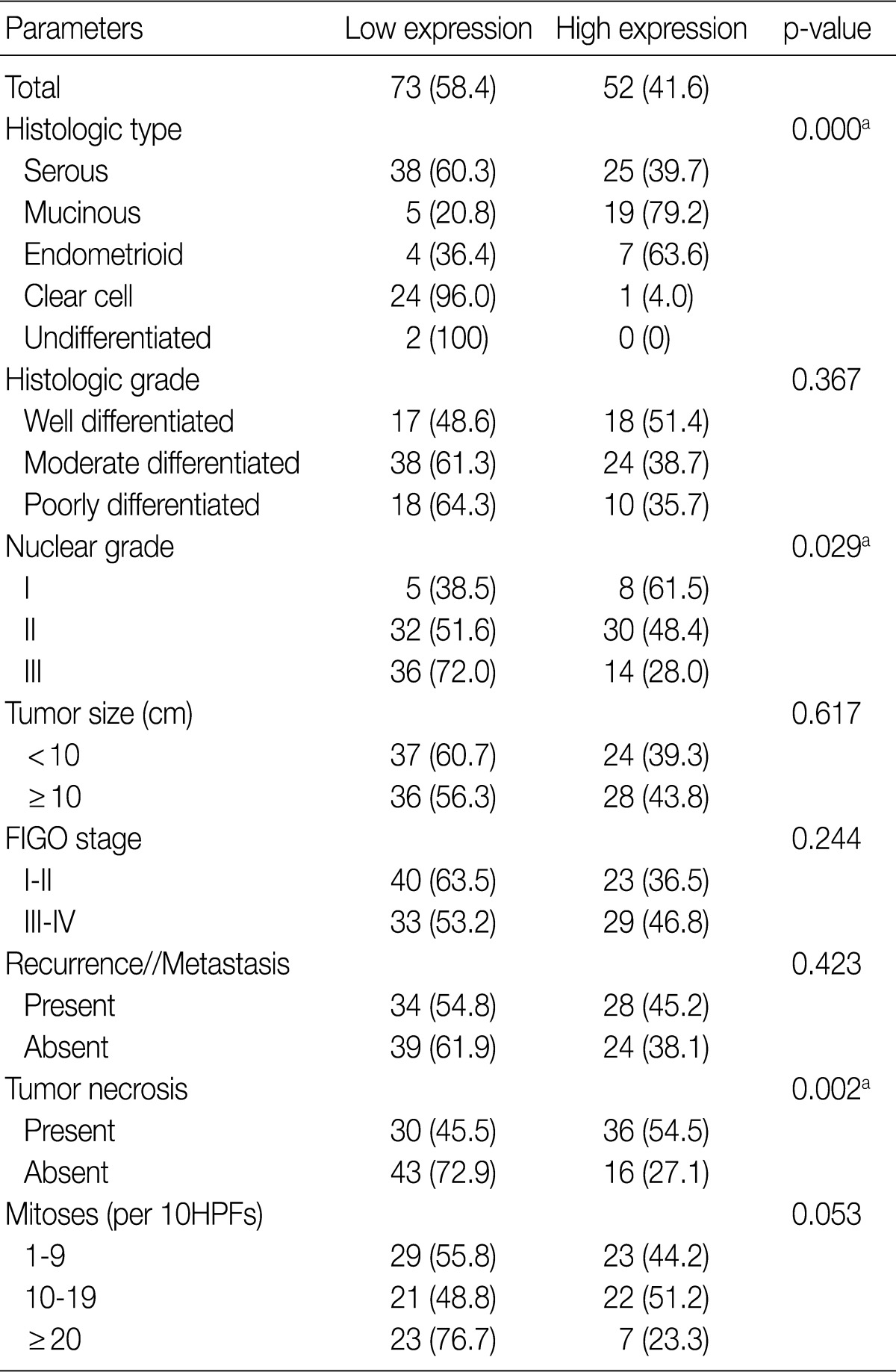
- Table 2 summarizes the association of CA IX expression in OECs with clinicopathological parameters. High CA IX expression was associated with the histological tumor type, nuclear grade, and tumor necrosis. Mucinous and endometrioid carcinomas exhibited higher CA IX expression levels more frequently than serous and clear cell carcinomas (p=0.000). The expression of CA IX was lower when the nuclear grade was higher (p=0.029). Table 3 summarizes the association of GLUT-1 expression in OECs with clinicopathological parameters. GLUT-1 showed a positive correlation with tumor necrosis and mitosis. Correlation of immunohistochemical results of VEGF with clinicopathological parameters is summarized in Table 4. VEGF expression showed a positive correlation only with disease recurrence or metastasis. No statistical significance was observed between VEGF expression and the histologic type, histologic grade, nuclear grade, tumor size, FIGO stage, tumor necrosis, and mitosis (p>0.05).
- Correlation between high expression of HRFs and OS
- Follow-up was available for 125 patients (median, 38 months; range, 1 to 140 months). Thirty patients (24.0% of a total of 125 patients) died from the disease during the follow-up period. In univariate analyses, there were no significant correlations between high expressions of CA IX, GLUT-1, and VEGF and patient survival. Statistical significance was not seen (p>0.05), but a trend towards shorter OS in the high-expression groups was demonstrated. With respect to CA IX expression, the high-expression group showed a lower 5-year survival rate (YSR) compared to the low-expression group (61.0% vs. 69.0%, p=0.16). In the case of GLUT-1 expression, the high-expression group showed a significantly lower 5-YSR compared to the low-expression group (30.0% vs. 73.0%, p=0.244). The 5-YSR of the high VEGF expression group was lower than that of the low VEGF expression group (60% vs. 83%, respectively, p=0.110). However, these differences were not statistically significant.
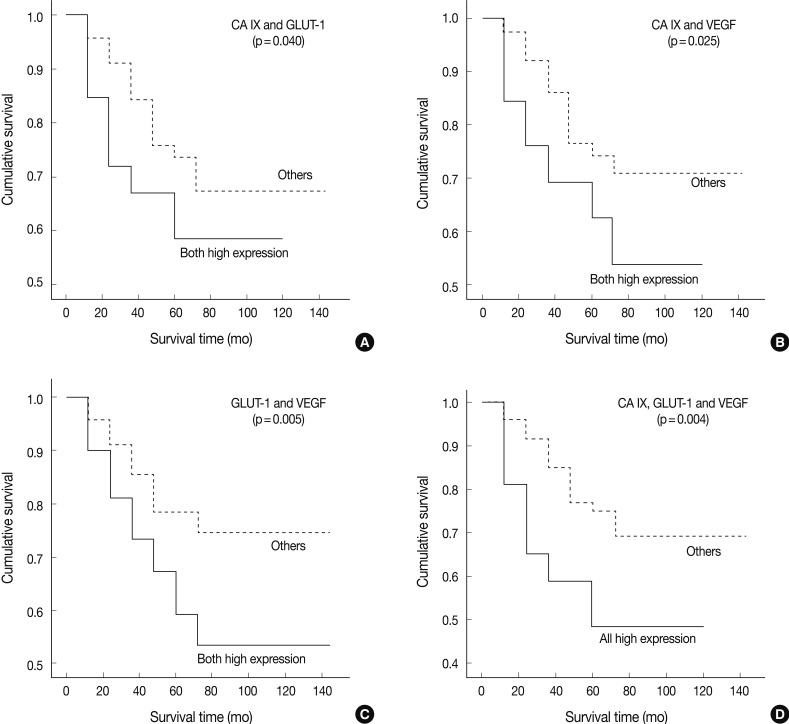
- We tried to explore whether the coexpression among these markers had any prognostic value. The high coexpression of CA IX/GLUT-1 was observed in 26 cases (20.8% of a total of 125 patients). The high coexpression of CA IX/GLUT-1 was significantly correlated with histologic type (p=0.012) and mitosis (p=0.019). Mucinous and endometrioid carcinomas were the most common type of cancers (37.5% vs. 36.4%, respectively). No correlation was observed between high CA IX/GLUT-1 coexpression and other clinical factors, such as tumor grade, nuclear grade, tumor size, FIGO stage, disease recurrence, and tumor necrosis. The high coexpression of CA IX/VEGF was seen in 40 patients (32% of total 125 patients). There was a significant correlation only between CA IX/VEGF coexpression and the histologic type (p=0.000). Similarly, a high coexpression of CA IX/VEGF was noted in mucinous and endometrioid carcinomas (62.5% vs. 45.5%, respectively). The high coexpression of CA IX/VEGF was associated with tumor stage (p=0.018), disease recurrence (p=0.018), tumor necrosis (p=0.008), and mitosis (p=0.021), but not with tumor grade, nuclear grade, or tumor size (p>0.05). There were 50 patients (40% of a total of 125 patients) who showed a high expression of both GLUT-1 and VEGF. In these cases, there were no correlations with any of the clinicopathological factors (p>0.05).
- In univariate survival analyses, using the Kaplan-Meier survival curves and log rank test, all three categories showed a significantly negative prognostic impact on OS (Fig. 4). The 5-YSR according to each coexpression pattern was as follows (high expression of two factors vs. the others): CA IX/GLUT-1, 59.0% vs. 67.0%; CA IX/VEGF, 54.0% vs. 71.0%; and GLUT-1/VEGF, 53.0% vs. 74.0%, respectively.
RESULTS
- Clinical investigations have clearly demonstrated that the prevalence of hypoxic tissue areas is a characteristic pathophysiological property of locally advanced solid tumors and a relevant factor of tumor pathophysiology. Tumor hypoxia has been found in a wide range of malignancies such as cancers of the breast, uterine cervix, vulva, head and neck, prostate, lung, and kidney.21,22 When tumors develop, they often become more malignant over time. Substantial data suggest that tumor hypoxia and the HIF-system are intensely involved in the processes conferring a growth advantage to tumor cells and the development of a more malignant phenotype.4 In addition, tumor hypoxia is classically associated with resistance to radiotherapy, and it has also been shown to decrease the efficacy of certain forms of chemotherapy. Evidence has accumulated showing that up to 50-60% of locally advanced solid tumors may exhibit hypoxic tissue areas that are heterogeneously distributed within the tumor mass. On this basis, there is convincing evidence of the clinical importance of tumor hypoxia.
- HIF-1 has been known to be a key gene for adapting cells to microenvironmental conditions via the up-regulation of the transcription response to hypoxia.23 Through the HIF-1-mediated pathway, various endogenous hypoxia markers used for the estimation of the oxygenation status of total OECs are activated; among them, CA IX, GLUT-1, and VEGF have been known to play an important role.24,25 Previous studies have indicated that HIF-1α and its target genes act as positive regulators of tumor growth in many solid tumors, including OECs.26 We tried to analyze the association between the prognosis and the overexpression of HRFs, including CA IX, GLUT-1, and VEGF in various histological types of OECs.
- Necrotic areas, most probably induced by hypoxia, were observed more frequently in tissue samples of poorly differentiated OECs, compared to those in tissue samples of well-differentiated OECs. The high expression of these markers was identified specifically in the perinecrotic zones that mark areas of avascularity and hypoxia and was correlated with tumor necrosis in this study. The distribution pattern of overexpression suggests that the oxygen level within the tumor regulates CA IX, GLUT-1, and VEGF expression in OECs.
- Several studies evaluated the correlation of HRFs with the histological subtypes.15,27 In these reports, the patients were grouped into histologic subtypes based on the most frequent immunoreactivity: mucinous and endometrioid carcinomas expressing CA IX, clear cell and serous carcinomas expressing GLUT-1, and clear cell and mucinous carcinomas expressing VEGF. Our results showed that CA IX was overexpressed in a substantial proportion of mucinous and endometrioid type OECs and GLUT-1 overexpression was observed more frequently in serous and clear cell type OECs compared to that in the other types of OECs. One of the reasons for this is that each of these subtypes is reportedly associated with different molecular pathogenetic events and different responses.15 Therefore, it may be useful to evaluate the patient's prognosis using a single, hypoxic marker according to the histologic subtype of OECs. This suggests a specific application of each marker, namely, CA IX in mucinous and endometrioid carcinomas, and GLUT-1 in clear cell carcinomas. However, we could not establish that CA IX, GLUT-1, and VEGF were each correlated with OS in this study.
- Some positive links have been drawn between the clinical outcome and the expression of these HRFs.15,16,27,28 GLUT-1 overexpression was demonstrated in OECs,15,16 and it was related to a shorter disease-free survival, while high VEGF expression predicted poor prognosis in OECs.28 Overexpression of CA IX was reported to be an unfavorable prognostic marker in endometrioid ovarian cancer in one study.27 In the present study, there were no significant associations between each of these markers and patients' outcomes. Even though the OS rate tended to decrease when each marker was highly expressed, it is questionable whether CA IX, GLUT-1, and VEGF can be used independently as prognostic markers in OECs. We also assessed the patients who were assigned to the coexpression groups according to the combination of the three markers. It was found that the survival rate of patients with combined high coexpression (of CA IX/GLUT-1, CA IX/VEGF, and GLUT-1/VEGF, respectively) was lower compared to those without. The high expression of all three markers had an increase detrimental effect on patient prognosis. Particularly, the coexpression of CA IX/GLUT-1 and CA IX/VEGF were related to shorter OS in mucinous and endometroid type OECs. CA IX/VEGF coexpression also showed a correlation with advanced tumor stage and tumor recurrence in mucinous and endometrioid type OECs. In the present study, there were no substantial, single predictors of HRF expression in OECs; instead, it appears that the coexpression of two markers may provide a better predictive value in OECs.
- In conclusion, the expressions of HRFs differed based on the histologic type of OECs and HRFs, including CA IX, GLUT-1, and VEGF, which are not always independent prognostic factors in OECs when they are used individually, but the high coexpression of HRFs may provide useful prognostic information and have a significantly negative impact on patient survival. Identifying two or three HRFs and inhibiting CA IX, GLUT-1, and VEGF may be a key strategy for an attractive anticancer therapy, comprising of the combination of two or three markers.
DISCUSSION
Acknowledgments
Acknowledgments
- 1. Siegel R, Ward E, Brawley O, Jemal A. Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin 2011; 61: 212-236. PubMed
- 2. Jung KW, Park S, Kong HJ, et al. Cancer statistics in Korea: incidence, mortality, survival, and prevalence in 2008. Cancer Res Treat 2011; 43: 1-11. ArticlePubMedPMC
- 3. Murdoch WJ, McDonnel AC. Roles of the ovarian surface epithelium in ovulation and carcinogenesis. Reproduction 2002; 123: 743-750. ArticlePubMed
- 4. Le QT, Denko NC, Giaccia AJ. Hypoxic gene expression and metastasis. Cancer Metastasis Rev 2004; 23: 293-310. ArticlePubMed
- 5. Ruan K, Song G, Ouyang G. Role of hypoxia in the hallmarks of human cancer. J Cell Biochem 2009; 107: 1053-1062. ArticlePubMed
- 6. Harrison L, Blackwell K. Hypoxia and anemia: factors in decreased sensitivity to radiation therapy and chemotherapy? Oncologist 2004; 9(Suppl 5): 31-40. ArticlePubMedPDF
- 7. Kaluz S, Kaluzová M, Liao SY, Lerman M, Stanbridge EJ. Transcriptional control of the tumor- and hypoxia-marker carbonic anhydrase 9: a one transcription factor (HIF-1) show? Biochim Biophys Acta 2009; 1795: 162-172. ArticlePubMedPMC
- 8. Behrooz A, Ismail-Beigi F. Dual control of glut1 glucose transporter gene expression by hypoxia and by inhibition of oxidative phosphorylation. J Biol Chem 1997; 272: 5555-5562. ArticlePubMed
- 9. Kim JW, Gao P, Dang CV. Effects of hypoxia on tumor metabolism. Cancer Metastasis Rev 2007; 26: 291-298. ArticlePubMedPDF
- 10. Greijer AE, van der Groep P, Kemming D, et al. Up-regulation of gene expression by hypoxia is mediated predominantly by hypoxia-inducible factor 1 (HIF-1). J Pathol 2005; 206: 291-304. ArticlePubMed
- 11. Semenza GL. HIF-1: mediator of physiological and pathophysiological responses to hypoxia. J Appl Physiol 2000; 88: 1474-1480. ArticlePubMed
- 12. Ivanov S, Liao SY, Ivanova A, et al. Expression of hypoxia-inducible cell-surface transmembrane carbonic anhydrases in human cancer. Am J Pathol 2001; 158: 905-919. ArticlePubMedPMC
- 13. Giatromanolaki A, Koukourakis MI, Sivridis E, et al. Expression of hypoxia-inducible carbonic anhydrase-9 relates to angiogenic pathways and independently to poor outcome in non-small cell lung cancer. Cancer Res 2001; 61: 7992-7998. PubMed
- 14. Loncaster JA, Harris AL, Davidson SE, et al. Carbonic anhydrase (CA IX) expression, a potential new intrinsic marker of hypoxia: correlations with tumor oxygen measurements and prognosis in locally advanced carcinoma of the cervix. Cancer Res 2001; 61: 6394-6399. PubMed
- 15. Yasuda M, Miyazawa M, Fujita M, et al. Expression of hypoxia inducible factor-1alpha (HIF-1alpha) and glucose transporter-1 (GLUT-1) in ovarian adenocarcinomas: difference in hypoxic status depending on histological character. Oncol Rep 2008; 19: 111-116. PubMed
- 16. Cantuaria G, Fagotti A, Ferrandina G, et al. GLUT-1 expression in ovarian carcinoma: association with survival and response to chemotherapy. Cancer 2001; 92: 1144-1150. ArticlePubMed
- 17. Ferrara N. The role of vascular endothelial growth factor in pathological angiogenesis. Breast Cancer Res Treat 1995; 36: 127-137. ArticlePubMedPDF
- 18. Shen GH, Ghazizadeh M, Kawanami O, et al. Prognostic significance of vascular endothelial growth factor expression in human ovarian carcinoma. Br J Cancer 2000; 83: 196-203. ArticlePubMedPMC
- 19. Silverberg SG. Histopathologic grading of ovarian carcinoma: a review and proposal. Int J Gynecol Pathol 2000; 19: 7-15. ArticlePubMed
- 20. Miyazawa M, Yasuda M, Fujita M, et al. Therapeutic strategy targeting the mTOR-HIF-1alpha-VEGF pathway in ovarian clear cell adenocarcinoma. Pathol Int 2009; 59: 19-27. PubMed
- 21. Giatromanolaki A, Harris AL. Tumour hypoxia, hypoxia signaling pathways and hypoxia inducible factor expression in human cancer. Anticancer Res 2001; 21: 4317-4324. PubMed
- 22. Semenza GL. Hypoxia, clonal selection, and the role of HIF-1 in tumor progression. Crit Rev Biochem Mol Biol 2000; 35: 71-103. ArticlePubMed
- 23. Semenza GL, Wang GL. A nuclear factor induced by hypoxia via de novo protein synthesis binds to the human erythropoietin gene enhancer at a site required for transcriptional activation. Mol Cell Biol 1992; 12: 5447-5454. ArticlePubMedPMCPDF
- 24. Airley RE, Mobasheri A. Hypoxic regulation of glucose transport, anaerobic metabolism and angiogenesis in cancer: novel pathways and targets for anticancer therapeutics. Chemotherapy 2007; 53: 233-256. ArticlePubMedPDF
- 25. Jubb AM, Pham TQ, Hanby AM, et al. Expression of vascular endothelial growth factor, hypoxia inducible factor 1alpha, and carbonic anhydrase IX in human tumours. J Clin Pathol 2004; 57: 504-512. ArticlePubMedPMC
- 26. Zhong H, De Marzo AM, Laughner E, et al. Overexpression of hypoxia-inducible factor 1alpha in common human cancers and their metastases. Cancer Res 1999; 59: 5830-5835. PubMed
- 27. Choschzick M, Oosterwijk E, Müller V, et al. Overexpression of carbonic anhydrase IX (CAIX) is an independent unfavorable prognostic marker in endometrioid ovarian cancer. Virchows Arch 2011; 459: 193-200. ArticlePubMedPDF
- 28. Duncan TJ, Al-Attar A, Rolland P, et al. Vascular endothelial growth factor expression in ovarian cancer: a model for targeted use of novel therapies? Clin Cancer Res 2008; 14: 3030-3035. ArticlePubMedPDF
REFERENCES
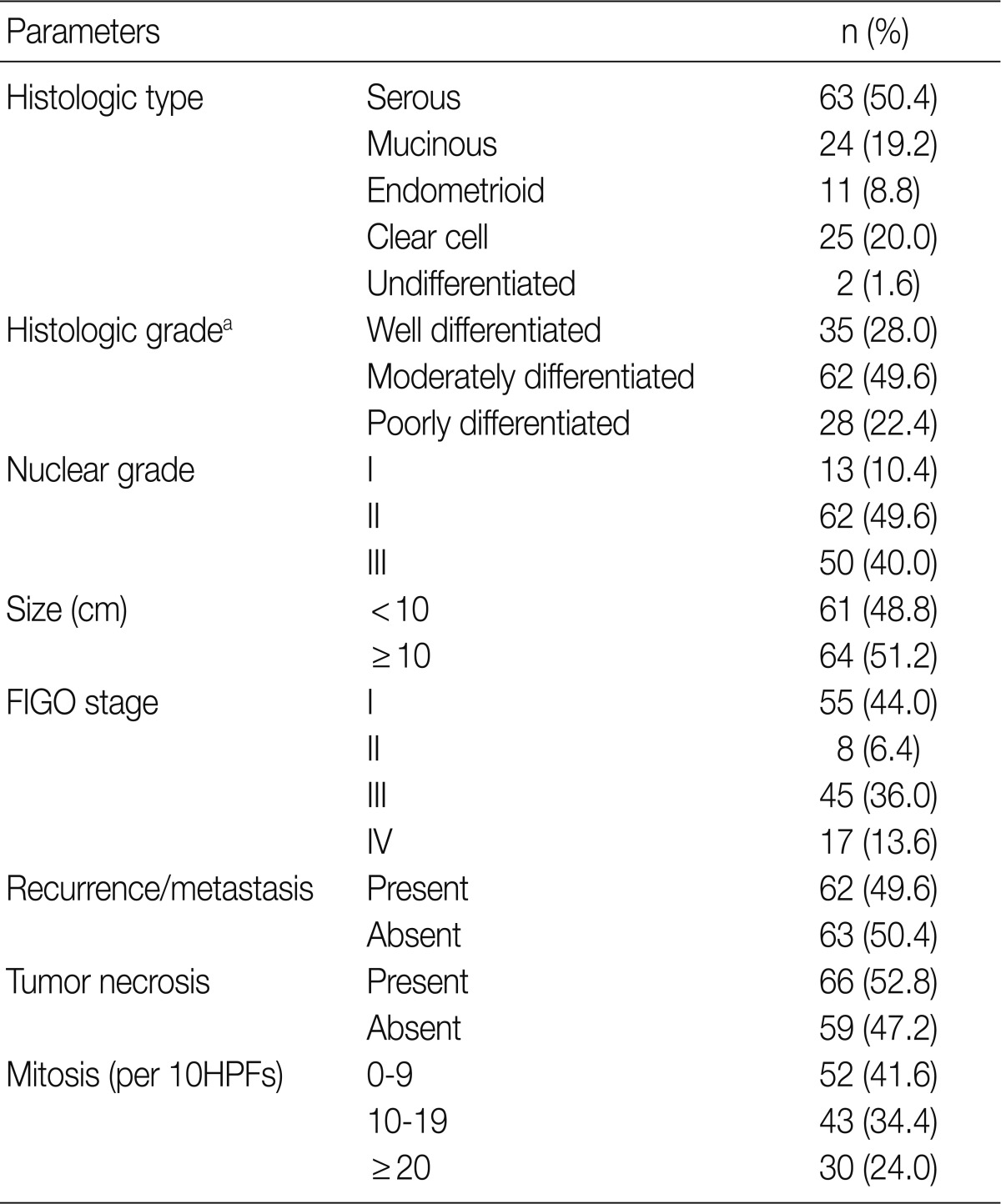
FIGO, Federation of Gynecology and Obstetrics; HPF, high power field.
aSilverberg tumor grade system.19
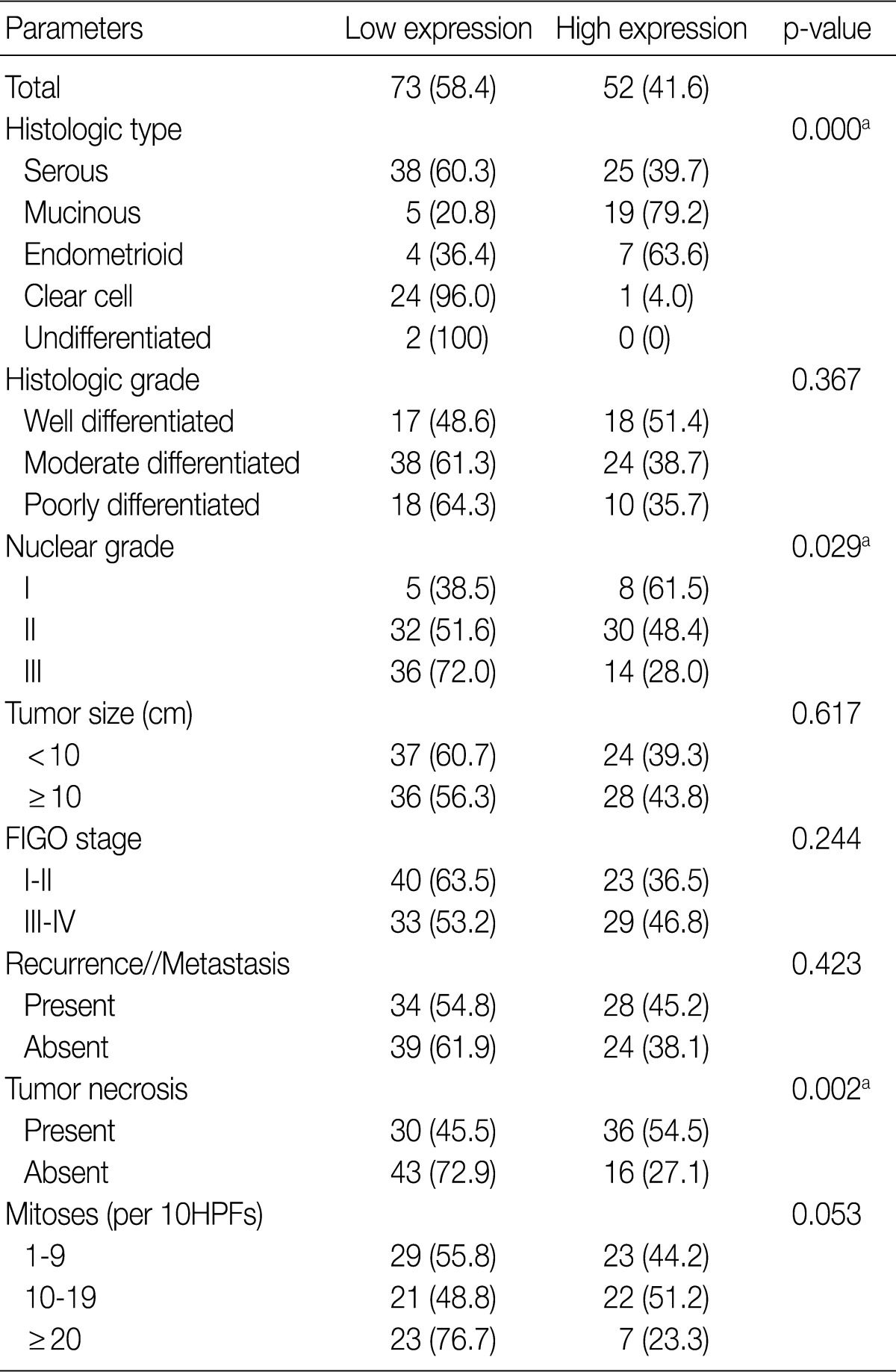
Values are presented as number (%).
Low expression, less than 10% positive staining of tumor cells; high expression, more than 10% positive staining of tumor cells.
CA IX, carbonic anhydrase IX; FIGO, Federation of Gynecology and Obstetrics; HPF, high power field.
aStatistically significance (Pearson's chi-squared test).
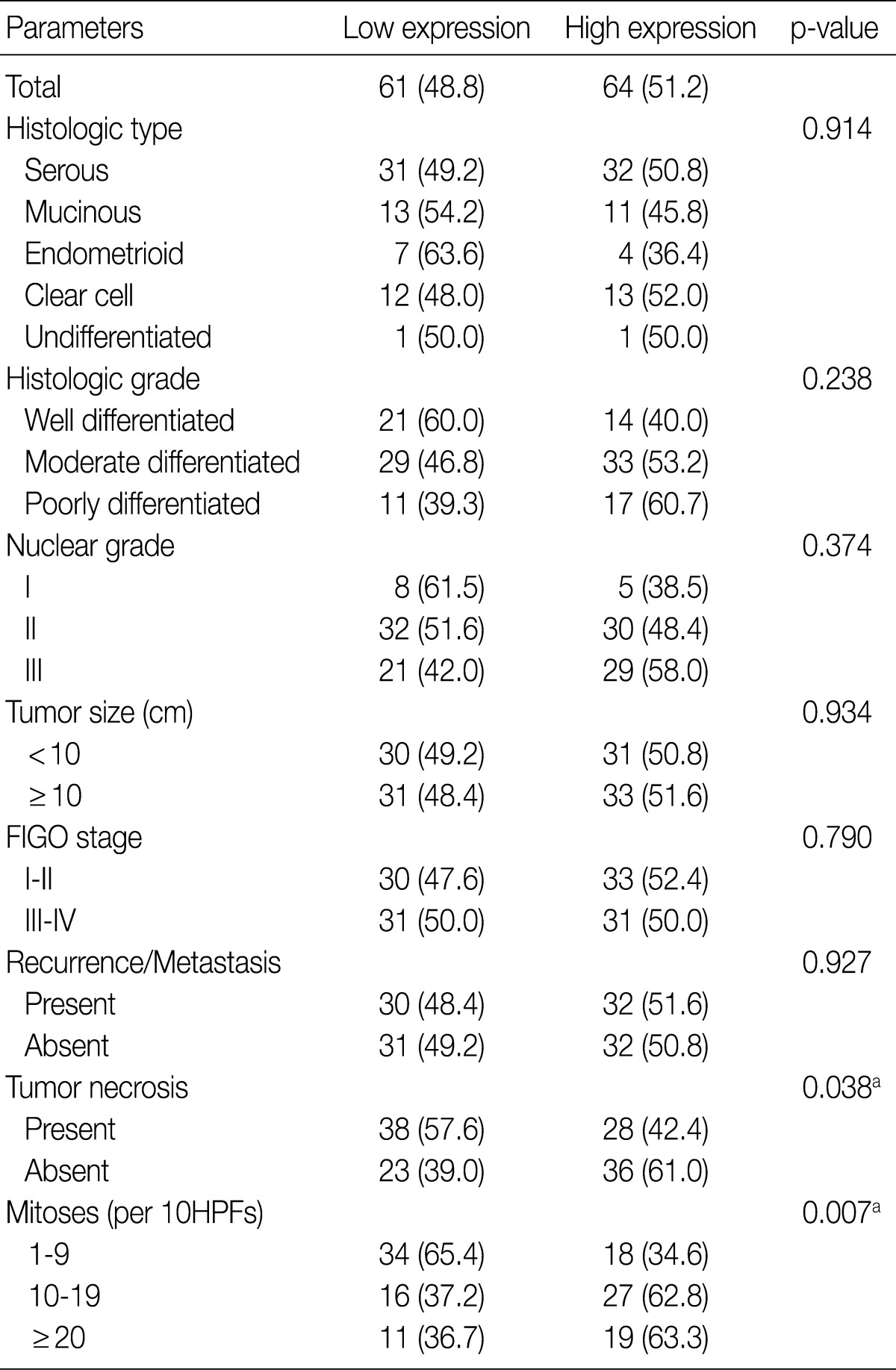
Values are presented as number (%).
Low expression, less than 10% positive staining of tumor cells; high expression, more than 10% positive staining of tumor cells.
GLUT-1, glucose transporter-1; FIGO, Federation of Gynecology and Obstetrics; HPF, high power field.
aStatistically significance (Pearson's chi-squared test).
Figure & Data
References
Citations

- Glucose-transporter 1 (GLUT1) as a prognostic biomarker: evidence from 14,966 human tumors across 134 cancer types
Seyma Büyücek, Katharina Möller, Florian Viehweger, Ria Schlichter, Anne Menz, Andreas M Luebke, Viktor Reiswich, Martina Kluth, Claudia Hube-Magg, Andrea Hinsch, Florian Lutz, Sören Weidemann, Frank Jacobsen, Maximilian Lennartz, David Dum, Christian Ber
BMC Cancer.2026;[Epub] CrossRef - Role of Carbonic Anhydrase IX Expression in Triple-negative Breast Cancer
Hemali B Kotadiya, Nupur A Patel, Hemangini H Vora
Gujarat Cancer Society Research Journal.2026; 27(2): 31. CrossRef - Evaluation of the effect of Endogenous and Exogenous Antioxidants on Hypoxia Induced Breast Adenocarcinoma Cell Invasion and Migration:
In vitro
Subarnarekha Maitra, Poulami Sen, Dibya Sinha, Tathagata Roy
Research Journal of Pharmacy and Technology.2025; : 3153. CrossRef - Effect of Ultrasound Therapy at the ST11 on Sympathetic Nervous System Change: A Prospective Randomized Controlled Study
Shinwoo Kang, Dongho Keum
Journal of Korean Medicine Rehabilitation.2023; 33(4): 167. CrossRef - A systematic review verified by bioinformatic analysis based on TCGA reveals week prognosis power of CAIX in renal cancer
Zikuan Zhang, Bo Wu, Yuan Shao, Yongquan Chen, Dongwen Wang, Lucia Magnelli
PLOS ONE.2022; 17(12): e0278556. CrossRef - Effect of Acupuncture at the Field of the Auricular Branch of the Vagus Nerve on Autonomic Nervous System Change
Sunjoo An, Dongho Keum
Journal of Korean Medicine Rehabilitation.2021; 31(2): 81. CrossRef - Responses of Autonomic Nervous System and Gastrointestinal Function to Acupuncture at Abdominal Anterior Cutaneous Nerve : A Pilot Study
Seohyun Park, Hojun Kim, Dongho Keum
Journal of Korean Medicine.2019; 40(1): 99. CrossRef - Omega-3 polyunsaturated fatty acid docosahexaenoic acid and its role in exhaustive-exercise-induced changes in female rat ovulatory cycle
Abeer F. Mostafa, Shereen M. Samir, R.M. Nagib
Canadian Journal of Physiology and Pharmacology.2018; 96(4): 395. CrossRef - Clear cell carcinomas of the ovary and kidney: clarity through genomics
Jennifer X Ji, Yi Kan Wang, Dawn R Cochrane, David G Huntsman
The Journal of Pathology.2018; 244(5): 550. CrossRef - Prognostic Significance of Carbonic Anhydrase IX Expression in Cancer Patients: A Meta-Analysis
Simon J. A. van Kuijk, Ala Yaromina, Ruud Houben, Raymon Niemans, Philippe Lambin, Ludwig J. Dubois
Frontiers in Oncology.2016;[Epub] CrossRef - Review of Research Topics on Abdominal Examination
Jihye Kim, Jeong Hwan Park, Keun Ho Kim
Journal of Korean Medicine.2016; 37(3): 1. CrossRef - Expression of hypoxic markers and their prognostic significance in soft tissue sarcoma
JEUNG IL KIM, KYUNG UN CHOI, IN SOOK LEE, YOUNG JIN CHOI, WON TACK KIM, DONG HOON SHIN, KYUNGBIN KIM, JEONG HEE LEE, JEE YEON KIM, MEE YOUNG SOL
Oncology Letters.2015; 9(4): 1699. CrossRef - Evaluation of a Hypoxia Regulated Gene Panel in Ovarian Cancer
Amanda F. Baker, Scott W. Malm, Ritu Pandey, Cindy Laughren, Haiyan Cui, Denise Roe, Setsuko K. Chambers
Cancer Microenvironment.2015; 8(1): 45. CrossRef - Sulforaphane reduces molecular response to hypoxia in ovarian tumor cells independently of their resistance to chemotherapy
MICHAL PASTOREK, VERONIKA SIMKO, MARTINA TAKACOVA, MONIKA BARATHOVA, MARIA BARTOSOVA, LUBA HUNAKOVA, OLGA SEDLAKOVA, SONA HUDECOVA, OLGA KRIZANOVA, FRANCK DEQUIEDT, SILVIA PASTOREKOVA, JAN SEDLAK
International Journal of Oncology.2015; 47(1): 51. CrossRef - Overexpression of Glucose Transporter-1 (GLUT-1) Predicts Poor Prognosis in Epithelial Ovarian Cancer
Hanbyoul Cho, You Sun Lee, Julie Kim, Joon-Yong Chung, Jae-Hoon Kim
Cancer Investigation.2013; 31(9): 607. CrossRef - Towards Lipidomics of Low-Abundant Species for Exploring Tumor Heterogeneity Guided by High-Resolution Mass Spectrometry Imaging
Jonathan Cimino, David Calligaris, Johann Far, Delphine Debois, Silvia Blacher, Nor Sounni, Agnès Noel, Edwin De Pauw
International Journal of Molecular Sciences.2013; 14(12): 24560. CrossRef

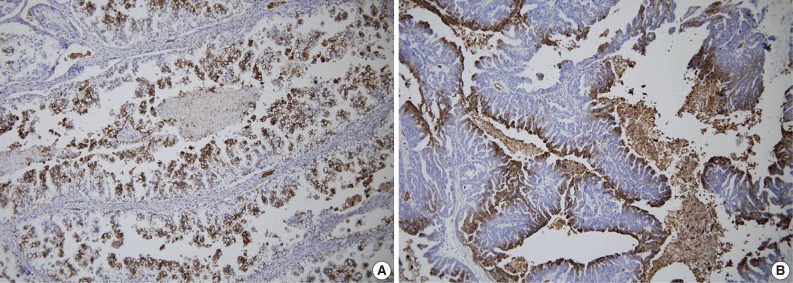
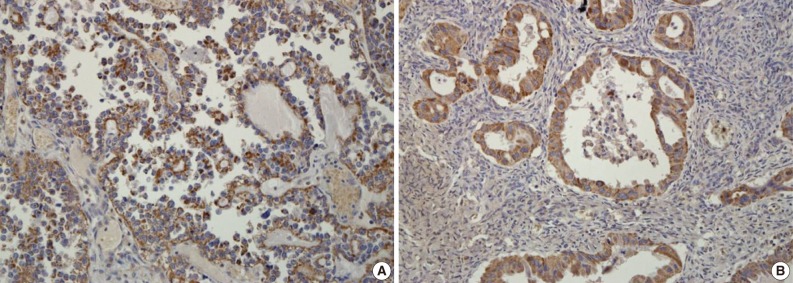
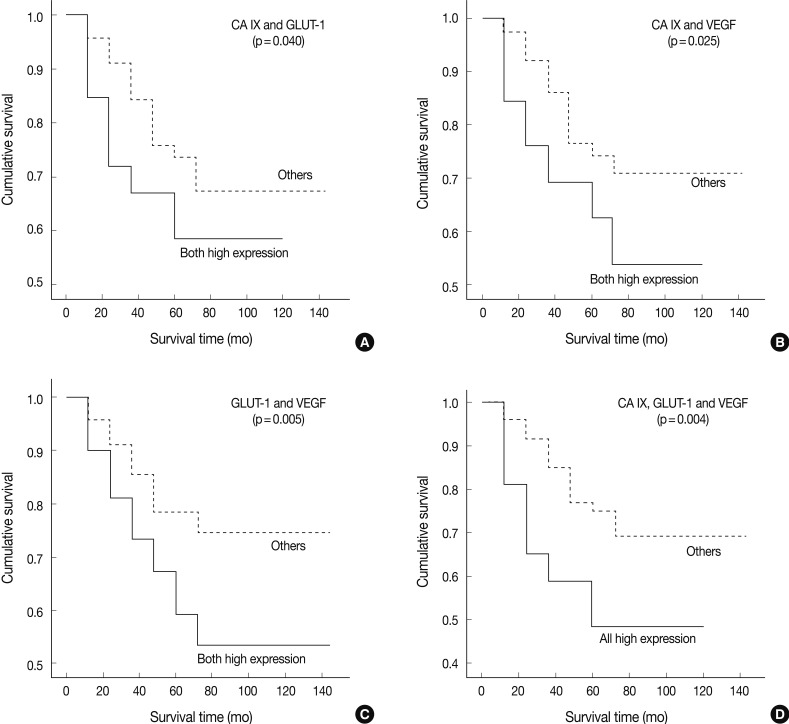
Fig. 1
Fig. 2
Fig. 3
Fig. 4
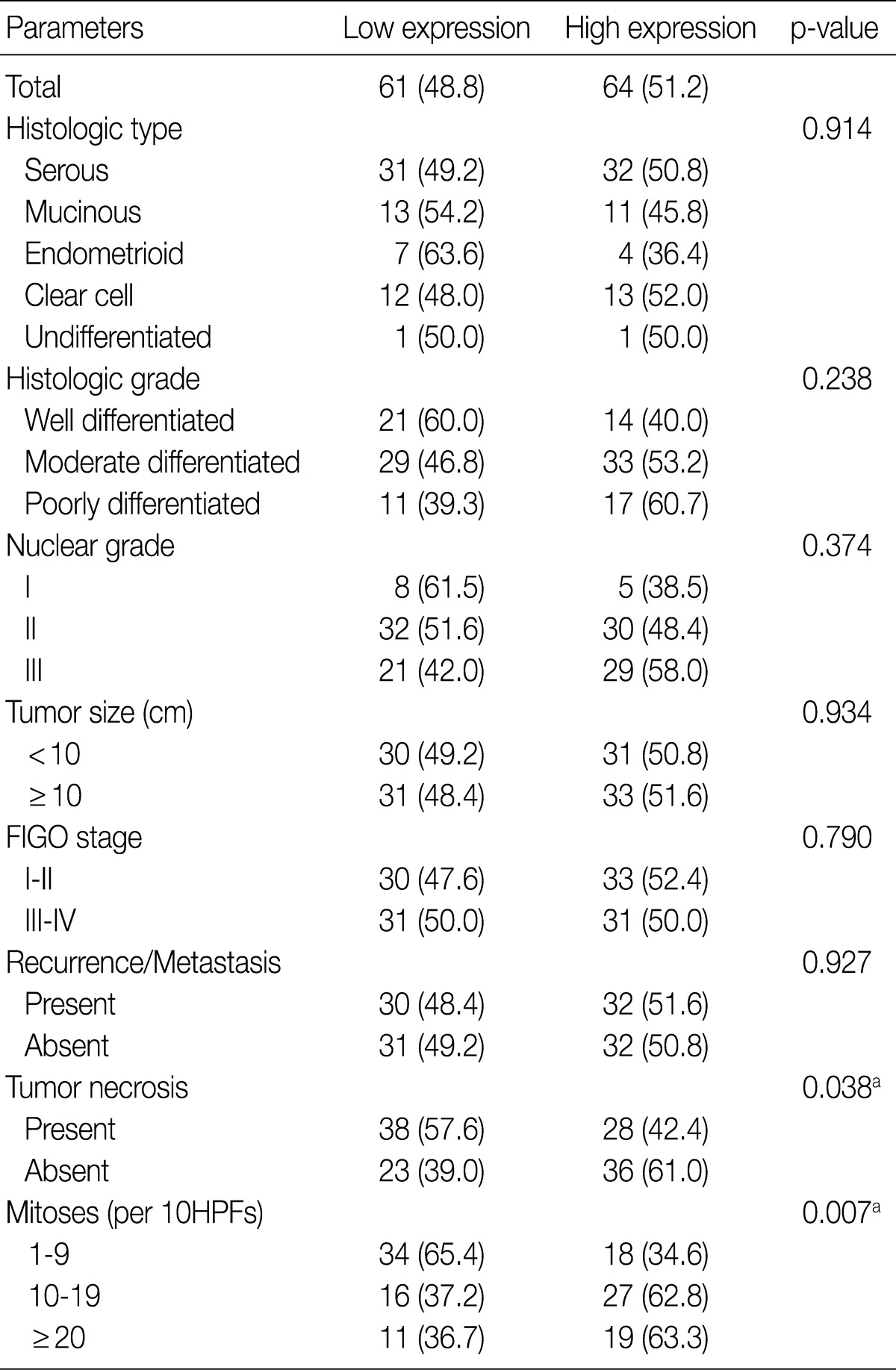
FIGO, Federation of Gynecology and Obstetrics; HPF, high power field. aSilverberg tumor grade system.
Values are presented as number (%). Low expression, less than 10% positive staining of tumor cells; high expression, more than 10% positive staining of tumor cells. CA IX, carbonic anhydrase IX; FIGO, Federation of Gynecology and Obstetrics; HPF, high power field. aStatistically significance (Pearson's chi-squared test).
Values are presented as number (%). Low expression, less than 10% positive staining of tumor cells; high expression, more than 10% positive staining of tumor cells. GLUT-1, glucose transporter-1; FIGO, Federation of Gynecology and Obstetrics; HPF, high power field. aStatistically significance (Pearson's chi-squared test).
Low expression, less than 10% positive staining of tumor cells; high expression, more than 10% positive staining of tumor cells. VEGF, vascular endothelial growth factor; FIGO, Federation of Gynecology and Obstetrics; HPF, high power field. aStatistically significance (Pearson's chi-squared test).

 E-submission
E-submission
 PubReader
PubReader Cite this Article
Cite this Article





