Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 46(5); 2012 > Article
-
Review
Pathologic Review of Cystic and Cavitary Lung Diseases - Na Rae Kim, Joungho Han1
-
Korean Journal of Pathology 2012;46(5):407-414.
DOI: https://doi.org/10.4132/KoreanJPathol.2012.46.5.407
Published online: October 25, 2012
Department of Pathology, Gachon University Gil Medical Center, Incheon, Korea.
1Department of Pathology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- Corresponding Author: Joungho Han, M.D. Department of Pathology, Samsung Medical Center, Sungkyunkwan University School of Medicine, 81 Irwon-ro, Gangnam-gu, Seoul 135-710, Korea. Tel: +82-2-3410-2765, Fax: +82-2-3410-6393, hanjho@skku.edu
© 2012 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- Pulmonary cystic and cavitary lesions caused by diverse etiologies are commonly encountered in chest imaging. The terms "cyst" and "cavity" are used to describe air-filled regions in the center of a nodule or consolidation of the lung. To date, only radiologic aspects of these lesions have been addressed. The morphologies of pulmonary cystic and cavitary lesions exhibit a broad spectrum, ranging from benign to malignant pulmonary diseases of acquired or congenital origin, including variable infectious diseases. In this review, we summarized the differential diagnosis of pathological entities to provide pathologists and radiologists with an overview of the diseases most commonly associated with pulmonary cystic and cavitary lesions in adults and children. The results showed slightly different patterns in the distribution of the diseases in the two groups. The most common causes of cavitary lesions include malignancy and infection in adults, and congenital malformation in children. Therefore, identification of pathologic entities correlating with the radiologic findings, clinical course, and location of the lesion is important in the evaluation of cystic and cavitary lung lesions in order to avoid unnecessary surgical procedures or delayed treatment.
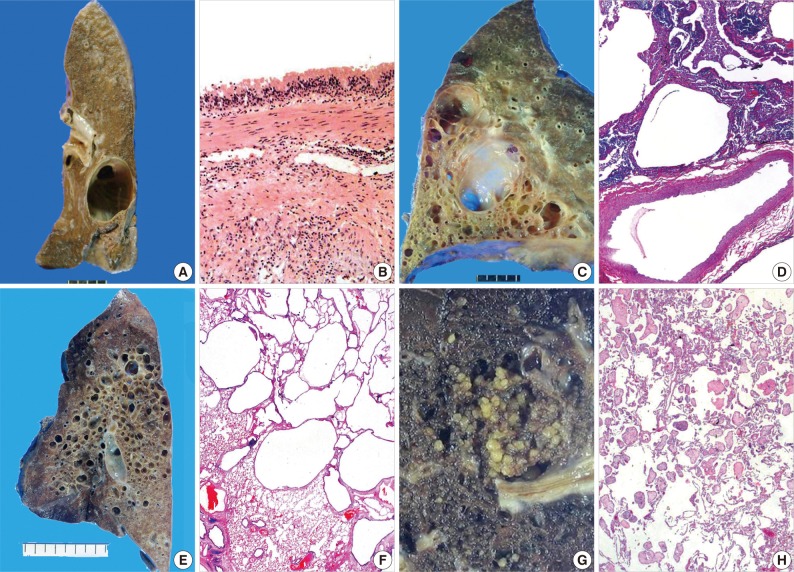
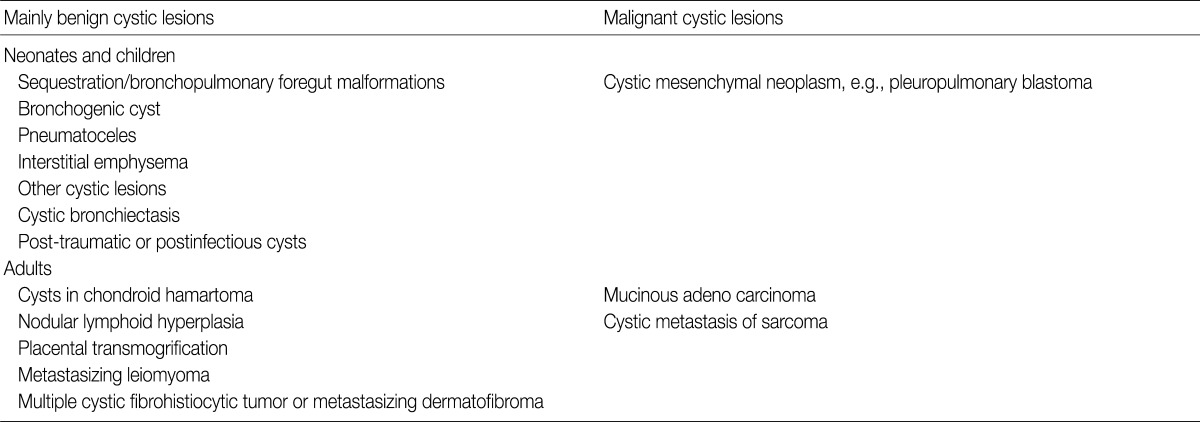
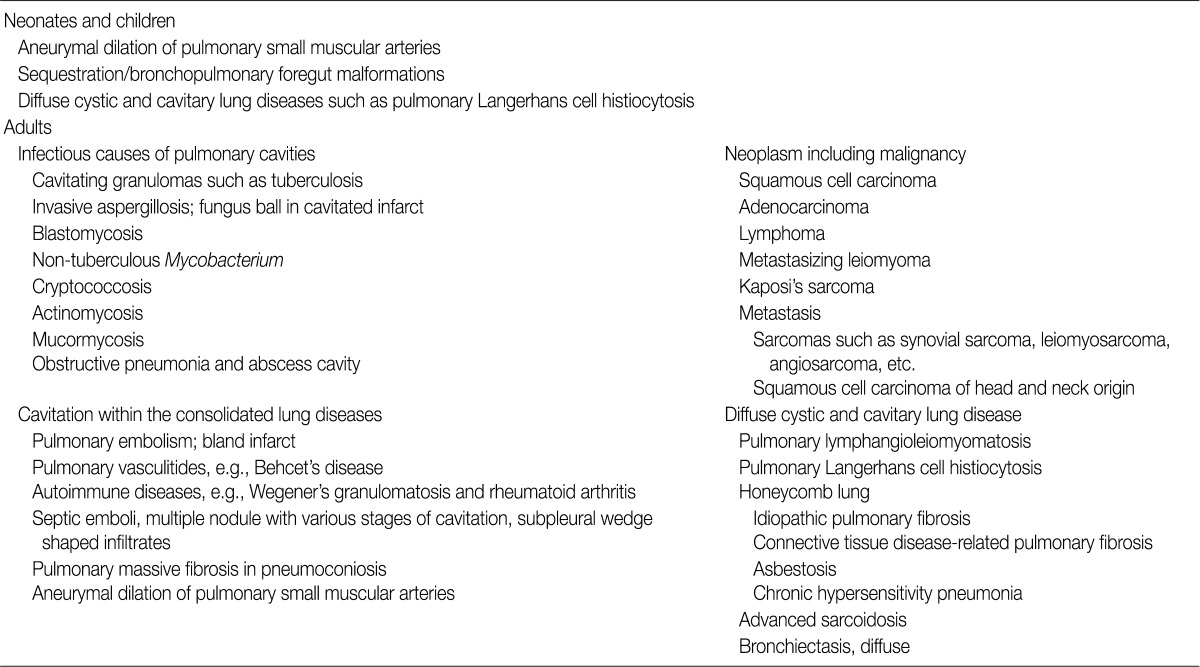
- In children, the most common cause of cystic pulmonary lesions is congenital, such as bronchopulmonary foregut malformation.10-12 This condition includes bronchogenic cysts, pulmonary sequestration, congenital pulmonary airway malformation (CPAM), congenital lobar emphysema or congenital lobar over-inflation, bronchial atresia, and congenital pulmonary cysts. Cystic and cavitary lesions produced by bronchogenic cysts are lined with pseudostratified respiratory epithelium containing mucous glands, cartilage, and smooth muscle, and are filled with clear or proteinaceous material (Fig. 1A, B).13 Pulmonary sequestration of the intralobar (80%) and extralobar (20%) types exhibits dilated bronchioles containing a mucous plug, which is the same pattern as bronchiolitis obliterans organizing pneumonia (Fig. 1C, D). In addition, fibrosis is evident in peribronchiolar parenchyma and in systemic arteries with adjacent bronchi. CPAM is composed of a solid and cystic mass of pulmonary tissue, and is subclassified into types 0 to 4. The lesions are lined by flattened type 1 and 2 pneumocytes or pseudostratified columnar epithelium, with or without underlying skeletal muscle or cartilage (Fig. 1E, F).10,14 In particular, CPAM types 0 and 4 appear as large cystic pulmonary lesions.11 Emphysema is characterized by permanently enlarged airspaces distal to terminal bronchioles with destruction of alveolar walls, which radiologically appears as a focal, low attenuated area usually without visible walls. In the case of panacinar emphysema (i.e., panlobular emphysema, associated with alpha-1-antitrypsin deficiency), decreased attenuation causes a more diffuse lesion. Histology showing bronchial atresia and deficiency of cartilage in the involved lobe suggests developmental emphysema rather than an acquired one.14 Lymphoid interstitial pneumonia (LIP) is a rare disease with a pathology of diffuse pulmonary lymphohistiocytic proliferation and characterized by predominant interstitial involvement sparing blood vessels and airways as well as relative preservation of the pulmonary architecture.15 LIP is the most common form of classically described interstitial pneumonia in children and is most often associated with underlying autoimmune disease and immunodeficiency status.16 Thin-walled perivascular cysts may be found in the advanced stage of LIP.17 Peripheral cyst of pulmonary maldevelopment in Down syndrome consists of rare intrathoracic cystic lesions in neonates and children.10 Neoplasms, such as pleuropulmonary blastoma or post-traumatic or post-infectious cysts,1 are also rare causes of cystic lesions in children, and aneurysmal dilation of pulmonary small arteries is one of the congenital causes of cavitary pulmonary lesions in neonates and children.1,18
- The causes of cystic and cavitary pulmonary lesions differ in adults in whom a simple cystic pathology is most common.3 In some cases, CPAM may be a rare congenital malformation diagnosed during adulthood in patients that usually present with recurrent respiratory infections and thin-walled multi-septated cystic lesions.14 However, these congenital malformations are not prevalent in adults. Other causes of focal or multifocal cystic lung lesions are: bullae (or blebs), pneumatocele, penetrating or closed chest trauma, LIP, lung lesions associated with Down syndrome, Ehlers-Danlos syndrome, follicular bronchiolitis, or metastatic diseases including metastasizing leiomyoma, in rare cases.1,2,13,14 Placental transmogrification is a rarely encountered cystic lesion in adults, and is a rare histologic subtype of bullous emphysema that has no connection with the cystic lesion and bronchial tree (Fig. 1G, H). Diverse histological patterns, such as myxoid villous structures mimicking placental chorionic villi with large, thin-walled emphysematous bullae, may be interpreted as a cystic lesion upon radiological examination.19 Examples of these cystic lesions are listed in Table 1.
CYSTIC PULMONARY LESIONS
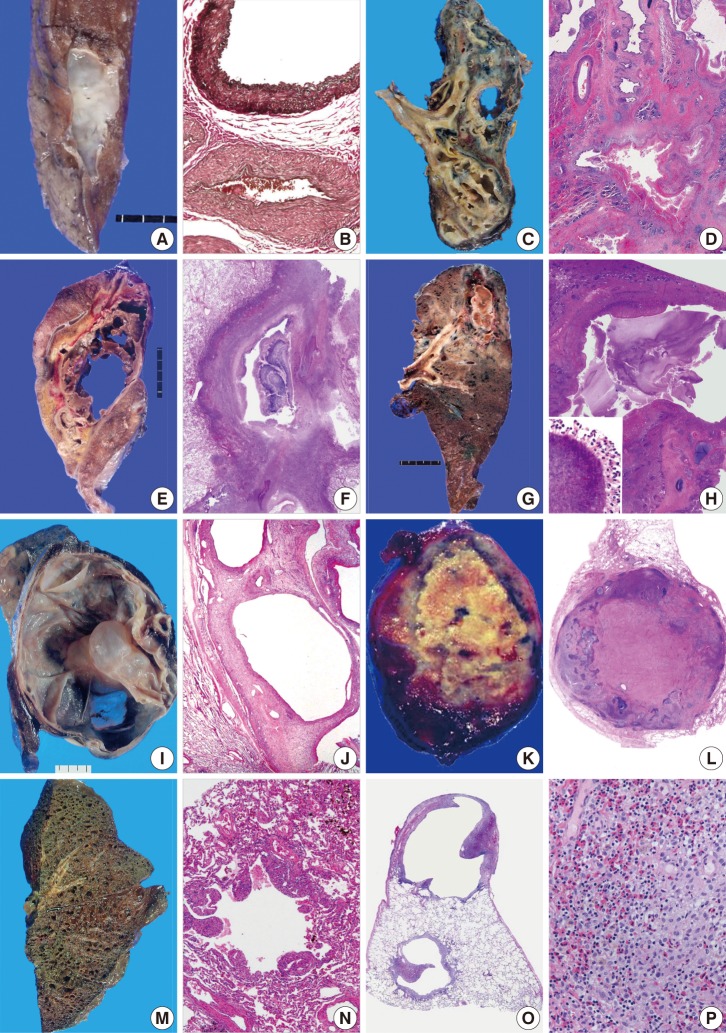
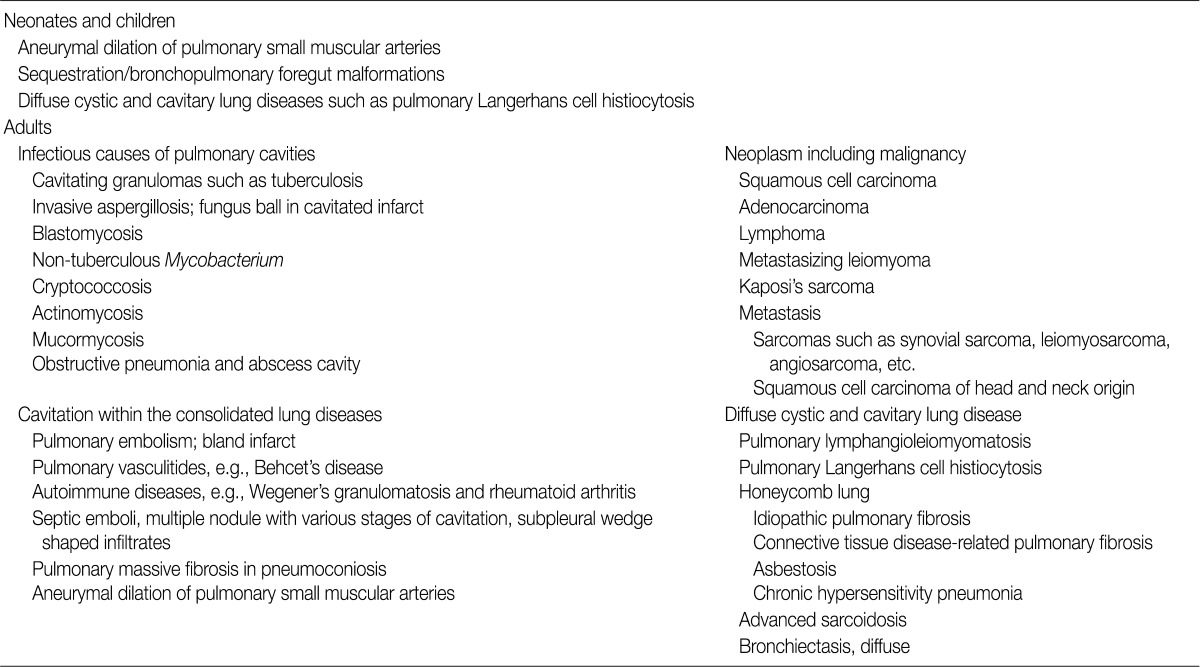
- Cavity-forming pulmonary lesions are uncommon in the absence of a concomitant underlying disease because cavities are pathologically formed by necrotic tissue expelled by an underlying lesion.2 Different mechanisms have been proposed to explain necrotizing pneumonia, lung abscess formation, and pulmonary septic embolic processes.20 In septic emboli transferred from other infectious foci, septic occlusion of small, peripheral, pulmonary arterial branches results in cavitary pulmonary lesions.21 Aneurysmal dilation of pulmonary small muscular arteries in arteriovenous malformation (Fig. 2A, B) may elicit different pathogenetic causes of cavitary lesions in neonates.1,18
- In adults, the two main causes of cavitary pulmonary lesions include malignancy and infection. The thickness of the thickest portion of cavity wall, the characteristics of its inner lining (irregular or smooth), and its nature and location are important for determining the benignity of cavitary pulmonary lesions by radiology.1-3,22 For example, cavity-forming carcinomas can have a smooth or irregular inner contour, and cavities associated with an abscess are shaggy. Cystic or cavitary lesions with a maximal wall thickness of 1 mm based on radiologic findings are generally regarded as benign, those with a maximal wall thickness of >1 to <4 mm are usually regarded as benign, and lesions with maximal wall thicknesses of 5 to 15 mm are equally divided between benignity and malignancy. For lesions with a maximal wall thickness >15 mm, 95% of them are likely to be malignant, although it should be noted that there is an overlap in wall thickness between malignant and benign lesions, such as in aspergilloma.23,24 In addition to these factors, epidemiologic differences also exist. Tuberculosis is the predominant infectious cause of cystic and cavitary pulmonary lesions in Korea, which predominantly occurs in patients less than 50 years of age. Large cavities in the upper lobe are significantly associated with mycobacterial infection,25 but the location is of limited help in the differential diagnosis of most cavitary lesions (Fig. 2C, D). Invasive aspergillosis is a common infection cause of cavitary pulmonary lesions (Fig. 2E, F),1,2,24 and gram-positive and gram-negative bacterial pneumonia, such as Streptococcus pneumoniae or Haemophilus influenza, can cause the cavitations, although community-acquired pneumonia is not typically accompanied with cavity formation in the lung.1,2,25,26 A lung abscess is a common bacterial cause of cavitary pulmonary lesions. Pulmonary actinomycosis (Fig. 2G, H) is a rare disease and is commonly misdiagnosed as other chronic suppurative cavitary lung diseases.4 Together with infectious etiologies, the incidence of pulmonary malignancy has increased as another important cause of cavitary lesions that pathologists and radiologists commonly encounter; 161,920 incident cases were reported in Korea in 2009,27 and 221,130 incident cases in the United States in 2011.28 Cavitation has been noted in 7-20% of primary lung cancers that have thick cavity walls.2,29 Cavitation may be ascribed to treatment-related necrosis, internal cyst formation, or internal desquamation of tumor cells and subsequent liquefaction. The process of cavitation in lung tumors has been associated with a poor prognosis,30 and more than 80% of cavity-forming pulmonary tumors associated with epidermal growth factor receptor overexpression exhibit rapid growth.31 Non-small cell carcinoma, especially squamous cell carcinoma, is the most common histologic type of cavity forming tumor, whereas small cell carcinoma has never been reported to cavitate.32 Rarely, cavitation or multiloculated cystic changes have been reported in lung adenocarcinoma.33 The most common origin of cavitating pulmonary metastases is squamous cell carcinoma of the head and neck,34 while the second most common form is metastatic adenocarcinoma. Hematologic or mesenchymal malignancies occur less frequently as cystic and cavitary pulmonary lesions, including metastasizing leiomyoma (Fig. 2I, J), leiomyosarcoma, synovial sarcoma, epithelioid sarcoma, endometrial stromal sarcoma, malignant lymphoma, angiosarcoma, and Kaposi's sarcoma.1,35-38 In particular, immunocompromised patients, such as human immunodeficiency virus (HIV)-infected or post-transplant recipients, have more diverse cavity-forming etiologies than do immunocompetent individuals. Kaposi's sarcoma, which has a histology of anastomosing vascular channels, appears as peribronchovascular interstitial thickening. Pulmonary non-Hodgkin's lymphoma is rare, and may manifest as cavitating nodules in immunocompromised patients.39 In one study of HIV-infected patients with primary pulmonary lymphoma, 5 of 12 (41.7%) patients presented with a pulmonary cavitary lesion.40 Primary pulmonary lymphoma is a rare entity usually formed of B-cells, and is usually low-grade lymphoma composed of mucosal-bronchial-associated lymphoid tissue.41 High-grade primary pulmonary lymphomas often occur in immunodeficient patients, which mostly appear as multiple nodular masses with cavitation.42 Lymphomatoid granulomatosis associated with Epstein-Barr virus infection frequently presents with pulmonary cavities, and rarely, bronchioloalveolar carcinoma manifests as cystic or cavitary disease.2,43 Among infections, Pneumocystis carinii infection is a common problem in HIV-infected individuals and infrequently manifests as a cavitary infiltrate.44 Mycobacterium, histoplasmosis, and cryptococcal pneumonitis are uncommon causes of cavitary infections, and viral infections caused by cytomegalovirus or influenza viruses can appear as cavitary lung lesions at a delayed stage in these immunocompromised groups.45
CAVITARY PULMONARY LESIONS
- Various uncommon non-infectious etiology of cavitary lesions is pulmonary embolism, especially in adults.2 Traumatic pulmonary pseudocysts, formed after blunt thoracic trauma that causes tearing or laceration of the lung parenchyma, are also a rare cause of cavitary pulmonary lesions,8 but obtaining the history of the patient usually facilitates an early diagnosis. Systemic diseases, such as connective tissue disease, Behcet's disease, and systemic autoimmune vasculitis such as Wegener's granulomatosis, which affects arteries and veins of all sizes, can involve lung parenchyma, the pulmonary artery, or large vessels (Fig. 2K, L).10,46 Pulmonary Behcet's disease can present as hemorrhage, infarction, or multifocal cavitary lesions. Cystic and cavitary lesions are rare in sarcoidosis, and therefore can be confusing in the manifestations of sarcoidosis, especially in active and severe sarcoidosis.47 Both lymphangioleiomyomatosis (LAM) and Langerhans cell histiocytosis (LCH) involve all lobes of both lungs, and therefore appear as diffuse cystic and cavitary lesions, though there is radiologic preponderance of the lower lung. Pulmonary LCH and LAM predominantly affect adults.48 Pathologically, LAM is characterized by peribronchial, perivascular, and perilymphatic proliferation of myoid cells resulting in vascular and airway obstructions as well as cyst formation (Fig. 2M, N). Pulmonary LCH may appear as cystic with nodules, and Erdheim-Chester disease (ECD) shares pathologic features, such as xanthogranulomatous inflammation with foamy macrophages (Fig. 2O, P), and radiologically can present as symmetric, diffuse, interstitial lung disease with interlobular septal thickening.49 Pulmonary involvement of LCH is much common in adults, but occurs in nearly half of young children with multisystem LCH. LCH can be differentiated from ECD based on an immunohistochemical profile of CD68+, Langerin-, CD1a-, and S-100 protein+/-. Bronchocentric granulomatosis may be present in the nodule, which attests to the likelihood of an infectious etiology.50 Examples of these lesions are listed in Table 2.
OTHER UNCOMMON CYSTIC OR CAVITARY PULMONARY LESIONS
- In summary, we have reviewed diverse etiologies of pathologic entities showing cystic and cavitary pulmonary lesions in adults and children. The distribution of the diseases showed a slightly different pattern in the two groups. Identification of pathologic findings that correlate with radiologic findings, clinical progression, and location is important in the evaluation of cystic and cavitary lung lesions in order to avoid unnecessary procedure or delayed treatment.
CONCLUSION
- 1. Ryu JH, Swensen SJ. Cystic and cavitary lung diseases: focal and diffuse. Mayo Clin Proc 2003; 78: 744-752. ArticlePubMed
- 2. Gadkowski LB, Stout JE. Cavitary pulmonary disease. Clin Microbiol Rev 2008; 21: 305-333. ArticlePubMedPMCPDF
- 3. Papagiannopoulos K, Hughes S, Nicholson AG, Goldstraw P. Cystic lung lesions in the pediatric and adult population: surgical experience at the Brompton Hospital. Ann Thorac Surg 2002; 73: 1594-1598. ArticlePubMed
- 4. Mabeza GF, Macfarlane J. Pulmonary actinomycosis. Eur Respir J 2003; 21: 545-551. ArticlePubMed
- 5. Kim TS, Koh WJ, Han J, et al. Hypothesis on the evolution of cavitary lesions in nontuberculous mycobacterial pulmonary infection: thin-section CT and histopathologic correlation. AJR Am J Roentgenol 2005; 184: 1247-1252. ArticlePubMed
- 6. Seaman DM, Meyer CA, Gilman MD, McCormack FX. Diffuse cystic lung disease at high-resolution CT. AJR Am J Roentgenol 2011; 196: 1305-1311. ArticlePubMed
- 7. Castañer E, Alguersuari A, Gallardo X, et al. When to suspect pulmonary vasculitis: radiologic and clinical clues. Radiographics 2010; 30: 33-53. ArticlePubMed
- 8. Athanassiadi K, Gerazounis M, Kalantzi N, Kazakidis P, Fakou A, Kourousis D. Primary traumatic pulmonary pseudocysts: a rare entity. Eur J Cardiothorac Surg 2003; 23: 43-45. ArticlePubMed
- 9. Vourtsi A, Gouliamos A, Moulopoulos L, et al. CT appearance of solitary and multiple cystic and cavitary lung lesions. Eur Radiol 2001; 11: 612-622. ArticlePubMedPDF
- 10. Jones KD, Colby TV. In: Leslie KO, Wick MR, eds. Congenital and developmental diseases. Practical pulmonary pathology: a diagnostic approach. 2005; Philadelphia: Churchill Livingston, 51-70.
- 11. Stocker JT. In: Dali DH, Hammer SP, eds. Congenital and developmental diseases. Pulmonary pathology. 1994; 2nd ed. New York: Springer-Verlag, 155-190. Article
- 12. Austin JH, Müller NL, Friedman PJ, et al. Glossary of terms for CT of the lungs: recommendations of the Nomenclature Committee of the Fleischner Society. Radiology 1996; 200: 327-331. ArticlePubMed
- 13. Aktoğu S, Yuncu G, Halilçolar H, Ermete S, Buduneli T. Bronchogenic cysts: clinicopathological presentation and treatment. Eur Respir J 1996; 9: 2017-2021. ArticlePubMed
- 14. Zylak CJ, Eyler WR, Spizarny DL, Stone CH. Developmental lung anomalies in the adult: radiologic-pathologic correlation. Radiographics 2002; 22 Spec No: S25-S43. Article
- 15. American Thoracic Society. European Respiratory Society. American Thoracic Society/European Respiratory Society International Multidisciplinary Consensus Classification of the Idiopathic Interstitial Pneumonias. This joint statement of the American Thoracic Society (ATS), and the European Respiratory Society (ERS) was adopted by the ATS board of directors, June 2001 and by the ERS Executive Committee, June 2001. Am J Respir Crit Care Med 2002; 165: 277-304. ArticlePubMed
- 16. Joseph E, Rubin G, Murray J. Lymphocytic interstitial pneumonitis (pneumonia) in HIV-positive adults. S Afr J Radiol 2005; 9: 14-16. ArticlePDF
- 17. Cha SI, Fessler MB, Cool CD, Schwarz MI, Brown KK. Lymphoid interstitial pneumonia: clinical features, associations and prognosis. Eur Respir J 2006; 28: 364-369. ArticlePubMed
- 18. Castañer E, Gallardo X, Rimola J, et al. Congenital and acquired pulmonary artery anomalies in the adult: radiologic overview. Radiographics 2006; 26: 349-371. ArticlePubMed
- 19. Santana AN, Canzian M, Stelmach R, Jatene FB. Placental transmogrification of the lung presenting as giant bullae with soft-fatty components. Eur J Cardiothorac Surg 2008; 33: 124-126. ArticlePubMed
- 20. Kiwamoto T, Endo T, Sekizawa K. A case of septic pulmonary embolism induced by urinary tract infection. Nihon Kokyuki Gakkai Zasshi 2004; 42: 89-93. PubMed
- 21. Huang RM, Naidich DP, Lubat E, Schinella R, Garay SM, McCauley DI. Septic pulmonary emboli: CT-radiographic correlation. AJR Am J Roentgenol 1989; 153: 41-45. ArticlePubMed
- 22. Honda O, Tsubamoto M, Inoue A, et al. Pulmonary cavitary nodules on computed tomography: differentiation of malignancy and benignancy. J Comput Assist Tomogr 2007; 31: 943-949. PubMed
- 23. Woodring JH, Fried AM, Chuang VP. Solitary cavities of the lung: diagnostic implications of cavity wall thickness. AJR Am J Roentgenol 1980; 135: 1269-1271. ArticlePubMed
- 24. Park Y, Kim TS, Yi CA, Cho EY, Kim H, Choi YS. Pulmonary cavitary mass containing a mural nodule: differential diagnosis between intracavitary aspergilloma and cavitating lung cancer on contrast-enhanced computed tomography. Clin Radiol 2007; 62: 227-232. ArticlePubMed
- 25. Yang YW, Kang YA, Lee SH, et al. Aetiologies and predictors of pulmonary cavities in South Korea. Int J Tuberc Lung Dis 2007; 11: 457-462. PubMed
- 26. Robbins AW, Arora NS. Pulmonary cavitation caused by Haemophilus influenzae in adults. South Med J 1981; 74: 225-227. ArticlePubMed
- 27. Ministry for Health and Welfare. 2009 Annual report of national cancer registration and statistics in Korea. 2011; Seoul: Ministry for Health and Welfare.
- 28. Siegel R, Ward E, Brawley O, Jemal A. Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin 2011; 61: 212-236. PubMed
- 29. Mouroux J, Padovani B, Elkaïm D, Richelme H. Should cavitated bronchopulmonary cancers be considered a separate entity? Ann Thorac Surg 1996; 61: 530-532. ArticlePubMed
- 30. Kolodziejski LS, Dyczek S, Duda K, Góralczyk J, Wysocki WM, Lobaziewicz W. Cavitated tumor as a clinical subentity in squamous cell lung cancer patients. Neoplasma 2003; 50: 66-73. PubMed
- 31. Onn A, Choe DH, Herbst RS, et al. Tumor cavitation in stage I non-small cell lung cancer: epidermal growth factor receptor expression and prediction of poor outcome. Radiology 2005; 237: 342-347. ArticlePubMed
- 32. Gasinska A, Kolodziejski L, Niemiec J, Dyczek S. Clinical significance of biological differences between cavitated and solid form of squamous cell lung cancer. Lung Cancer 2005; 49: 171-179. ArticlePubMed
- 33. Yoshida T, Harada T, Fuke S, et al. Lung adenocarcinoma presenting with enlarged and multiloculated cystic lesions over 2 years. Respir Care 2004; 49: 1522-1524. ArticlePubMedPDF
- 34. Miura H, Taira O, Hiraguri S, Hagiwara M, Kato H. Cavitating adenocarcinoma of the lung. Ann Thorac Cardiovasc Surg 1998; 4: 154-158. PubMed
- 35. Murphey MD, Gibson MS, Jennings BT, Crespo-Rodriguez AM, Fanburg-Smith J, Gajewski DA. From the archives of the AFIP: Imaging of synovial sarcoma with radiologic-pathologic correlation. Radiographics 2006; 26: 1543-1565. ArticlePubMed
- 36. Lillo-Gil R, Albrechtsson U, Jakobsson B. Pulmonary leiomyosarcoma appearing as a cyst: report of one case and review of the literature. Thorac Cardiovasc Surg 1985; 33: 250-252. ArticlePubMed
- 37. Pérez-Montiel D, Salmeron AA, Domínguez Malagon H. Multicystic endometrial stromal sarcoma. Ann Diagn Pathol 2004; 8: 213-218. ArticlePubMed
- 38. Weissferdt A, Moran CA. Primary vascular tumors of the lungs: a review. Ann Diagn Pathol 2010; 14: 296-308. ArticlePubMed
- 39. Olsen SR, Bhutani M. Multiple cavitating nodules in a renal transplant recipient. Can Respir J 2009; 16: 195-197. ArticlePubMedPMCPDF
- 40. Gallant JE, Ko AH. Cavitary pulmonary lesions in patients infected with human immunodeficiency virus. Clin Infect Dis 1996; 22: 671-682. ArticlePubMed
- 41. Martínez Rivera C, Bonnin Vilaplana M, Simón Adiego C, Palacín Forgué A, Puig Zuza J, Sampablo Lauro I. Primary pulmonary lymphoma presenting as a pulmonary mass with cavitation. Arch Bronconeumol 2004; 40: 94-96. ArticlePubMed
- 42. Corti M, Villafañe MF, Trione N, Schtirbu R, Narbaitz M. Primary pulmonary AIDS-related lymphoma. Rev Inst Med Trop Sao Paulo 2005; 47: 231-234. ArticlePubMed
- 43. McCloskey M, Catherwood M, McManus D, Todd G, Cuthbert R, Riley M. A case of lymphomatoid granulomatosis masquerading as a lung abscess. Thorax 2004; 59: 818-819. ArticlePubMedPMC
- 44. Ferré C, Báguena F, Podzamczer D, et al. Lung cavitation associated with Pneumocystis carinii infection in the acquired immunodeficiency syndrome: a report of six cases and review of the literature. Eur Respir J 1994; 7: 134-139. ArticlePubMed
- 45. Lin CY, Sun HY, Chen MY, et al. Aetiology of cavitary lung lesions in patients with HIV infection. HIV Med 2009; 10: 191-198. ArticlePubMed
- 46. Yoon SH, Son JW, Joung CI, Choi EG. A case of Behcet's disease with multiple cavitary lung lesion. Tuberc Respir Dis 2006; 61: 65-69. Article
- 47. Haramati LB, Lee G, Singh A, Molina PL, White CS. Newly diagnosed pulmonary sarcoidosis in HIV-infected patients. Radiology 2001; 218: 242-246. ArticlePubMed
- 48. Kitaichi M, Nishimura K, Itoh H, Izumi T. Pulmonary lymphangioleiomyomatosis: a report of 46 patients including a clinicopathologic study of prognostic factors. Am J Respir Crit Care Med 1995; 151(2 Pt 1): 527-533. ArticlePubMed
- 49. Protopapadakis C, Antoniou KM, Nicholson AG, et al. Erdheim-Chester disease: pulmonary presentation in a case with advanced systemic involvement. Respiration 2009; 77: 337-340. ArticlePubMedPDF
- 50. Ward S, Heyneman LE, Flint JD, Leung AN, Kazerooni EA, Müller NL. Bronchocentric granulomatosis: computed tomographic findings in five patients. Clin Radiol 2000; 55: 296-300. ArticlePubMed
REFERENCES
Modified from Ryu JH, Swensen SJ.1 Cystic and cavitary lung diseases: focal and diffuse. Mayo Clin Proc 2003; 78: 744-52.
Modified from Ryu JH, Swensen SJ.1 Cystic and cavitary lung diseases: focal and diffuse. Mayo Clin Proc 2003; 78: 744-52.
Figure & Data
References
Citations

- Cavitary pulmonary metastasis from renal pelvic urothelial carcinoma with pathological evidence of bronchiolar obstruction supporting a check-valve mechanism: a case report
Kei Nakano, Tomoki Nakagawa, Haruka Kishi, Shota Fujino, Takashi Ishihara, Masaya Ohara, Kazuhiro Matsuo, Tomoki Higeta, Kie Maita, Hirohito Kobayashi, Masatoshi Yamada, Ryota Masuda
Respiratory Medicine Case Reports.2026; 62: 102431. CrossRef - Managing Cavitary Coccidioidomycosis Expert Opinions for Improving Patient Outcomes
Fariba M. Donovan, George R. Thompson, Janis E. Blair, Royce H. Johnson, Josh Malo, Waseem Albasha, Stephanie G. Worrell, Staci E. Beamer, Kavitha Yaddanapudi, John N. Galgiani, Neil M. Ampel
CHEST.2025; 167(5): 1311. CrossRef - Causes and outcome of pulmonary cysts in pediatric age group among Egyptian patients, case-series study
Erini Fawzy, Mai El-Mahdy, Nevine Elhelaly, Iman Abdulaziz, Muhammad Hegazy, Amr Mustafa
Egyptian Pediatric Association Gazette.2025;[Epub] CrossRef - Weakly-Supervised Segmentation-Based Quantitative Characterization of Pulmonary Cavity Lesions in CT Scans
Wenyu Xing, Yanping Yang, Yanan Zhou, Tao Jiang, Yifang Li, Yuanlin Song, Dongni Hou, Dean TA
IEEE Journal of Translational Engineering in Health and Medicine.2024; 12: 457. CrossRef - Chemotherapy-induced cavitating Wilms' tumor pulmonary metastasis: Active disease or scarring? A case report and literature review
Angelo Zarfati, Cristina Martucci, Alessandro Crocoli, Annalisa Serra, Giorgio Persano, Alessandro Inserra
Frontiers in Pediatrics.2023;[Epub] CrossRef - High-Resolution Computed Tomography of Cystic Lung Disease
Joanna E. Kusmirek, Cristopher A. Meyer
Seminars in Respiratory and Critical Care Medicine.2022; 43(06): 792. CrossRef - Miliary tuberculosis in a paediatric patient with psoriasis
Jacob Kilgore, Jonathon Pelletier, Bradford Becken, Stephen Kenny, Samrat Das, Lisa Parnell
BMJ Case Reports.2021; 14(3): e237580. CrossRef - Atypical pulmonary metastases in children: the spectrum of radiologic findings
Michal Scolnik, Luda Glozman, Ronen Bar-Yoseph, Michal Gur, Yazeed Toukan, Lea Bentur, Anat Ilivitzki
Pediatric Radiology.2021; 51(10): 1907. CrossRef - Radiographic and CT appearance of cavitary pulmonary lesions in a lamb
J Kan, J Bauquier, D Tyrrell, K O'Byrne, AW Stent, B Brosnan
Australian Veterinary Journal.2021; 99(12): 529. CrossRef - Community-acquired Achromobacter xylosoxidans infection presenting as a cavitary lung disease in an immunocompetent patient
Chan Hee Hwang, Woo Jin Kim, Hye Young Jwa, Sung Heon Song
Yeungnam University Journal of Medicine.2020; 37(1): 54. CrossRef - Clinical Research of Pulmonary Langerhans Cell Histiocytosis in Children
Dong Wang, Lei Cui, Zhi-Gang Li, Li Zhang, Hong-Yun Lian, Hong-Hao Ma, Yun-Ze Zhao, Xiao-Xi Zhao, Tian-You Wang, Rui Zhang
Chinese Medical Journal.2018; 131(15): 1793. CrossRef - Benign features of infection‐related tumor‐like lesions of the lung: A retrospective imaging review study
Chun‐Chao Huang, Sho‐Ting Hung, Wei‐Chin Chang, Chin‐Yin Sheu
Journal of Medical Imaging and Radiation Oncology.2017; 61(4): 481. CrossRef - Cavitary lung disease in renal transplant recipients: A single center experience
Gizem Kumru, Serkan Akturk, Siyar Erdogmus, Aysegul Gursoy Coruh, Acar Tuzuner, Sule Sengul, Kenan Keven
Transplantation Reports.2017; 2(4): 19. CrossRef - Solitary lung cavities: CT findings in malignant and non-malignant disease
C.S. Nin, V.V.S. de Souza, G.R.T. Alves, R.H. do Amaral, K.L. Irion, E. Marchiori, B. Hochhegger
Clinical Radiology.2016; 71(11): 1132. CrossRef - Radial endobronchial ultrasound with a guide sheath for diagnosis of peripheral cavitary lung lesions: a retrospective study
Manabu Hayama, Norio Okamoto, Hidekazu Suzuki, Motohiro Tamiya, Takayuki Shiroyama, Ayako Tanaka, Takuji Nishida, Takashi Nishihara, Nobuko Uehara, Naoko Morishita, Kunimitsu Kawahara, Tomonori Hirashima
BMC Pulmonary Medicine.2016;[Epub] CrossRef - An infant with a hyperlucent chest mass: An unexpected diagnosis
Zarmina Ehsan, Jaimie D. Nathan, Carolyn M. Kercsmar
Pediatric Pulmonology.2015; 50(12): E52. CrossRef - The Pseudocavitation Sign of Lung Adenocarcinoma
Tina D. Tailor, Rodney A. Schmidt, Keith D. Eaton, Douglas E. Wood, Sudhakar N. J. Pipavath
Journal of Thoracic Imaging.2015; 30(5): 308. CrossRef - A Case of Pulmonary Artery Sarcoma Presented as Cavitary Pulmonary Lesions
Daniel Min, Ji-Hyun Lee, Hye-Cheol Jeong, Jung-Hyun Kim, Suk-Pyo Shin, Hong-Min Kim, Kyu Hyun Han, Hye Yun Jeong, Eun-Kyung Kim
Tuberculosis and Respiratory Diseases.2014; 76(3): 136. CrossRef
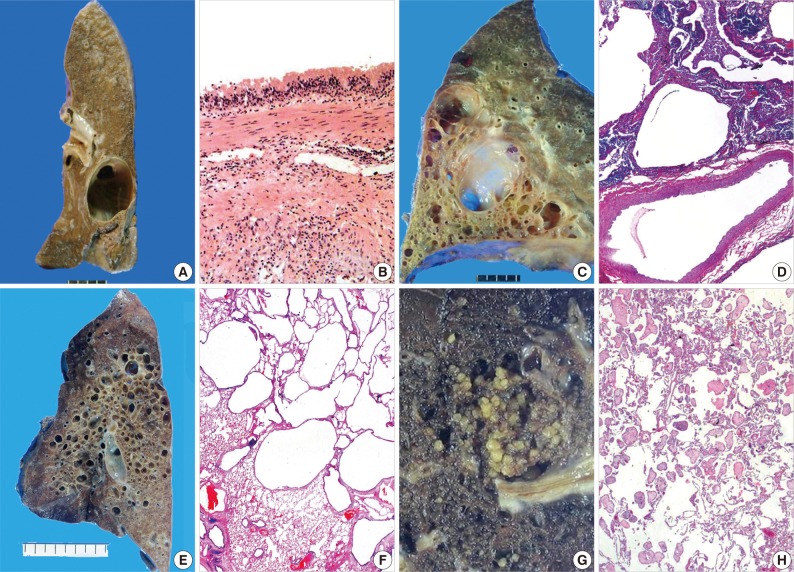
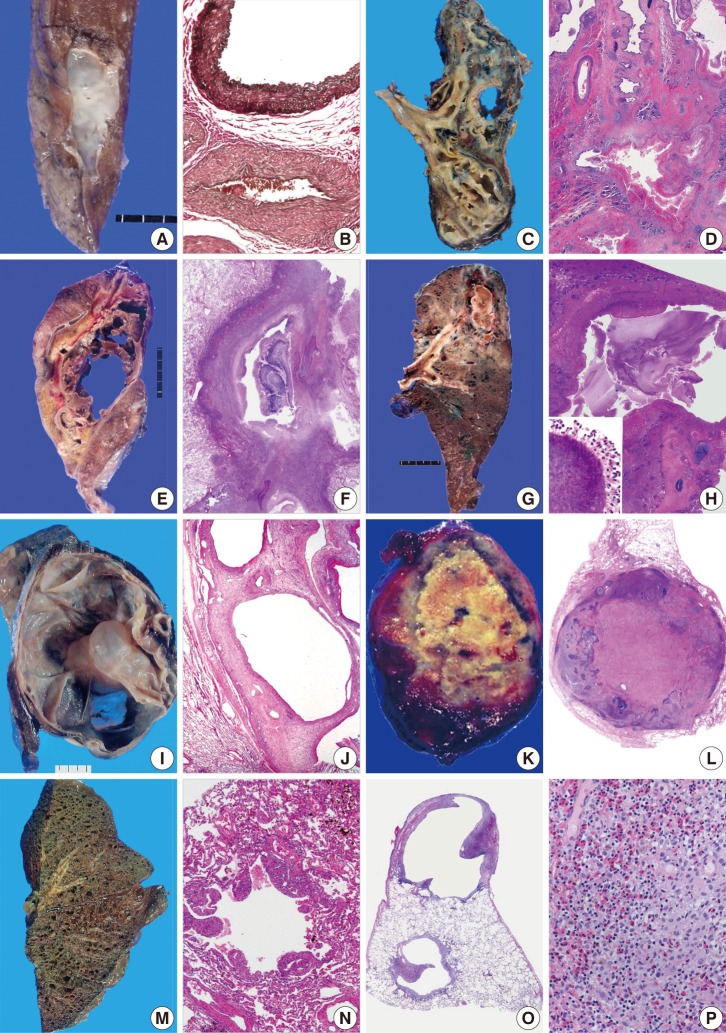
Fig. 1
Fig. 2

Modified from Ryu JH, Swensen SJ.
Modified from Ryu JH, Swensen SJ.

 E-submission
E-submission
 PubReader
PubReader Cite this Article
Cite this Article



