Articles
- Page Path
- HOME > J Pathol Transl Med > Forthcoming articles > Article
-
Original Article
Aquaporin 1 promotes proliferation and migration of tumor by up-regulating claudin-1 expression in colon cancer -
Wei Wei Xie,*
 , Lin Xu,*
, Lin Xu,* , Qian Li
, Qian Li , Dao Quan Zhang
, Dao Quan Zhang , Yu Bao Zhou
, Yu Bao Zhou
-
DOI: https://doi.org/10.4132/jptm.2026.01.01
Published online: March 20, 2026
Department of Digestive Medicine, The Second Affiliated Hospital of Anhui Medical University, Hefei, China
- Corresponding Author: Yu-Bao Zhou, MSc Department of Digestive Medicine, The Second Affiliated Hospital of Anhui Medical University, No.678 Furong Road, Economic and Technological Development Zone, Hefei City, Anhui Province 230000, China Tel: +86-15215513030, Fax: +86-551-63869400, E-mail: zhouyubao@fy.ahmu.edu.cn
- *Wei Wei Xie and Lin Xu contributed equally to this work.
© The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (https://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 632 Views
- 63 Download
Abstract
-
Background
- With the rising incidence of colon cancer, several studies have indicated that aquaporin 1 (AQP1) expression is associated with the development of colon cancer. This study aims to elucidate the potential molecular mechanisms between them.
-
Methods
- We screened data from The Cancer Genome Atlas (TCGA) database and retrospectively examined AQP1 protein expression in 127 colon cancer patients to analyze the relationship between AQP1 expression and pathological stages, prognosis. We created stable colon cancer cell lines with differential AQP1 expression, the effect of AQP1 expression on the proliferation and migration of colon cancer cells was assessed by in vitro and in vivo studies, and explored potential molecular mechanisms through Western blotting.
-
Results
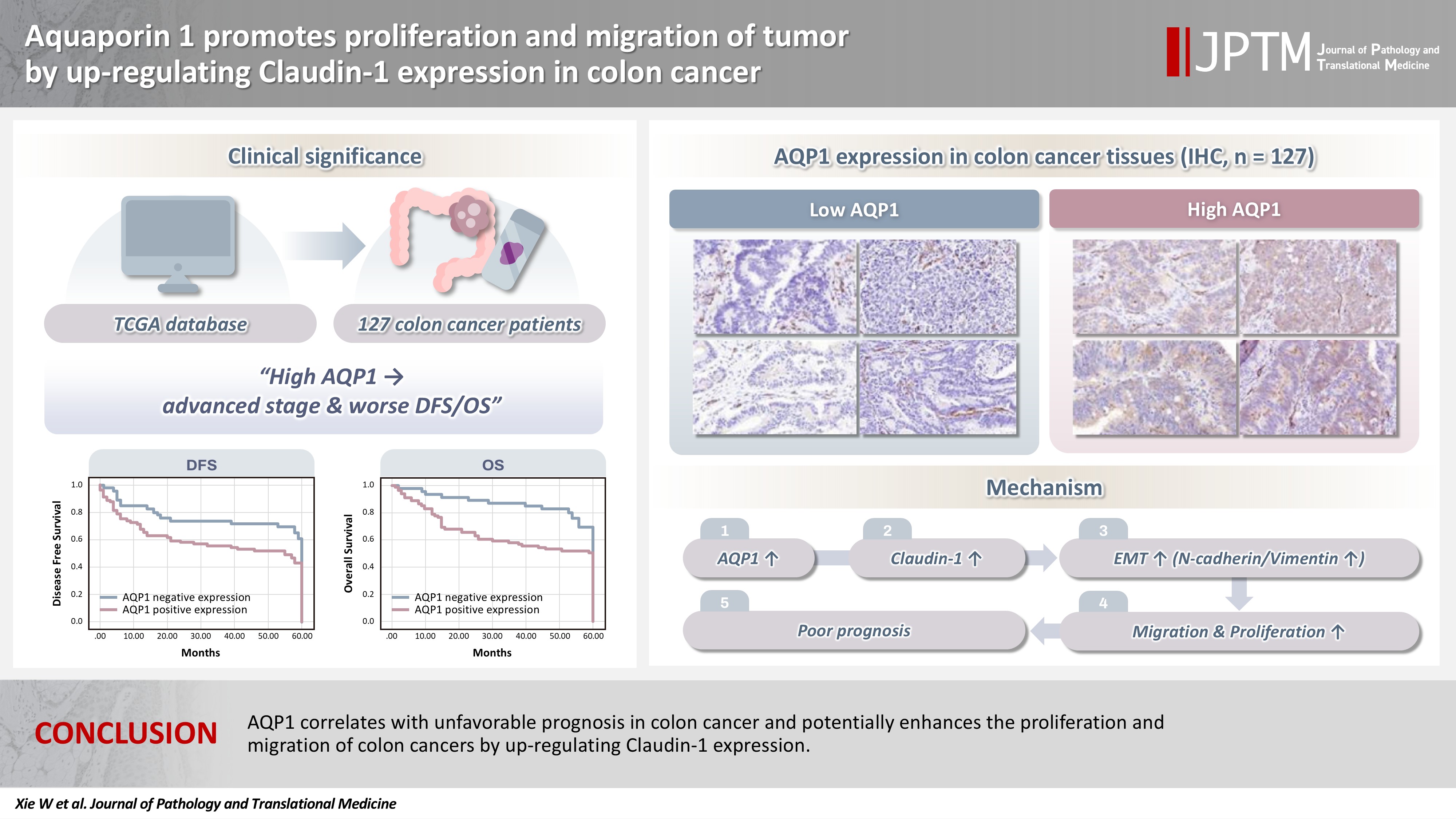
- High AQP1 expression was associated with poorer survival (overall survival [OS], p = .028) in colon cancer patients from the TCGA database. Similarly, retrospective clinical data indicated that high AQP1 expression was associated with reduced disease-free survival and OS (p = .036 and p = .017, respectively). The low-expressing AQP1 colon cancer cells exhibited a decrease in proliferation and migration ability of colon cancer cells compared to the overexpressing AQP1 group (p < .05) in vitro and in vivo. Immunohistochemistry and western blotting experiments validated heightened expression of N-cadherin, vimentin, and claudin-1 in the tumor tissues of the overexpressing AQP1 group. Conversely, reduced AQP1 expression resulted in decreased expression of claudin-1.
-
Conclusions
- AQP1 correlates with unfavorable prognosis in colon cancer and potentially enhances the proliferation and migration of colon cancer by up-regulating claudin-1 expression.
- Colorectal cancer (CRC) stands as the most prevalent malignant tumor within the digestive system and is the third most common malignant tumor globally [1]. According to the 2022 Global Cancer Observatory (GLOBOCAN) data, CRC specifically accounts for 9.7% of total new cancer cases, and 9.4% of total cancer-related deaths [2]. Surgical resection remains the primary treatment for stages I–III colon cancer, while stage IV disease requires individualized therapeutic approaches [3]. Nevertheless, up to 50% of patients after treatment develop liver metastases, contributing to a poor prognosis [4]. Therefore, there is a necessity to identify a novel therapeutic target for long-term use in treating colon cancer.
- The aquaporin family (AQPs) comprises small-molecule membrane proteins that promote cell migration, expansion and adhesion [5]. AQP1 expression is elevated in various tumor types, which can increase the risk of death in some cancer patients. In diseases such as lung adenocarcinoma, pleural mesothelioma, colorectal cancer, breast cancer, and prostate cancer, the hazard ratios for AQP1 are 4.0, 2.7, 2.6 to 3.4, 2.85, and 3.35, respectively [6,7]. AQP1 is expressed in colon cancer tissues but not in normal colon mucosa. Furthermore, its expression undergoes upregulation from the early to advanced stages of colon cancer development [8,9]. Evidence has been presented illustrating a correlation between the severity of the disease and the increased expression of AQP1 in colon cancer [10]. Increasing evidence suggests that AQP1 can influence cancer cell migration, invasion, and angiogenesis, potentially contributing to the development and progression of various cancers via tumor cell proliferation. Nevertheless, the precise signaling pathways remain elusive. Therefore, the aim of this study is to explore the effect of AQP1 on the clinicopathology and prognosis of colon cancer patients by database analysis and retrospective clinical study, and elucidate the potential molecular mechanisms through which AQP1 modulates the biological behaviors of colon cancer cells, providing potential novel therapeutic targets for colon cancer.
INTRODUCTION
- Materials
- Human colon cancer cell lines RKO and SW620 were procured from Procell Life Science and Technology Co. (Wuhan, China). Dulbecco's modified Eagle's medium (DMEM) culture medium and bovine serum were procured from Gibco (Grand Island, NY, USA). Antibodies against AQP1 (cat No. 20333-1-AP), actin (cat No. 66009-1-Ig), claudin-1 (cat No. 13050-1-AP), N-cadherin (cat No. 22018-1-AP), vimentin (cat No. 10366-1-AP), and Ki-67 (cat No. 27309-1-AP) were purchased from Proteintech (Wuhan, China).
- The Cancer Genome Atlas database analysis
- The relationship between AQPs and pathological stages of colon cancer patients was analyzed from The Cancer Genome Atlas (TCGA) database, and the correlation between AQP1 with different degrees of expression and the prognosis of colon cancer patients was observed.
- Patients, samples, and clinicopathological data
- A retrospective cohort study of 127 colon cancer patients who underwent first surgical resection from December 2017 to March 2020 at the Second Affiliated Hospital of Anhui Medical University was included in this study. Inclusion criteria are as follows: (1) patients aged between 18 and 80 years; (2) pathologically confirmed colon cancer patients undergoing initial radical resection; (3) availability of complete clinicopathological data and follow-up information, along with signed informed consent for this study. Exclusion criteria are as follows: (1) presence of other primary malignant tumors or severe systemic diseases; (2) incomplete clinical data; (3) unwillingness to participate in this study. All colon cancer patients were tested for AQP1 protein expression data by immunohistochemistry. Demographic and clinicopathologic information was retrospectively obtained from patients' medical records.
- Cell culture
- RKO and SW620 cells were cultivated in DMEM medium enriched with 10% (v/v) fetal bovine serum (Gibco) and 1% (v/v) antibiotic/antifungal solution (Beijing Beyotime Biotech Co. Inc, Beijing, China). All these cells underwent incubation in a 37°C environment with humidified air containing 5% CO2. Adherent cultured cells were passaged every 1–2 days. And cells in the logarithmic growth phase were utilized for the experiments.
- Lentiviral infection
- Lentiviral delivery of low-expressing AQP1, overexpressing AQP1, and negative control viral RNA were constructed by Genechem Co. Ltd. (Shanghai, China). RKO and SW620 cell lines were transduced with lentiviruses, plated in 6-well plates at a density of 4 × 105 cells/mL 24 hours before transfection, and then rinsed with serum-free, antibiotic-free DMEM medium. Low-expressing AQP1, overexpressing AQP1, and negative control viral RNA, along with the viral transfection solution, were appropriately diluted in serum-free, antibiotic-free DMEM medium following reagent instructions. These two dilutions were combined to create a transfection complex. The multiplicity of infection was set at 20, and infection was carried out with polybrene (6 µg/mL) for 8 hours.
- Western blotting assays
- Protein lysate (RIPA lysate:protease inhibitor:phosphatase inhibitor at 100:1:1) was added to each group of cells to be lysed on ice for 30 minutes to obtain whole-cell extracts and centrifuged in a centrifuge at 13,200 rpm for 25 minutes at 4°C. Supernatants were collected and protein concentrations were measured using the BCA Protein Assay Kit (Beijing Beyotime Biotech Co. Inc.), and proteins were denatured at 100°C for 10 minutes of denaturation. Equal amounts of proteins were electrophoresed by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis for 90 minutes, membrane transfer for 80 minutes, blocking solution was blocked and incubated with specific primary antibody overnight. Membrane was washed with TBST and incubated with secondary antibody then secondary antibody (Affinit Goat Anti-Rabbit IgG (H+L) HRP S0001, Goat Anti-Mouse IgG (H+L) HRP S0002) were incubated to form immune complexes, then the membranes were photographed with a gel imager and the results were analyzed. Quantification was performed using Image-J (1.53k, National Institutes of Health, Bethesda, MD, USA).
- Cell migration assay
- The AQP1 differentially expressing RKO and SW620 cell lines were plated into 6-well plates and allowed to reach confluence. Linear wounds were gently created using the tip of a 200 µL sterile yellow pipette. After rinsing with phosphate buffered saline (PBS) to remove debris, the cells were incubated with fetal bovine serum-free medium at 37°C and 5% CO2 for 48 hours. Images of the wound area were obtained using phase contrast microscopy, and the images were then analyzed to quantify migration, quantifying wound closure for each sample as the area covered by cells over 48 hours.
- A total of 1 × 104 cells were mixed with 200 µL of serum-free DMEM and added homogeneously to the inner chamber of a transwell and the lower chamber was loaded with 800 µL of medium containing 30% fetal bovine serum. The cells were incubated in 5% CO2 at 37°C for 48 hours. After incubation, the cells were removed from the upper surface of the membrane with a cotton swab, fixed with 10% paraformaldehyde for 10 minutes, and stained with 1% crystal violet for 30 minutes to count the migrating cells left on the bottom surface, which were analyzed by microscopic observation.
- Cell proliferation experiment
- Cells in logarithmic growth phase were inoculated into 6-well plates, each experimental group was inoculated with 1,000 cells/well. And the culture was continued until the number of cells was greater than 50 for 14 days or until the number of cells was greater than 50 in the majority of individual clones, and then fixation staining and photographs were taken. The rate of clone formation was calculated using Image-J software.
- Colon cancer cells in the logarithmic growth stage were seeded into 96-well plates at a density of 1–2 × 103 cells and cultured in 100 µL of DMEM per well. After the cells were attached to the wall, respectively, continue to incubate for 0, 12, 24, 48, 60, 72 hours to add the Cell Counting Kit-8 (CCK8) reagent (5 mg/mL) (Beijing Beyotime Biotech Co. Inc.) 10 µL/well, continue to incubate for 2 hours and then measure the cell formation rate with the spectrophotometer. The absorbance values at 450 nm were measured by spectrophotometer, respectively.
- In vivo studies
- All animal experiments were performed in accordance with the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals and approved by the Institutional Animal Care and Use Committee of Anhui Medical University (Hefei, China) (approval number: LLSC20241736). Four-week-old male nude mice were purchased from Hangzhou Ziyuan Laboratory Animal Science and Technology Co. (Hangzhou, China). Mice were housed in a pathogen-free animal facility with controlled temperature and humidity. About 1 × 107 SW620-shAQP1, SW620-control and SW620-ovAQP1 cells were suspended in 0.1 mL of PBS. And the above cell lines were injected subcutaneously into the mice, which were euthanized four weeks later, and all the tumors were carefully excised, their sizes were recorded, and the tumors were embedded in 10% formalin and paraffin for immunohistochemical staining.
- Immunohistochemistry
- Tumor tissue specimens from mice were fixed in formalin for 12 hours and dehydrated. After paraffin embedding, the tissues were cut into 4-μm microsections. Tissue sections were dewaxed, rehydrated, and then soaked in methanol containing 0.3% hydrogen peroxide for 30 minutes to block endogenous peroxidase activity. The sections were then heated in a pressure cooker containing 10 mM ethylenediaminetetraacetic acid buffer (pH 8.0) for 2 minutes. After cooling, the sections were incubated in 1% blocking serum for 30 minutes to reduce nonspecific binding. The primary anti-AQP1 polyclonal antibody was diluted 1:250 and incubated with the sections overnight at 4°C. The sections were then incubated with the biotinylated secondary antiserum followed by horseradish peroxidase–conjugated streptavidin-biotin complex. Finally, the sections were developed with diaminobenzidine and restained with haematoxylin.
- Using the German Immune Response Score, the immune response score (IRS) evaluates stained sections in an uninformative manner without prior information. The IRS scores the distribution (0–4) and intensity (0–3) of the immune response, which are then multiplied to produce an IRS score. The percentage of positive cells was scored as follows: "0" (<5%), "1" (5%–25%), "2" (25%–50%), "3" (50%–75%), and "4" (>75%). The intensity of staining was scored as follows: "0" (no staining), "1" (weak staining), "2" (moderate staining), and "3" (strong staining). The final AQP1 expression score was calculated using the percentage of positivity score multiplied the staining intensity score, ranging from 0 to 12. We estimated the IRS by averaging the values of eight fields of view for each sample at 400× magnification. AQP1 expression was defined as follows: low expression (0–6/0–3 score) and high expression (>6/4–6 score). Immunohistochemical analysis and scoring were performed by two independent investigators.
- Statistical analysis
- Disease-free survival (DFS) was measured from the date of surgery to the date of recurrence. Overall survival (OS) time was defined as the period from the date of colectomy to the date of death. Follow-up of patients was continued until death or 5 years after surgery, whichever occurred first. Statistical analyses were performed using SPSS ver. 25.0 for Windows (IBM Corp., Armonk, NY, USA). Qualitative variables were analyzed using the Pearson χ2 test or Fisher’s exact test. OS and DFS were calculated using the Kaplan-Meier method and Life Table method. A comparison between groups was conducted using Student’s t-test and the Mann-Whitney test. A value of p < .05 was considered statistically significant.
- The experimental data were analyzed statistically using GraphPad Prism 9.0 (GraphPad Software Inc., San Diego, CA, USA), and the data were expressed as mean ± standard deviation. Differences between two groups were analyzed using t-test, and comparisons between multiple groups were made using one-way ANOVA. All experiments were repeated 3 times, and the difference was considered statistically significant at p < .05.
MATERIALS AND METHODS
Wound healing assay
Migration assay
Plate cloning assay
Cell Counting Kit-8 assay
- AQP1 expression correlates with an unfavorable prognosis in colon patients
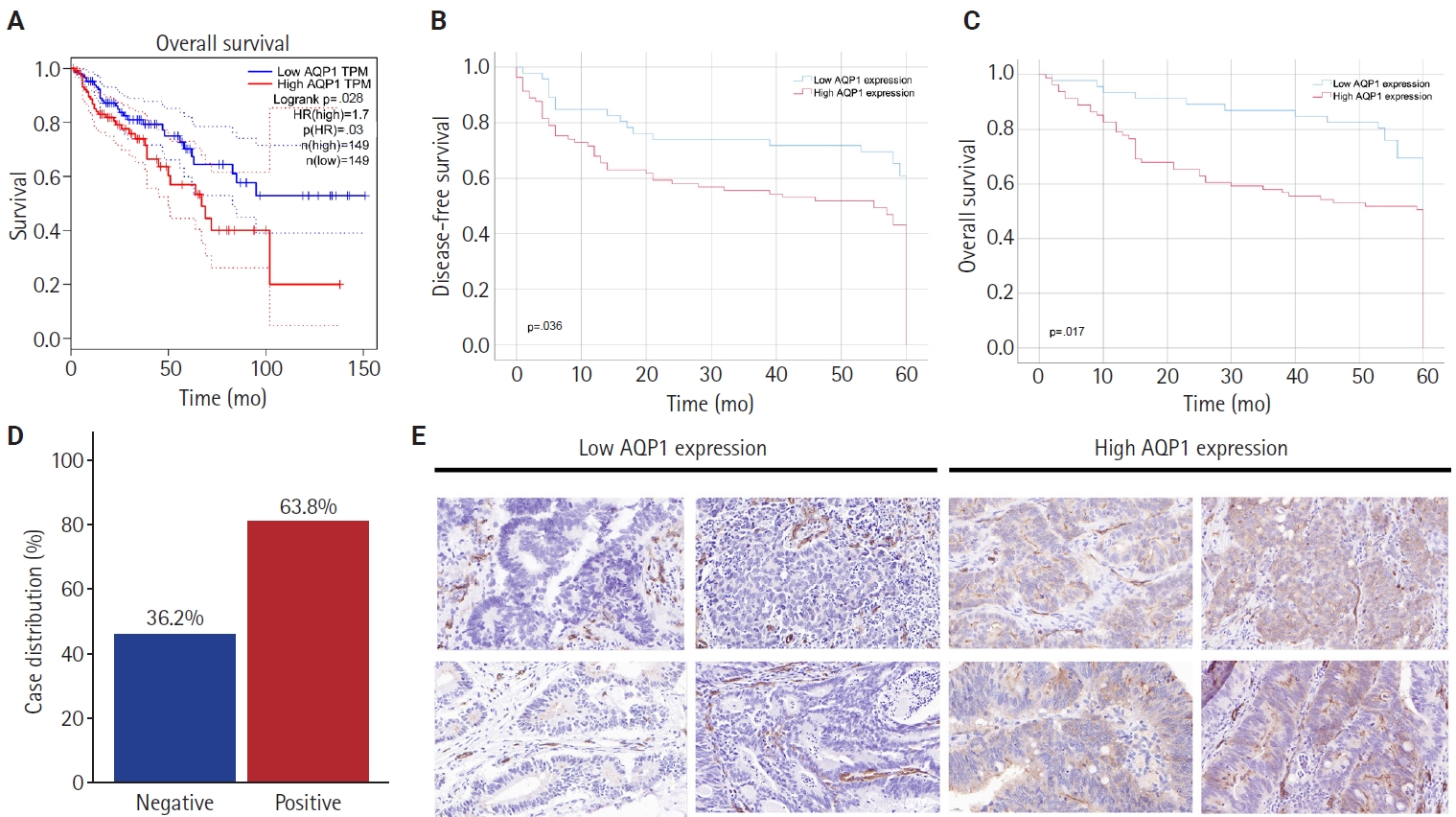
- Analysis of the relationship between the AQPs and pathological staging of colon cancer patients by the TCGA database revealed that only AQP1 expression levels were associated with pathological staging (p < .001), and high AQP1 expression means a later pathological staging (Table 1) and lymphatic vessel invasion in colon cancer (p = .046) (AQP1 expression levels were graded by median). Survival analysis showed that the OS of patients with high AQP1 expression were statistically different from those of the low expression group (p = .028) (Fig. 1A).
- Association of AQP1 protein expression with demographic characteristics, clinicopathological features, and survival of colon cancer patients
- Demographic, clinical and pathological data of 127 patients with colon cancer are shown in Table 2. The median age was 65.0 years old (range, 21 to 88 years). The male-to-female ratio was comparable. Of the 127 patients, 17 (13.4%) had intestinal obstruction, 86 (67.7%) had intestinal stenosis, 17 (13.4%) had hydroperitoneum, and 71 (55.9%) had lesions located in the right colon. According to the 8th edition of the American Joint Committee on Cancer/Union for International Cancer Control staging system, the tumor stages were distributed as follows: stage I in six cases (4.7%); stage Ⅱ in 70 cases (55.1%); stage Ⅲ in 41 cases (32.3%); and stage Ⅳ in 10 cases (7.9%) (Table 2).
- High AQP1 expression was found in 63.8% (81/127) of the patients (Fig. 1D). The association between high AQP1 expression and tumor location in the right colon (p = .034), poorly differentiated (p = .020), neurological invasion (p = .001), lymph node metastasis (p = .021), vascular embolism (p = .004), and pathologic tumor stage (p = .027) (Table 2) were significant. The mean follow-up was 44.2 months (range, 1 to 60 months), with recurrence occurring in 61 (43.6%) of the cases. Statistical analysis of survival data showed that high AQP1 expression was associated with poorer DFS and OS (p = .036 and p = .017, respectively) (Fig. 1B, C).
- Effect of AQP1 on migration of RKO and SW620 cells
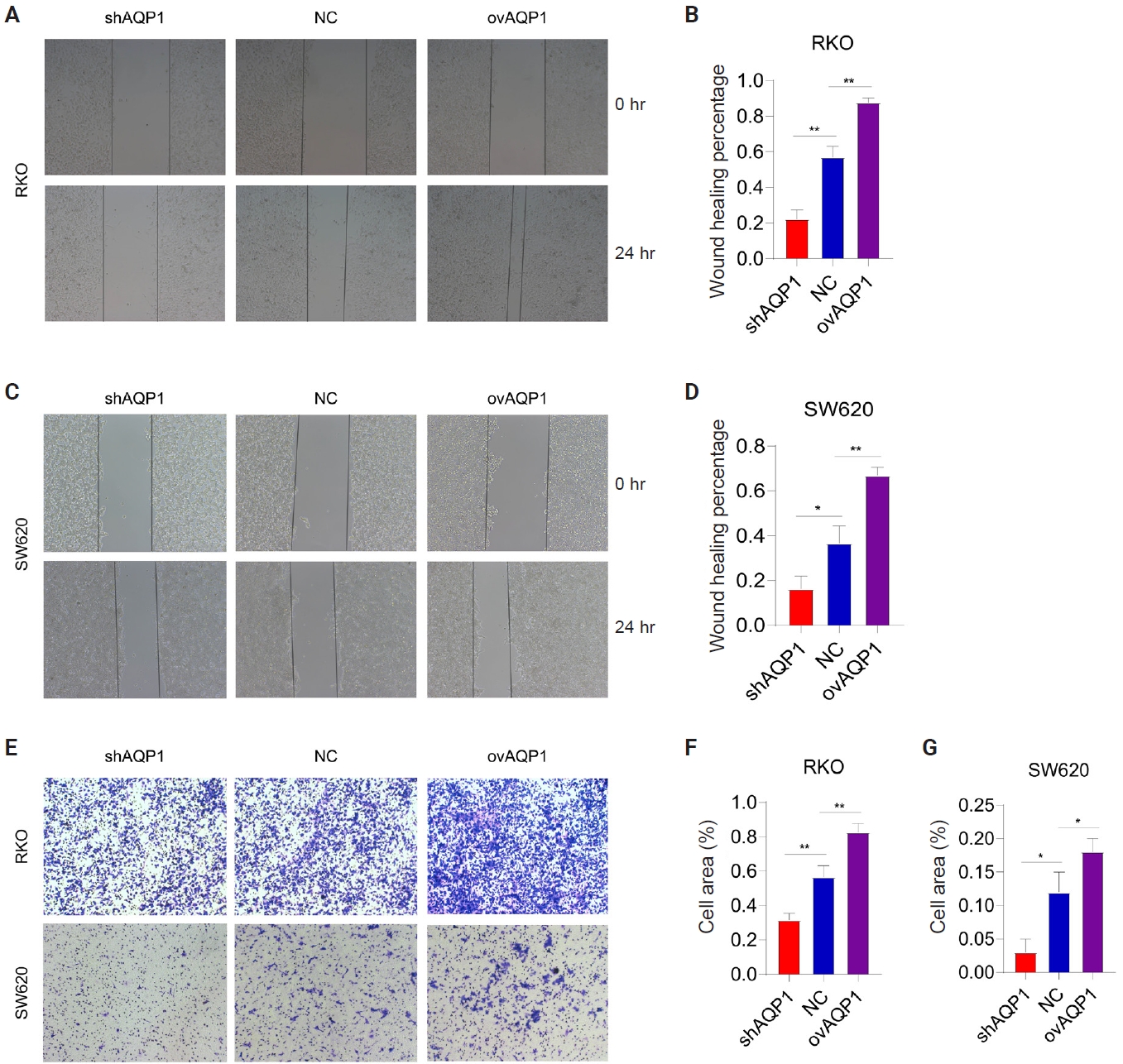
- To clarify the role of AQP1 in colon cancer metastasis, we constructed stably transfected RKO and SW620 cell lines with low and overexpressing of AQP1. Western blotting results showed that RKO and SW620 cells had been successfully transfected with lentivirus.
- Wound healing assay showed that the percentage of wound healing was decreased in low-expressing AQP1 cells compared to overexpressing AQP1 cells (p < .05) (Fig. 2A–D). Migration assay of RKO and SW620 also showed that the decrease in the number of transmembrane cells in the low-expressing AQP1 cells inhibited the metastasis of colon cancer cells, whereas the increase in the number of transmembrane cells in the overexpressing AQP1 cells promoted the colon cancer migration (Fig. 2E–G). These results suggest that elevated AQP1 in colon cancer cells promotes colon cancer cell migration in vitro.
- Effect of AQP1 on proliferation of RKO and SW620 cells
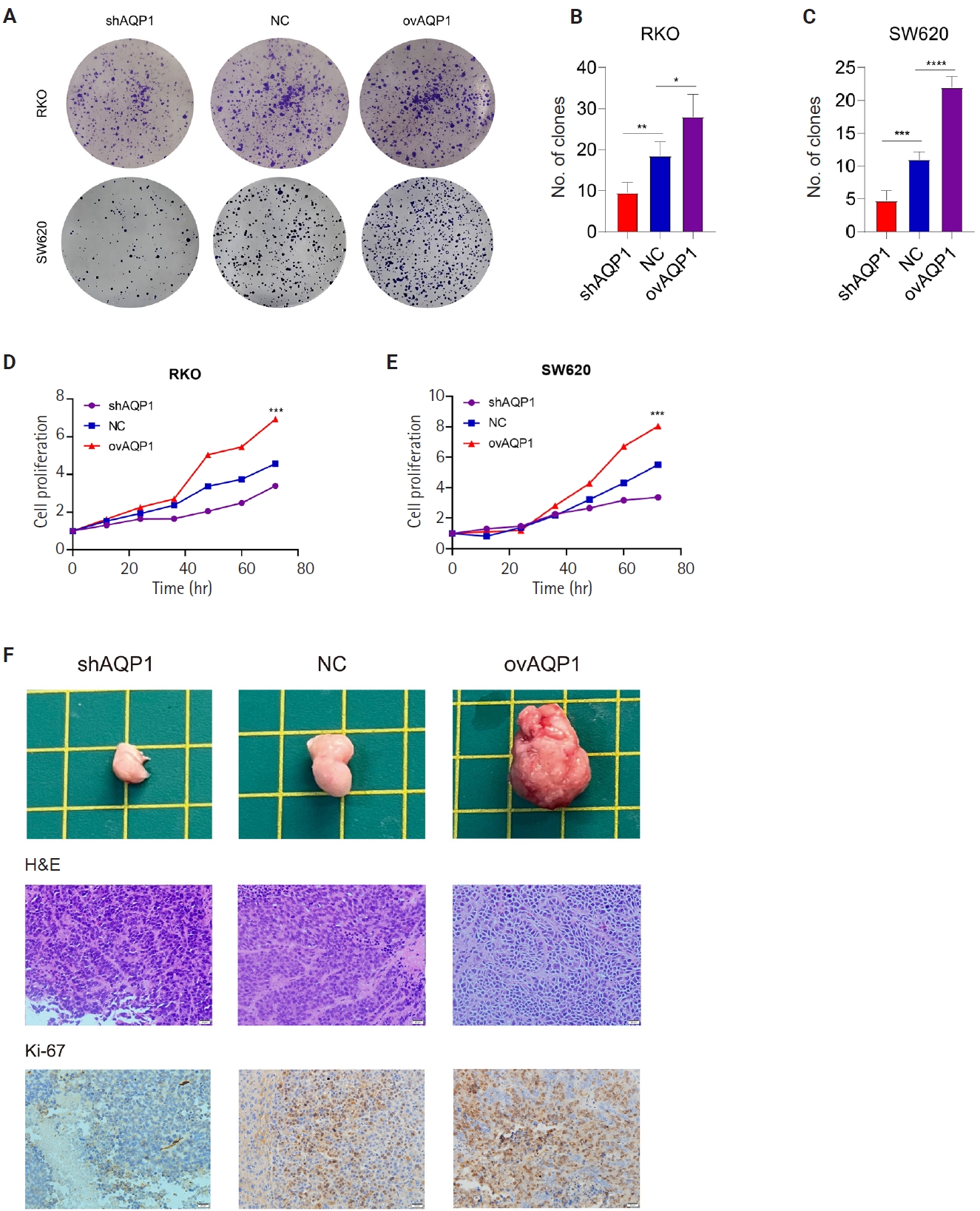
- Plate cloning assays showed a reduction in the number of clones of low-expressing AQP1 cells compared to overexpressing AQP1 cells (p < .05) (Fig. 3A–C). We used CCK8 reagent to assess the effect of different expression levels of AQP1 on the proliferative ability of colon cancer cells (Fig. 3D, E). The cell proliferation curves showed elevated AQP1 expression at the same time and increased cell number, representing enhanced cell proliferation ability. These results suggest that AQP1 promotes the proliferation of colon cancer cells in vitro.
- Effect of AQP1 on colon cancer cell proliferation ability in vivo
- To confirm the role of AQP1 in growth and proliferation in vivo, we injected tumor cells (SW620-shAQP1, SW620-NC, and SW620-ovAQP1) into the subcutaneous tissue of nude mice and established a subcutaneous tumorigenic model. After 30 days, we found that the nude mice in the SW620-ovAQP1 group had a larger volume of subcutaneous tumors when compared to the SW620-NC group; on the contrary, subcutaneous tumor volume was smaller in nude mice injected with SW620-shAQP1 group compared with SW620-NC group (Fig. 3F). And the expression levels of Ki-67 in the subcutaneous tumor tissues of nude mice were examined by immunohistochemistry, results demonstrate that Ki-67 levels are higher in the ovAQP1 group compared to the shAQP1 group (Fig. 3F). These results suggest that increased AQP1 expression promotes the proliferative capacity of colon cancer in vivo.
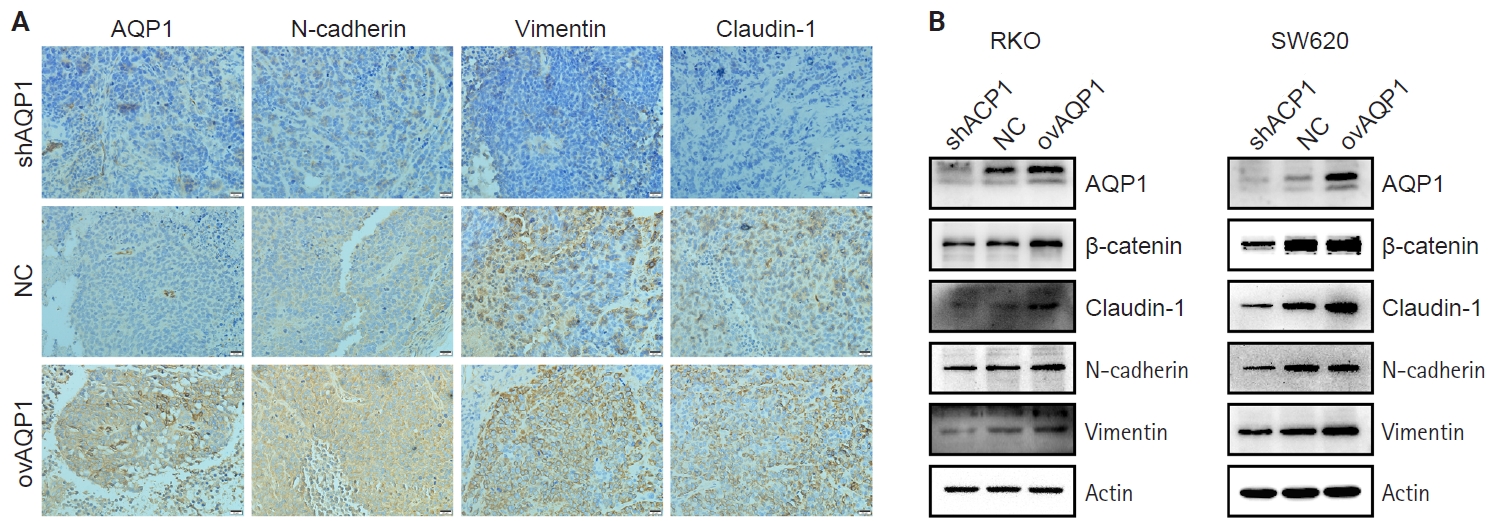
- Next, the expression levels of AQP1, N-cadherin, vimentin, and claudin-1 in the subcutaneous tumor tissues of nude mice were examined by immunohistochemistry. The expression levels of N-cadherin, vimentin, and claudin-1 were higher in the ovAQP1 group than in the shAQP1 group (Fig. 4A). Therefore, we hypothesized that AQP1 may promote epithelial-mesenchymal transition (EMT) by promoting the expression of claudin-1.
- AQP1 may promote EMT by up-regulating claudin-1 expression
- Based on the above results, we hypothesized that AQP1 may regulate tumor proliferation and metastasis through EMT. To determine the possible mechanism of action of AQP1 on colon cancer cell migration, we examined the expression of EMT-related genes by Western blotting experiments. With elevated AQP1 expression, the expression of claudin-1 was upregulated, as was the expression level of the epithelial cell marker, N-cadherin, whereas the low-expressing AQP1 resulted in the opposite (Fig. 4B). These results suggest that AQP1 may promote the EMT process by up-regulating the expression of claudin-1.
RESULTS
- Colon cancer frequently invades neighboring tissues and metastasizes via lymphatic and blood vessels [11], with distant metastases present in 25% of patients at diagnosis and high recurrence rates post-surgery [12]. Although screening has reduced the overall incidence rate, the incidence rate among young people and the mortality rate among patients under 65 years of age are still rising [13]. Despite therapies targeting proliferation, angiogenesis, and the tumor microenvironment, metastasis remains a major cause of death, with a 5-year survival rate below 15% for advanced cases [14,15]. Understanding the molecular mechanisms driving progression is critical for developing more effective treatments.
- AQP1 elevation is more pronounced in middle and advanced, poorly differentiated colon cancer tissues [16]. The expression of AQP1 is linked to lymph node metastasis, lymphovascular infiltration, and vascular infiltration in colon cancer, establishing it as an independent predictor of poor prognosis [17]. Similar to these findings, we derived that among the AQPs, only AQP1 was associated with pathological stage of colon cancer patients (p < .001) and lymphovascular invasion (p = .046) according to TCGA. Survival analysis showed that patients with high AQP1 expression had shorter OS (p = .028). In this study, 63.8% of cases were high AQP1 expression, which was significantly correlated with aggressive tumor characteristics including degree of differentiation, perineural invasion, lymphatic invasion, choroidal cancer embolism, and tumor stage. Patients with high AQP1 expression had shorter DFS and OS than those with low AQP1 expression. These findings suggest that AQP1 plays a tumor-promoting role in colon cancer.
- Numerous studies demonstrate that the dual-channel functionality of AQP1, involving water and ions, enhances the migration and invasion of cancer cells in vitro [18,19]. Ion channel inhibitors like 5HMF and AqB011, along with water pore channel inhibitors like Bacopaside II and AqB013, have demonstrated the ability to impair the in vitro migration of colon cancer cells. When employed in combination, AQP1 blockers exhibit a synergistic effect, efficiently decelerating the progression of cancer. Hence, they are regarded as a promising tool for enhancing cancer treatment [19-21]. In the realm of medical possibilities, AQP1 blockers show promise for treating congestive heart failure, refractory edema associated with cirrhosis, and specific cancer therapies [22].
- Colon cancer employ various mechanisms for spreading, with the most central being the transition of cells from a non-motile, quiescent epithelial phenotype to a mesenchymal cell-like form characterized by traits such as migration, invasion, and resistance—commonly known as EMT [23]. Throughout EMT, there is typically a reduction in the levels of epithelial cell markers, coupled with an upregulation of mesenchymal cell-associated markers [24]. To unravel the specific mechanism, we constructed colon cancer cell lines exhibiting varied expression of AQP1. Within this study, it was demonstrated that AQP1 promotes the proliferative and metastatic ability of colon cancer in vitro and in vivo. Western blotting revealed a gradual increase in the expression of N-cadherin as the content of AQP1 increased. Recognizing N-cadherin as a promoter of tumor metastasis and a major intracellular marker of EMT, its heightened expression correlates directly with tumor invasiveness, spread, and resistance to cell death. Elevated N-cadherin levels are considered an independent prognostic factor for poor outcomes in solid tumor cells, including colon cancer [25,26]. Therefore, our conclusion is that the metastatic capability of colon cancer cells escalates in tandem with the increased expression of AQP1, affirming the promotional role of AQP1 in colon cancer metastasis. Simultaneously, we observed an elevation in claudin-1 expression with heightened AQP1 expression. In normal epithelial cells, claudin-1 plays a role in preserving the mucosal barrier of the normal colon epithelium [27,28]; nevertheless, in the context of colon cancer, its overexpression promotes the transformation, invasion, and metastasis of cancer cells [29]. Given the alteration in transcription factors linked to EMT, we hypothesized that AQP1 influences the EMT process in colon cancer by up-regulating claudin-1 expression.
- AQP1 is expressed in various gastrointestinal tumors, but studies indicate its functions vary across different contexts. For instance, reports indicate AQP1 exhibits tumor-suppressing effects in intrahepatic cholangiocarcinoma, potentially regulating tumor growth by modulating Snail expression [30]. Conversely, it promotes tumorigenesis in gastric, pancreatic, and hepatocellular carcinomas [31-33]. This study further demonstrates its oncogenic role in colorectal cancer. Thus, we conclude that AQP1 exhibits distinct biological behaviors across different gastrointestinal malignancies. No studies have yet elucidated the potential reasons for this discrepancy. Future research will investigate whether AQP1 mutations exist in different gastrointestinal tumors and further explore the causes of these mutations.
- The precise mechanism by which AQP1 regulates claudin-1 in colon cancer progression remains unclear. Current evidence primarily relies on in vitro and single-center retrospective studies, which lack robust mechanistic depth. Future studies should employ multicenter prospective cohorts, AQP1-knockout/transgenic mouse models, and metastatic xenograft models to clarify its role in tumor invasion and metastasis. Additionally, advanced molecular techniques are needed to dissect the AQP1–claudin-1 interaction, including upstream regulators and downstream effectors to identify potential therapeutic targets.
- In conclusion, the findings of this study propose that AQP1 potentially stimulates the proliferation, metastasis, and EMT of colon cancer cells by upregulating claudin-1 expression, indicating an association with unfavorable outcomes in colon cancer. The reduction in AQP1 expression hindered the proliferation and migration abilities of colon cancer cells. AQP1 emerges as a potential molecular marker for predicting the prognosis of colon cancer patients and serves as a potential target for the treatment of colon cancer.
DISCUSSION
Ethics Statement
All procedures performed in the current study were approved by Institutional Review Board (IRB) of the Second Affiliated Hospital of Anhui Medical University (approval number: YX2024-213) in accordance with the 1964 Helsinki declaration and its later amendments. Informed consent was obtained from all individual participants included in the study. The data did not contain any patient-identifiable information.
Availability of Data and Material
The datasets generated or analyzed during the study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
Author Contributions
Conceptualization: WWX, DQZ, YBZ. Data curation: LX, QL. Formal analysis: WWX, LX, QL. Funding acquisition: YBZ. Investigation: WWX, LX, QL. Methodology: WWX, LX, DQZ, YBZ. Project administration: DQZ, YBZ. Resources: YBZ. Software: WWX, LX. Supervision: DQZ, YBZ. Validation: WWX, LX, QL. Visualization: LX, QL. Writing—original draft: WWX, LX, QL. Writing—review & editing: YBZ, DQZ. Approval of final manuscript: all authors.
Conflicts of Interest
The authors declare that they have no potential conflicts of interest.
Funding Statement
This project was supported by Clinical Research Cultivation Program of the Second Affiliated Hospital of Anhui Medical University (2020LCZD13) and Scientific Research Program of Higher Education Institutions in Anhui Province (Natural Science) (2024AH050791).
| Factor | Total (n = 127) | AQP1 expression | p-value | |
|---|---|---|---|---|
| High (n = 81) | Low (n = 46) | |||
| Sex (M/F) | 66/61 | 44/37 | 22/24 | .481 |
| Age (yr) | 127 | 60.57 ± 12.59 | 63.41 ± 12.38 | .220 |
| Hemoglobin (g/L) | 127 | 105.80 ± 24.05 | 104.37 ± 25.79 | .754 |
| CEA | 102 (68/34)a | 13.37 ± 36.06 | 11.57 ± 21.62 | .790 |
| CA19-9 | 99 (65/34)a | 23.29 ± 35.26 | 19.85 ± 20.61 | .602 |
| CA72-4 | 98 (65/33)a | 5.82 ± 9.77 | 4.07 ± 5.57 | .345 |
| Intestinal obstruction | .087 | |||
| No | 110 (86.6) | 67 (82.7) | 43 (93.5) | |
| Yes | 17 (13.4) | 14 (17.3) | 3 (6.5) | |
| Intestinal stenosis | .128 | |||
| No | 41 (32.3) | 30 (37.0) | 11 (23.9) | |
| Yes | 86 (67.7) | 51 (63.0) | 35 (76.1) | |
| Hydroperitoneum | .242 | |||
| No | 110 (86.8) | 68 (84.0) | 42 (91.3) | |
| Yes | 17 (13.4) | 13 (16.0) | 4 (8.7) | |
| Location | .034 | |||
| Right colon | 71 (55.9) | 51 (63.0) | 20 (43.5) | |
| Left colon | 56 (44.1) | 30 (37.0) | 26 (56.5) | |
| Tumor differentiation | .020b | |||
| Poorly | 19 (15.0) | 17 (21.0) | 2 (4.3) | |
| Moderately | 106 (83.5) | 63 (77.8) | 43 (93.5) | |
| Well | 2 (1.6) | 1 (1.2) | 1 (2.2) | |
| Perineural invasion | .001 | |||
| No | 43 (33.9) | 19 (23.5) | 24 (52.2) | |
| Yes | 84 (66.1) | 62 (76.5) | 22 (47.8) | |
| Lymphatic invasion | .021 | |||
| No | 80 (63.0) | 45 (55.6) | 35 (76.1) | |
| Yes | 47 (37.0) | 36 (44.4) | 11 (23.9) | |
| Vascular thrombosis | .004 | |||
| No | 56 (44.1) | 28 (34.6) | 28 (60.9) | |
| Yes | 71 (55.9) | 53 (65.4) | 18 (39.1) | |
| TNM stage | .027b | |||
| I | 6 (4.7) | 3 (3.7) | 3 (6.5) | |
| Ⅱ | 70 (55.1) | 38 (46.9) | 32 (69.6) | |
| Ⅲ | 41 (32.3) | 31 (38.3) | 10 (21.7) | |
| Ⅳ | 10 (7.9) | 9 (11.1) | 1 (2.2) | |
Values are presented as mean ± SD or number (%).
AQP1, aquaporin 1; CEA, carcinoembryonic antigen; CA19-9, glycoconjugate antigen 19-9; CA72-4, glycoconjugate antigen 72-4; SD, standard deviation.
aNumbers in parentheses indicate cases with high AQP1 expression/low AQP1 expression;
bFisher’s exact test; statistically significant differences (p < .05).
- 1. Liu S, Peng X, Wu X, et al. Construction of a new immune-related lncRNA model and prediction of treatment and survival prognosis of human colon cancer. World J Surg Oncol 2022; 20: 71.ArticlePubMedPMCPDF
- 2. Bray F, Laversanne M, Sung H, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2024; 74: 229-63. ArticlePubMed
- 3. Benson AB, Venook AP, Al-Hawary MM, et al. NCCN guidelines insights: colon cancer, version 2.2018. J Natl Compr Canc Netw 2018; 16: 359-69. ArticlePubMedPMC
- 4. Brown KG, Koh CE. Surgical management of recurrent colon cancer. J Gastrointest Oncol 2020; 11: 513-25. ArticlePubMedPMC
- 5. Wang L, Zhang Y, Wu X, Yu G. Aquaporins: new targets for cancer therapy. Technol Cancer Res Treat 2016; 15: 821-8. ArticlePubMedPDF
- 6. Mobasheri A, Airley R, Hewitt SM, Marples D. Heterogeneous expression of the aquaporin 1 (AQP1) water channel in tumors of the prostate, breast, ovary, colon and lung: a study using high density multiple human tumor tissue microarrays. Int J Oncol 2005; 26: 1149-58. ArticlePubMed
- 7. Chow PH, Bowen J, Yool AJ. Combined systematic review and transcriptomic analyses of mammalian aquaporin classes 1 to 10 as biomarkers and prognostic indicators in diverse cancers. Cancers (Basel) 2020; 12: 1911.ArticlePubMedPMC
- 8. Moon C, Soria JC, Jang SJ, et al. Involvement of aquaporins in colorectal carcinogenesis. Oncogene 2003; 22: 6699-703. ArticlePubMedPDF
- 9. Smith E, Tomita Y, Palethorpe HM, et al. Reduced aquaporin-1 transcript expression in colorectal carcinoma is associated with promoter hypermethylation. Epigenetics 2019; 14: 158-70. ArticlePubMedPMC
- 10. El Hindy N, Bankfalvi A, Herring A, et al. Correlation of aquaporin-1 water channel protein expression with tumor angiogenesis in human astrocytoma. Anticancer Res 2013; 33: 609-13. PubMed
- 11. Pretzsch E, Bosch F, Neumann J, et al. Mechanisms of metastasis in colorectal cancer and metastatic organotropism: hematogenous versus peritoneal spread. J Oncol 2019; 2019: 7407190.ArticlePubMedPMCPDF
- 12. Young PE, Womeldorph CM, Johnson EK, et al. Early detection of colorectal cancer recurrence in patients undergoing surgery with curative intent: current status and challenges. J Cancer 2014; 5: 262-71. ArticlePubMedPMC
- 13. Siegel RL, Wagle NS, Cercek A, Smith RA, Jemal A. Colorectal cancer statistics, 2023. CA Cancer J Clin 2023; 73: 233-54. ArticlePubMedPDF
- 14. Van Cutsem E, Cervantes A, Adam R, et al. ESMO consensus guidelines for the management of patients with metastatic colorectal cancer. Ann Oncol 2016; 27: 1386-422. ArticlePubMed
- 15. Khan S, Ricciardelli C, Yool AJ. Targeting aquaporins in novel therapies for male and female breast and reproductive cancers. Cells 2021; 10: 215.ArticlePubMedPMC
- 16. Hong Y, Chen Z, Li N, Zhang M. Prognostic value of serum aquaporin-1, aquaporin-3 and galectin-3 for young patients with colon cancer. Ann Clin Biochem 2020; 57: 404-11. ArticlePubMedPDF
- 17. Yoshida T, Hojo S, Sekine S, et al. Expression of aquaporin-1 is a poor prognostic factor for stage II and III colon cancer. Mol Clin Oncol 2013; 1: 953-8. ArticlePubMedPMC
- 18. Pei JV, Kourghi M, De Ieso ML, et al. Differential inhibition of water and ion channel activities of mammalian aquaporin-1 by two structurally related bacopaside compounds derived from the medicinal plant Bacopa monnieri. Mol Pharmacol 2016; 90: 496-507. ArticlePubMed
- 19. De Ieso ML, Pei JV, Nourmohammadi S, et al. Combined pharmacological administration of AQP1 ion channel blocker AqB011 and water channel blocker Bacopaside II amplifies inhibition of colon cancer cell migration. Sci Rep 2019; 9: 12635.ArticlePubMedPMC
- 20. Chow PH, Kourghi M, Pei JV, Nourmohammadi S, Yool AJ. 5-Hydroxymethyl-furfural and structurally related compounds block the ion conductance in human aquaporin-1 channels and slow cancer cell migration and invasion. Mol Pharmacol 2020; 98: 38-48. ArticlePubMed
- 21. Dorward HS, Du A, Bruhn MA, et al. Pharmacological blockade of aquaporin-1 water channel by AqB013 restricts migration and invasiveness of colon cancer cells and prevents endothelial tube formation in vitro. J Exp Clin Cancer Res 2016; 35: 36.ArticlePubMedPMC
- 22. Verkman AS, Anderson MO, Papadopoulos MC. Aquaporins: important but elusive drug targets. Nat Rev Drug Discov 2014; 13: 259-77. ArticlePubMedPMCPDF
- 23. Skarkova V, Vitovcova B, Matouskova P, et al. Role of N-cadherin in epithelial-to-mesenchymal transition and chemosensitivity of colon carcinoma cells. Cancers (Basel) 2022; 14: 5146.ArticlePubMedPMC
- 24. Wang J, Zhang N, Han Q, et al. Pin1 inhibition reverses the acquired resistance of human hepatocellular carcinoma cells to Regorafenib via the Gli1/Snail/E-cadherin pathway. Cancer Lett 2019; 444: 82-93. ArticlePubMedPMC
- 25. Yan X, Yan L, Liu S, Shan Z, Tian Y, Jin Z. N-cadherin, a novel prognostic biomarker, drives malignant progression of colorectal cancer. Mol Med Rep 2015; 12: 2999-3006. ArticlePubMed
- 26. Ye Z, Zhou M, Tian B, Wu B, Li J. Expression of lncRNA-CCAT1, E-cadherin and N-cadherin in colorectal cancer and its clinical significance. Int J Clin Exp Med 2015; 8: 3707-15. PubMedPMC
- 27. Gunzel D, Yu AS. Claudins and the modulation of tight junction permeability. Physiol Rev 2013; 93: 525-69. ArticlePubMedPMC
- 28. Singh AB, Uppada SB, Dhawan P. Claudin proteins, outside-in signaling, and carcinogenesis. Pflugers Arch 2017; 469: 69-75. ArticlePubMedPMCPDF
- 29. Bhat AA, Ahmad R, Uppada SB, Singh AB, Dhawan P. Claudin-1 promotes TNF-alpha-induced epithelial-mesenchymal transition and migration in colorectal adenocarcinoma cells. Exp Cell Res 2016; 349: 119-27. ArticlePubMedPMC
- 30. Zhuang MQ, Jiang XL, Liu WD, et al. Aquaporin 1 is a prognostic marker and inhibits tumour progression through downregulation of Snail expression in intrahepatic cholangiocarcinoma. Dig Liver Dis 2023; 55: 1133-40. ArticlePubMed
- 31. Wang Z, Wang Y, He Y, Zhang N, Chang W, Niu Y. Aquaporin-1 facilitates proliferation and invasion of gastric cancer cells via GRB7-mediated ERK and Ras activation. Anim Cells Syst (Seoul) 2020; 24: 253-9. ArticlePubMedPMC
- 32. Lopes PA, Fonseca E, da Silva IV, Vigia E, Paulino J, Soveral G. Aquaporins transcripts with potential prognostic value in pancreatic cancer. Genes (Basel) 2023; 14: 1694.ArticlePubMedPMC
- 33. Pelagalli A, Nardelli A, Fontanella R, Zannetti A. Inhibition of AQP1 hampers osteosarcoma and hepatocellular carcinoma progression mediated by bone marrow-derived mesenchymal stem cells. Int J Mol Sci 2016; 17: 1102.ArticlePubMedPMC
REFERENCES
Figure & Data
References
Citations

 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
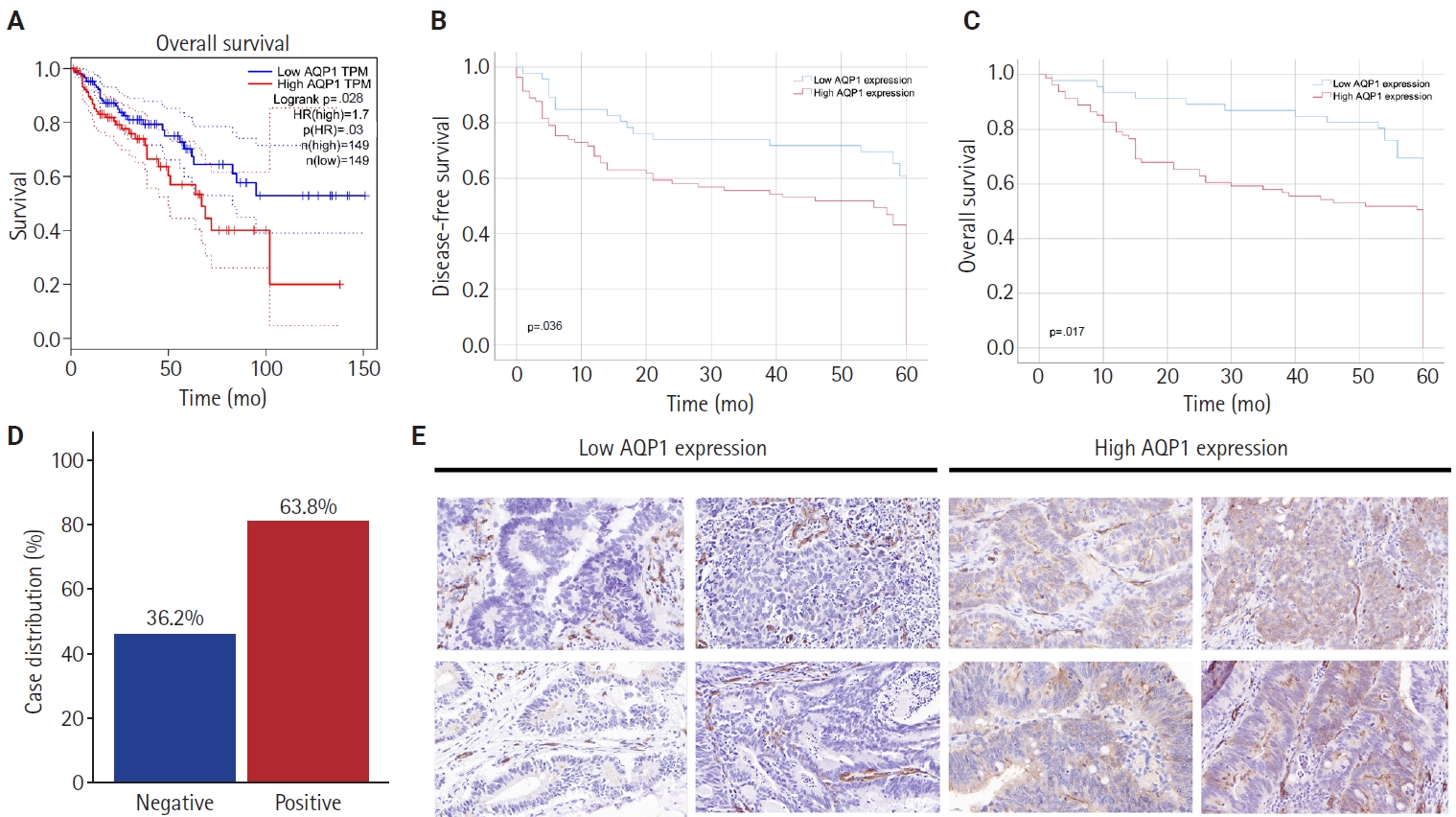
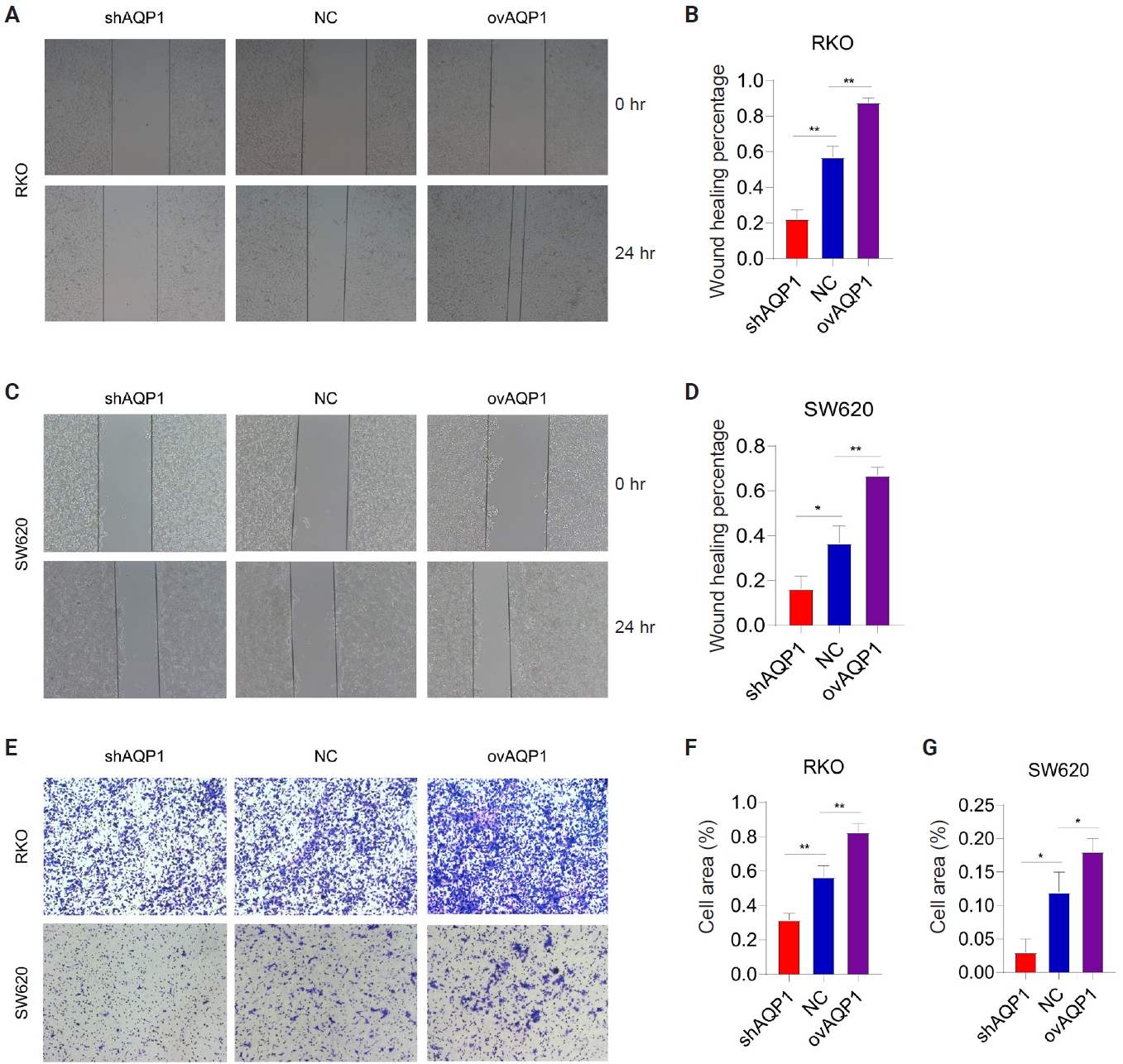
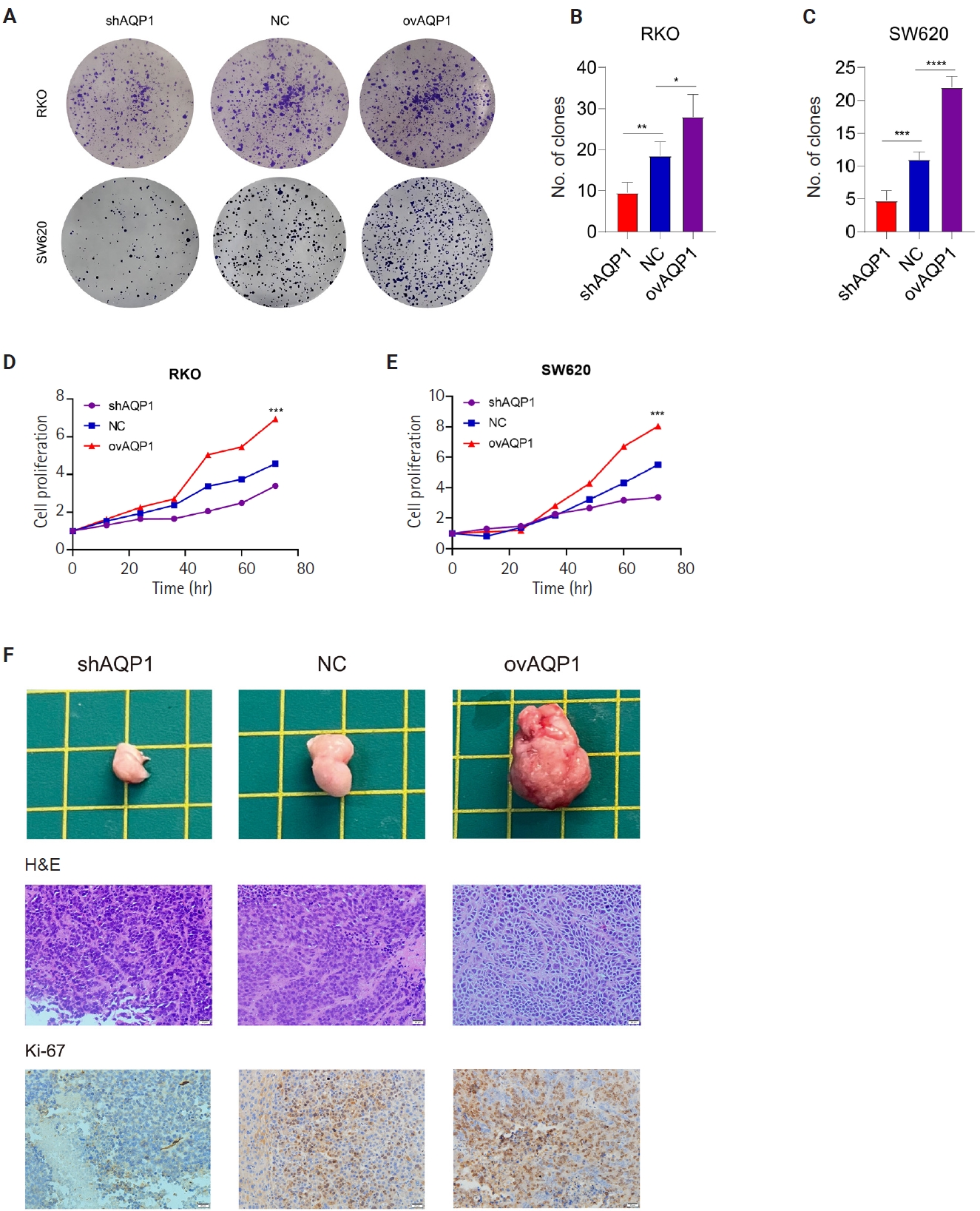
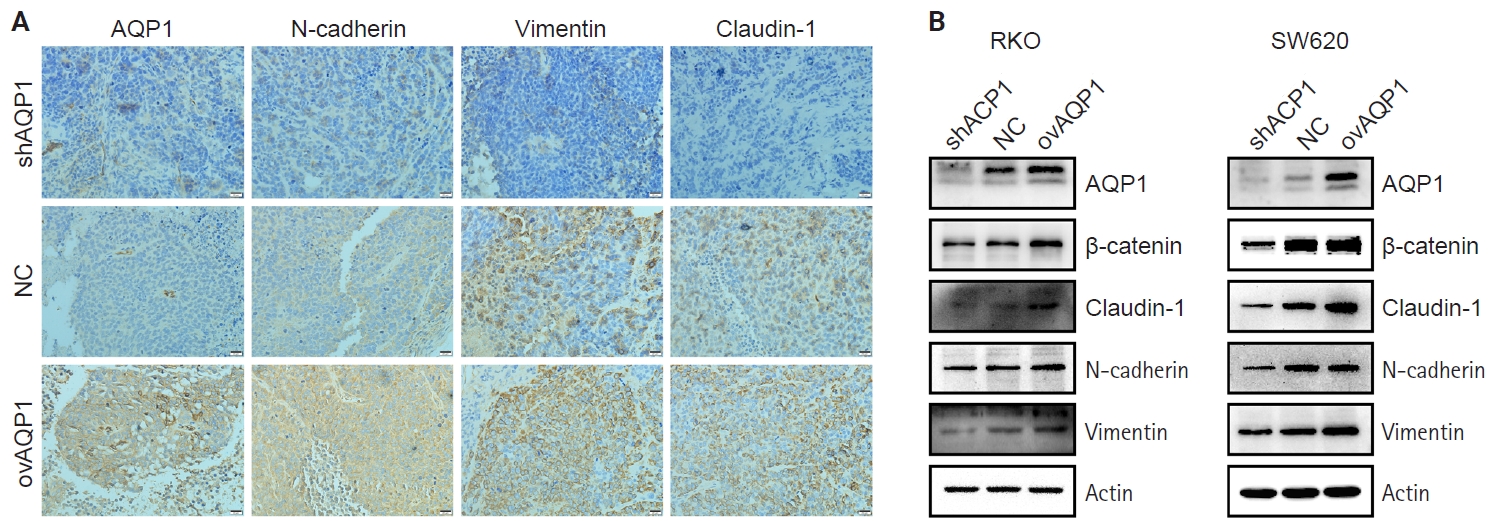
Fig. 1.
Fig. 2.
Fig. 3.
Fig. 4.
Graphical abstract
| No. | TNM stage | F value | p-value | ||||
|---|---|---|---|---|---|---|---|
| Ⅰ | Ⅱ | Ⅲ | Ⅳ | ||||
| AQP1 | 298 | 9.73 ± 1.17 | 10.02 ± 1.37 | 10.17 ± 1.17 | 10.77 ± 1.03 | 7.935 | <.001 |
| AQP2 | 176 | 0.87 ± 0.49 | 1.35 ± 0.90 | 1.41 ± 1.20 | 1.47 ± 0.84 | 2.364 | .073 |
| AQP3 | 298 | 8.46 ± 1.87 | 8.19 ± 1.68 | 8.35 ± 1.86 | 8.43 ± 1.76 | 0.373 | .773 |
| AQP4 | 142 | 0.94 ± 0.72 | 1.05 ± 0.72 | 1.11 ± 0.77 | 1.42 ± 1.67 | 1.196 | .314 |
| AQP5 | 276 | 2.84 ± 2.80 | 3.14 ± 2.86 | 3.62 ± 3.21 | 3.02 ± 2.91 | 0.762 | .516 |
| AQP6 | 286 | 2.74 ± 1.33 | 2.77 ± 1.36 | 3.05 ± 1.47 | 2.98 ± 1.36 | 0.844 | .471 |
| AQP7 | 298 | 4.19 ± 1.22 | 4.27 ± 1.65 | 4.54 ± 1.51 | 4.84 ± 1.68 | 2.378 | .070 |
| AQP8 | 270 | 4.78 ± 3.40 | 5.07 ± 3.87 | 5.32 ± 3.78 | 4.94 ± 3.70 | 0.206 | .892 |
| AQP9 | 298 | 4.91 ± 2.43 | 5.41 ± 2.35 | 5.37 ± 2.33 | 5.58 ± 2.04 | 0.790 | .501 |
| AQP10 | 156 | 1.32 ± 1.15 | 1.19 ± 0.89 | 1.43 ± 0.92 | 1.10 ± 0.79 | 0.916 | .435 |
| AQP11 | 298 | 5.66 ± 0.73 | 5.73 ± 0.91 | 5.56 ± 0.81 | 5.55 ± 0.83 | 0.925 | .429 |
| AQP12 | 288 | 3.58 ± 1.61 | 3.81 ± 1.81 | 4.01 ± 1.73 | 4.07 ± 1.59 | 0.933 | .425 |
| Factor | Total (n = 127) | AQP1 expression | p-value | |
|---|---|---|---|---|
| High (n = 81) | Low (n = 46) | |||
| Sex (M/F) | 66/61 | 44/37 | 22/24 | .481 |
| Age (yr) | 127 | 60.57 ± 12.59 | 63.41 ± 12.38 | .220 |
| Hemoglobin (g/L) | 127 | 105.80 ± 24.05 | 104.37 ± 25.79 | .754 |
| CEA | 102 (68/34) |
13.37 ± 36.06 | 11.57 ± 21.62 | .790 |
| CA19-9 | 99 (65/34) |
23.29 ± 35.26 | 19.85 ± 20.61 | .602 |
| CA72-4 | 98 (65/33) |
5.82 ± 9.77 | 4.07 ± 5.57 | .345 |
| Intestinal obstruction | .087 | |||
| No | 110 (86.6) | 67 (82.7) | 43 (93.5) | |
| Yes | 17 (13.4) | 14 (17.3) | 3 (6.5) | |
| Intestinal stenosis | .128 | |||
| No | 41 (32.3) | 30 (37.0) | 11 (23.9) | |
| Yes | 86 (67.7) | 51 (63.0) | 35 (76.1) | |
| Hydroperitoneum | .242 | |||
| No | 110 (86.8) | 68 (84.0) | 42 (91.3) | |
| Yes | 17 (13.4) | 13 (16.0) | 4 (8.7) | |
| Location | .034 | |||
| Right colon | 71 (55.9) | 51 (63.0) | 20 (43.5) | |
| Left colon | 56 (44.1) | 30 (37.0) | 26 (56.5) | |
| Tumor differentiation | .020 |
|||
| Poorly | 19 (15.0) | 17 (21.0) | 2 (4.3) | |
| Moderately | 106 (83.5) | 63 (77.8) | 43 (93.5) | |
| Well | 2 (1.6) | 1 (1.2) | 1 (2.2) | |
| Perineural invasion | .001 | |||
| No | 43 (33.9) | 19 (23.5) | 24 (52.2) | |
| Yes | 84 (66.1) | 62 (76.5) | 22 (47.8) | |
| Lymphatic invasion | .021 | |||
| No | 80 (63.0) | 45 (55.6) | 35 (76.1) | |
| Yes | 47 (37.0) | 36 (44.4) | 11 (23.9) | |
| Vascular thrombosis | .004 | |||
| No | 56 (44.1) | 28 (34.6) | 28 (60.9) | |
| Yes | 71 (55.9) | 53 (65.4) | 18 (39.1) | |
| TNM stage | .027 |
|||
| I | 6 (4.7) | 3 (3.7) | 3 (6.5) | |
| Ⅱ | 70 (55.1) | 38 (46.9) | 32 (69.6) | |
| Ⅲ | 41 (32.3) | 31 (38.3) | 10 (21.7) | |
| Ⅳ | 10 (7.9) | 9 (11.1) | 1 (2.2) | |
Values are presented as mean ± SD. AQP, aquaporin; TCGA, The Cancer Genome Atlas; SD, standard deviation.
Values are presented as mean ± SD or number (%). AQP1, aquaporin 1; CEA, carcinoembryonic antigen; CA19-9, glycoconjugate antigen 19-9; CA72-4, glycoconjugate antigen 72-4; SD, standard deviation. Numbers in parentheses indicate cases with high AQP1 expression/low AQP1 expression; Fisher’s exact test; statistically significant differences (p < .05).

 E-submission
E-submission