Articles
- Page Path
- HOME > J Pathol Transl Med > Forthcoming articles > Article
-
Case Study
Multidimensional analysis of concurrent proximal bronchiolar adenoma and lung carcinoma -
Lu-Yao Li1,*
 , Gong-Ming Dong2,*
, Gong-Ming Dong2,* , Yun-Peng Zhang1
, Yun-Peng Zhang1 , Ting-Ting Wang1
, Ting-Ting Wang1 , Fu-Quan Jia3
, Fu-Quan Jia3 , Guan-Jun Zhang1
, Guan-Jun Zhang1
-
DOI: https://doi.org/10.4132/jptm.2025.12.31
Published online: March 23, 2026
1Department of Pathology, The First Affiliated Hospital of Xi’an Jiaotong University, Xi’an, China
2Health Science Center of Xi'an Jiaotong University, Xi’an, China
3College of Basic Medical Science, Inner Mongolia Medical University, Hohhot, China
-
Corresponding Author: Guan-Jun Zhang, PhD Department of Pathology, The First Affiliated Hospital of Xi’an Jiaotong University, No. 277 Yanta West Road, 710061, Xi’an, China Tel, Fax: +86-029-85323251, E-mail: zgjxjtu@163.com
Fu-Quan Jia, PhD College of Basic Medical Science, Inner Mongolia Medical University, No. 5 Xinhua Street, 010059, Hohhot, China Tel, Fax: +86-0471-6636017, E-mail: jiafuquan915@163.com - *Lu-Yao Li and Gong-Ming Dong contributed equally to this work.
© The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (https://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 477 Views
- 55 Download
Abstract
- Bronchiolar adenoma (BA) is a rare type of lung tumor characterized by bilayered epithelial cells having a continuous basal layer and a luminal layer. It resembles mucinous adenocarcinoma (MA) on frozen section, with difficulty in distinguishing the basal layer. Immunohistochemistry is the best choice for verifying the diagnosis. This study aimed to comprehensively characterize three cases of BA-combined carcinoma using clinical, histopathological, and genetic features. BA and carcinoma sections were subjected to next-generation sequencing, respectively. It was hypothesized that while different mutation forms matched different regions, BA and lung adenocarcinoma shared the same gene mutation when they co-occurred in the same location. BA with extensive carcinoma is extremely rare and presents diagnostic challenges due to its overlap with conditions such as MA. Because of its distinctive morphological characteristics, BA may be regarded as a low-grade malignancy, particularly during a confusing evaluation. A multifaceted examination of clinical, radiological, immunohistochemical, and genetic data is necessary for an accurate diagnosis.
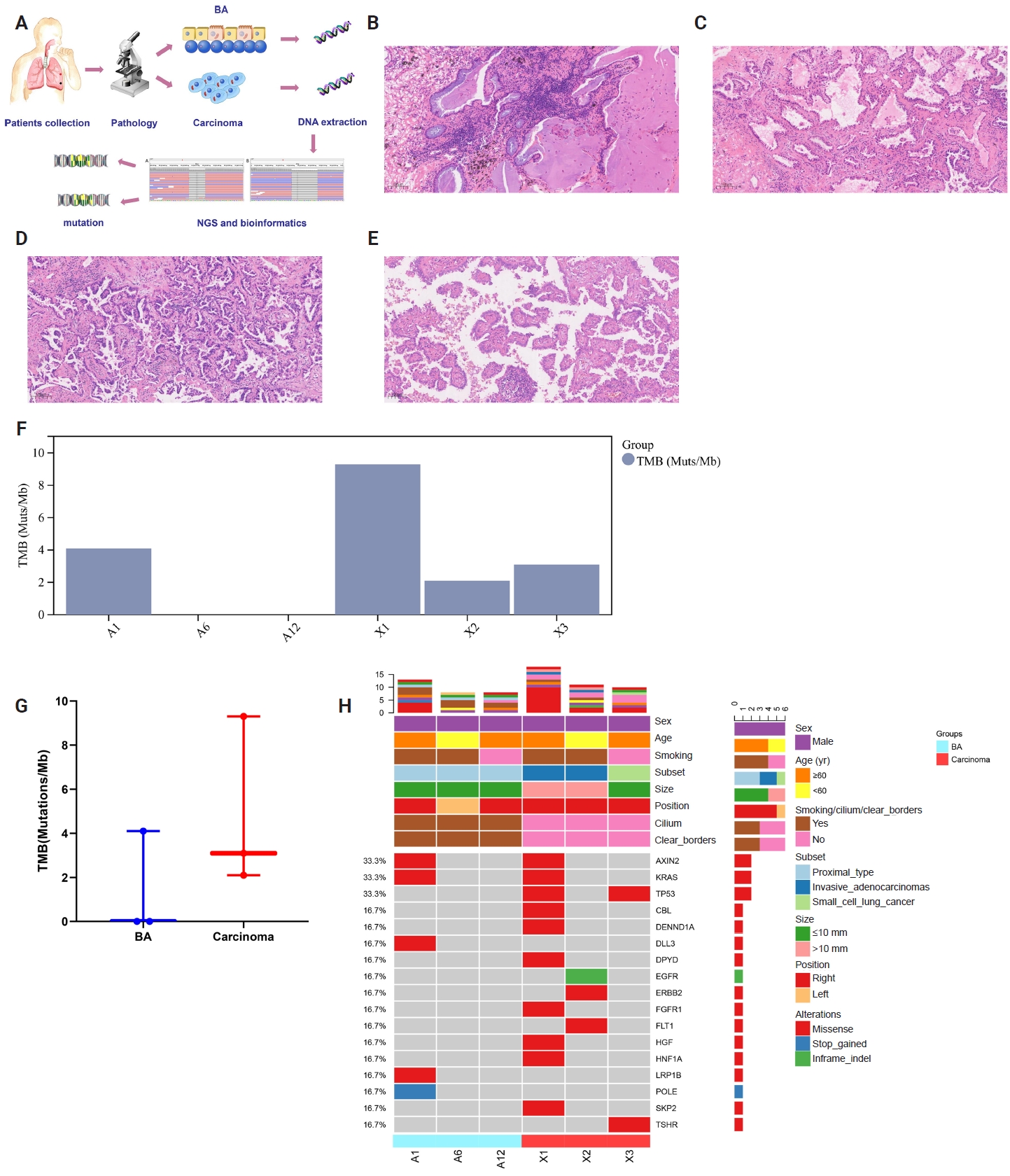
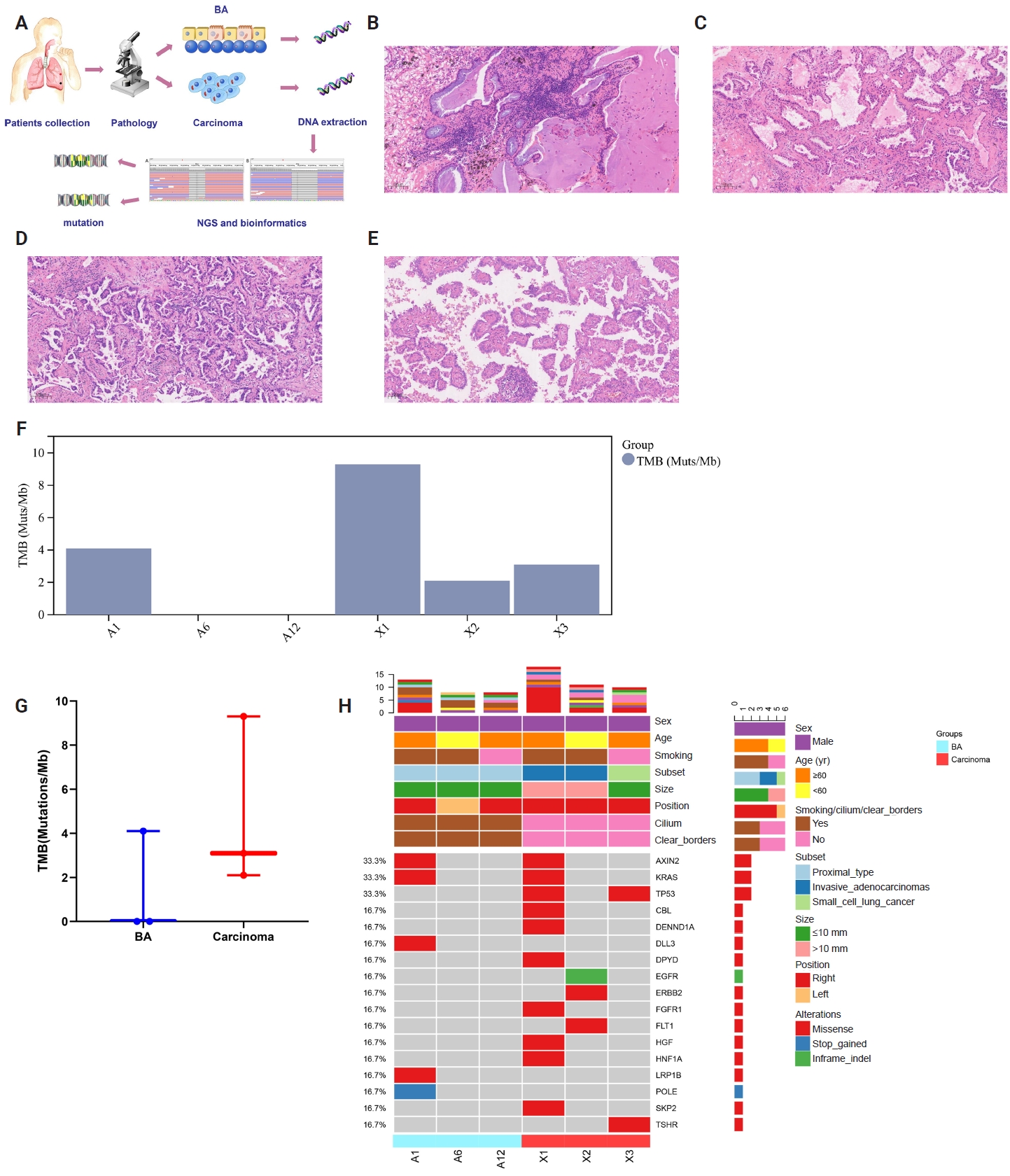
- Bronchiolar adenomas (BAs) have recently been recognized as a group of apparently benign neoplasms arising from the epithelial cell components of pulmonary bronchioles [1]. In 2021, BA or ciliated muconodular papillary tumor was defined in the 5th World Health Organization based on the characteristic bilayered architecture and a continuous basal cell layer [2]. Based on the degree of similarity to the cell proportion of bronchioles, BA was divided into proximal and distal types [2]. The continuous basal layer is a reliable histological characteristic for differentiating BA from carcinoma. Additionally, the presence of ciliated cells in frozen tissue sections consistently indicates BA rather than carcinoma [3]. During subsequent follow-ups, given its favorable prognosis, including the absence of lymph node or distant metastases in earlier studies, researchers believe that BA should be a benign tumor [4]. The possibility of BA turning into a malignant tumor could not be ruled out yet. Rapid and accurate postoperative discrimination of BA based on our proposal is crucial and will help prevent needless treatment. Additionally, no report has yet confirmed whether BA and carcinoma were accidentally mixed or had a common origin when they coexisted in the same patient. In practice, pathologists frequently misdiagnose and underdiagnose BA [5]. Different morphological characteristics of lung tumors may be identified using histologic and genetic features to presumably reveal biological behavior, transformation, and prognosis [6]. In this study, we recruited three bilayered BA cases combined with carcinoma, all of which had continuous basal cells. To determine whether the coexistence of BA and lung carcinoma suggests the potential for malignant differentiation of BA and the formation of a new specific subtype, we seek to identify the multifaceted similarities and differences between BA and carcinoma components. Investigation of molecular features, in particular, is a prerequisite for examining the relationship between BA and carcinoma, as well as possible mechanisms of tumor progression. Comprehensive evaluation of clinical, morphologic, immunohistochemical, and molecular characteristics was conducted (Fig. 1A).
INTRODUCTION
- Case 1
- A 63-year-old male was asymptomatic and diagnosed incidentally with nodules during physical examination. The patient had a smoking history of 30 years with no family history of lung cancer. Radiologically, computed tomography (CT) revealed mixed density of nodules in the lower lobe of the right lung and the upper lobe of the left lung. No pleural effusion was observed. Complete blood count and serum tumor markers were normal. However, pulmonary function tests revealed a drop in the oxygenation index to 249 mmHg (reference range, 400–500 mmHg). Echocardiography, head CT scan, and abdominal ultrasound displayed no abnormalities. The patient underwent thoracoscopic resection of the right lung's lower lobe and frozen section analysis following the completion of preoperative evaluations.
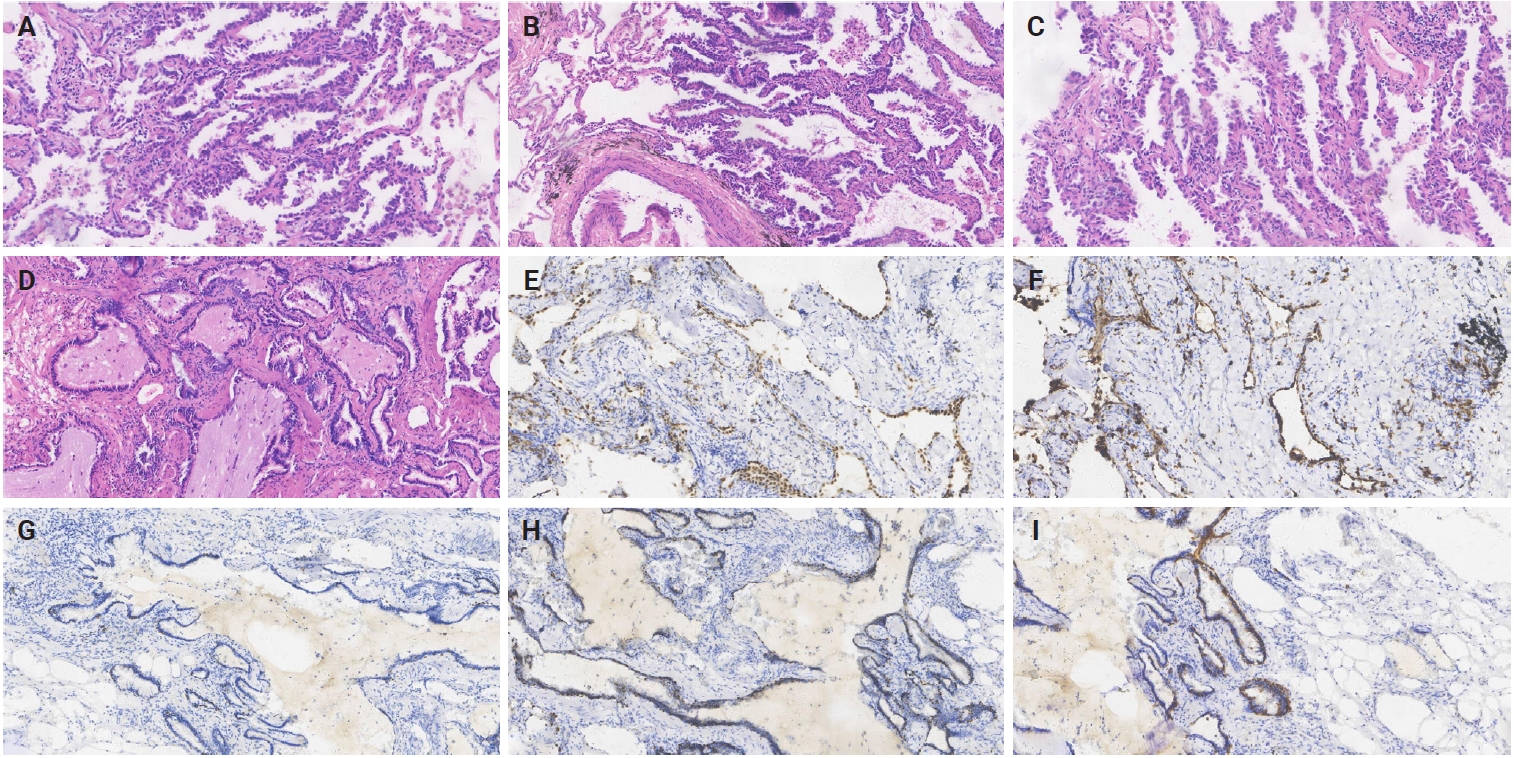
- The dimensions of the removed specimen were 10 × 8 × 3 cm. In the lung's periphery, a gray-white nodular mass (2.7 × 2.5 × 1.6 cm) was observed, invading the local pleura. A gray-red nodule (5 mm) was inadvertently found near the mass during sampling. The distance between the two was 1.5 cm. Lung adenocarcinoma or BA was first suggested based on intraoperative frozen-section analysis. The postoperative pathological findings revealed the coexistence of BA and invasive adenocarcinoma. BA was mainly flat with a few papillary structures, according to histological analysis. Under a microscope, BA displayed two distinct epithelial layers with distinct cell component ratios. Prominent mucinous cells comprised the luminal cell layer (Fig. 1B). Notably, the tumor depicted significant destruction of alveolar structure, and interstitial fibrous tissue proliferation was observed. B-Raf proto-oncogene, serine/threonine kinase (BRAF) and mucin 5AC (MUC5AC) were not expressed, according to immunohistochemistry (IHC) staining. In this instance, the invasive adenocarcinoma region displayed structures with 50% acinar (Fig. 1C), 40% papillary (Fig. 1D), and 10% micropapillary (Fig. 1E).
- To identify molecular characteristics, we compared the genetic changes between the concurrent primary carcinoma and BA. Supplementary Tables S1–S3 present the genetic alterations that were discussed. Out of 15 changes found in this study, 13 were unique (Supplementary Table S4). The most common mutations found in BA and invasive adenocarcinoma were Kirsten rat sarcoma viral oncogene homolog (KRAS) G12C and Axin family member 2 (AXIN2). Furthermore, only invasive adenocarcinoma harbored a TP53 mutation. To some extent, tumor mutation burden (TMB) was accurately reflected by single-nucleotide mutations and insertions or deletions (Indels). TMB was 9.3 in carcinoma and 4.1 in BA (Fig. 1F). The patient was diagnosed with invasive adenocarcinoma without pleural invasion after undergoing a thoracoscopic resection of the upper lobe of the right lung more than four months later. After 23 months, the patient exhibited no signs of metastasis or postsurgical recurrence.
- Case 2
- A 58-year-old male was admitted with abnormal lung CT manifestations. The patient had smoked for 40 years without experiencing any respiratory problems. Neuron-specific enolase (NSE) and cytokeratin 19-fragments (CYFRA21-1), two serum tumor markers, were marginally elevated. There was a slight decrease in the oxygenation index (377 mmHg). A chest CT scan revealed a fibrous rope band in the left upper lung lobe and a 14 × 18 mm nodular shadow with a spicule sign in the right upper lung lobe. He had a bilateral pulmonary wedge resection after evaluation.
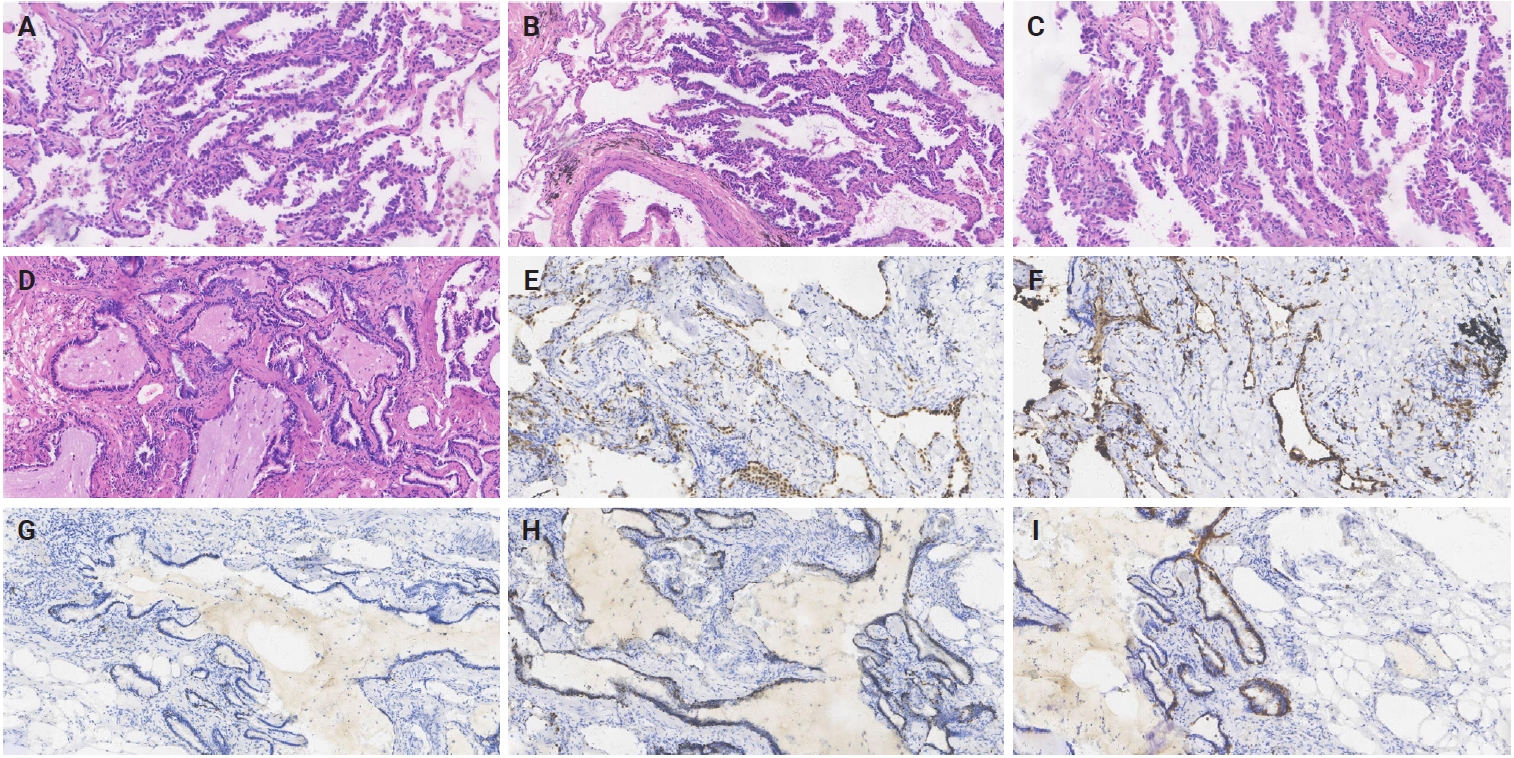
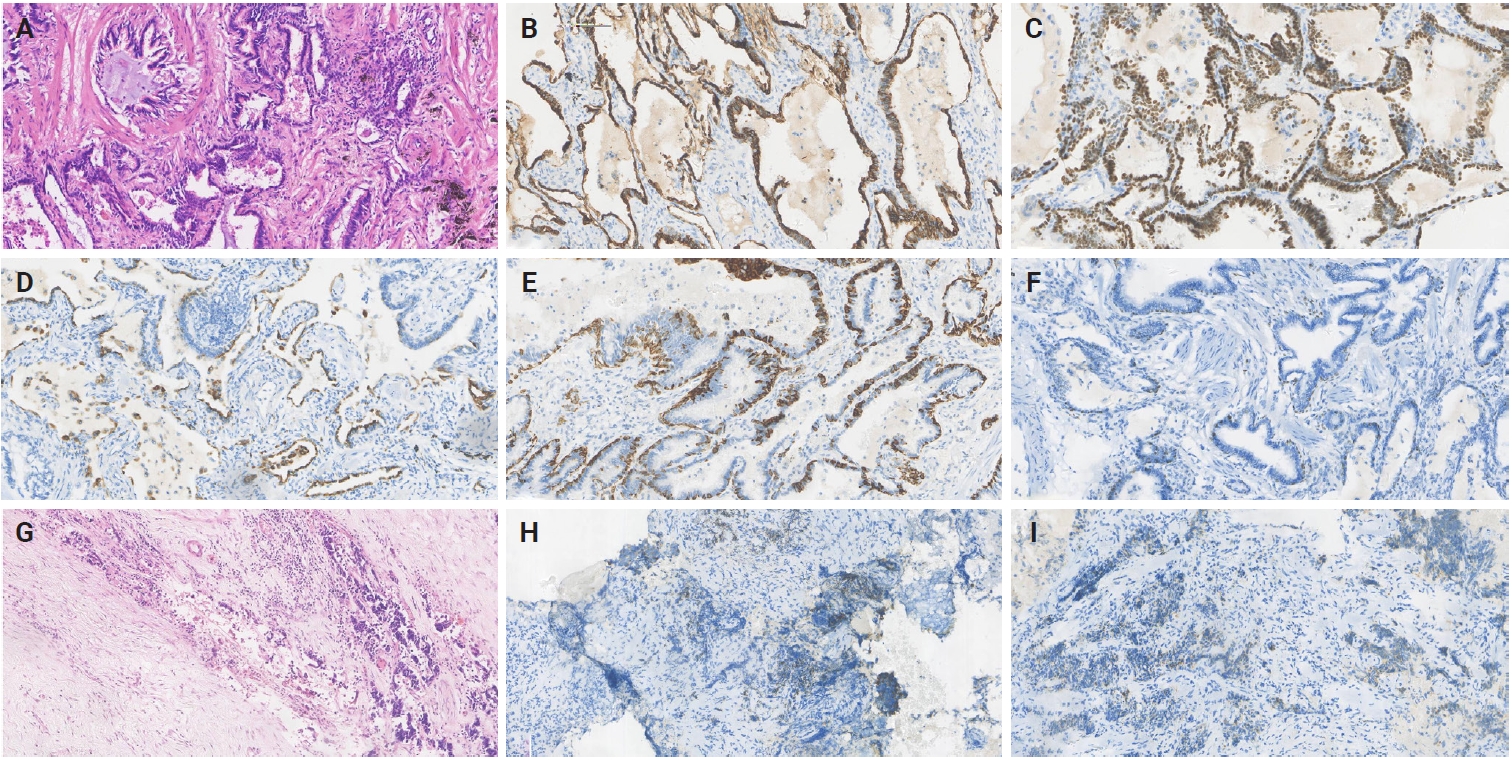
- The left upper lung lobe of the wedge-resected specimen had a gray nodule (10 × 6 × 5 mm). In comparison, the right upper lung lobe had a mass (17 × 14 × 10 mm). The 60% acinar (Fig. 2A), 20% papillary (Fig. 2B), and 20% adherent subtype (Fig. 2C) were found in the invasive adenocarcinoma region. The absence of cilia and basal cells, cell atypia, and an infiltrating interstitial reaction characterize invasive adenocarcinoma. The luminal epithelium inside the bilayered BA was positive for thyroid transcription factor-1 (TTF-1) (Fig. 2E), napsin A (Fig. 2F), p40 (Fig. 2G), p63 (Fig. 2H), and cytokeratin (CK) 5/6 (Fig. 2I) expressed in basal cells. BA was discovered by accident and lined with a spot of mucinous cells with abundant mucus in alveoli (Fig. 2D). MUC5AC and BRAF were negative. A pathology diagnosis of proximal-type BA was made based on the presence of numerous ciliated columnar cells and fragments of mucus cells in the luminal cell layer, as well as positivity for p63, p40, and CK5/6 in the basal cells, which verifies the preservation of the continuous basal cell architecture. KRAS, epidermal growth factor receptor (EGFR), anaplastic lymphoma kinase (ALK), c-ros oncogene 1, receptor tyrosine kinase (ROS1), BRAF, and other genes were not mutated or fused in BA. Only the invasive adenocarcinoma component contained EGFR (exon 19 deletion), Fms-like tyrosine kinase 1 (FLT1), and Erb-B2 receptor tyrosine kinase 2 (ERBB2). TMB was 2.1 in invasive adenocarcinoma and 0 in BA (Fig. 1F). After a 24-month follow-up, the patient made a full recovery with no distant metastases or recurrence.
- Case 3
- A 71-year-old male was admitted for chest tightness. Other clinical symptoms, such as cough or chest pain, were absent. The patient had no prior history of cancer or smoking. A routine CT scan revealed multiple fiber rope bands in both lungs and a 10 mm × 10 mm anterior basal segment nodule in the right lower lung lobe. No anomalies were found in the remaining tests, except for CYFRA21-1 and NSE, which were marginally elevated. He underwent surgery without frozen section pathology following laboratory testing and other preoperative examinations.
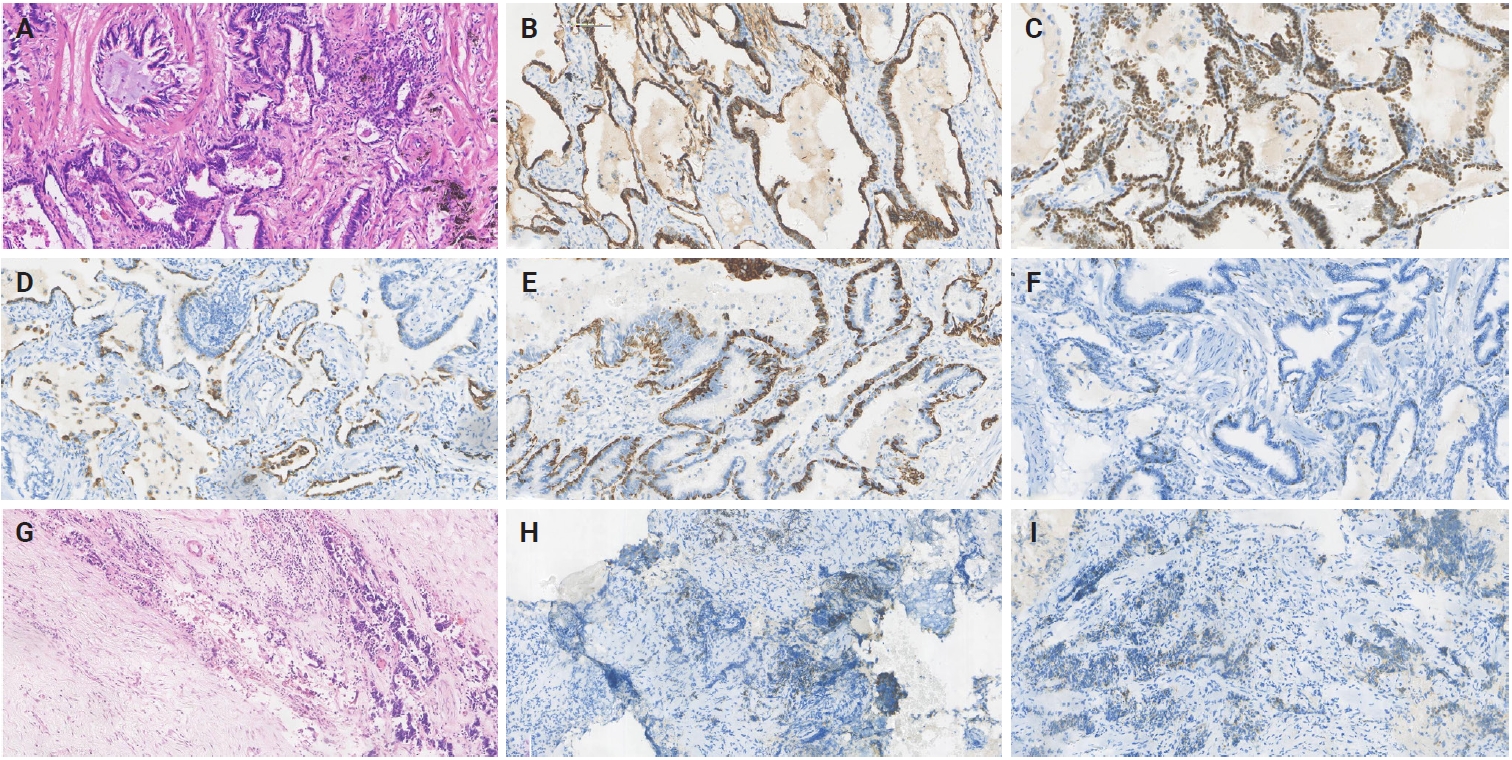
- The removed specimen consisted of right middle and lower lobes of the lung, measuring 90 × 80 × 30 mm. A calcified nodule (10 mm) and a gray-red solid area (10 mm × 10 mm × 3 mm) were noted grossly, and the microscopic examination revealed the tumor to be composed of flat luminal and basal cell. The cellular constituents of the luminal epithlium were mucinous cells and ciliated columnar cells. Some BA foci displayed central fibrosis and focal lymphocyte infiltration (Fig. 3A). Notably, the tumor cells displayed no signs of necrosis, mitosis, or atypia. IHC revealed that luminal cells were negative for CK5/6, p40, and p63 but positive for CK7 (Fig. 3B), TTF-1 (Fig. 3C), and napsin A (Fig. 3D). Basal cells were negative for BRAF and MUC5AC and expressed CK5/6 (Fig. 3E) and p40 (Fig. 3F). Small cell lung cancer (SCLC) was identified and elaborated in Fig. 3G, which invaded the peripheral lung tissue and the entire bronchial wall layer without invading the lung pleura. Strong and widespread positive expression of tumor cells in SCLC was demonstrated by CgA (Fig. 3H) and Syn (Fig. 3I). Next-generation sequencing also depicts that TP53 and TSHR were detected in BA, but no mutations or fusions were found. TMB was 3.3 in SCLC and 0 in BA (Fig. 1F). These patients' carcinoma components clearly had comparatively higher TMB, but there was no statistically significant difference (p = .259) (Fig. 1G). Overall description of the mutations were provided in Fig. 1H. The patient was lost to follow-up after 61 months of survival without metastasis or recurrence.
CASE REPORT
- The characteristic double-layered epithelial cells and a continuous layer of basal cells are the main features of BA, a recently identified benign or potentially malignant lung tumor [7]. Lately, BA has been recognized as a distinct entity, setting it apart from other pulmonary lesions and lung carcinoma [8]. According to earlier research, the most reliable histological indicator for distinguishing BA from carcinoma is a continuous layer of basal cells [1,5]. Pathogenesis analysis has not yet revealed a clear correlation between BA and a specific histological subtype of lung carcinoma [9]. Case reports of BA are still uncommon in the current study, and BA coexisting with lung carcinoma are even less common. The current report provides a thorough analysis of radiological, morphological, and molecular features in patients with BA combined with lung carcinoma, based on the compared studies.
- Accurate differential diagnosis and thorough and methodical understanding of BA are essential. Pathology has been the primary focus of research on BA, and its clinical and imaging features still remain poorly understood. Most elderly, lethargic individuals with BA do not exhibit symptoms like coughing, expectoration, or chest pain. Therefore, regular follow-up is crucial. BA shows various imaging features, most of which are irregular peripheral solitary nodules (≤2 cm) with distinct boundaries [10]. They can occasionally be misdiagnosed as adenocarcinoma in situ due to shallow lobulation or ground-glass components, features that overlap with the early signs of lung adenocarcinoma [10,11]. Additionally, thin-slice CT examinations frequently depict BA with pseudocavity formation; therefore, imaging observations must be closely monitored for early warning signs of malignant transformation, such as pleural traction or necrosis [11].
- The well-known histologic characteristics of BA are flat, glandular, or papillary. To confirm the basal layer by IHC, a bilayered structure consisting of a continuous basal cell layer and a luminal cell layer is often required. The basal layer displays positive expression of common basal cell markers, including CK5/6, p40, and p63. Interestingly, lung carcinomas had a relatively high Ki-67 proliferating index (10%–40%), whereas BAs had a low Ki-67 proliferating index (5%–10%). However, Ki-67 could not be used as a criterion to distinguish between benign and malignant tumors. IHC is therefore an essential tool for differential diagnosis [12]. When cilia are clearly visible, even diagnosis on frozen section can be straightforward. It is very uncommon for a malignant tumor to show cilia in the lung. Histologically, the presence of cilia and a basal cell layer is the main distinguishing feature of BA from carcinoma, and the luminal cells in BA typically consist of varying proportions of mucinous and ciliated cells. Based on the aforementioned features, BA was artificially separated into proximal and distal types [5]. A portion of distal-type BAs may develop into adenocarcinoma, according to several studies [5,6]. In contrast, BA, mixed-type BAs with monolayered lesions, and monolayered BA-like lesions displayed progressive loss of a continuous basal layer, morphologically approaching adenocarcinoma, suggesting the possibility of a malignant transition from BA to carcinoma [10,13]. The morphology of bilayered BA in these three proximal cases with carcinoma components suggests a potential for malignant transformation. While basal cells and mild cell morphology indicated that bilayered BA is probably benign [14], low-grade malignant features, such as central fibrosis and focal lymphocyte infiltration, were somewhat similar to those of lung adenocarcinoma. Intriguingly, BA and SCLC exhibit few histological similarities, suggesting that their tissue origins differ. The histological origin of BA may be closer to that of invasive adenocarcinoma, and the co-occurrence of SCLC and BA is thought to be coincidental.
- We are now considering whether the bilayered structures represent adenocarcinoma extending to bronchiolar epithelium without total loss of basal cells in light of the two cases of bilayered BA with malignant transformation. Consequently, it is not possible to classify classical bilayered BA as benign tumors. The genetic changes in BA were first identified and described by Liu et al. [15]. The most prevalent mutational feature in BA (38%) is the BRAF V600E mutation. EGFR mutation [10,16], KRAS mutation [10,17], AKT1 mutation [17], ALK gene rearrangement [18,19], and other molecular profiles have also been reported. They are all relatively common in lung cancer, suggesting that there are some genetic similarities between BA and lung cancer. The same KRAS G12C and AXIN2 mutations, which are frequently found in invasive adenocarcinomas [19], were identified in both sections of case 1, suggesting that the adenocarcinomas and BAs may have shared a similar genetic background. The aforementioned findings might lend credence to BA's malignant transformation. Simultaneously, one of the current cases revealed EGFR 19del in the carcinoma component to be distinct from the BA, suggesting that the pathogenesis of BA and adenocarcinoma may differ (case 2). Based on the molecular analysis, we hypothesized that if lung adenocarcinoma and BA are found in the same focus, they might share a driver gene mutation (case 1) [9]; on the other hand, if BA and adenocarcinoma co-occur at different locations, they might have different forms (case 2). We hypothesize that the inconsistent genetic detection of BA and adenocarcinoma in distinct foci can be attributed to two factors. First, gene mutations may accompany tumor cells of the same origin during development and metastasis, which encourages the formation of cells with different histological manifestations. The same genetic mutation was found in both bilayered BA and adenocarcinoma, confirming the same cell origin and the potential for BA to undergo malignant transformation. Second, distinct cell types are produced by the unique genetic profiles of tissue cells from various sources. In terms of non-fixed loci, it is hypothesized that mutations may have occurred in BA linked to lung carcinoma in the two cases (cases 2 and 3). Variant allele frequencies between the two components were AXIN2 (33.3% in BA, 33.3% in carcinoma), KRAS (33.3% in BA, 33.3% in carcinoma), TP53 (0% in BA, 66.7% in carcinoma), CBL (0% in BA, 33.3% in carcinoma), DENND1A (0% in BA, 33.3% in carcinoma), DLL3 (33.3% in BA, 0% in carcinoma), DPYD (0% in BA, 33.3% in carcinoma), EGFR (0% in BA, 33.3% in carcinoma), ERBB2 (0% in BA, 33.3% in carcinoma), FGFR1 (0% in BA, 33.3% in carcinoma), FLT1 (0% in BA, 33.3% in carcinoma), HGF (0% in BA, 33.3% in carcinoma), HNF1A (0% in BA, 33.3% in carcinoma), LRP1B (33.3% in BA, 0% in carcinoma), POLE (33.3% in BA, 0% in carcinoma), SKP2 (0% in BA, 33.3% in carcinoma), and TSHR (0% in BA, 33.3% in carcinoma).
- As in our study, BA can be found coincidentally in lung carcinomas. Treatment guidelines for carcinoma can serve as a starting point. Nevertheless, during long-term follow-ups, none of the three cases under study has experienced metastasis or postsurgical recurrence. BA with carcinoma appeared to be low-grade malignant. Therefore, unlike classical bilayered BA and lung adenocarcinoma, the case with BA and malignant components may be an independent variant with unique histological and molecular variation. The invasiveness and malignancy of carcinoma may be diminished as the tumor progresses due to the addition of BA components in this particular variant. Notably, due to sample limitations, no comparison between proximal and distal types was performed, and all BAs with carcinoma in the current study were proximal.
- In conclusion, histological and genetic evidence for the possible malignant transformation of BAs was presented by both this study and recent case reports [20]. The findings in our study indicate that BA is associated with malignant differentiation, particularly when co-occurring with carcinoma. Genetics and histopathology remain essential diagnostic methods. Future research on the clinical and biological significance of specific alterations and related changes is necessary, given the small sample size. To determine whether BA is a possible precursor to carcinoma based on its long-term prognosis, further research is required.
DISCUSSION
Supplementary Information
Ethics Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Ethics Committee at the First Affiliated Hospital of Xi'an Jiaotong University (No. XJTU1AF2024LSYY-472 and date of approval is December 6, 2024). Informed consent was obtained from the individuals for the publication of any potentially identifiable data included in this article.
Availability of Data and Material
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
Code Availability
Not applicable.
Author Contributions
Conceptualization: GJZ. Data curation: LYL, GMD, TTW. Supervision: GJZ, FQJ. Writing—original draft: LYL, GMD, YPZ. Writing—review & editing: all authors. Approval of final manuscript: all authors.
Conflicts of Interest
The authors declare that they have no potential conflicts of interest.
Funding Statement
This study was supported by the Xi'an Jiaotong University Medical Development Fund (No. XJYG2025-SFJJ011).
- 1. Kao T, Yeh Y. Ciliated muconodular papillary tumor/bronchiolar adenoma of the lung. Semin Diagn Pathol 2021; 38: 62-71. ArticlePubMed
- 2. Nicholson AG, Tsao MS, Beasley MB, et al. The 2021 WHO classification of lung tumors: impact of advances since 2015. J Thorac Oncol 2022; 17: 362-87. ArticlePubMed
- 3. Kamata T, Yoshida A, Kosuge T, Watanabe S, Asamura H, Tsuta K. Ciliated muconodular papillary tumors of the lung: a clinicopathologic analysis of 10 cases. Am J Surg Pathol 2015; 39: 753-60. ArticlePubMed
- 4. Yang C, Wang X, Da J, Ma K. Distal-type bronchiolar adenoma of the lung harboring an EGFR exon 21 p.L858R mutation: a case report. Thorac Cancer 2020; 11: 3596-8. ArticlePubMedPMC
- 5. Chang JC, Montecalvo J, Borsu L, et al. Bronchiolar adenoma: expansion of the concept of ciliated muconodular papillary tumors with proposal for revised terminology based on morphologic, immunophenotypic, and genomic analysis of 25 cases. Am J Surg Pathol 2018; 42: 1010-26. ArticlePubMedPMC
- 6. Chen F, Ren F, Zhao H, Xu X, Chen J. Mucinous adenocarcinoma caused by cancerization from a ciliated multinodular papilloma tumor: a case report. Thorac Cancer 2021; 12: 1629-33. ArticlePubMedPMCPDF
- 7. Shao K, Wang Y, Xue Q, et al. Clinicopathological features and prognosis of ciliated muconodular papillary tumor. J Cardiothorac Surg 2019; 14: 143.ArticlePubMedPMCPDF
- 8. Shao J, Yin JC, Bao H, et al. Morphological, immunohistochemical, and genetic analyses of bronchiolar adenoma and its putative variants. J Pathol Clin Res 2021; 7: 287-300. ArticlePubMedPMCPDF
- 9. Han X, Hao J, Ding S, Wang E, Wang L. Bronchiolar adenoma transforming to invasive mucinous adenocarcinoma: a case report. Onco Targets Ther 2021; 14: 2241-6. ArticlePubMedPMCPDF
- 10. Cao L, Wang Z, Gong T, et al. Discriminating between bronchiolar adenoma, adenocarcinoma in situ and minimally invasive adenocarcinoma of the lung with CT. Diagn Interv Imaging 2020; 101: 831-7. ArticlePubMed
- 11. Li F, He Y, Yang H, et al. Pseudocavity on thin-slice CT can be a suggestion of bronchiolar adenoma: a preliminary study on 80 cases with bronchiolar adenoma of the lung. J Comput Assist Tomogr 2025; 49: 934-42. ArticlePubMedPMC
- 12. Gao Z, Feng Q, Wang Y, Jiang Y, Han D, Xu W. Case report: two cases of bronchiolar adenoma/ciliated muconodular papillary tumor characterized by significant basal cell hyperplasia and squamous metaplasia. Front Oncol 2025; 15: 1617720.ArticlePubMedPMC
- 13. Zhu M, Yang Q, Zhan S, Liu W, Guo L, Huang S. Clinicopathological analysis of bronchiolar adenoma combined with lung adenocarcinoma: report of eight cases and literature review. Histol Histopathol 2024; 39: 783-94. ArticlePubMed
- 14. Cheung FM, Guan J, Luo QG, Sihoe AD, Shen XP. Ciliated muconodular papillary tumour of the lung mimicking mucinous adenocarcinoma: a case report and literature review. Hong Kong Med J 2019; 25: 71-3. ArticlePubMed
- 15. Liu L, Aesif SW, Kipp BR, et al. Ciliated muconodular papillary tumors of the lung can occur in Western patients and show mutations in BRAF and AKT1. Am J Surg Pathol 2016; 40: 1631-6. ArticlePubMed
- 16. Zheng Q, Hou L, Shang G, et al. Frequent EGFR exon 20 insertion in the so-called peripheral-type squamous cell neoplasm of uncertain malignant potential: a variant of bronchiolar adenoma or under-recognised entity? Histopathology 2023; 83: 178-92. ArticlePubMedPDF
- 17. Udo E, Furusato B, Sakai K, et al. Ciliated muconodular papillary tumors of the lung with KRAS/BRAF/AKT1 mutation. Diagn Pathol 2017; 12: 62.ArticlePubMedPMCPDF
- 18. Jin Y, Shen X, Shen L, Sun Y, Chen H, Li Y. Ciliated muconodular papillary tumor of the lung harboring ALK gene rearrangement: case report and review of the literature. Pathol Int 2017; 67: 171-5. ArticlePubMedPDF
- 19. Di Federico A, Hong L, Elkrief A, et al. Lung adenocarcinomas with mucinous histology: clinical, genomic, and immune microenvironment characterization and outcomes to immunotherapy-based treatments and KRAS(G12C) inhibitors. Ann Oncol 2025; 36: 297-308. ArticlePubMed
- 20. Li X, Wu Y, Hui D, et al. Multiple bronchiolar adenomas with malignant transformation and CCNE1 mutation: a case report and literature review. J Cardiothorac Surg 2021; 16: 307.ArticlePubMedPMCPDF
REFERENCES
Figure & Data
References
Citations

 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure

 E-submission
E-submission





