Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 53(4); 2019 > Article
-
Original Article
Association between Expression of 8-OHdG and Cigarette Smoking in Non-small Cell Lung Cancer -
Ae Ri An
 , Kyoung Min Kim
, Kyoung Min Kim , Ho Sung Park
, Ho Sung Park , Kyu Yun Jang
, Kyu Yun Jang , Woo Sung Moon
, Woo Sung Moon , Myoung Jae Kang
, Myoung Jae Kang , Yong Chul Lee1
, Yong Chul Lee1 , Jong Hun Kim2
, Jong Hun Kim2 , Han Jung Chae3
, Han Jung Chae3 , Myoung Ja Chung
, Myoung Ja Chung
-
Journal of Pathology and Translational Medicine 2019;53(4):217-224.
DOI: https://doi.org/10.4132/jptm.2019.02.20
Published online: March 11, 2019
Department of Pathology, Research Institute of Clinical Medicine of Chonbuk National University, Biomedical Research Institute of Chonbuk National University Hospital, Research Institute for Endocrine Sciences, Chonbuk National University Medical School, Jeonju, Korea
1Department of Internal Medicine, Research Institute of Clinical Medicine of Chonbuk National University, Biomedical Research Institute of Chonbuk National University Hospital, Research Institute for Endocrine Sciences, Chonbuk National University Medical School, Jeonju, Korea
2Department of Thoracic and Cardiovascular Surgery, Research Institute of Clinical Medicine of Chonbuk National University, Biomedical Research Institute of Chonbuk National University Hospital, Research Institute for Endocrine Sciences, Chonbuk National University Medical School, Jeonju, Korea
3Department of Pharmacology, Research Institute of Clinical Medicine of Chonbuk National University, Biomedical Research Institute of Chonbuk National University Hospital, Research Institute for Endocrine Sciences, Chonbuk National University Medical School, Jeonju, Korea
- Corresponding Author Myoung Ja Chung, MD Department of Pathology, Chonbuk National University Medical School, 567 Baekje-daero, Deokjin-gu, Jeonju 54896, Korea Tel: +82-63-270-3072 Fax: +82-63-270-3135 E-mail: mjchung@jbnu.ac.kr
© 2019 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Exposure to cigarette smoking (CS) is a major risk factor for the development of lung cancer. CS is known to cause oxidative DNA damage and mutation of tumor-related genes, and these factors are involved in carcinogenesis. 8-Hydroxydeoxyguanosine (8-OHdG) is considered to be a reliable biomarker for oxidative DNA damage. Increased levels of 8-OHdG are associated with a number of pathological conditions, including cancer. There are no reports on the expression of 8-OHdG by immunohistochemistry in non-small cell lung cancer (NSCLC).
-
Methods
- We investigated the expression of 8-OHdG and p53 in 203 NSCLC tissues using immunohistochemistry and correlated it with clinicopathological features including smoking.
-
Results
- The expression of 8-OHdG was observed in 83.3% of NSCLC. It was significantly correlated with a low T category, negative lymph node status, never-smoker, and longer overall survival (p < .05) by univariate analysis. But multivariate analysis revealed that 8-OHdG was not an independent prognostic factor for overall survival in NSCLC patients. The aberrant expression of p53 significantly correlated with smoking, male, squamous cell carcinoma, and Ki-67 positivity (p < .05).
-
Conclusions
- The expression of 8-OHdG was associated with good prognostic factors. It was positively correlated with never-smokers in NSCLC, suggesting that oxidative damage of DNA cannot be explained by smoking alone and may depend on complex control mechanisms.
- Patients and follow-up
- This study evaluated NSCLC patients who underwent surgical resection between January 2007 and December 2012. Among them, 203 NSCLC had complete available medical records, original hematoxylin and eosin slides, and paraffin-embedded tissue blocks. Clinical information including smoking history and postoperative follow-up was obtained by reviewing the medical records. The pathologic diagnosis and staging were reviewed according to the 8th American Joint Committee on Cancer staging system and World Health Organization classification [7]. The classification of smoking status follows the Centers for Disease Control and Prevention guidelines, and the status categories were “Never,” “Former” (adults who have smoked at least 100 cigarettes in their lifetime, but say they currently do not smoke), or “Current” (adults who have smoked 100 cigarettes in their lifetime and currently smoke cigarettes). This study was approved by the institutional review board of Chonbuk National University Hospital (CUH 2017-09-029-001). All samples derived from the National Biobank of Korea were obtained with informed consent under institutional review board-approved protocols.
- Immunohistochemical staining and scoring
- Formalin-fixed paraffin-embedded tissue of NSCLC patients were used to establish tissue microarrays. Original hematoxylin and eosin slides were reviewed, and one 3.0-mm-sized core from the representative area was taken. The tissue sections were deparaffinized and boiled in an antigen retrieval solution (citrate, pH 6.0) for 20 minutes in a cooker. The primary antibodies for 8-OHdG (1:200, N45.1, JaiCA, Shizuoka, Japan), p53 (1:800, DO-7, Leica, London, UK), and Ki-67 (1:100, MIB-1, Dako, Glustrup, Denmark) were used for immunohistochemical staining. The slides that had been stained for 8-OHdG, p53, and Ki-67 were evaluated by two pathologists (M.J.C. and A.R.A.) who had no clinicopathologic information on the patients. Immunohistochemical staining was evaluated to estimate the nuclear positivity of tumor cells for each marker, and the stained area was semiquantitatively recorded for the area of positive cells in 5% increments.
- Immunohistochemical staining for 8-OHdG and Ki-67 was considered positive when ≥ 5% and ≥ 10% of the tumor cells showed moderate to strong nuclear staining, respectively. The cut-off points for the immunohistochemical staining score were determined by receiver operating characteristic (ROC) curve analysis. It has recently been reported that immunohistochemical staining for p53 can be used as a surrogate for TP53 mutation analysis [8,9]. Complete negative and high expression showed strong correlation with TP53 mutation [8,9]. We categorized immunohistochemical results for p53 into three groups as negative, low (1%– 50% positively stained nuclei), and high (> 50% positively stained nuclei) as previously reported [10]. Then, p53 immunohistochemical staining was defined as “aberrant expression” in the p53 negative and high expression group and “wild type expression” in the p53 low expression group [10].
- Statistical analysis
- ROC curve analysis was used to determine the cut-off points for immunohistochemical staining. The cutoff points for 8-OHdG– and Ki-67–positivity were determined at the point with the highest area under the curve (AUC) to estimate survival of NSCLC patients. Because the ROC curve is a plot of the true positive rate (sensitivity) versus the false positive rate (1–specificity) for determination of patient death, the cutoff level for the ideal test is presented as sensitivity 1 and specificity 1 (AUC, 1.000). Therefore, we chose the cut-off point at the highest AUC value.
- The date of last follow-up was recorded as the last contact or the death of patients through August 2017. The prognosis of NSCLC patients was evaluated by analyzing overall survival (OS). The death of the patient from lung cancer was considered an event for OS analysis. SPSS software ver. 22.0 (IBM Corp., Armonk, NY, USA) was used to perform statistical analysis for the univariate and multivariate Cox proportional hazards regression analyses, Kaplan-Meier survival analysis, and Pearson’s chi-square test. The p-values less than 0.05 were considered statistically significant.
MATERIALS AND METHODS
- Correlations between 8-OHdG expression and clinicopathologic parameters of NSCLC patients
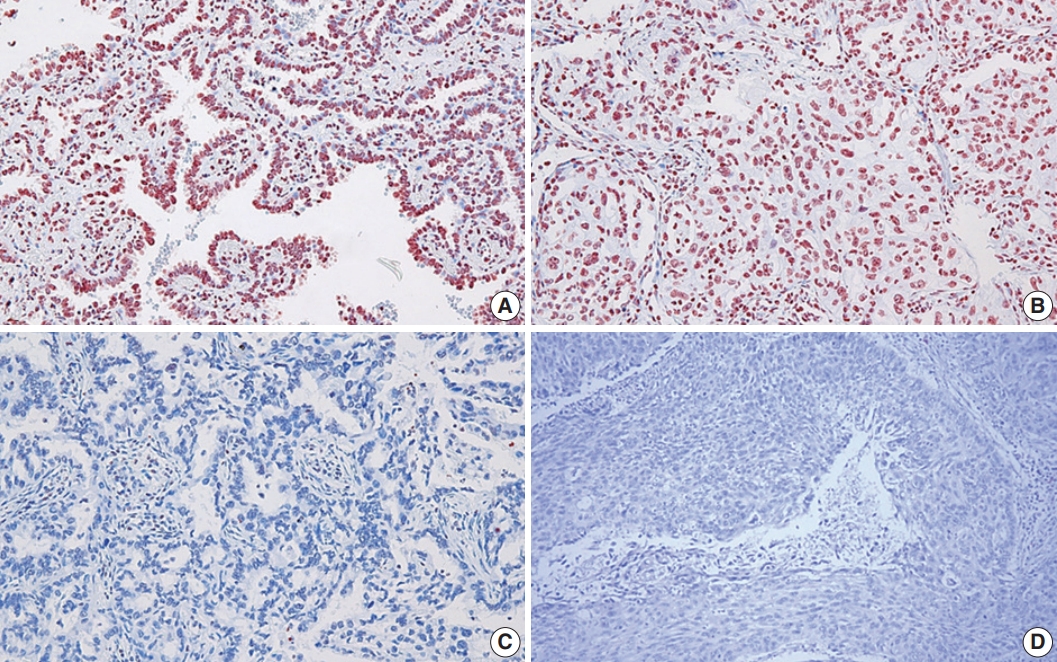
- In this study, the NSCLC used consisted of 94 squamous cell carcinomas and 109 adenocarcinomas. The mean age of the study subjects was 65 years. In all, 72.4% of the subjects were male, and 27.3% were female. Expression of 8-OHdG was found exclusively in nuclei of tumor cells and was positive in 83.3% (169/203) of NSCLC samples (Table 1, Fig. 1). The positive expression of 8-OHdG was significantly associated with female, adenocarcinoma, smaller tumor size, lower T group, negative lymph node status, and never-smoker (p < .05) (Table 1). The expression of 8-OHdG was high in never-smoker (89.5%) compared to smoker (73.4%) (p = .003).
- For p53 immunohistochemistry, 18% (37/203), 30% (60/203), and 52% (106/203) of cases were categorized as p53 negative, p53 low, and p53 high groups, respectively. Analysis was performed on the correlations between p53 staining pattern and clinicopathological features of NSCLC patients, and p53 aberrant expression was significantly correlated with male, smoker, squamous cell carcinoma, and Ki-67 positivity (p < .05) (Table 2). The p53 wild-type expression was high in never-smoker (39.5%) compared to smoker (13.9%), and p53 aberrant expression was 86.1% (68/79) in smoker and 60.5% (75/124) in never-smoker (p < .001). The p53 aberrant expression rate was 87.2% in squamous cell carcinoma and 56% in lung adenocarcinoma. In the 8-OHdG negative group, p53 aberrant expression pattern was more frequent (82.4%, 28/34) than in the 8-OHdG positive group (68%, 115/169) but not to an extent that was statistically significant (p = .095). Comparing the p53 wild-type and the p53 aberrant patients, there was no significant difference in tumor stage or lymph node stage.
- NSCLC patients with negative 8-OHdG expression had shorter OS by univariate analysis
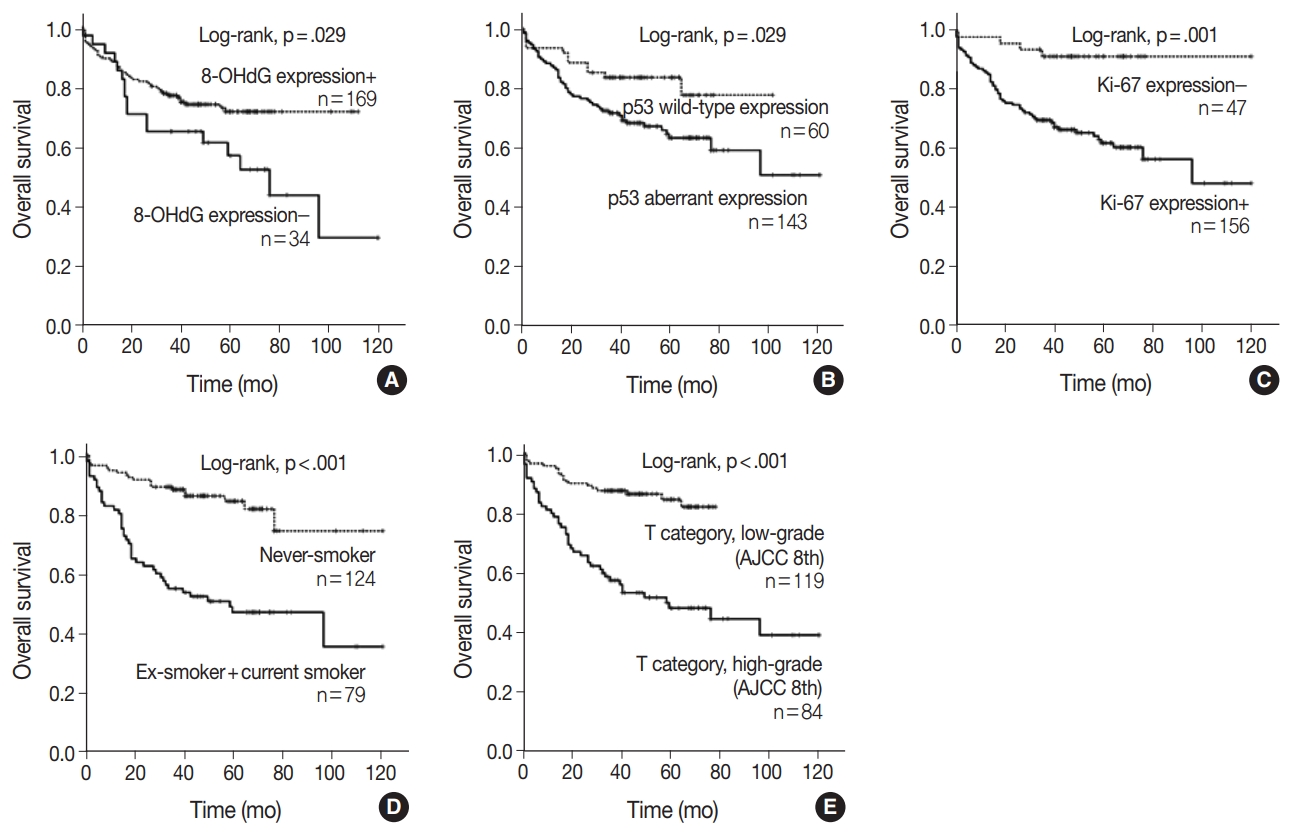
- When univariate Cox proportional regression analysis was performed for the survival of NSCLC patients, older age (≥ 65), male, smoker, higher tumor stage, presence of lymph node metastasis, 8-OHdG negativity and p53 aberrant expression, and Ki-67 positivity showed significant association with shorter OS of NSCLC patients by univariate analysis (Table 3). The patients with 8-OHdG negativity showed 1.847-fold (95% confidence interval [95% CI], 1.054 to 3.236; p = .032) greater risk of death. Smoking (former and current) predicted a 3.926-fold (95% CI, 2.302 to 6.697; p < .001) greater risk of death than the never-smoker group. The p53 aberrant expression group predicted a 2.031-fold (95% CI, 1.057 to 3.900; p = .033) greater risk of death than the p53 wild-type group. Kaplan-Meier survival analysis was used to analyze the prognostic impact of the expression of 8-OHdG, p53, and Ki-67 and smoking and tumor stage on OS, and the results are presented in Fig. 2.
- The multivariate analysis included the factors that showed p-value lower than .05 in univariate analysis (sex, age, smoking, T category, lymph node stage, p53 expression, Ki-67 expression, and 8-OHdG expression). Age older than 65, smoker, and higher T category in the TNM stage system were independent poor prognostic factors for OS of NSCLC patients (Table 4). However, multivariate analysis did not indicate that 8-OHdG expression was an independent prognostic factor associated with OS for NSCLC.
RESULTS
- In this study, we investigated the immunohistochemical expression of 8-OHdG and p53 in human NSCLC tissues. Our results show that (1) expression of 8-OHdG was observed in 83.3% of NSCLC tissue; (2) expression of 8-OHdG was significantly associated with low T category, negative lymph node status, and never-smoker; (3) expression of 8-OHdG was significantly associated with longer OS by univariate and Kaplan-Meier survival analysis; (4) p53 aberrant expression closely correlated with smoker, male, squamous cell carcinoma, and Ki-67 positivity.
- CS induces oxidative stress, which can cause severe DNA damage and is known as an important carcinogenic mechanism linked to NSCLC. It is challenging to perform a direct measurement of reactive oxygen species (ROS) because of their short lifetime and rapid degradation and removal by cellular defense mechanisms. Therefore, a useful method to assess ROS is to use antibodies against the specific “footprints” of oxidative damage. The oxidative damage of DNA by ROS produces 8-OHdG, and this serves as an established footprint of oxidative stress. Increased levels of 8-OHdG are associated with the aging process as well as with a number of pathological conditions including cancer, diabetes, and hypertension [2].
- The overexpression of 8-OHdG has been reported in a variety of cancers, including breast cancer, lung cancer, bladder cancer, colorectal cancer, renal cell carcinoma, prostate cancer, and gastric adenocarcinoma [4,11-15]. Reports have predominantly indicated that overexpression of 8-OHdG is associated with unfavorable prognostic factors and/or poor patient prognosis. However, some studies reported different results. Negative 8-OHdG expression was associated with an aggressive breast cancer phenotype, and some reports have found no difference in 8-OHdG level between cancer patients and healthy subjects [4,16-18]. Therefore, there is controversy surrounding the clinical significance of 8-OHdG expression. Our results showed that tumors with positive expression of 8-OHdG were significantly associated with favorable clinicopathologic factors and longer OS in NSCLC patients. We considered the following possibilities as the cause of the different results. In many of the previous studies, 8-OHdG was measured in urine or at the serum level, not in cancer tissue. It is known that the method of measuring the absolute amount of 8-OHdG in urine or serum can be influenced by artificial production of 8-OHdG in the sample preparation stage [19,20]. Also, a previous study showed that serum and urinary levels of 8-OHdG did not associate with those found in the tissues [21]. We detected 8-OHdG expression using immunohistochemistry because immunohistochemical procedures allow detection of 8-OHdG at the single-cell level, which has the advantage of directly confirming its expression in cancer cells in tissues composed of different cell populations [22]. To the best of our knowledge, this is the first study to use immunohistochemistry to investigate the correlation between 8-OHdG expression and prognosis in NSCLC patients.
- Several possible mechanisms may be behind the inverse association of 8-OHdG level and tumor aggressiveness. Sova et al. [4] have insisted that the increase in ROS induces the overproduction of antioxidant enzymes, thereby avoiding the death of tumor cells and inducing tumor growth. Overproduction of antioxidant enzymes would prevent ROS interaction with DNA, leading to decreased formation of 8-OHdG at the tissue level, as suggested by their results [4]. Another reasonable possibility we suggest is that cancer tissue is generally hypoxic, which is predicted to be the reason for low ROS in cancer cells, because oxygen is needed for production of ROS. Thus, ROS production is low in rapidly growing cancers, leading to a decrease in the production of 8-OHdG, which may explain the poor prognosis of negative 8-OHdG in NSCLC patients in this study. Although there was no statistical significance, the association between positive 8-OHdG and negative Ki-67 (p = .084) in this study may support this hypothesis. Previous studies showed that ROS-mediated DNA damage plays a role in the initiation of carcinogenesis as well as in malignant transformation [17,23]. The expression of 8-OHdG decreased significantly in invasive breast carcinomas compared to non-invasive lesions, namely usual ductal hyperplasia, atypical ductal hyperplasia, and ductal carcinoma in situ [17]. To support the hypothesis that ROS-mediated DNA damage may be involved in the early stages of cancer development rather than cancer progression, examination of 8-OHdG expression in pre-cancerous lesions or pre-invasive lung cancer lesions is necessary. In addition, a DNA repair enzyme assessment is needed to clarify the fundamental background behind the current results.
- In this study, expression of 8-OHdG was significantly associated with never-smoker (p = .003). We cannot explain the exact mechanism for the relationship between never-smokers and 8-OHdG expression in this study. However, we inferred some possibilities based on the results of other researchers. The formation of carboxyhemoglobin in smokers can lower the ability of hemoglobin to deliver O2 (toxic hypoxia) and result in hypoxia. In turn, this can reduce ROS formation and lower the expression of 8-OHdG [24,25]. The association between smoking and low 8-OHdG may also be explained by smoking-induced oxidative stress, i.e., overproduction of antioxidant enzymes. The state of the smoking-ROS-8-OHdG axis in the mechanism of NSCLC development by smoking is thought to be regulated by more complex regulatory mechanisms, including the functional status of ROS and antioxidant enzymes and the hypoxic environment of cancer.
- DNA adduct formation and oncogenic mutation in lung epithelial cells are caused by chronic exposure to carcinogens in CS, and the accumulation of mutations is known to be involved in lung cancer development. TP53 mutation is an important mutation associated with CS exposure, and loss of wild-type p53 function and increased mutant p53 function due to TP53 mutation are important in NSCLC carcinogenesis. It has recently been reported that immunohistochemical staining for p53 can be used as a surrogate for TP53 mutation analysis [8]. In this study, p53 immunohistochemical staining was classified as wild or aberrant expression, and the correlation between p53 staining pattern and clinicopathologic features of NSCLC patients was analyzed. The p53 aberrant expression rate was 87.2% in squamous cell carcinoma and 56% in lung adenocarcinoma (data not shown). Our results are consistent with another report that found p53 mutations in 46% of lung adenocarcinomas and 81% of squamous cell carcinomas [5]. The p53 aberrant expression is closely correlated with smoker (p = .000), male (p = .000), squamous cell carcinoma (p = .000), and Ki-67 positivity (p = .003). Further studies are needed to determine the concordance of p53 immunohistochemistry with TP53 mutations in NSCLC to confirm our assertion.
- In conclusion, this study has demonstrated the expression of 8-OHdG and p53 in NSCLC and its relationship with clinicopathologic factors and patient survival. Univariate analysis revealed that expression of 8-OHdG was significantly associated with longer OS. Smoking is an important inducer of oxidative stress, but it was not associated with increased expression of 8-OHdG in this study, suggesting that more complex regulatory mechanisms regulate 8-OHdG expression in cancer, including the functional status of the antioxidant enzyme and the hypoxic environment of cancer. The aberrant expression patterns of p53 immunohistochemical staining showed significant associations with smoking status.
DISCUSSION
Author Contributions
Conceptualization; ARA, KMK, HSP, KYJ, WSM, MJK, YCL, JHK, HJC, MJC.
Data curation: ARA, KMK, HSP, MJC.
Formal analysis: ARA, KMK, HJC, MJC.
Investigation: SCL, CWY.
Funding acquisition: MJC.
Methodology: ARA, HJC, MJC, YCL.
Writing—original draft: ARA, KMK, HSP, KYJ, WSM, MJK, YCL, JHK, MJC.
Writing—review & editing: ARA, KMK, MJC.
Conflicts of Interest
The authors declare that they have no potential conflicts of interest.
Acknowledgments
- 1. Ferlay J, Soerjomataram I, Ervik M, et al. Cancer incidence and mortality worldwide: IARC CancerBase No. 11. Lyon: International Agency for Research on Cancer, 2014.
- 2. David SS, O'Shea VL, Kundu S. Base-excision repair of oxidative DNA damage. Nature 2007; 447: 941-50. ArticlePubMedPMCPDF
- 3. Matsumoto K, Satoh Y, Sugo H, et al. Immunohistochemical study of the relationship between 8-hydroxy-2'-deoxyguanosine levels in noncancerous region and postoperative recurrence of hepatocellular carcinoma in remnant liver. Hepatol Res 2003; 25: 435-41. ArticlePubMed
- 4. Sova H, Jukkola-Vuorinen A, Puistola U, Kauppila S, Karihtala P. 8-Hydroxydeoxyguanosine: a new potential independent prognostic factor in breast cancer. Br J Cancer 2010; 102: 1018-23. ArticlePubMedPMCPDF
- 5. Gibbons DL, Byers LA, Kurie JM. Smoking, p53 mutation, and lung cancer. Mol Cancer Res 2014; 12: 3-13. ArticlePubMedPMCPDF
- 6. Murtas D, Piras F, Minerba L, et al. Nuclear 8-hydroxy-2'-deoxyguanosine as survival biomarker in patients with cutaneous melanoma. Oncol Rep 2010; 23: 329-35. PubMed
- 7. Rami-Porta R, Asamura H, Travis WD, Rusch VW. Lung. In: Amin MB, ed. AJCC cancer staging manual. Cham: Springer, 2017; 431-56. 8th.
- 8. Yemelyanova A, Vang R, Kshirsagar M, et al. Immunohistochemical staining patterns of p53 can serve as a surrogate marker for TP53 mutations in ovarian carcinoma: an immunohistochemical and nucleotide sequencing analysis. Mod Pathol 2011; 24: 1248-53. ArticlePubMedPDF
- 9. Cha YJ, Kim HR, Lee CY, Cho BC, Shim HS. Clinicopathological and prognostic significance of programmed cell death ligand-1 expression in lung adenocarcinoma and its relationship with p53 status. Lung Cancer 2016; 97: 73-80. ArticlePubMed
- 10. Shim HS, Kenudson M, Zheng Z, et al. Unique genetic and survival characteristics of invasive mucinous adenocarcinoma of the lung. J Thorac Oncol 2015; 10: 1156-62. ArticlePubMed
- 11. Shen J, Deininger P, Hunt JD, Zhao H. 8-Hydroxy-2'-deoxyguanosine (8-OH-dG) as a potential survival biomarker in patients with nonsmall-cell lung cancer. Cancer 2007; 109: 574-80. ArticlePubMed
- 12. Kaczmarek P, Blaszczyk J, Fijalkowski P, et al. Assessment of 8-hydroxy-2'-deoxyguanosine concentrations in bladder cancer patients treated with intravesical BCG instillation. Pol Merkur Lekarski 2005; 19: 526-8. PubMed
- 13. Oliva MR, Ripoll F, Muniz P, et al. Genetic alterations and oxidative metabolism in sporadic colorectal tumors from a Spanish community. Mol Carcinog 1997; 18: 232-43. ArticlePubMed
- 14. Okamoto K, Toyokuni S, Uchida K, et al. Formation of 8-hydroxy-2'-deoxyguanosine and 4-hydroxy-2-nonenal-modified proteins in human renal-cell carcinoma. Int J Cancer 1994; 58: 825-9. ArticlePubMed
- 15. Miyake H, Hara I, Kamidono S, Eto H. Oxidative DNA damage in patients with prostate cancer and its response to treatment. J Urol 2004; 171: 1533-6. ArticlePubMed
- 16. Nagashima M, Tsuda H, Takenoshita S, et al. 8-hydroxydeoxyguanosine levels in DNA of human breast cancer are not significantly different from those of non-cancerous breast tissues by the HPLC-ECD method. Cancer Lett 1995; 90: 157-62. PubMed
- 17. Jaloszynski P, Jaruga P, Olinski R, et al. Oxidative DNA base modifications and polycyclic aromatic hydrocarbon DNA adducts in squamous cell carcinoma of larynx. Free Radic Res 2003; 37: 231-40. ArticlePubMed
- 18. Karihtala P, Kauppila S, Puistola U, Jukkola-Vuorinen A. Divergent behaviour of oxidative stress markers 8-hydroxydeoxyguanosine (8-OHdG) and 4-hydroxy-2-nonenal (HNE) in breast carcinogenesis. Histopathology 2011; 58: 854-62. ArticlePubMed
- 19. Toyokuni S, Tanaka T, Hattori Y, et al. Quantitative immunohistochemical determination of 8-hydroxy-2'-deoxyguanosine by a monoclonal antibody N45.1: its application to ferric nitrilotriacetate-induced renal carcinogenesis model. Lab Invest 1997; 76: 365-74. PubMed
- 20. Lindahl T. Instability and decay of the primary structure of DNA. Nature 1993; 362: 709-15. ArticlePubMedPDF
- 21. Soini Y, Haapasaari KM, Vaarala MH, Turpeenniemi-Hujanen T, Kärjä V, Karihtala P. 8-hydroxydeguanosine and nitrotyrosine are prognostic factors in urinary bladder carcinoma. Int J Clin Exp Pathol 2011; 4: 267-75. PubMedPMC
- 22. Takahashi S, Hirose M, Tamano S, et al. Immunohistochemical detection of 8-hydroxy-2'-deoxyguanosine in paraffin-embedded sections of rat liver after carbon tetrachloride treatment. Toxicol Pathol 1998; 26: 247-52. ArticlePubMedPDF
- 23. Valavanidis A, Vlachogianni T, Fiotakis K, Loridas S. Pulmonary oxidative stress, inflammation and cancer: respirable particulate matter, fibrous dusts and ozone as major causes of lung carcinogenesis through reactive oxygen species mechanisms. Int J Environ Res Public Health 2013; 10: 3886-907. ArticlePubMedPMC
- 24. Vaupel P, Harrison L. Tumor hypoxia: causative factors, compensatory mechanisms, and cellular response. Oncologist 2004; 9 Suppl 5: 4-9. ArticlePubMedPDF
- 25. Archer SL, Huang J, Henry T, Peterson D, Weir EK. A redox-based O2 sensor in rat pulmonary vasculature. Circ Res 1993; 73: 1100-12. ArticlePubMed
REFERENCES
Figure & Data
References
Citations

- N-acetyl-cysteine alleviates nandrolone decanoate-induced hippocampal cell apoptosis in rats via reversing protein expressions of S1P1, Akt and FOXO3a signaling pathway
Alireza Shirpoor, Zahra Zarrini, Roya Naderi
Steroids.2026; 228: 109759. CrossRef - The distinct roles of ROS in tumor immunity: from mechanisms to immunotherapeutic applications
Jiayi Li, Chen Huang, Pan Tang, Ruiyan Wu, Quanyou Wu, Chenliang Zhang
Journal of Hematology & Oncology.2026;[Epub] CrossRef - Sustainable framework for automated segmentation and prediction of lung cancer in CT image using CapsNet with U-net segmentation
S.R. Vijayakumar, S. Aarthy, D. Deepa, P. Suresh
Biomedical Signal Processing and Control.2025; 99: 106873. CrossRef - Endolysosomal cation channel MCOLN as the novel regulator of redox homeostasis
Yahao Gao, Lei Xu, Ying Chen
Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease.2025; 1871(7): 167910. CrossRef - Catalase: The golden key to regulate oxidative stress in breast cancer
Jia-Wei Liu, Wen-Jia Chen, Yang-Zheng Lan, Jing Liu
World Journal of Clinical Oncology.2025;[Epub] CrossRef - Association of sirtuin 1 rs10997868 and rs730821 polymorphisms with sirtuin 1 and hydroxy-2′-deoxyguanosine levels in healthy smokers: A case–control study
Samar Sultan
Journal of International Medical Research.2025;[Epub] CrossRef - Increased pretreatment triglyceride glucose-body mass index associated with poor prognosis in patients with advanced non-small cell lung cancer
Shaoming Guo, Yi Zhao, Yue Jiang, Huaping Ye, Ying Wang
Clinical Nutrition ESPEN.2024; 59: 412. CrossRef - Oxidative Damage and Telomere Length as Markers of Lung Cancer Development among Chronic Obstructive Pulmonary Disease (COPD) Smokers
Elizabeth Córdoba-Lanús, Luis M. Montuenga, Angélica Domínguez-de-Barros, Alexis Oliva, Delia Mayato, Ana Remírez-Sanz, Francisca Gonzalvo, Bartolomé Celli, Javier J. Zulueta, Ciro Casanova
Antioxidants.2024; 13(2): 156. CrossRef - Automated determination of 8-OHdG in cells and tissue via immunofluorescence using a specially created antibody
Tobias Jung, Nicole Findik, Bianca Hartmann, Katja Hanack, Kai Grossmann, Dirk Roggenbuck, Marc Wegmann, René Mantke, Markus Deckert, Tilman Grune
Biotechnology Reports.2024; 42: e00833. CrossRef - Combination treatment of zinc and selenium intervention ameliorated BPA-exposed germ cell damage in SD rats: elucidation of molecular mechanisms
Chittaranjan Sahu, Gopabandhu Jena
Naunyn-Schmiedeberg's Archives of Pharmacology.2024; 397(9): 6685. CrossRef - Interplay of arsenic exposure and cigarette smoking on oxidative DNA damage in healthy males
Sepideh Nemati-Mansour, Mohammad Mosaferi, Javad Babaie, Asghar Mohammadpoorasl, Reza Dehghanzadeh, Leila Nikniaz, Mohammad Miri
Environmental Sciences Europe.2024;[Epub] CrossRef - The role of tissue persistent organic pollutants and genetic polymorphisms in patients with benign and malignant kidney tumors
Rasih Kocagöz, İlgen Onat, Merve Demirbügen Öz, Burak Turna, Banu Sarsık Kumbaracı, Mehmet Nurullah Orman, Halit Sinan Süzen, Hilmi Orhan
Environmental Toxicology and Pharmacology.2024; 110: 104495. CrossRef - Mitochondrial Plasticity and Glucose Metabolic Alterations in Human Cancer under Oxidative Stress—From Viewpoints of Chronic Inflammation and Neutrophil Extracellular Traps (NETs)
Hui-Ting Lee, Chen-Sung Lin, Chao-Yu Liu, Po Chen, Chang-Youh Tsai, Yau-Huei Wei
International Journal of Molecular Sciences.2024; 25(17): 9458. CrossRef - Oxidative DNA Damage and Arterial Hypertension in Light of Current ESC Guidelines
Radka Hazuková, Zdeněk Zadák, Miloslav Pleskot, Petr Zdráhal, Martin Pumprla, Miloš Táborský
International Journal of Molecular Sciences.2024; 25(23): 12557. CrossRef - Significance of 8-OHdG Expression as a Predictor of Survival in Colorectal Cancer
Myunghee Kang, Soyeon Jeong, Sungjin Park, Seungyoon Nam, Jun-Won Chung, Kyoung Oh Kim, Jungsuk An, Jung Ho Kim
Cancers.2023; 15(18): 4613. CrossRef - Serum 8-Hydroxy-2′-deoxyguanosine Predicts Severity and Prognosis of Patients with Acute Exacerbation of Chronic Obstructive Pulmonary Disease
Peng Cao, Chen Zhang, Dong-Xu Hua, Meng-Die Li, Bian-Bian Lv, Lin Fu, Hui Zhao
Lung.2022; 200(1): 31. CrossRef - Redox signaling at the crossroads of human health and disease
Jing Zuo, Zhe Zhang, Maochao Luo, Li Zhou, Edouard C. Nice, Wei Zhang, Chuang Wang, Canhua Huang
MedComm.2022;[Epub] CrossRef - Assessment of MDA and 8-OHdG expressions in ovine pulmonary adenocarcinomas by immunohistochemical and immunofluorescence methods
Emin Karakurt, Enver Beytut, Serpil Dağ, Hilmi Nuhoğlu, Ayfer Yıldız, Emre Kurtbaş
Acta Veterinaria Brno.2022; 91(3): 235. CrossRef - Dietary Antioxidants and Lung Cancer Risk in Smokers and Non-Smokers
Naser A. Alsharairi
Healthcare.2022; 10(12): 2501. CrossRef - Targeting oxidative stress in disease: promise and limitations of antioxidant therapy
Henry Jay Forman, Hongqiao Zhang
Nature Reviews Drug Discovery.2021; 20(9): 689. CrossRef - Association between tobacco substance usage and a missense mutation in the tumor suppressor gene P53 in the Saudi Arabian population
Mikhlid H. Almutairi, Bader O. Almutairi, Turki M. Alrubie, Sultan N. Alharbi, Narasimha R. Parine, Abdulwahed F. Alrefaei, Ibrahim Aldeailej, Abdullah Alamri, Abdelhabib Semlali, Alvaro Galli
PLOS ONE.2021; 16(1): e0245133. CrossRef - Measurement of uranium concentrations in urine samples of adult healthy groups in Najaf governorate with estimation of urine concentrations of 8-OHdG compound as biomarker for DNA damage
Samia K. Abbas, Dhuha S. Saleh, Hayder S. Hussain
Journal of Physics: Conference Series.2021; 1879(3): 032097. CrossRef - Common Data Model and Database System Development for the Korea Biobank Network
Soo-Jeong Ko, Wona Choi, Ki-Hoon Kim, Seo-Joon Lee, Haesook Min, Seol-Whan Oh, In Young Choi
Applied Sciences.2021; 11(24): 11825. CrossRef - EVALUATION OF OXIDATIVE STATUS IN PATIENTS WITH CHRONIC PERIODONTITIS AND ADDITIONAL TOBACCO ABUSE: A CROSS-SECTIONAL STUDY
Didem ÖZKAL EMİNOĞLU, Varol ÇANAKÇI
Atatürk Üniversitesi Diş Hekimliği Fakültesi Dergisi.2020; : 1. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
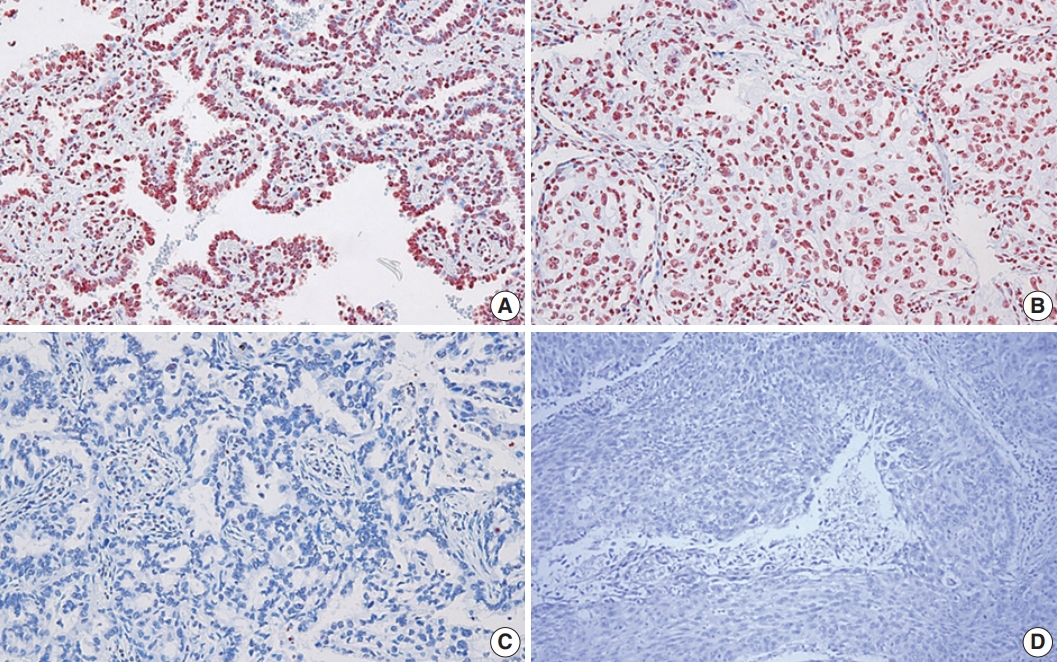
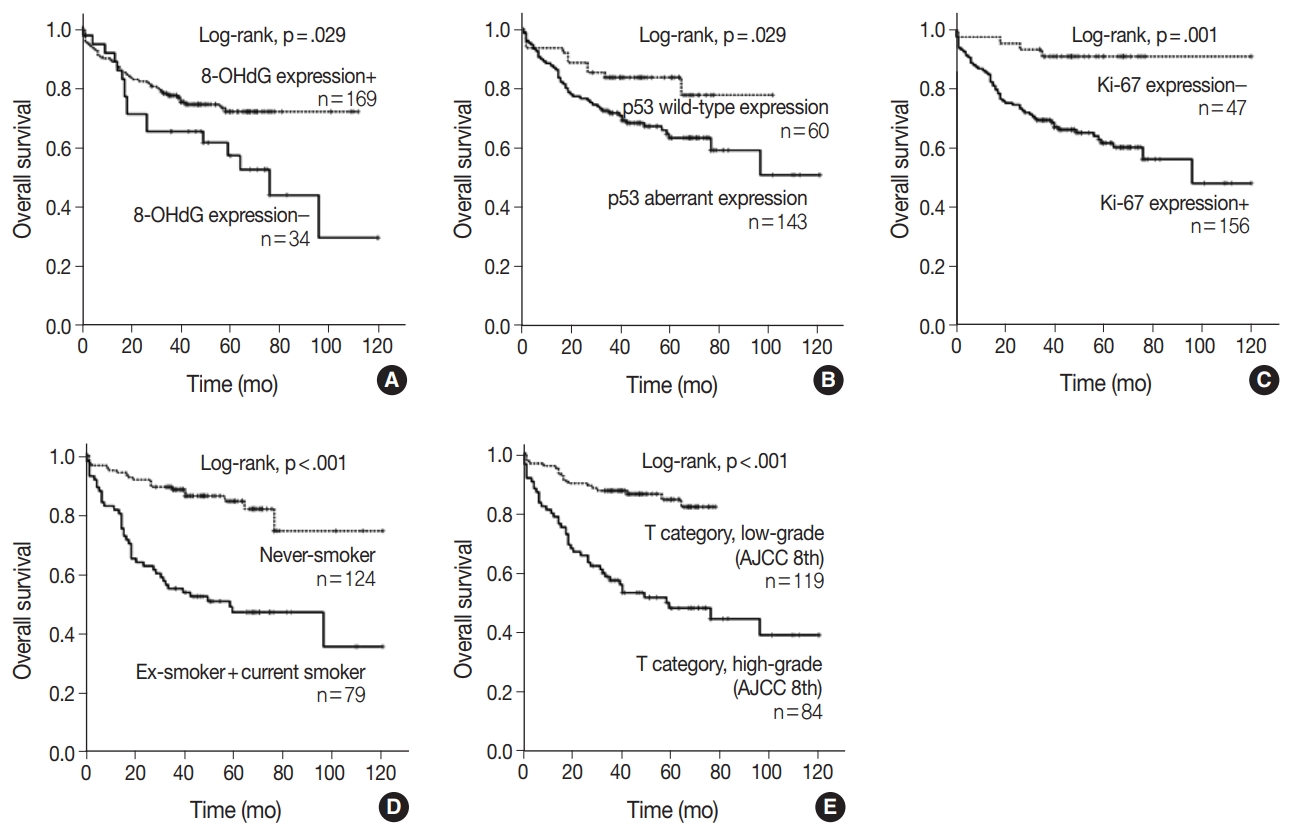
Fig. 1.
Fig. 2.
| Characteristic | No. | 8-OHdG (+) | 8-OHdG (–) | p-value |
|---|---|---|---|---|
| Age (yr) | ||||
| < 65 | 84 | 71 (84.5) | 13 (15.5) | .683 |
| ≥ 65 | 119 | 98 (82.4) | 21 (17.6) | |
| Sex | ||||
| Male | 144 | 115 (79.9) | 29 (20.1) | .030 |
| Female | 59 | 54 (91.5) | 5 (8.5) | |
| Histologic type | ||||
| SqCC | 94 | 73 (77.7) | 21 (22.3) | .037 |
| ADC | 109 | 96 (89.1) | 13 (10.9) | |
| T category | ||||
| 1 + 2 | 119 | 109 (91.6) | 10 (8.4) | < .001 |
| 3 + 4 | 84 | 60 (71.4) | 24 (28.6) | |
| N category | ||||
| N0 | 108 | 100 (92.6) | 8 (7.4) | < .001 |
| N1 | 95 | 69 (72.6) | 26 (27.4) | |
| Pleural invasion | ||||
| Negative | 135 | 117 (86.7) | 18 (13.3) | .066 |
| Positive | 68 | 52 (76.5) | 16 (23.5) | |
| Smoking | ||||
| Never | 124 | 111 (89.5) | 13 (10.5) | .003 |
| Former + current | 79 | 58 (73.4) | 21 (26.6) | |
| p53 | ||||
| Wild-type | 60 | 54 (90.0) | 6 (10.0) | .095 |
| Aberrant | 143 | 115 (80.4) | 28 (19.6) | |
| Ki-67 | ||||
| Negative | 47 | 43 (91.5) | 4 (8.5) | .084 |
| Positive | 156 | 126 (80.8) | 30 (19.2) |
| Characteristic | p53 |
p-value | ||
|---|---|---|---|---|
| No. | Wild-type | Aberrant | ||
| Age (yr) | ||||
| < 65 | 84 | 24 (28.6) | 60 (71.4) | .796 |
| ≥ 65 | 119 | 36 (30.3) | 19 (16.0) | |
| Sex | ||||
| Male | 144 | 32 (22.2) | 112 (77.8) | < .001 |
| Female | 59 | 28 (47.5) | 8 (13.6) | |
| Histologic type | ||||
| SqCC | 94 | 12 (12.8) | 82 (87.2) | < .001 |
| ADC | 109 | 48 (44.0) | 61 (56.0) | |
| T category 8th | ||||
| 1 + 2 | 119 | 40 (33.6) | 79 (66.4) | .132 |
| 3 + 4 | 84 | 20 (23.8) | 64 (76.2) | |
| N category | ||||
| N0 | 108 | 36 (33.3) | 72 (66.7) | .301 |
| N1 | 95 | 24 (25.3) | 71 (74.4) | |
| Pleural invasion | ||||
| Negative | 135 | 41 (30.4) | 94 (69.6) | .720 |
| Positive | 68 | 19 (27.9) | 49 (72.1) | |
| Smoking | ||||
| Never | 124 | 49 (39.5) | 75 (60.5) | < .001 |
| Former + current | 79 | 11 (13.9) | 68 (86.1) | |
| 8-OHdG | ||||
| Negative | 34 | 6 (17.6) | 28 (82.4) | .095 |
| Positive | 169 | 54 (32.0) | 115 (68.0) | |
| Ki-67 | ||||
| Negative | 47 | 22 (46.8) | 25 (53.2) | .003 |
| Positive | 156 | 38 (24.4) | 118 (75.6) | |
| Characteristic | OS (n=203) |
|
|---|---|---|
| HR (95% CI) | p-value | |
| Age, ≥ 65 yr (vs < 65 yr) | 2.153 (1.231–3.764) | .007 |
| Sex, female (vs male) | 0.324 (0.154–0.681) | .003 |
| T category, T3+4 (vs 1 + 2) | 4.027 (2.318–6.993) | < .001 |
| N category, N1 (vs N0) | 3.620 (2.042–6.418) | < .001 |
| Pleural invasion, positive (vs negative) | 1.403 (0.844–2.331) | .191 |
| Smoking, former + current (vs never) | 3.926 (2.302–6.697) | < .001 |
| 8-OHdG negative (vs positive) | 1.847 (1.054–3.236) | .032 |
| p53 aberrant (vs wild-type) | 2.031 (1.057–3.900) | .033 |
| Ki-67 positive (vs negative) | 4.982 (1.808–13.731) | .002 |
| Total (n = 203) |
||
|---|---|---|
| HR (95% CI) | p-value | |
| T category, T 3,4 (vs 1,2) | 2.346 (1.267–4.343) | .007 |
| Smoking, former + current (vs never) | 2.371 (1.347–4.172) | .003 |
| Age, ≥ 65 yr (vs < 65 yr) | 2.154 (1.220–3.804) | .008 |
| 8-OHdG negative (vs positive) | 1.486 (0.520–4.246) | .460 |
| p53 aberrant (vs wild-type) | 1.211 (0.671–2.184) | .525 |
Values are presented as number (%). 8-OhdG, 8-hydroxy-2-deoxy guanosine; SqCC, squamous cell carcinoma; ADC, adenocarcinoma.
Values are presented as number (%). SqCC, squamous cell carcinoma; ADC, adenocarcinoma; 8-OHdG, 8-hydroxy-2-deoxy guanosine.
NSCLC, non-small cell lung cancer; 8-OHdG, 8-hydroxy-2-deoxy guanosine.
Variables considered in the model were age, lymph node status, smoking history, T category (American Joint Committee on Cancer 8th edition), 8-OHdG expression and p53, Ki-67 expression. NSCLC, non-small cell lung cancer; 8-OHdG, 8-hydroxy-2-deoxy guanosine.

 E-submission
E-submission




