Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 53(3); 2019 > Article
-
Original Article
Potential Role for a Panel of Immunohistochemical Markers in the Management of Endometrial Carcinoma -
Amany Salama1
 , Mohammad Arafa1,2
, Mohammad Arafa1,2 , Eman ElZahaf3, Abdelhadi Mohamed Shebl1
, Eman ElZahaf3, Abdelhadi Mohamed Shebl1 , Azmy Abd El-Hameed Awad1, Sylvia A. Ashamallah1
, Azmy Abd El-Hameed Awad1, Sylvia A. Ashamallah1 , Reda Hemida4
, Reda Hemida4 , Anas Gamal4
, Anas Gamal4 , Abd AlRahman Foda1
, Abd AlRahman Foda1 , Khaled Zalata1
, Khaled Zalata1 , El-Said M. Abdel-Hady4
, El-Said M. Abdel-Hady4
-
Journal of Pathology and Translational Medicine 2019;53(3):164-172.
DOI: https://doi.org/10.4132/jptm.2019.02.12
Published online: February 28, 2019
1Department of Pathology, Faculty of Medicine, Mansoura University, Mansoura, Egypt
2Department of Pathology, College of Medicine and Health Sciences, Sultan Qaboos University, Muscat, Oman
3Department of Clinical Oncology and Nuclear Medicine, Faculty of Medicine, Mansoura University, Mansoura, Egypt
4Department of Obstetrics and Gynecology, Faculty of Medicine, Mansoura University, Mansoura, Egypt
- Corresponding Author Mohammad Arafa, MD, PhD Department of Pathology, Faculty of Medicine, Mansoura University, 35516 Mansoura, Egypt Tel: +2-050-226-59-22 Fax: +2-050-226-37-17 E-mail: marafa8@yahoo.com
- This study was partially presented as an abstract in the XXXI Congress of the International Academy of Pathology and the 28th Congress of the European Society of Pathology, Cologne, Germany, 2016.
© 2019 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- In order to improve the efficacy of endometrial carcinoma (EC) treatment, identifying prognostic factors for high risk patients is a high research priority. This study aimed to assess the relationships among the expression of estrogen receptors (ER), progesterone receptors (PR), human epidermal growth factor receptor 2 (HER2), Ki-67, and the different histopathological prognostic parameters in EC and to assess the value of these in the management of EC.
-
Methods
- We examined 109 cases of EC. Immunohistochemistry for ER, PR, HER2, and Ki-67 were evaluated in relation to age, tumor size, International Federation of Gynecology and Obstetrics (FIGO) stage and grade, depth of infiltration, cervical and ovarian involvement, lymphovascular space invasion (LVSI), and lymph node (LN) metastasis.
-
Results
- The mean age of patients in this study was 59.8 ± 8.2 years. Low ER and PR expression scores and high Ki-67 expression showed highly significant associations with non-endometrioid histology (p = .007, p < .001, and p < .001, respectively) and poor differentiation (p = .007, p < .001, and p <. 001, respectively). Low PR score showed a significant association with advanced stage (p = .009). Low ER score was highly associated with LVSI (p = .006), and low PR scores were associated significantly with LN metastasis (p = .026). HER2 expression was significantly related to advanced stages (p = .04), increased depth of infiltration (p = .02), LVSI (p = .017), ovarian involvement (p = .038), and LN metastasis (p = .038). There was a close relationship between HER2 expression and uterine cervical involvement (p = .009). Higher Ki-67 values were associated with LN involvement (p = .012).
-
Conclusions
- The over-expression of HER2 and Ki-67 and low expression of ER and PR indicate a more malignant EC behavior. An immunohistochemical panel for the identification of high risk tumors can contribute significantly to prognostic assessments.
- Sample selection
- Archival material of randomly-selected hysterectomy specimens of 109 EC cases were retrieved from the Pathology Department. These cases were diagnosed in the period between 2005 and 2017. Corresponding files of these cases were retrieved from the Clinical Oncology and Nuclear Medicine Departments at Mansoura University. The histological types were endometrioid (89 cases), serous (12 cases), undifferentiated (one case), dedifferentiated (one case), and carcinosarcoma (three cases). The remaining three cases showed mixed patterns. The major component in two was endometrioid; the other was serous carcinoma. Hematoxylin and eosin (H&E) stained slides for every case were reviewed by two independent pathologists. International Federation of Gynecology and Obstetrics (FIGO) revised criteria in 2009 were used for grading and staging of cases [11]. All procedures performed in the current study were approved by the ethical committee of Mansoura University (Institutional Review Board [IRB] code number MD15.09.08, dated 18/09/2015) in accordance with the 1964 Declaration of Helsinki and its later amendments. Formal written informed consent was not required with a waiver by the IRB.
- Tissue microarray construction
- The tissue microarray (TMA) was constructed as previously published [12]. Briefly, a representative slide for each tumor was selected and an area of the tumor was circled. Using the manual tissue arrayer (MTA-1, Estigen, Tartu, Estonia), the areas of interest of a donor block were cored using tissue punches of 0.6 mm diameter. The cores were then transferred into the recipient block. Three cores were taken from each tumour. In carcinosarcoma cases, only the epithelial component was assessed. Sections from these microarrays were then H&E stained and tested for spot adequacy.
- Immunohistochemistry
- Sections from the microarray were stained with antibodies against ER, PR, HER2 (Rabbit, monoclonal, Genemed, South San Francisco, CA, USA) and Ki-67 (mouse, monoclonal, Genemed) according to the instructions of the manufacturers. The positive control for ER and PR in this study was normal endometrial glands and stroma where these receptors show nuclear expression. The positive control for HER2 was positive breast carcinoma tissue. The positive internal control for Ki-67 was tonsillar lymphoid follicles.
- Evaluation of the staining
- Slides were examined by two independent pathologists blinded to patient characteristics and outcome. For ER and PR, we applied a scoring system that depended on immunoreactivity distribution and intensity [13,14]. The percentage of stained cells was scored as follows: 1, 0%–25%; 2, 26%–75%, and 3, ≥ 76%. The intensity of staining was also reported as 1, absent or weak; 2, moderate; and 3, strong. The sum of the two values equalled the score. Tumours were then subdivided into three categories depending on this immunohistochemical score. Category I corresponded to a score of 2, category II to a score of 3–4, and category III to a score of 5–6.
- U.S. Food and Drug Administration criteria were used for evaluation of HER2 scoring [15]. The scoring was 3+ if complete with strong membranous staining in more than 10% of tumor cells; 2+ if complete, weak to moderately intense staining of the membrane was seen in greater than 10% of tumor cells; 1+ if incomplete staining of the membrane was found in more than 10% of tumor cells and a score of 0 was assigned when no staining or membranous staining in less than 10% of tumor cells was present. A score of 3+ was considered positive, a score of 2+ was equivocal positive and scores of 1+ and 0 were negative.
- Ki-67 was evaluated as the percentage of cells showing positive nuclear reactivity in at least 500 histologically recognized tumour cells counted at × 400 magnification.
- For TMA validation purposes, the originally recorded immunohistochemical results from the initial routine histopathology reports of ten patients were compared to those of the current experiment. Similar findings were observed in the TMAs compared to full tissue sections.
- Statistical analysis
- Data were analysed by IBM SPSS software package ver. 20.0 (IBM Corp., Armonk, NY, USA). Qualitative data were described as number and percent. Quantitative data were described using median (minimum and maximum) and interquartile range for non-parametric data and mean and standard deviation for parametric data after testing for normality using the Kolmogrov-Smirnov test. The significance of the results obtained was judged at the 5% level. The tests used were chi-square, Monte Carlo, Fisher exact, Student t-, F- (ANOVA), Mann-Whitney, Kruskal-Wallis, and Spearman correlation.
MATERIALS AND METHODS
- Clinicopathological features of the studied cases
- Patient ages ranged from 37 to 79 years with a mean age of 59.8 ± 8.2 years. Most of the cases (88 patients, 80.7%) in this study were postmenopausal.
- Tumors ranged from 1 to 14 cm in largest dimension with a median value of 3 cm. There were 36 cases of grade 1 (33%), 43 cases of grade 2 (39.4%), and 30 cases were high grade carcinomas (27.5%) including grade 3 endometrioid, serous, mixed, undifferentiated and dedifferentiated carcinomas, and carcinosarcomas. In three cases (2.7%) the tumour was limited to the endometrium, 69 (63.3%) cases showed infiltration of the inner myometrial half, the tumour infiltrated the outer half in 26 cases (24%), and the serosa was infiltrated in three cases (2%). Cervical involvement was found in 20 cases (18%), 71 cases (65%) were free from cervical infiltration and in 14 cases (12.8%) cervical involvement was not determined due to suboptimal surgery. Adnexal metastases were found in 11 cases (10%), 83 cases (76%) were free from adnexal infiltration, and in 15 cases (13.7%) adnexal infiltration was unknown due to suboptimal surgery. There were 71 cases (65.1%) in stage I (56 stage IA and 15 stage IB), 15 cases in stage II (13.8%), 10 cases (9%) in stage IIIA, only two cases were stage IIIB, and one case was stage IVA. Lymphovascular emboli were found in 29 cases (26.6%).
- The association of immunohistochemical results with histopathological prognostic parameters
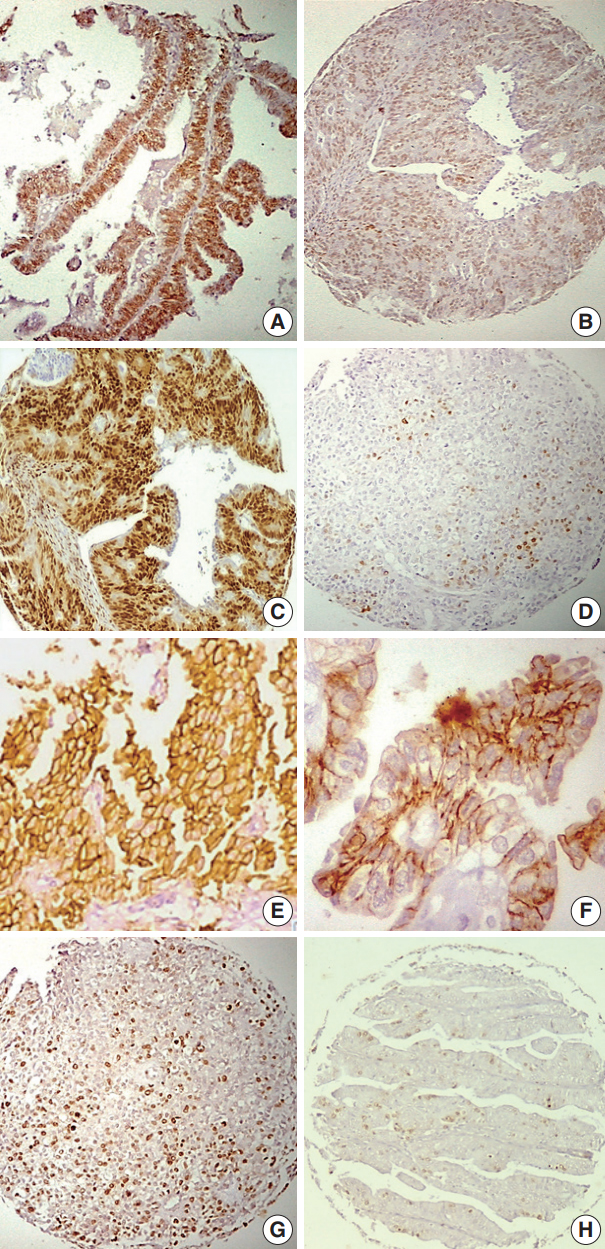
- The distribution of immunohistochemical data in relation to individual histopathological parameters is presented in Tables 1–4. The relationships among ER, PR expression, and other markers (HER2-neu and Ki-67) as well as the relationship between HER2-neu expression and Ki-67 expression are presented in Tables 5. Representative examples of the different expression patterns are show in Fig. 1.
- ER and PR scores were statistically associated (p < .001). There were significant relationships between low ER scores and nonendometrioid histology (p = .007) and higher grade of endometrial cancer (p = .007). The ER score tended to decrease with advanced stage (p = .057). Low ER score was associated with ovarian involvement (p = .025), lymphovascular space invasion (LVSI) (p = .006), and higher Ki-67 values (p = .024).
- Low PR expression score was associated with non-endometrioid histology (p < .001), higher tumour grade (p < .001), advanced stage (p = .009), and ovarian involvement (p < .007). The PR score decreased with LVSI (p = .06), and lower score was associated with lymph node metastasis (p = .026). Ki-67 values were higher with low PR score (p = .025).
- HER2 expression was significantly associated with advanced tumour stages (p = .04), increased depth of myometrial infiltration (p = .02), greater incidence of LVSI (p = .017), ovarian involvement (p = .038), and lymph node metastasis (p = .038). There was a notable relationship between HER2 expression and cervical involvement (p = .009).
- A positive correlation was found between tumour size and Ki-67 index (p = .02). Higher Ki-67 index was linked to more aggressive features such as non-endometrioid histotype (p < .001) and poor differentiation grade (p < .001). There was a strong relationship between higher Ki-67 values and lymph node involvement (p = .012).
- Median Ki-67 index value was higher in HER2-neu–positive cases than that of negative cases (p = .482, Mann-Whitney test).
RESULTS
- EC is the most common gynaecologic cancer worldwide and the incidence is increasing [2,3,16]. EC may not always fit into the dual model of type I and type II cancers: those can be vague clinicopathological designations rather than firm diagnostic entities. Tumours display varying degrees of conformity with both types and have different behaviours and prognoses [17-19]. According to the National Cancer Comprehensive Network guidelines for management of EC, the treatment strategy depends on surgical staging, depth of infiltration and the presence of adverse risk factors such as age, tumour size, LVSI and lower uterine involvement. Adjuvant therapy determinations are made on the basis of pathologic findings in the postoperative specimen. Superficially invasive, low grade (G 1–2) carcinomas in the absence of adverse risk factors can be treated by surgery with post-operative observation. However, in the presence of adverse risk factors, patients need adjuvant radiotherapy. High grade carcinomas with no adverse risk factors may be spared from adjuvant chemotherapy [20].
- Both breast and endometrial cancers are among the commonest cancers in females, and both are largely considered to be hormonedependent tumours. In breast cancer, a simple immunohistochemical panel of ER, PR, HER2, and Ki-67 is routinely performed on preoperative or postoperative specimens yielding valuable therapeutic and prognostic information. Similar to breast cancer, this panel may be of value when assessing EC specimens. The information attained may be helpful in guiding patient management and in providing prognostic information about tumour behaviour [7].
- In the current work, we assessed the immunohistochemical expression of the same panel of biological markers (ER, PR, HER2, and Ki-67) on 109 cases of EC and their association with histopathological prognostic characteristics. The presence of hormone receptors in ECs correlates with the clinical disease stage, histological grade, and overall survival. The absence of hormone receptors is considered to indicate aggressive tumour behaviour and poor prognosis [21,22]. A recent systematic review and meta-analysis revealed that higher levels of ER and PR were associated with favourable prognosis and longer overall survival [23]. This study showed close associations between low ER and PR scores, nonendometrioid histology and high grade endometrial cancer. Moreover, low PR score was significantly associated with advanced tumour stage. These findings agree with previous studies [21,24,25]. While not statistically significant, the ER score tended to be lower with advanced stage. Some studies failed to show associations between ER and PR expression and tumour stage [26,27]. Our data revealed significant associations between ovarian involvement and low ER and PR scores, an observation in contrast to previous observations [6,28]. This discrepancy may be due to differences in sample size, primary antibody used, and the method of scoring the immunohistochemical results. ER and PR did not show significant association with the depth of myometrial invasion or cervical infiltration as previously reported [25,26]. Low ER score was significantly associated with LVSI; low PR score tended to be associated with LVSI as well, but the strength of the low PR association did not match that of low ER. This agrees with the findings of a previous study [24]. Low PR scores were significantly associated with lymph node metastasis as reported earlier [26]. Consistent with previous studies, high ER and PR scores were highly associated while lower scores were associated with higher Ki-67 values [24,27,29].
- The increased expression of HER2 correlates with worse prognosis in various malignant tumours. In their extensive study (483 cases), Morrison et al. [30] demonstrated that the over-expression of HER2 was an independent prognostic factor that correlated with worse survival. Our work confirms a close relationship between HER2 overexpression and some of the traditional prognostic factors of endometrial cancer. In partial agreement with previous studies, we found HER2 expression to be associated with advanced tumour stages and increased depth of myometrial invasion [31-33]. We have not observed, however, any substantial relationship between HER2 overexpression and the grading of ECs. Some previous studies did not show a significant association between HER2 expression and the prognostic parameters [6,33]. In contrast to this, our study revealed that HER2 overexpression was significantly associated with a greater incidence of ovarian and cervical involvement, lymphovascular emboli and LN metastasis, findings in line with a previous observation [4]. We did not find HER2 over-expression to be significantly associated with ER, PR, or Ki-67 expression, a finding inconsistent with that of a study showing significant correlation between HER2 over-expression and high Ki-67 index [4].
- Increased Ki-67 expression indicates higher mitotic activity and greater tumour cell proliferation. Some studies revealed that Ki-67 could be an independent prognostic marker of survival in EC [34,35]. On the other hand, Pansare et al. [36] did not find correlations between Ki-67, histological type, grading, and tumour clinical staging. An elevated Ki-67 expression in this study was strongly related to non-endometrioid histotype and poor differentiation. Higher Ki-67 index was also found to be associated with lymph node involvement but not tumour stage, depth of myometrial invasion, cervical infiltration, or ovarian involvement.
- Our proposed immunohistochemical panel (ER, PR, HER2, and Ki-67) may be of value for preoperative biopsies. Results may indicate tumour behaviour characteristics, presence of adverse risk factors such as lymphovascular emboli and cervical involvement, and the necessity for more radical surgery with pelvic and para-aortic lymph node dissection [37]. Moreover, the panel may also be performed on postoperative specimens. The panel may be included routinely as an adjunct consideration in the postoperative treatment decision making process. Low risk patients with low grade, superficially invasive tumours may be spared the morbidity of lymphadenectomy as well as the cost and morbidity of radiotherapy. The panel results can also assist in identifying high risk patients requiring more radical surgery, post-operative radiotherapy, and/or chemotherapy [38].
- In conclusion, low ER and PR expression scores (category I), together with HER2 overexpression (score + 3) and Ki-67 indices of more than 20%, were associated with more malignant behaviour of ECs. Further studies involving larger numbers of patients are needed to investigate the correlation between this immunohistochemical panel’s results and the recent molecular classification of EC.
DISCUSSION
Author contributions
Conceptualization: MA.
Data curation: AS, MA, AAF, SAA.
Formal analysis: AS, MA, AAF, SAA.
Funding acquisition: MA.
Investigation: AS, MA, AAF, SAA.
Methodology: AS, MA, AAF, SAA.
Project administration: MA, SAA, RH, AG, KZ, ESMAH.
Resources: MA, SAA, RH, AG, KZ, ESMAH.
Software: AS, MA, SAA.
Supervision: MA, AMS, EE, AAEHA.
Validation: AS, MA, SAA.
Visualization: AS, MA, AAF, SAA.
Writing—original draft: AS.
Writing—review & editing: AS, MA, AAF, SAA.
Conflicts of Interest
The authors declare that they have no potential conflicts of interest.
Acknowledgments
- 1. Le Gallo M, Bell DW. The emerging genomic landscape of endometrial cancer. Clin Chem 2014; 60: 98-110. ArticlePubMedPDF
- 2. Llaurado M, Ruiz A, Majem B, et al. Molecular bases of endometrial cancer: new roles for new actors in the diagnosis and the therapy of the disease. Mol Cell Endocrinol 2012; 358: 244-55. ArticlePubMed
- 3. Backes FJ, Walker CJ, Goodfellow PJ, et al. Estrogen receptor-alpha as a predictive biomarker in endometrioid endometrial cancer. Gynecol Oncol 2016; 141: 312-7. ArticlePubMedPMC
- 4. Yu CG, Jiang XY, Li B, Gan L, Huang JF. Expression of ER, PR, CerbB-2 and Ki-67 in endometrial carcinoma and their relationships with the clinicopathological features. Asian Pac J Cancer Prev 2015; 16: 6789-94. ArticlePubMedPDF
- 5. Arafa M, Somja J, Dehan P, et al. Current concepts in the pathology and epigenetics of endometrial carcinoma. Pathology 2010; 42: 613-7. ArticlePubMed
- 6. Markova I, Duskova M, Lubusky M, et al. Selected immunohistochemical prognostic factors in endometrial cancer. Int J Gynecol Cancer 2010; 20: 576-82. ArticlePubMed
- 7. Lapinska-Szumczyk S, Supernat A, Majewska H, et al. HER2-positive endometrial cancer subtype carries poor prognosis. Clin Transl Sci 2014; 7: 482-8. ArticlePubMedPMC
- 8. Cancer Genome Atlas Research Network, Kandoth C, Schultz N, et al. Integrated genomic characterization of endometrial carcinoma. Nature 2013; 497: 67-73. ArticlePubMedPMCPDF
- 9. Faria SC, Sagebiel T, Balachandran A, Devine C, Lal C, Bhosale PR. Imaging in endometrial carcinoma. Indian J Radiol Imaging 2015; 25: 137-47. ArticlePubMedPMC
- 10. Li M, Zhao L, Qi W, et al. Clinical implications and prognostic value of five biomarkers in endometrial carcinoma. Chin Ger J Clin Oncol 2013; 12: 586-91. ArticlePDF
- 11. Pecorelli S. Revised FIGO staging for carcinoma of the vulva, cervix, and endometrium. Int J Gynaecol Obstet 2009; 105: 103-4. ArticlePubMedPDF
- 12. Arafa M, Boniver J, Delvenne P. Progression model tissue microarray (TMA) for the study of uterine carcinomas. Dis Markers 2010; 28: 267-72. ArticlePubMedPMCPDF
- 13. Zannoni GF, Vellone VG, Arena V, et al. Does high-grade endometrioid carcinoma (grade 3 FIGO) belong to type I or type II endometrial cancer? A clinical-pathological and immunohistochemical study. Virchows Arch 2010; 457: 27-34. ArticlePubMedPDF
- 14. Kounelis S, Kapranos N, Kouri E, Coppola D, Papadaki H, Jones MW. Immunohistochemical profile of endometrial adenocarcinoma: a study of 61 cases and review of the literature. Mod Pathol 2000; 13: 379-88. ArticlePubMedPDF
- 15. Brunelli M, Manfrin E, Martignoni G, et al. HER-2/neu assessment in breast cancer using the original FDA and new ASCO/CAP guideline recommendations: impact on selecting patients for herceptin therapy. Am J Clin Pathol 2008; 129: 907-11. ArticlePubMedPDF
- 16. Binder PS, Mutch DG. Update on prognostic markers for endometrial cancer. Womens Health (Lond) 2014; 10: 277-88. ArticlePubMedPDF
- 17. Rutgers JK. Update on pathology, staging and molecular pathology of endometrial (uterine corpus) adenocarcinoma. Future Oncol 2015; 11: 3207-18. ArticlePubMed
- 18. Maiques O, Cuevas D, Garcia Dios DA, et al. FISH analysis of PTEN in endometrial carcinoma. Comparison with SNP arrays and MLPA. Histopathology 2014; 65: 371-88. ArticlePubMedPMCPDF
- 19. Garg K, Soslow RA. Strategies for distinguishing low-grade endometrioid and serous carcinomas of endometrium. Adv Anat Pathol 2012; 19: 1-10. ArticlePubMed
- 20. Koh WJ, Greer BE, Abu-Rustum NR, et al. Uterine neoplasms, version 1.2014. J Natl Compr Canc Netw 2014; 12: 248-80. PubMed
- 21. Ferrandina G, Ranelletti FO, Gallotta V, et al. Expression of cyclooxygenase-2 (COX-2), receptors for estrogen (ER), and progesterone (PR), p53, ki67, and neu protein in endometrial cancer. Gynecol Oncol 2005; 98: 383-9. ArticlePubMed
- 22. Jazaeri AA, Nunes KJ, Dalton MS, Xu M, Shupnik MA, Rice LW. Well-differentiated endometrial adenocarcinomas and poorly differentiated mixed mullerian tumors have altered ER and PR isoform expression. Oncogene 2001; 20: 6965-9. ArticlePubMedPDF
- 23. Zhang Y, Zhao D, Gong C, et al. Prognostic role of hormone receptors in endometrial cancer: a systematic review and meta-analysis. World J Surg Oncol 2015; 13: 208.ArticlePubMedPMCPDF
- 24. Engelsen IB, Stefansson IM, Akslen LA, Salvesen HB. GATA3 expression in estrogen receptor alpha-negative endometrial carcinomas identifies aggressive tumors with high proliferation and poor patient survival. Am J Obstet Gynecol 2008; 199: 543.e1-7. PubMed
- 25. Tomica D, Ramic S, Danolic D, et al. A correlation between the expression of estrogen receptors and progesterone receptors in cancer cells and in the myometrium and prognostic factors in endometrial cancer. Coll Antropol 2014; 38: 129-34. PubMed
- 26. Srijaipracharoen S, Tangjitgamol S, Tanvanich S, et al. Expression of ER, PR, and Her-2/neu in endometrial cancer: a clinicopathological study. Asian Pac J Cancer Prev 2010; 11: 215-20. PubMed
- 27. Sivridis E, Giatromanolaki A, Koukourakis M, Anastasiadis P. Endometrial carcinoma: association of steroid hormone receptor expression with low angiogenesis and bcl-2 expression. Virchows Arch 2001; 438: 470-7. ArticlePubMedPDF
- 28. Kobel M, Atenafu EG, Rambau PF, et al. Progesterone receptor expression is associated with longer overall survival within highgrade histotypes of endometrial carcinoma: a Canadian high risk endometrial cancer consortium (CHREC) study. Gynecol Oncol 2016; 141: 559-63. ArticlePubMed
- 29. Stoian SC, Simionescu C, Margaritescu C, Stepan A, Nurciu M. Endometrial carcinomas: correlation between ER, PR, Ki67 status and histopathological prognostic parameters. Rom J Morphol Embryol 2011; 52: 631-6. PubMed
- 30. Morrison C, Zanagnolo V, Ramirez N, et al. HER-2 is an independent prognostic factor in endometrial cancer: association with outcome in a large cohort of surgically staged patients. J Clin Oncol 2006; 24: 2376-85. ArticlePubMed
- 31. Ioffe OB, Papadimitriou JC, Drachenberg CB. Correlation of proliferation indices, apoptosis, and related oncogene expression (bcl-2 and c-erbB-2) and p53 in proliferative, hyperplastic, and malignant endometrium. Hum Pathol 1998; 29: 1150-9. ArticlePubMed
- 32. Williams JA Jr, Wang ZR, Parrish RS, Hazlett LJ, Smith ST, Young SR. Fluorescence in situ hybridization analysis of HER-2/neu, cmyc, and p53 in endometrial cancer. Exp Mol Pathol 1999; 67: 135-43. ArticlePubMed
- 33. Gul AE, Keser SH, Barisik NO, et al. The relationship of cerb B 2 expression with estrogen receptor and progesterone receptor and prognostic parameters in endometrial carcinomas. Diagn Pathol 2010; 5: 13.ArticlePubMedPMCPDF
- 34. Salvesen HB, Iversen OE, Akslen LA. Prognostic significance of angiogenesis and Ki-67, p53, and p21 expression: a populationbased endometrial carcinoma study. J Clin Oncol 1999; 17: 1382-90. ArticlePubMed
- 35. Geisler JP, Geisler HE, Miller GA, Wiemann MC, Zhou Z, Crabtree W. MIB-1 in endometrial carcinoma: prognostic significance with 5-year follow-up. Gynecol Oncol 1999; 75: 432-6. ArticlePubMed
- 36. Pansare V, Munkarah AR, Schimp V, et al. Increased expression of hypoxia-inducible factor 1alpha in type I and type II endometrial carcinomas. Mod Pathol 2007; 20: 35-43. PubMedPDF
- 37. Goebel EA, Vidal A, Matias-Guiu X, Blake Gilks C. The evolution of endometrial carcinoma classification through application of immunohistochemistry and molecular diagnostics: past, present and future. Virchows Arch 2018; 472: 885-96. ArticlePubMedPDF
- 38. Sundar S, Balega J, Crosbie E, et al. BGCS uterine cancer guidelines: Recommendations for practice. Eur J Obstet Gynecol Reprod Biol 2017; 213: 71-97. ArticlePubMed
REFERENCES
Figure & Data
References
Citations

- Tissue Microarray for Gynecological Pathology Studies: A Mini-Review
Mohammad Arafa, Abd AlRahman Foda, Amany Salama, Ola Shalaby, Muna Al-Jabri, Fatma Al Hinai, Afrah Al-Rashdi, Samya Al-Husaini, Suaad Al-Badi
Journal of Microscopy and Ultrastructure.2026; 14(1): 1. CrossRef - Clinicopathological Correlation of Hormone Receptors, Angiogenesis, and Tumor Budding in Endometrial Carcinoma: A Tertiary Care Center Study
Senjuti Dasgupta, Arpita Das, Ujjwal Bandyopadhyay
The Journal of Obstetrics and Gynecology of India.2025;[Epub] CrossRef - Multiparameter MRI-based radiomics analysis for preoperative prediction of type II endometrial cancer
Yingying Cao, Wei Zhang, Xiaorong Wang, Xiaojing Lv, Yaping Zhang, Kai Guo, Shuai Ren, Yuan Li, Zhongqiu Wang, Jingya Chen
Heliyon.2024; 10(12): e32940. CrossRef - Correlation of PD-L1 expression with different clinico-pathological and immunohistochemical features of ovarian surface epithelial tumors
Asem Shalaby, Ola Shalaby, Hazem Abdullah, Mohamed Rachid Boulassel, Mohammad Arafa
Clinical and Translational Oncology.2024; 27(2): 699. CrossRef - Estrogen/Progesterone Receptor Loss, CTNNB1 and KRAS Mutations Are Associated With Local Recurrence or Distant Metastasis in Low-Grade Endometrial Endometrioid Carcinoma
Rajni Chibbar, Sabrina Foerstner, Janarathnee Suresh, Richa Chibbar, Alexandre Piche, Deeksha Kundapur, Rani Kanthan, Vijayanand Kundapur, Cheng Han Lee, Anita Agrawal, Raymond Lai
Applied Immunohistochemistry & Molecular Morphology.2023; 31(3): 181. CrossRef - Exploring the Prognostic and Predictive Roles of Ki-67 in Endometrial Cancer
Laura Paleari, Mariangela Rutigliani, Oriana D’Ecclesiis, Sara Gandini, Irene Maria Briata, Tania Buttiron Webber, Nicoletta Provinciali, Andrea DeCensi
International Journal of Translational Medicine.2023; 3(4): 479. CrossRef - Analysis of human epidermal growth factor receptor 2 immunohistochemical expression in high-grade endometrial carcinomas and its association with variable clinical outcomes
Malames M. Faisal, Marwa M. Shakweer, Ghada Refaat, Khaled S. Mohammed, Tarek I. ElMallawy, Magda H. Nasreldin, Laila M. Farid, Mariam B. Abouelkhair
Egyptian Journal of Pathology.2023; 43(2): 119. CrossRef - Correlation of PD-L1 immunohistochemical expression with microsatellite instability and p53 status in endometrial carcinoma
Mohammad Arafa, Abdelhadi Mohamed Shebl, Amany Salama, Eman ElZahaf, Sylvia A. Ashamallah, Abd AlRahman Foda, AzmyAbd El-Hameed Awad, Asem Shalaby
European Journal of Obstetrics & Gynecology and Reproductive Biology: X.2022; 16: 100172. CrossRef - Immunohistochemical Expression of Oestrogen and Epidermal Growth Factor Receptors in Endometrial Cancerous in Sudanese Patients
Salwa Abdalraheem Abubaker, Mohamed Elfatih Abdelwadoud, Mutaz Mohamed Ali, Hadia Alhaj Ahmad, Abuobieda Mohamed Khlafalla, Osman Mohammed Elmahi, Hisham Ali Waggiallah
Journal Of Biochemical Technology.2021; 12(1): 58. CrossRef - Expression of ER/PR Receptor, Her-2/neu, Ki67 and p53 in Endometrial Carcinoma: Clinicopathological Implication and Prognostic Value
V. B. Shivkumar, Manisha A. Atram, Nitin M. Gangane
Indian Journal of Gynecologic Oncology.2020;[Epub] CrossRef - Immunohistochemical study of ER, PR, p53 and Ki67 expression in patients with endometrial adenocarcinoma and atypical endometrial hyperplasia
Rachana Lakhe, Ravi M Swami, Preeti Doshi, Manjiri N Karandikar, Ravindra Nimbargi
IP Archives of Cytology and Histopathology Research.2020; 5(4): 274. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
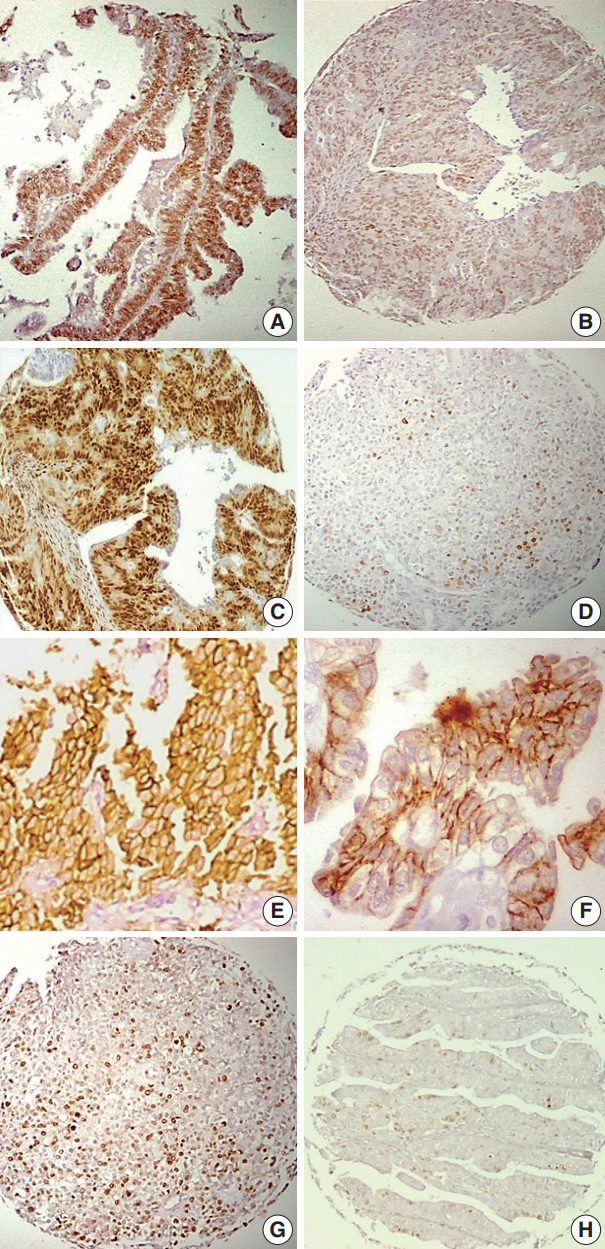
Fig. 1.
| ER score |
||||
|---|---|---|---|---|
| Category 1 (n = 45) | Category 2 (n = 40) | Category 3 (n = 20) | Test of significance | |
| Grade | ||||
| G1 | 10 (22.2) | 20 (50.0) | 6 (30.0) | MC, p = .021 |
| G2 | 15 (33.3) | 13 (32.5) | 11 (55.0) | χ2 = 2.9, p = .233 |
| G3 | 20 (44.4) | 7 (17.5) | 3 (15.0) | χ2 = 2.67, p = .007 |
| Stage | MC, p = .057 | |||
| I & II | 32 (78.0) | 35 (94.6) | 16 (94.1) | |
| III & IV | 9 (22.0) | 2 (5.4) | 1 (5.9) | |
| Depth | χ2 = 1.97, p = .362 | |||
| Inner half | 27 (65.9) | 28 (73.7) | 15 (83.3) | |
| Outer half | 14 (34.1) | 10 (26.3) | 3 (16.7) | |
| Cervical involvement | χ2 = 5.1, p = .081 | |||
| Absent | 30 (75.0) | 26 (72.2) | 15 (100) | |
| Present | 10 (25.0) | 10 (27.8) | 0 | |
| LVI | χ2 = 10.13, p = .006 | |||
| Present | 19 (42.2) | 7 (17.5) | 2 (10.0) | |
| Absent | 26 (57.8) | 33 (82.5) | 18 (90.0) | |
| Lymph node involvement | MC, p = .161 | |||
| Absent | 15 (71.4) | 18 (94.7) | 3 (75.0) | |
| Present | 6 (28.6) | 1 (5.3) | 1 (25.0) | |
| Ovarian involvement | MC, p = .025 | |||
| Absent | 30 (76.9) | 35 (97.2) | 14 (93.3) | |
| Present | 9 (23.1) | 1 (2.8) | 1 (6.7) | |
| Histology | MC, p = .007 | |||
| Non-endometrioid | 15 (33.3) | 3 (2.5) | 2 (10.0) | |
| Endometrioid | 30 (66.7) | 37 (92.5) | 18 (90.0) | |
| PR score |
||||
|---|---|---|---|---|
| Category 1 (n = 35) | Category 2 (n = 33) | Category 3 (n = 37) | Test of significance | |
| Grade | ||||
| G1 | 2 (5.7) | 12 (36.4) | 22 (59.5) | MC, p < .001 |
| G2 | 13 (37.1) | 13 (39.4) | 13 (35.1) | MC, p = .901 |
| G3 | 20 (57.1) | 8 (24.2) | 2 (5.4) | MC, p < .001 |
| Stage | ||||
| I & II | 25 (73.5) | 28 (93.3) | 30 (96.8) | MC, p = .009 |
| III & IV | 9 (26.5) | 2 (6.7) | 1 (3.2) | |
| Depth | ||||
| Inner half | 21 (63.6) | 24 (77.4) | 25 (75.8) | χ2 = 1.83, p = .401 |
| Outer half | 12 (36.4) | 7 (22.6) | 8 (24.20) | |
| Cervical involvement | ||||
| Absent | 23 (69.7) | 21 (75.0) | 27 (90.0) | χ2 = 3.9, p = .162 |
| Present | 10 (30.3) | 7 (25.0) | 3 (10.0) | |
| LVI | ||||
| Present | 13 (37.1) | 10 (30.3) | 5 (13.5) | χ2 = 5.46, p = .063 |
| Absent | 22 (62.9) | 23 (59.7) | 32 (86.5) | |
| Lymph node involvement | ||||
| Absent | 11 (64.7) | 10 (83.3) | 15 (100) | MC, p = .026 |
| Present | 6 (35.3) | 2 (16.7) | 0 | |
| Ovarian involvement | MC, p = .007 | |||
| Absent | 24 (75.0) | 25 (89.3) | 30 (100) | |
| Present | 8 (25.0) | 3 (10.7) | 0 | |
| Histology | χ2 = 21.89, p < .001 | |||
| Non-endometrioid | 15 (42.9) | 5 (15.2) | 0 | |
| Endometrioid | 20 (57.1) | 28 (84.8) | 37 (100) | |
| HER2 |
|||
|---|---|---|---|
| Negative (n = 102) | Positive (n = 3) | Fisher exact test p-value | |
| Grade | |||
| G1 | 36 (35.3) | 0 | .321 |
| G2 | 38 (37.3) | 1 (33.3) | > .992 |
| G3 | 28 (27.5) | 2 (66.7) | .192 |
| Stage | |||
| I & II | 82 (89.1) | 1 (33.3) | .042 |
| III & IV | 10 (10.9) | 2 (66.7) | |
| Depth | |||
| Inner half | 70 (74.5) | 0 | .022 |
| Outer half | 24 (25.5) | 3 (100) | |
| Cervical involvement | |||
| Absent | 71 (80.7) | 0 | .009 |
| Present | 17 (19.3) | 3 (100) | |
| LVI | |||
| Present | 25 (24.5) | 3 (100) | .017 |
| Absent | 77 (75.5) | 0 | |
| Lymph node involvement | |||
| Absent | 36 (85.7) | 0 | .032 |
| Present | 6 (14.3) | 2 (100) | |
| Ovarian involvement | |||
| Absent | 78 (89.7) | 1 (33.3) | .038 |
| Present | 9 (10.3) | 2 (66.7) | |
| Histology | |||
| Non-endometrioid | 19 (18.6) | 1 (33.3) | .473 |
| Endometrioid | 83 (81.4) | 2 (66.7) | |
| KI-67 |
||
|---|---|---|
| Median (min–max) | Test of significance | |
| Grade | ||
| G1 | 15.0 (0.5–90.0) | KW, p < .001 |
| G2 | 15.0 (0.5–75.0) | |
| G3 | 35.0 (0.5–80.0) | |
| Stage | ||
| I & II | 20.0 (0.5–90.0) | Z = 1.5, p = 0.132 |
| III & IV | 35.0 (2.0–80.0) | |
| Depth | ||
| Inner half | 17.0 (0.5–90.0) | Z = 0.11, p = .921 |
| Outer half | 23.0 (0.5–80.0) | |
| Cervical involvement | ||
| Absent | 20.0 (0.5–90.0) | Z = 0.18, p = .862 |
| Present | 20.0 (0.5–70.0) | |
| LVI | ||
| Present | 30.0 (0.5–80.0) | Z = 1.58, p = .113 |
| Absent | 18.5 (0.5–90.0) | |
| Lymph node involvement | ||
| Absent | 20.0 (0.5–70.0) | Z = 2.5, p = .012 |
| Present | 50.0 (8.0–80.0) | |
| Ovarian involvement | ||
| Absent | 20.0 (0.5–90.0) | Z = 1.36, p = .171 |
| Present | 30.0 (5.0–80.0) | |
| Histology | ||
| Non-endometrioid | 50.0 (5.0–80.0) | Z = 4.4, p < .001 |
| Endometrioid | 15.0 (0.5–90.0) | |
| ER score |
PR score |
|||||||
|---|---|---|---|---|---|---|---|---|
| Category 1 (n = 45) | Category 2 (n = 40) | Category 3 (n = 20) | Test of significance | Category 1 (n = 35) | Category 2 (n = 33) | Category 3 (n = 37) | Test of significance | |
| HER2 | MC, p = .812 | MC, p = .193 | ||||||
| Negative | 43 (95.6) | 39 (97.5) | 20 (100) | 34 (97.1) | 31 (93.3) | 37 (100) | ||
| Positive | 2 (4.4) | 1 (2.5) | 0 | 1 (2.9) | 2 (6.1) | 0 | ||
| Ki-67 | KW, p = .024 | KW, p = .025 | ||||||
| Median (min–max) | 30 (0.5–80) | 10 (0.5–80) | 25 (1–90) | 35.0 (0.5–80) | 15 (0.5–70) | 10 (0.5–90) | ||
Values are presented as number (%). ER, estrogen receptor; χ2, chi-square test; MC, Monte Carlo test; LVI, lymphovascular invasion.
Values are presented as number (%). PR, progesterone receptor; MC, Monte Carlo test; χ2, chi-square test; LVI, lymphovascular invasion.
Values are presented as number (%). HER2, human epidermal growth factor receptor 2; LVI, lymphovascular invasion.
KW, Kruskal-Walis test; Z, Mann-Whitney U test; LVI, lymphovascular invasion.
Values are presented as number (%). ER, estrogen receptor; PR, progesterone receptor; HER2, human epidermal growth factor receptor 2; MC, Monte Carlo test; KW, Kruskal-Walis test.

 E-submission
E-submission



