Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 53(2); 2019 > Article
-
Brief Case Report
Wharton Jelly Hair in a Case of Umbilical Cord Stricture and Fetal Death -
Eun Na Kim
 , Jae-Yoon Shim1
, Jae-Yoon Shim1 , Chong Jai Kim
, Chong Jai Kim
-
Journal of Pathology and Translational Medicine 2019;53(2):145-147.
DOI: https://doi.org/10.4132/jptm.2018.10.24
Published online: November 14, 2018
Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
1Department of Obstetrics and Gynecology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- Corresponding Author Chong Jai Kim, MD, PhD Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, 88 Olympic-ro 43-gil, Songpa-gu, Seoul 05505, Korea Tel: +82-2-3010-4516, Fax: +82-2-472-7898, E-mail: ckim@amc.seoul.kr
© 2019 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
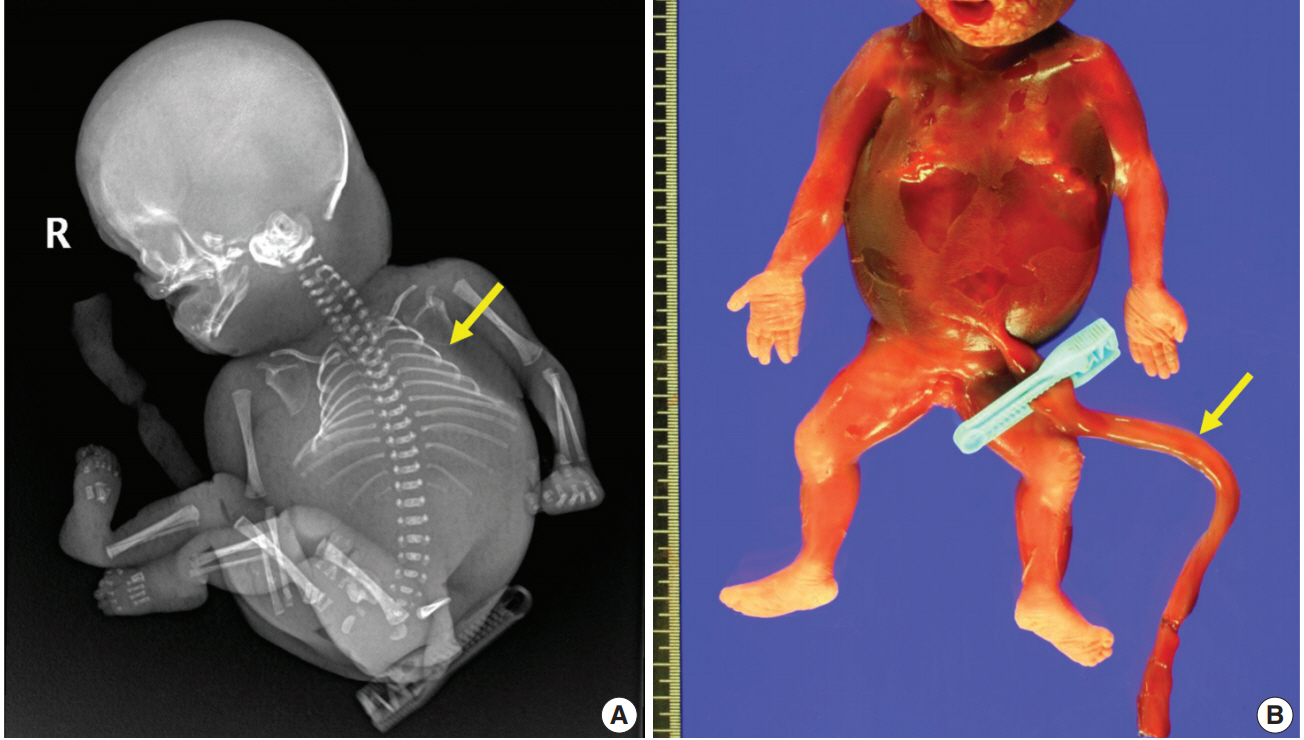
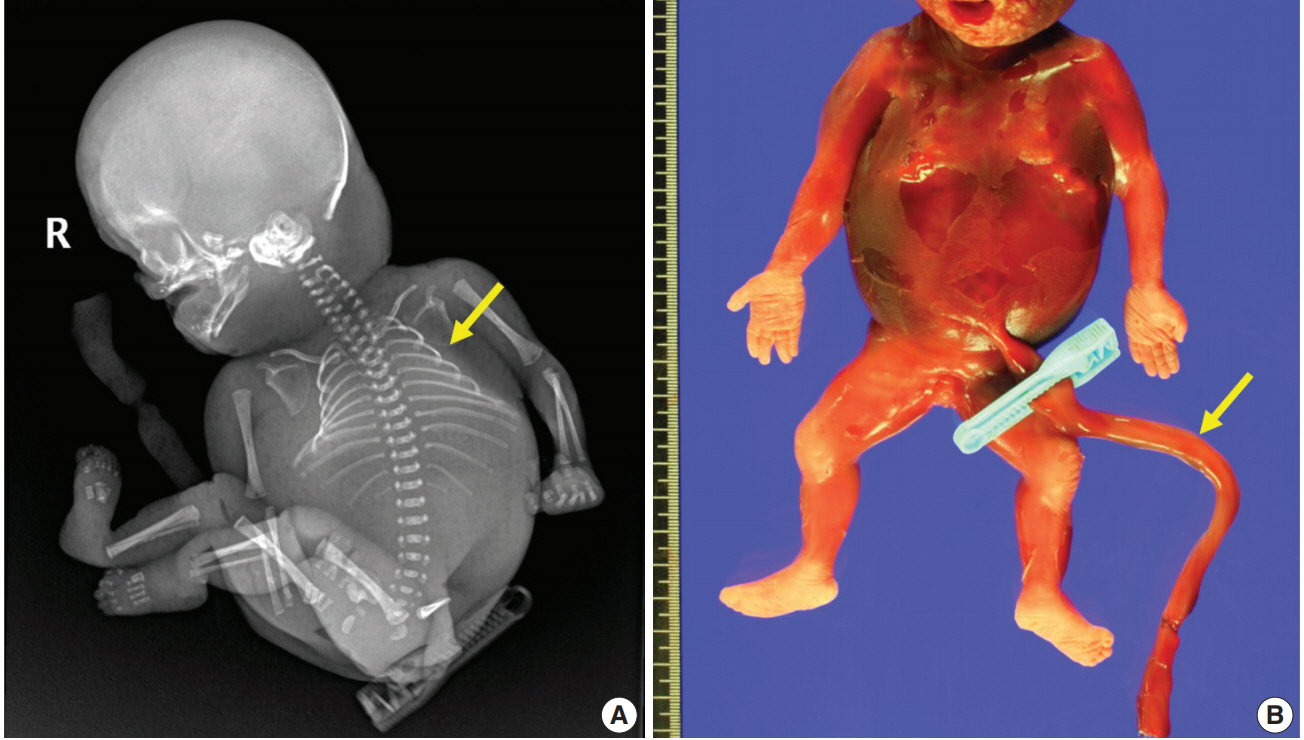
- A 28-year-old primigravida woman presented with severe fetal growth restriction and oligohydramnios at 23 weeks of gestational age. Five days later, the ultrasound examination revealed fetal death in utero, and a macerated female fetus weighing 350 g (< 3rd percentile) was delivered [5]. The circumference of the head was 18.0 cm (< 3rd percentile) [6], and that of the abdomen was 14.4 cm (< 3rd percentile) [6]. Infantogram showed mildly hypoplastic thoracic cage (Fig. 1A), and chest circumference was 14.6 cm (10th–25th percentile) [7]. Chromosomal analysis by amniocentesis performed at 18 weeks showed normal findings.
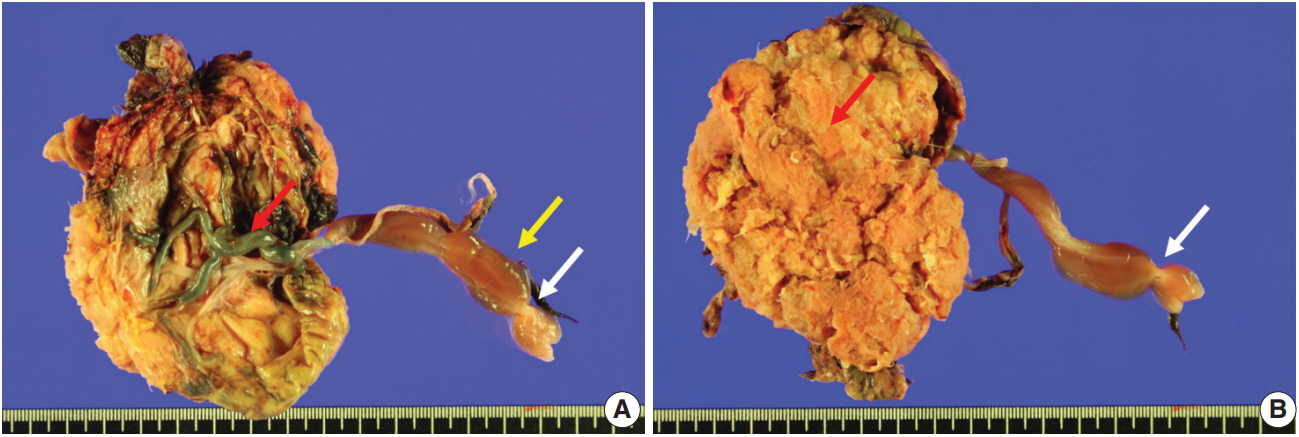
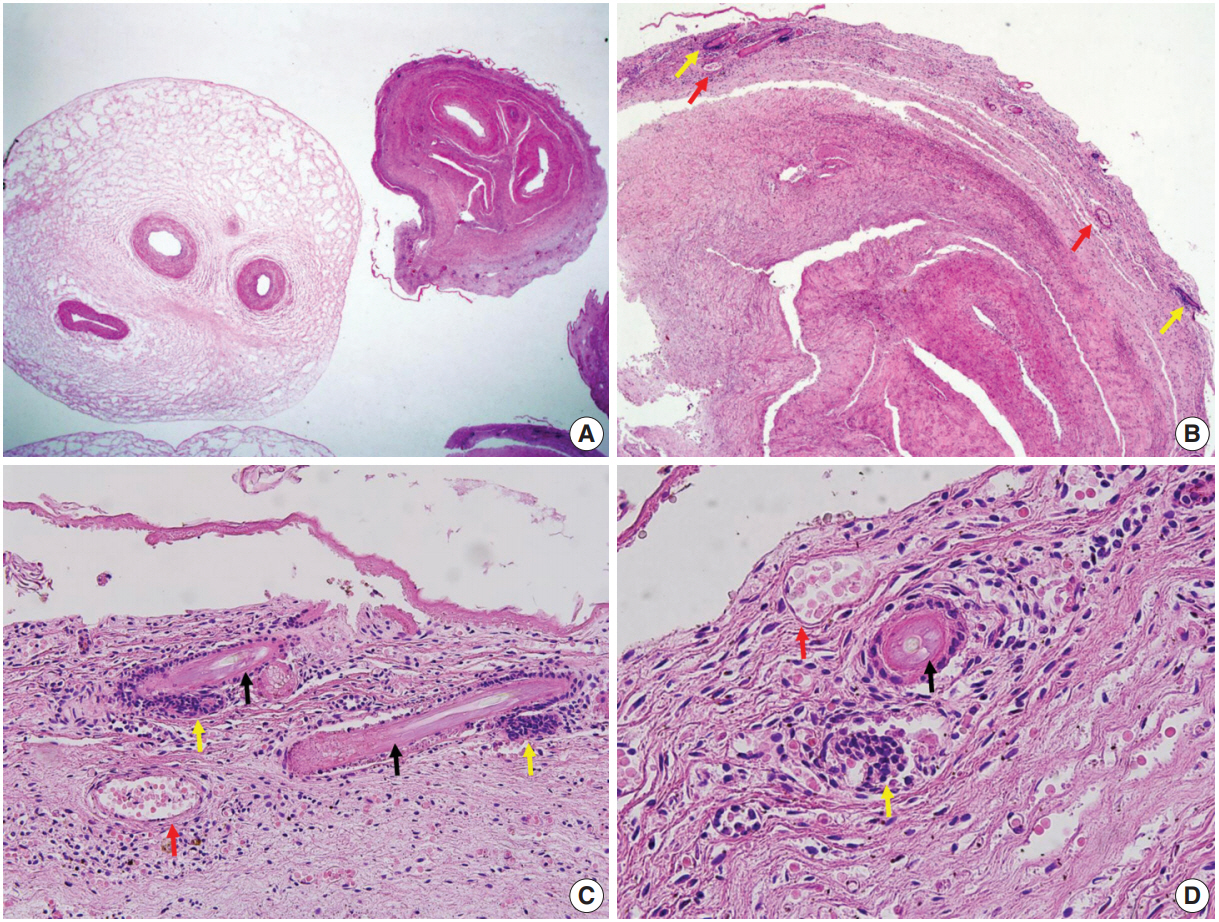
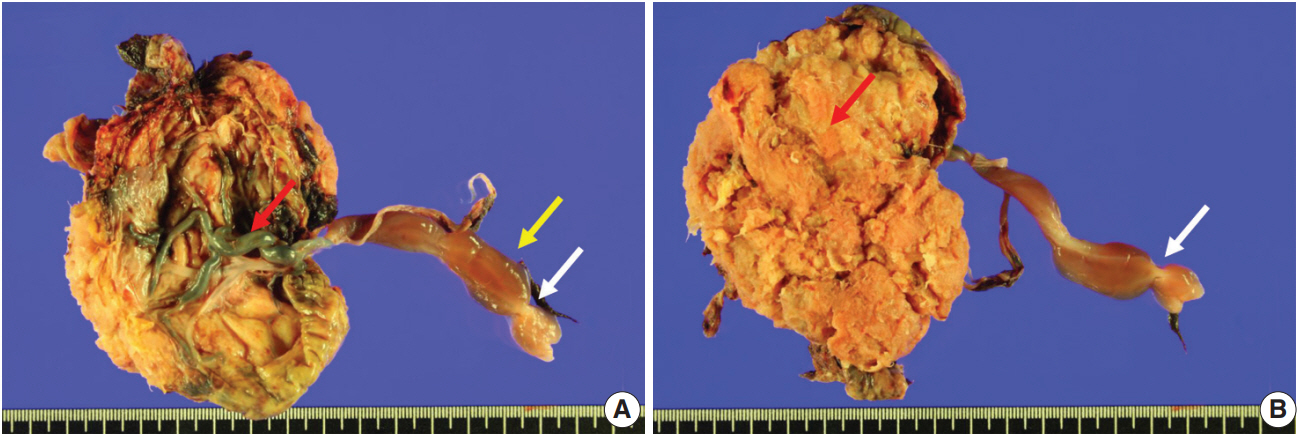
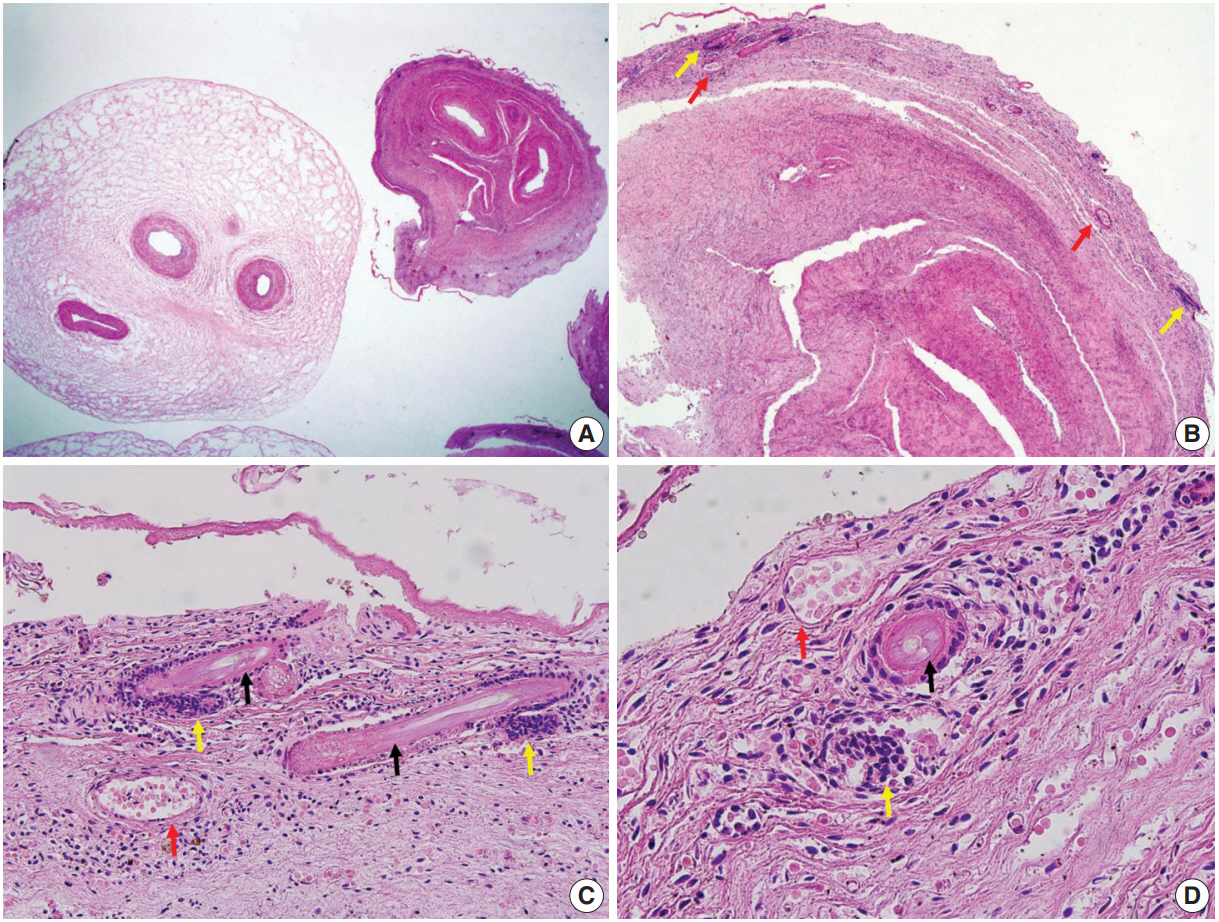
- Fetal autopsy and placental biopsy revealed hypocoiling of the umbilical cord (Fig. 1B) with a rather abrupt stricture located 6.5 cm from the placental insertion. Localized edema of the umbilical cord was associated with the stricture. The diameter of the umbilical cord at the stricture was 4 mm, which was lower than the 10th percentile of the normal range (Fig. 2) [1]. Microscopic examination of the umbilical cord stricture revealed hair follicles including hair shafts and papillary mesenchymal body in the firm and fibrotic Wharton jelly. Capillary vessel formation was also found in the peripheral region of the Wharton jelly (Fig. 3). Squames were detected in the alveoli of fetal lungs, and chorionic villi showed increased syncytial knots and distal villous hypoplasia. Overall, the umbilical cord stricture associated with unusual development of hair follicles and fibrotic Wharton jelly seems to have significantly contributed to the fetal demise.
- The mother has provided written informed consent for autopsy prior to the postmortem examinations. Since only the pathology and autopsy findings of an already deceased fetus and only deidentified personal information of the mother and the fetus were used, the Institutional Review Board (IRB) of Asan Medical Center determined exemption (IRB number: 2017-1192).
CASE REPORT
- The Wharton jelly is the umbilical cord stroma that originates from the extraembryonic mesoderm of allantois. The Wharton jelly is rich in mucopolysaccharides such as hyaluronic acid and chondroitin sulfate and thus protects umbilical vessels from compression [8]. Loss of protection by the Wharton jelly can lead to compromised fetoplacental circulation and subsequent fetal death [9]. Umbilical cord stricture is a known cause of fetal death [3]. Hypocoiling also renders umbilical cord vessels susceptible to kinking and acute obstruction.
- The Wharton jelly has been extensively studied as a reservoir of mesenchymal stem cells (MSCs) in the field of regenerative medicine. The Wharton jelly MSCs show a differentiation spectrum covering ectodermal and endodermal lineages [10]. To the best of our knowledge, the current case is the first example of hair growth and follicle formation in the umbilical cord. It should also be mentioned that hair formation overlaps with a fibrotic change in the Wharton jelly and the development of capillaries. Capillaries are not normally observed in the Wharton jelly, and the anatomical location of the stricture is apart from the umbilicus, where fetal skin tissue can coexist. It is unclear at this point whether the hair and capillaries are products of de novo differentiation of pluripotent Wharton jelly MSCs. The development of multiple capillaries in the periphery and fibrosis of Wharton jelly may be secondary consequences of stricture, and the capillaries can be considered collaterals for compensating for chronically compromised umbilical circulation.
- In addition to developmental uniqueness, lessons from the current case include that thorough histologic examination is necessary to determine the cause of fetal death and document significant findings.
DISCUSSION
Author contributions
Conceptualization: CJK.
Data curation: ENK.
Investigation: ENK.
Methodology: ENK.
Resources: JYS.
Supervision: CJK.
Visualization: CJK.
Writing—original draft: ENK.
Writing—review & editing: CJK, JYS.
Conflicts of Interest
The authors declare that they have no potential conflicts of interest.
- 1. Proctor LK, Fitzgerald B, Whittle WL, et al. Umbilical cord diameter percentile curves and their correlation to birth weight and placental pathology. Placenta 2013; 34: 62-6. ArticlePubMed
- 2. Baergen RN. Cord abnormalities, structural lesions, and cord “accidents”. Semin Diagn Pathol 2007; 24: 23-32. ArticlePubMed
- 3. Horn LC, Faber R, Stepan H, Simon E, Robel R, Wittekind C. Umbilical cord hypercoiling and thinning: a rare cause of intrauterine death in the second trimester of pregnancy. Pediatr Dev Pathol 2006; 9: 20-4. ArticlePubMedPDF
- 4. Benirschke K. Obstetrically important lesions of the umbilical cord. J Reprod Med 1994; 39: 262-72. PubMed
- 5. Lee JK, Jang HL, Kang BH, et al. Percentile distributions of birth weight according to gestational ages in Korea (2010-2012). J Korean Med Sci 2016; 31: 939-49. ArticlePubMedPMCPDF
- 6. Jung SI, Lee YH, Moon MH, et al. Reference charts and equations of Korean fetal biometry. Prenat Diagn 2007; 27: 545-51. ArticlePubMed
- 7. Chitkara U, Rosenberg J, Chervenak FA, et al. Prenatal sonographic assessment of the fetal thorax: normal values. Am J Obstet Gynecol 1987; 156: 1069-74. ArticlePubMed
- 8. Benirschke K, Burton GJ, Baergen RN. Pathology of the human placenta. 6th ed. New York: Springer-Verlag, 2016; 403.
- 9. Peng HQ, Levitin-Smith M, Rochelson B, Kahn E. Umbilical cord stricture and overcoiling are common causes of fetal demise. Pediatr Dev Pathol 2006; 9: 14-9. ArticlePubMedPDF
- 10. Aljitawi OS, Xiao Y, Zhang D, et al. Generating CK19-positive cells with hair-like structures from Wharton’s jelly mesenchymal stromal cells. Stem Cells Dev 2013; 22: 18-26. ArticlePubMed
REFERENCES
Figure & Data
References
Citations

- Placental and Umbilical Cord Anomalies Detected by Ultrasound as Clinical Risk Factors of Adverse Perinatal Outcomes. Case Series Review of Selected Conditions. Part 3: Vascular Anomalies of the Umbilical Cord and Fetoplacental Vascular Malperfusion
Gabriele Tonni, Mario Lituania, Gianpaolo Grisolia, Alessia Pinto, Maria Paola Bonasoni, Giuseppe Rizzo, Rodrigo Ruano, Edward Araujo Júnior, Heron Werner, Waldo Sepulveda, Gianluigi Pilu
Journal of Clinical Ultrasound.2025; 53(4): 863. CrossRef - What is your diagnosis?
Mrudula Chandrupatla, Mishu Mangla, Ariyanachi Kaliappan, Seetu Palo, Aparna Setty, Geetha Sulochana Gopidas, Punnapa Raviteja, Deepa G
Journal of the Turkish-German Gynecological Association.2025;[Epub] CrossRef - Histomorphometric analysis of Wharton's jelly size in pregnancies with normal and pre-eclamptic conditions, and its relation to baby weight
Ankit Jain, Rashmi Jain, Amarjyoti Chaturvedi
Indian Journal of Obstetrics and Gynecology Research.2024; 11(4): 647. CrossRef - Loss of Wharton's jelly and fibrosis in umbilical cord stricture area: A case report
Alžbeta Blichárová, Ľudmila Verbóová, Zuzana Benetinová, Tibor Mátyás, Richard Bašista, Vladimír Tancoš, Patrícia Kollárová
Journal of Forensic and Legal Medicine.2023; 96: 102512. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure

 E-submission
E-submission





