Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 52(5); 2018 > Article
-
Original Article
Significance of Intratumoral Fibrosis in Clear Cell Renal Cell Carcinoma - Jae Won Joung, Hoon Kyu Oh, Sun Jae Lee, Young Ah Kim, Hyun Jin Jung1
-
Journal of Pathology and Translational Medicine 2018;52(5):323-330.
DOI: https://doi.org/10.4132/jptm.2018.07.21
Published online: August 19, 2018
1Department of Pathology, Catholic University of Daegu School of Medicine, Daegu, Korea
2Department of Urology, Catholic University of Daegu School of Medicine, Daegu, Korea
- Corresponding Author Hoon Kyu Oh, MD Department of Pathology, Catholic University of Daegu School of Medicine, 33 Duryugongwon-ro 17-gil, Nam-gu, Daegu 42472, Korea Tel: +82-53-650-4156 Fax: +82-53-650-3456 E-mail: ap510@cu.ac.kr
© 2018 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Intratumoral fibrosis (ITF) is a frequent histologic finding in solid organ tumors. Renal cell carcinoma (RCC) is a highly vascularized tumor with different shapes and degrees of ITF and inflammation. ITF is a poor prognostic factor, especially in breast cancer, and is related to intratumoral necrosis (ITN) and intratumoral inflammation (ITI). However, the significance of ITF in RCC has not been fully studied. In this study, we evaluate the relationships between ITF and other clinicopathologic parameters associated with RCC prognosis.
-
Methods
- ITF was evaluated in 204 clear cell renal cell carcinoma (CCRCC) specimens according to presence and grade of fibrosis, degree of ITI, and presence of ITN. Lysyl oxidase (LOX) expression in tumor cells was also evaluated with clinicopathologic parameters.
-
Results
- Among 204 CCRCC cases, 167 (81.7%) showed ITF, 71 (34.8%) showed ITI, 35 (17.2%) showed ITN, and 111 (54.4%) showed LOX expression. ITF correlated with Fuhrman nuclear grade (p = .046), lymphovascular invasion (LVI) (p = .027), and ITN (p = .036). Patients with ITF had a poor five-year overall survival rate (p = .104).
-
Conclusions
- ITF is related to other poor prognostic factors in CCRCC, such as Fuhrman nuclear grade, ITN, and LVI, but ITF itself had no significant correlation with prognosis of CCRCC.
- Patients and materials
- This study was performed on 224 RCC samples diagnosed from 2003 to 2015 at Daegu Catholic University Medical Center. The specimens included 163 total nephrectomies and 61 partial nephrectomies. Follow-up data were started at the time of diagnosis until August 31, 2016. The average follow-up period was 66.3 months, excluding patients lost to follow-up. Two pathologists (H.K. Oh and J.W. Joung) reviewed all slides to evaluate the diagnosis, histologic type, Fuhrman nuclear grade, tumor diameter, lymphovascular invasion (LVI), ITF, ITI, and ITN. This study was approved by the Institutional Review Board of Daegu Catholic University Medical Center (CR-18-009-RES-001-R) and performed in accordance with the Helsinki declaration. Because this is a retrospective study, informed consent was waived. All patient information was anonymized prior to analysis.
- Tissue microarray and immunohistochemistry
- Tissue microarrays that contained representative tumor cores (2 mm in diameter) were used to evaluate LOX expression. Immunohistochemistry was performed using polyclonal rabbit LOX antibody (dilution 1:500, Novus Biologicals, Littleton, CO, USA) in the Leica Bond-Max automated immunostainer (Leica Biosystems, Wetzlar, Germany).
- Grading of intratumoral fibrosis, intratumoral inflammation, and LOX expression
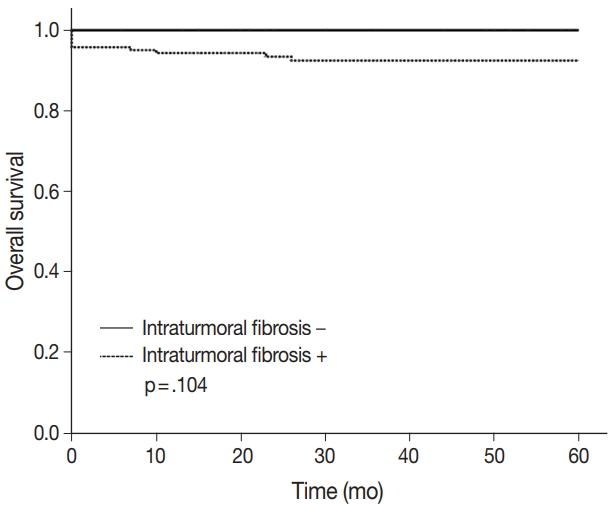
- ITF is composed of fibroblasts admixed with various volumes of collagen surrounded by tumor cells and located within the tumor boundary. Like many studies in breast cancer, ITF is defined when the size is more than 0.1 cm [15,16]. In this study, ITF was classified into grades 0 to 3. Grade 0 ITF was defined as no fibrosis within tumor. Grade 1 ITF was mainly composed of loose connective tissue, grade 2 ITF was composed of collagen fibers and loose connective tissue, and grade 3 ITF was mainly composed of thick sclerotic collagen fibers (Fig. 1).
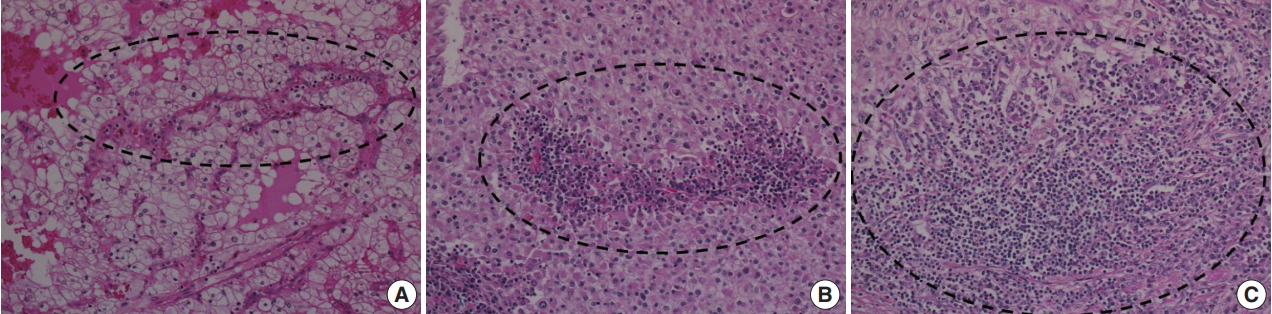
- To investigate the relationship between ITF and ITI, ITI was graded from 0 to 3 according to the amount of inflammatory cells within the tumor boundary. Grade 0 ITI had no inflammatory cell infiltration, while grade 1 ITI had a few scattered inflammatory cells. Grade 2 ITI was characterized by focal aggregation of inflammatory cells, and grade 3 ITI was defined as diffuse or nodular aggregation of inflammatory cells (Fig. 2). ITF grade 0 was classified as negative, and grades 1, 2, and 3 were classified as positive. ITI of 0 or 1 was classified as negative, and ITI grade 2 or 3 was classified as positive.
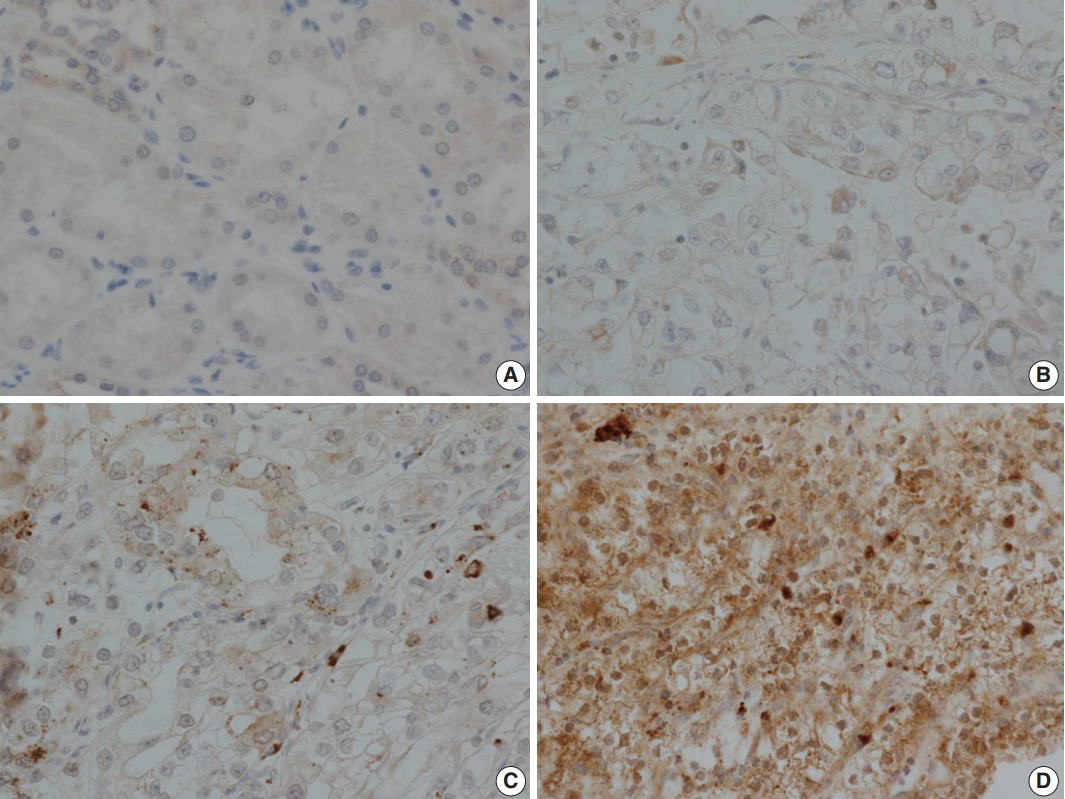
- LOX expression level was graded as 0 to 3 according to the degree of intensity and proportion of stained tumor cells: LOX expression is scored as 0 (no staining), 1 (minimal intensity, < 10% of cells), 2 (moderate intensity, 10%–49% of cells), or 3 (strong intensity, ≥ 50% of cells). LOX expression was then classified as negative (0 and 1) or positive (2 and 3). Every tissue microarray contained normal kidney as a control group. In normal kidneys, tubular cells show no reactivity to LOX (Fig. 3).
- Statistical analysis
- All statistical analyses were performed using SPSS software program ver. 19.0 for Windows (SPSS Inc., Chicago, IL, USA). The relationships between ITF, ITI, LOX expression, and clinicopathologic parameters were analyzed using the chi-square test. The 5-year overall survival rate according to presence of ITF was analyzed using the Kaplan-Meier method. The clinicopathologic parameters evaluated were patient age, sex, histologic type, Fuhrman nuclear grade, tumor size, pathologic staging, LVI, and ITN.
MATERIALS AND METHODS
- Clinicopathologic characteristics of patients
- In 224 cases of RCC, the three most common tumor types were CCRCC (204 cases), papillary RCC (11 cases), and chromophobe RCC (5 cases). Other types include mucinous RCC (one case), mucinous tubular and spindle-cell carcinoma (one case), oncocytoma (one case), and unclassified RCC (one case). CCRCC cases represented 91.1% of the total RCC cases (204/224).
- Of the 204 patients with CCRCC, 139 (68.1%) were male and 65 (31.9%) were female. The average age (mean ± standard deviation [SD]) at diagnosis was 59.7 ± 11.8 years and ranged between 27 and 87 years. Follow-up data were obtained from 191 patients (93.6%) with average follow-up period of 50.4 months. Based on the follow-up data available on patients, 11 patients (5.8%) died of disease, and 25 patients (13.1%) had metastasis to other organs.
- Average tumor diameter (mean ± SD) was 4.48 ± 3.01 cm (range 0.5 to 19 cm). Fuhrman nuclear grade distribution was 12 G1 (5.9%), 118 G2 (57.8%), 55 G3 (27.0%), and 19 G4 (9.3%). A total of 130 cases (63.7%) were low grade (grade 1 and 2), and 74 cases (36.3%) were high grade (grade 3 and 4). There were 154 cases of pT1 (75.5%), 19 cases of pT2 (9.3%), 30 cases of pT3 (14.7%), and one case of pT4 (0.5%). A total of 173 cases (84.8%) were low stage (stages 1 and 2), and 31 cases (15.2%) were high stage (stages 3 and 4). When performing statistical analysis, Fuhrman nuclear grade and pathologic T stage were divided into low (nuclear grades 1 and 2 or T stages 1 and 2) and high (nuclear grades 3 and 4 or T stages 3 and 4) groups. The clinicopathologic characteristics are listed in Table 1.
- Associations between ITF and clinicopathologic characteristics
- ITF was found in 167 cases (81.7%), indicating that ITF is a relatively common finding in CCRCC. Clinicopathologic parameters that are strongly associated with ITF are Fuhrman nuclear grade (p = .003), LVI (p = .027), and ITN (p = .036) according to presence (grades 1, 2 and 3) and absence (grade 0) of ITF. LOX expression status was not statistically associated with ITF (Table 2).
- Associations between ITI and clinicopathologic characteristics
- ITI was found in 71 (34.8%) cases. Clinicopathologic parameters that are strongly associated with ITI are tumor size (p < .001), Fuhrman nuclear grade (p < .001), pathologic stage (p = .004), LVI (p < .001), and ITN (p < .001). The LOX expression status was not statistically associated with ITI (Table 3).
- Associations between LOX expression and clinicopathologic characteristics
- LOX expression was found in 111 cases (54.4%). No clinicopathologic parameters evaluated were statistically associated with LOX expression (Table 4).
- Associations between ITF and clinicopathologic characteristics
- ITF size was on average 0.56 cm with a SD of 0.42 cm, ranging between 0.1 and 2.0 cm. The median value was 0.5 cm. ITF size was divided into two groups: small was defined as 0.5 cm or less, large was defined as over 0.5 cm. Large ITF size is associated with a higher Fuhrman nuclear grade (p = .007) (Table 5).
- Association between ITF and patient prognosis
- The 5-year overall survival rate of patients excluding loss to follow-up was 94.2% (180/191). Patients without ITF had 100% (36/36) 5-year overall survival rate, and patients with ITF had 92.9% (144/155) 5-year overall survival rate. The p-value was .104 (Fig. 4). Statistically, ITF and 5-year overall survival rate showed no significant correlation.
RESULTS
- There are many similar mechanisms between tumor development, tumor metastases, and formation of fibrosis. One of these mechanisms is ECM remodeling [17,18], which occurs predominantly while forming fibrosis and results in many changes in the ECM. These changes also activate other signaling pathways [10,11,19].
- Inflammatory cell infiltration in tumors and the tumor microenvironment is a significant factor in tumor progression. Recent studies have shown that tumor-associated immune cells play a role in RCC initiation and development [20,21]. Some inflammatory cells in RCC, such as T lymphocytes and macrophages, may affect tumor progression and invasion [22]. Yang et al. [23] reported that infiltrating macrophages could accelerate cancer invasion by increasing the epithelial to mesenchymal transition. This ability of macrophages is thought to be associated with activation of the AKT/mammalian target of rapamycin signal. Further research on this pathway could be helpful in developing a new target therapy for RCC.
- There is increasing evidence that ITF is associated with poor prognosis in breast cancers [15,16]. ITF is related to many factors, such as higher histologic grade, pathologic T stage, lymph node metastasis, and increased risk of recurrence in breast cancer. In many studies, ITF as a prognostic factor independently correlates with other prognostic factors [24]. In 1987, Delahunt and Nacey [25] studied prognostic factors of RCC with 102 cases. They accessed many clinicopathological parameters to find any relationship with prognosis. They categorized fibrosis into three groups: slight or absent, moderate, and severe. Their study showed that fibrosis was not correlated with prognosis of RCC.
- In our study, ITF is related to higher histologic grade, LVI, ITN, and ITI in CCRCC. Frank et al. [26] reported that ITN is a prognostic factor according to the SSIGN (tumor stage, size, grade, and necrosis) scoring system. Our result shows that ITF and ITI are strongly associated with ITN. We statistically analyzed two groups of ITF to identify differences between the presence and grades of ITF. In group 1, ITF 0 was classified as negative, and ITF 1, 2, and 3 were classified as positive. In group 2, ITF 0 and 1 were considered negative, and ITF 2 and 3 were considered positive. Statistically, no significant differences were identified between these two groups, probably because the presence of ITF itself is a meaningful finding rather than ITF grade. The large ITF size is related to a higher Fuhrman nuclear grade. The 5-year overall survival rate tends to be better for patients who do not have ITF, but statistical significance has not been identified (p = .104).
- LOX modifies collagen and elastin fibers in the ECM [27]. Collagen I is the most common substrate used by LOX. The most important factor in forming fibrosis is increased synthesis of ECM protein by fibroblast activation, and collagen type I is the most commonly synthesized protein during this process. When collagen I is present at a high level in vital organs, it sometimes causes severe morbidity and mortality. Many studies have shown that collagen I plays a major role in cancer development and metastases [28]. In breast cancers, the formation of fibrosis by remodeling of the ECM was also found to be associated with gene expression patterns related to adverse prognosis and cancer metastasis [29-32]. Jensen et al. [33] reported that, in recurrent breast cancer, an increased level of the collagen I precursor, procollagen I, could be detected in patient serum.
- In this study, we investigated the relationship between LOX expression and ITF formation. Our hypothesis was that LOX participates in formation of ITF in CCRCC. However, our results show no correlation between LOX expression and ITF formation or other clinicopathologic features. Hence, ITF in CCRCC may be formed by other cytokines during ECM remodeling. Further study of other fibrogenic cytokines, such as transforming growth factor β and tumor necrosis factor α, is needed.
- Our results show no relationship of ITF or ITI with LOX expression; there may be some reasons for this result. First, LOX is not involved in forming ITF of CCRCC. Second, there could be other classifications for grading LOX expression. We assumed that all normal tubular cells show no reactivity to LOX, although some normal tubular cells showed grade 1 expression. If we adjust LOX grading, different results may be obtained. We also found relatively low rates of death by CCRCC because the follow-up period was short and there was a high proportion of low-grade patients (Fuhrman nuclear grade 1 or 2).
- Our results show that ITF is a pathologic finding related to other adverse prognostic factors, including higher Fuhrman nuclear grade, ITN, and LVI. However, ITF itself shows no direct correlation with poor prognosis of CCRCC. LOX expression shows no correlation with ITF or any other clinicopathologic parameters in this study. Further evaluation of other fibrogenic cytokines in addition to LOX should be considered.
DISCUSSION
| Clinicopathological parameter |
Intratumoral fibrosis |
p-value | |
|---|---|---|---|
| Absent | Present | ||
| Age (yr) | |||
| < 60 | 21 (20.8) | 80 (79.2) | .330 |
| ≥ 60 | 16 (15.5) | 87 (84.5) | |
| Sex | |||
| Male | 26 (18.7) | 113 (81.3) | .758 |
| Female | 11 (16.9) | 54 (83.1) | |
| Tumor size (cm) | |||
| ≤ 7 | 34 (20.2) | 134 (79.8) | .093 |
| > 7 | 3 (8.3) | 33 (91.7) | |
| Fuhrman nuclear grade | |||
| Low (grade 1 and 2) | 31 (23.8) | 99 (76.2) | .003** |
| High (grade 3 and 4) | 6 (8.1) | 68 (91.9) | |
| Pathologic stage | |||
| Low (stage 1 and 2) | 33 (19.1) | 140 (80.9) | .295 |
| High (stage 3 and 4) | 4 (12.9) | 27 (87.1) | |
| Lymphovascular invasion | |||
| Absent | 36 (20.6) | 139 (79.4) | .027* |
| Present | 1 (3.4) | 28 (96.6) | |
| Tumor necrosis | |||
| Absent | 35 (20.7) | 134 (79.3) | .036* |
| Present | 2 (5.7) | 33 (94.3) | |
| Intratumoral inflammation | |||
| Absent | 25 (18.8) | 108 (81.2) | .738 |
| Present | 12 (16.9) | 59 (83.1) | |
| LOX expression | |||
| Absent | 16 (17.2) | 77 (82.8) | .752 |
| Present | 21 (18.9) | 90 (81.1) | |
| Clinicopathological parameter |
Intratumoral fibrosis |
p-value | |
|---|---|---|---|
| Absent | Present | ||
| Age (yr) | |||
| < 60 | 67 (66.3) | 34 (33.7) | .735 |
| ≥ 60 | 66 (64.1) | 37 (35.9) | |
| Sex | |||
| Male | 85 (61.2) | 54 (38.8) | .076 |
| Female | 48 (73.8) | 17 (26.2) | |
| Tumor size (cm) | |||
| ≤ 7 | 120 (71.4) | 48 (28.6) | < .001*** |
| > 7 | 13 (36.1) | 23 (63.9) | |
| Fuhrman nuclear grade | |||
| Low (grade 1 and 2) | 102 (78.5) | 28 (21.5) | < .001*** |
| High (grade 3 and 4) | 31 (41.9) | 43 (58.1) | |
| Pathologic stage | |||
| Low (stage 1 and 2) | 120 (69.4) | 53 (30.6) | .004** |
| High (stage 3 and 4) | 13 (41.9) | 18 (58.1) | |
| Lymphovascular invasion | |||
| Absent | 124 (70.9) | 51 (29.1) | < .001*** |
| Present | 9 (31.0) | 20 (69.0) | |
| Tumor necrosis | |||
| Absent | 124 (73.4) | 45 (26.6) | < .001*** |
| Present | 9 (25.7) | 26 (74.3) | |
| Intratumoral fibrosis | |||
| Absent | 25 (67.6) | 12 (32.4) | .738 |
| Present | 108 (64.7) | 59 (35.3) | |
| LOX expression | |||
| Absent | 60 (64.5) | 33 (35.5) | .852 |
| Present | 73 (65.8) | 38 (34.8) | |
| Clinicopathological parameter |
Intratumoral fibrosis size (cm) |
p-value | |
|---|---|---|---|
| ≤ 0.5 | > 0.5 | ||
| Age (yr) | |||
| < 60 | 57 (71.3) | 23 (28.7) | .523 |
| ≥ 60 | 58 (66.7) | 29 (33.3) | |
| Sex | |||
| Male | 82 (72.6) | 31 (27.4) | .135 |
| Female | 33 (61.1) | 21 (38.9) | |
| Tumor size (cm) | |||
| ≤ 7 | 96 (71.6) | 38 (28.4) | .118 |
| > 7 | 19 (57.6) | 14 (42.4) | |
| Fuhrman nuclear grade | |||
| Low (grade 1 and 2) | 76 (76.8) | 23 (23.2) | .007** |
| High (grade 3 and 4) | 39 (57.4) | 29 (42.6) | |
| Pathologic stage | |||
| Low (stage 1 and 2) | 97 (69.3) | 43 (30.7) | .475 |
| High (stage 3 and 4) | 18 (66.7) | 9 (33.3) | |
| Lymphovascular invasion | |||
| Absent | 100 (71.9) | 39 (28.1) | .055 |
| Present | 15 (53.6) | 13 (46.4) | |
| Tumor necrosis | |||
| Absent | 95 (70.9) | 39 (29.1) | .253 |
| Present | 20 (60.6) | 13 (39.4) | |
| Intratumoral inflammaion | |||
| Absent | 78 (72.2) | 30 (27.8) | .205 |
| Present | 37 (62.7) | 22 (37.3) | |
| LOX expression | |||
| Absent | 50 (64.9) | 27 (35.1) | .311 |
| Present | 65 (72.2) | 25 (27.8) | |
- 1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin 2016; 66: 7-30. ArticlePubMedPDF
- 2. Joung JY, Lim J, Oh CM, et al. Current trends in the incidence and survival rate of urological cancers in Korea. Cancer Res Treat 2017; 49: 607-15. ArticlePubMedPDF
- 3. Srigley JR, Delahunt B, Eble JN, et al. The International Society of Urological Pathology (ISUP) Vancouver classification of renal neoplasia. Am J Surg Pathol 2013; 37: 1469-89. ArticlePubMed
- 4. Gerlinger M, Rowan AJ, Horswell S, et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med 2012; 366: 883-92. PubMedPMC
- 5. Beksac AT, Paulucci DJ, Blum KA, Yadav SS, Sfakianos JP, Badani KK. Heterogeneity in renal cell carcinoma. Urol Oncol 2017; 35: 507-15. ArticlePubMed
- 6. Hiley C, de Bruin EC, McGranahan N, Swanton C. Deciphering intratumor heterogeneity and temporal acquisition of driver events to refine precision medicine. Genome Biol 2014; 15: 453.ArticlePubMedPMCPDF
- 7. Palsdottir HB, Hardarson S, Petursdottir V, et al. Incidental detection of renal cell carcinoma is an independent prognostic marker: results of a long-term, whole population study. J Urol 2012; 187: 48-53. ArticlePubMed
- 8. Steeg PS. Tumor metastasis: mechanistic insights and clinical challenges. Nat Med 2006; 12: 895-904. ArticlePubMedPDF
- 9. Cox TR, Bird D, Baker AM, et al. LOX-mediated collagen crosslinking is responsible for fibrosis-enhanced metastasis. Cancer Res 2013; 73: 1721-32. ArticlePubMedPMCPDF
- 10. Bissell MJ, Labarge MA. Context, tissue plasticity, and cancer: are tumor stem cells also regulated by the microenvironment? Cancer Cell 2005; 7: 17-23. ArticlePubMedPMC
- 11. Paszek MJ, Zahir N, Johnson KR, et al. Tensional homeostasis and the malignant phenotype. Cancer Cell 2005; 8: 241-54. ArticlePubMed
- 12. Cox TR, Erler JT. Remodeling and homeostasis of the extracellular matrix: implications for fibrotic diseases and cancer. Dis Model Mech 2011; 4: 165-78. ArticlePubMedPMCPDF
- 13. Lucero HA, Kagan HM. Lysyl oxidase: an oxidative enzyme and effector of cell function. Cell Mol Life Sci 2006; 63: 2304-16. ArticlePubMedPMCPDF
- 14. Chang HM, Cheng JC, Liu Y, Klausen C, Xu C, Leung PC. Activin A-induced increase in LOX activity in human granulosa-lutein cells is mediated by CTGF. Reproduction 2016; 152: 293-301. ArticlePubMed
- 15. Mujtaba SS, Ni YB, Tsang JY, et al. Fibrotic focus in breast carcinomas: relationship with prognostic parameters and biomarkers. Ann Surg Oncol 2013; 20: 2842-9. ArticlePubMedPDF
- 16. Hasebe T, Sasaki S, Imoto S, Mukai K, Yokose T, Ochiai A. Prognostic significance of fibrotic focus in invasive ductal carcinoma of the breast: a prospective observational study. Mod Pathol 2002; 15: 502-16. ArticlePubMedPDF
- 17. Wynn TA. Common and unique mechanisms regulate fibrosis in various fibroproliferative diseases. J Clin Invest 2007; 117: 524-9. ArticlePubMedPMC
- 18. Keeley EC, Mehrad B, Strieter RM. Fibrocytes: bringing new insights into mechanisms of inflammation and fibrosis. Int J Biochem Cell Biol 2010; 42: 535-42. ArticlePubMed
- 19. Butcher DT, Alliston T, Weaver VM. A tense situation: forcing tumour progression. Nat Rev Cancer 2009; 9: 108-22. ArticlePubMedPMCPDF
- 20. Geissler K, Fornara P, Lautenschläger C, Holzhausen HJ, Seliger B, Riemann D. Immune signature of tumor infiltrating immune cells in renal cancer. Oncoimmunology 2015; 4: e985082.ArticlePubMedPMC
- 21. Fang LY, Izumi K, Lai KP, et al. Infiltrating macrophages promote prostate tumorigenesis via modulating androgen receptor-mediated CCL4-STAT3 signaling. Cancer Res 2013; 73: 5633-46. ArticlePubMedPDF
- 22. Minárik I, Lašt’ovicˇka J, Budinský V, et al. Regulatory T cells, dendritic cells and neutrophils in patients with renal cell carcinoma. Immunol Lett 2013; 152: 144-50. ArticlePubMed
- 23. Yang Z, Xie H, He D, Li L. Infiltrating macrophages increase RCC epithelial mesenchymal transition (EMT) and stem cell-like populations via AKT and mTOR signaling. Oncotarget 2016; 7: 44478-91. ArticlePubMedPMC
- 24. Hasebe T, Mukai K, Tsuda H, Ochiai A. New prognostic histological parameter of invasive ductal carcinoma of the breast: clinicopathological significance of fibrotic focus. Pathol Int 2000; 50: 263-72. ArticlePubMed
- 25. Delahunt B, Nacey JN. Renal cell carcinoma. II. Histological indicators of prognosis. Pathology 1987; 19: 258-63. ArticlePubMed
- 26. Frank I, Blute ML, Cheville JC, Lohse CM, Weaver AL, Zincke H. An outcome prediction model for patients with clear cell renal cell carcinoma treated with radical nephrectomy based on tumor stage, size, grade and necrosis: the SSIGN score. J Urol 2002; 168: 2395-400. ArticlePubMed
- 27. Kagan HM, Li W. Lysyl oxidase: properties, specificity, and biological roles inside and outside of the cell. J Cell Biochem 2003; 88: 660-72. ArticlePubMed
- 28. Egeblad M, Rasch MG, Weaver VM. Dynamic interplay between the collagen scaffold and tumor evolution. Curr Opin Cell Biol 2010; 22: 697-706. ArticlePubMedPMC
- 29. Ma XJ, Dahiya S, Richardson E, Erlander M, Sgroi DC. Gene expression profiling of the tumor microenvironment during breast cancer progression. Breast Cancer Res 2009; 11: R7.ArticlePubMedPMCPDF
- 30. Ramaswamy S, Ross KN, Lander ES, Golub TR. A molecular signature of metastasis in primary solid tumors. Nat Genet 2003; 33: 49-54. ArticlePubMedPDF
- 31. Calvo A, Catena R, Noble MS, et al. Identification of VEGF-regulated genes associated with increased lung metastatic potential: functional involvement of tenascin-C in tumor growth and lung metastasis. Oncogene 2008; 27: 5373-84. ArticlePubMedPMCPDF
- 32. Qiu TH, Chandramouli GV, Hunter KW, Alkharouf NW, Green JE, Liu ET. Global expression profiling identifies signatures of tumor virulence in MMTV-PyMT-transgenic mice: correlation to human disease. Cancer Res 2004; 64: 5973-81. PubMed
- 33. Jensen BV, Johansen JS, Skovsgaard T, Brandt J, Teisner B. Extracellular matrix building marked by the N-terminal propeptide of procollagen type I reflect aggressiveness of recurrent breast cancer. Int J Cancer 2002; 98: 582-9. ArticlePubMed
REFERENCES
Figure & Data
References
Citations

- Mutational scanning reveals oncogenic CTNNB1 mutations have diverse effects on signaling
Anagha Krishna, Alison Meynert, Karamjit Singh Dolt, Martijn Kelder, Agavni Mesropian, Ailith Ewing, Conny Brouwers, Jill WC Claassens, Margot M. Linssen, Shahida Sheraz, Gillian CA Taylor, Philippe Gautier, Anna Ferrer-Vaquer, Graeme Grimes, Hannes Beche
Nature Genetics.2026; 58(2): 366. CrossRef - Postoperative prognostic assessment using ECV fraction derived from equilibrium contrast-enhanced CT in thymomas
Koji Takumi, Hiroto Hakamada, Hiroaki Nagano, Ryota Nakanosono, Fumiko Kanzaki, Masanori Nakajo, Kiyohisa Kamimura, Masatoyo Nakajo, Daigo Nagano, Kazuhiro Ueda, Takashi Yoshiura
European Journal of Radiology.2025; 184: 111978. CrossRef - FAP+ fibroblasts orchestrate tumor microenvironment remodeling in renal cell carcinoma with tumor thrombus
Jiacheng Ma, Yan Huang, Jie Chen, Yang Li, Rongyan Yao, Xiubin Li, Qiyang Liang, Xinran Chen, Cheng Peng, Kan Liu, Yuanjun Zhai, Xu Zhang, Xin Ma, Xiaowen Wang, Qingbo Huang, Fuchu He
Nature Communications.2025;[Epub] CrossRef - New insights into fibrotic signaling in renal cell carcinoma
Jiao-Yi Chen, Wai-Han Yiu, Patrick Ming-Kuen Tang, Sydney Chi-Wai Tang
Frontiers in Cell and Developmental Biology.2023;[Epub] CrossRef - Tumor-associated fibrosis impairs the response to immunotherapy
Angha Naik, Andrew Leask
Matrix Biology.2023; 119: 125. CrossRef - Decreased renal expression of PAQR5 is associated with the absence of a nephroprotective effect of progesterone in a rat UUO model
P. A. Abramicheva, D. S. Semenovich, L. D. Zorova, I. B. Pevzner, I. A. Sokolov, V. A. Popkov, E. P. Kazakov, D. B. Zorov, E. Y. Plotnikov
Scientific Reports.2023;[Epub] CrossRef - Intratumoral fibrosis and patterns of immune infiltration in clear cell renal cell carcinoma
Songchen Han, Wenbo Yang, Caipeng Qin, Yiqing Du, Mengting Ding, Huaqi Yin, Tao Xu
BMC Cancer.2022;[Epub] CrossRef - Siglec-F–expressing neutrophils are essential for creating a profibrotic microenvironment in renal fibrosis
Seungwon Ryu, Jae Woo Shin, Soie Kwon, Jiwon Lee, Yong Chul Kim, Yoe-Sik Bae, Yong-Soo Bae, Dong Ki Kim, Yon Su Kim, Seung Hee Yang, Hye Young Kim
Journal of Clinical Investigation.2022;[Epub] CrossRef - The atypical sphingosine 1‐phosphate variant, d16:1 S1P, mediates CTGF induction via S1P2 activation in renal cell carcinoma
Melanie Glueck, Alexander Koch, Robert Brunkhorst, Nerea Ferreiros Bouzas, Sandra Trautmann, Liliana Schaefer, Waltraud Pfeilschifter, Josef Pfeilschifter, Rajkumar Vutukuri
The FEBS Journal.2022; 289(18): 5670. CrossRef - The Synergistic Cooperation between TGF-β and Hypoxia in Cancer and Fibrosis
Pramod Mallikarjuna, Yang Zhou, Maréne Landström
Biomolecules.2022; 12(5): 635. CrossRef - In Vitro Characterization of Renal Drug Transporter Activity in Kidney Cancer
Pedro Caetano-Pinto, Nathanil Justian, Maria Dib, Jana Fischer, Maryna Somova, Martin Burchardt, Ingmar Wolff
International Journal of Molecular Sciences.2022; 23(17): 10177. CrossRef - PD-1 immunobiology in glomerulonephritis and renal cell carcinoma
Colleen S. Curran, Jeffrey B. Kopp
BMC Nephrology.2021;[Epub] CrossRef - Intratumoral Fibrosis in Facilitating Renal Cancer Aggressiveness: Underlying Mechanisms and Promising Targets
Chao Hu, Yufeng Zhao, Xuanchuan Wang, Tongyu Zhu
Frontiers in Cell and Developmental Biology.2021;[Epub] CrossRef - What Mediates Fibrosis in the Tumor Microenvironment of Clear Renal Cell Carcinoma
Wenbo Yang, Caipeng Qin, Jingli Han, Songchen Han, Wenjun Bai, Yiqing Du, Tao Xu
Frontiers in Genetics.2021;[Epub] CrossRef - Kidney Cancer and Chronic Kidney Disease: Too Close for Comfort
Pedro Caetano Pinto, Cindy Rönnau, Martin Burchardt, Ingmar Wolff
Biomedicines.2021; 9(12): 1761. CrossRef - The Significance of Fibrosis Quantification as a Marker in Assessing Pseudo-Capsule Status and Clear Cell Renal Cell Carcinoma Prognosis
Caipeng Qin, Huaqi Yin, Huixin Liu, Feng Liu, Yiqing Du, Tao Xu
Diagnostics.2020; 10(11): 895. CrossRef - The challenges of adoptive cell transfer in the treatment of human renal cell carcinoma
Zuzana Strizova, Jirina Bartunkova, Daniel Smrz
Cancer Immunology, Immunotherapy.2019; 68(11): 1831. CrossRef - Procollagen-lysine, 2-oxoglutarate 5-dioxygenases 1, 2, and 3 are potential prognostic indicators in patients with clear cell renal cell carcinoma
Wen-Hao Xu, Yue Xu, Jun Wang, Xi Tian, Junlong Wu, Fang-Ning Wan, Hong-Kai Wang, Yuan-Yuan Qu, Hai-Liang Zhang, Ding-Wei Ye
Aging.2019; 11(16): 6503. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
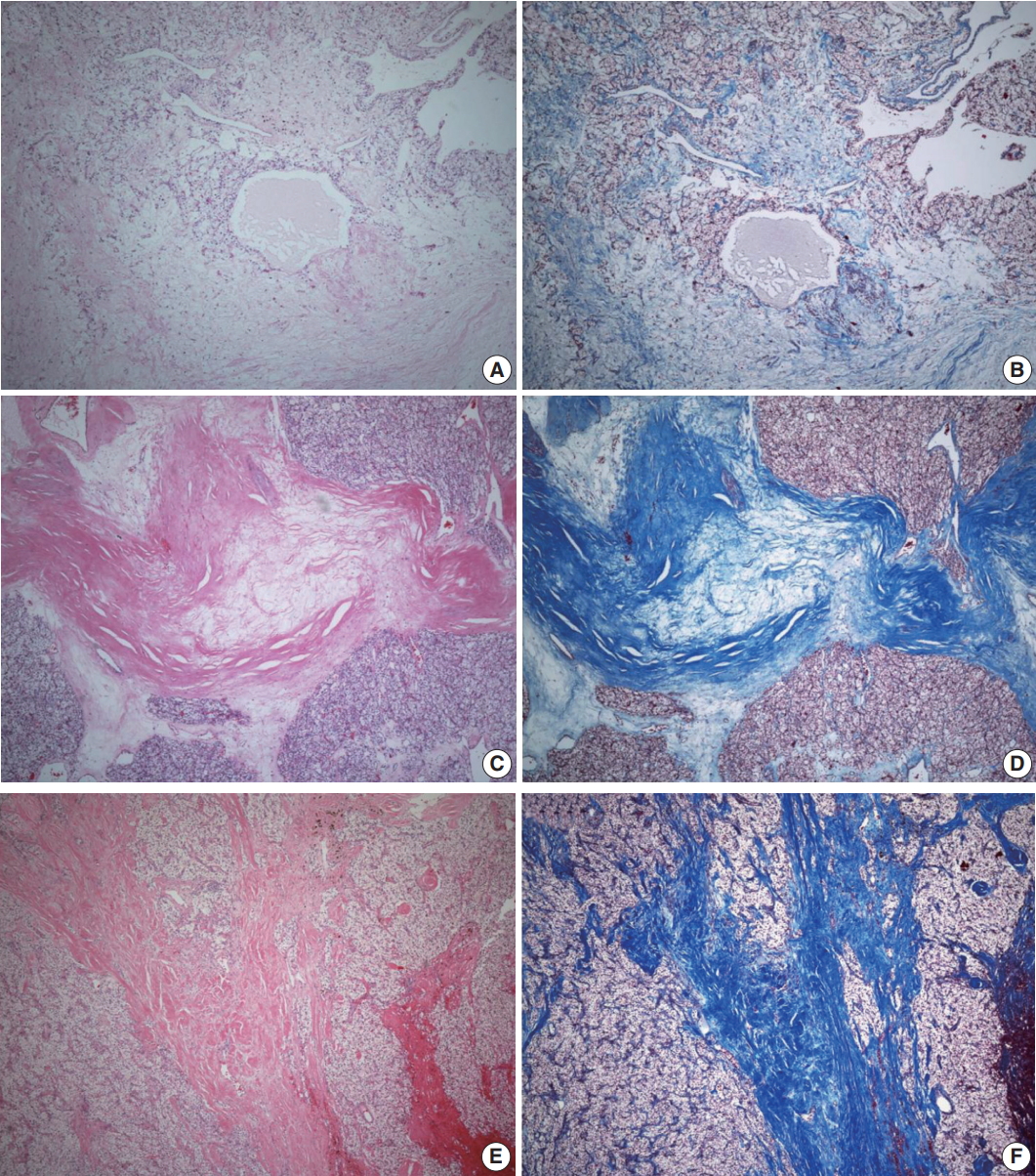
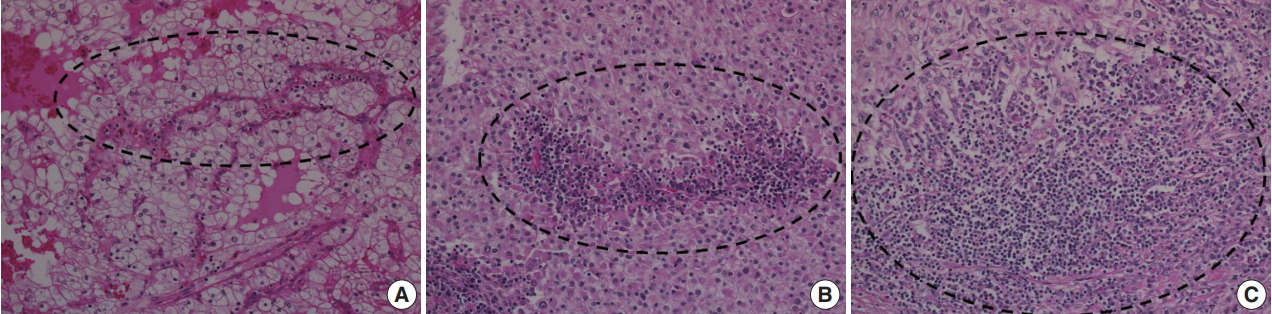
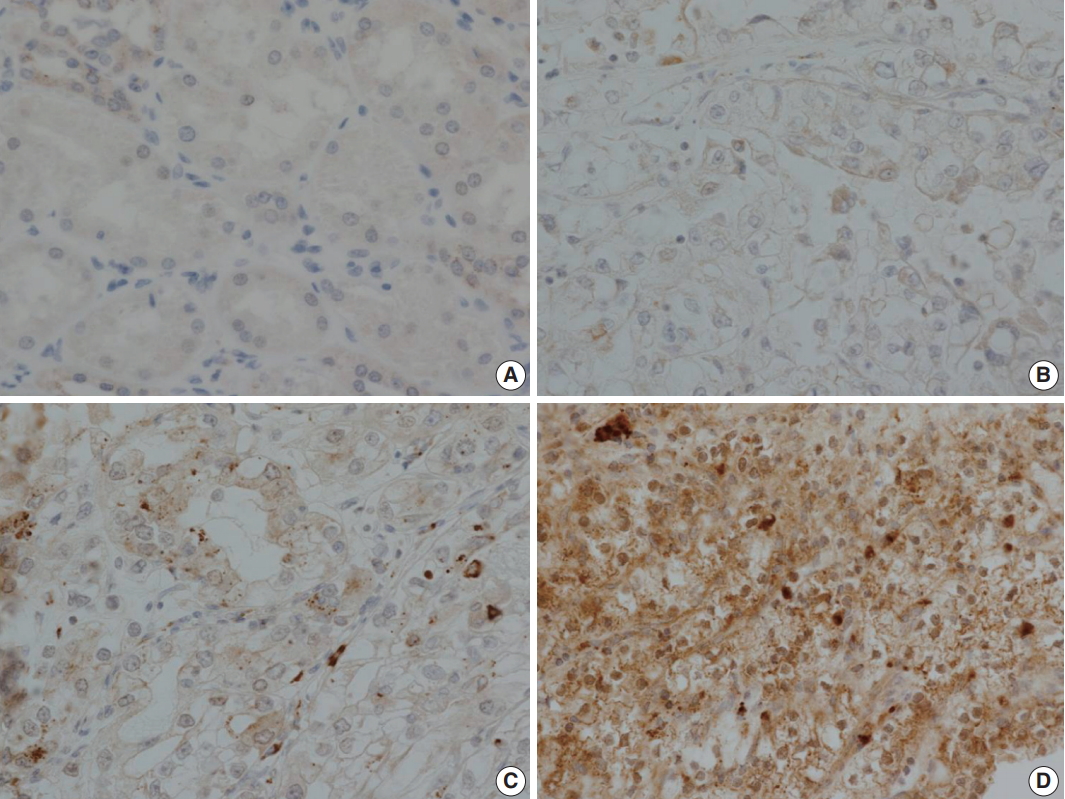
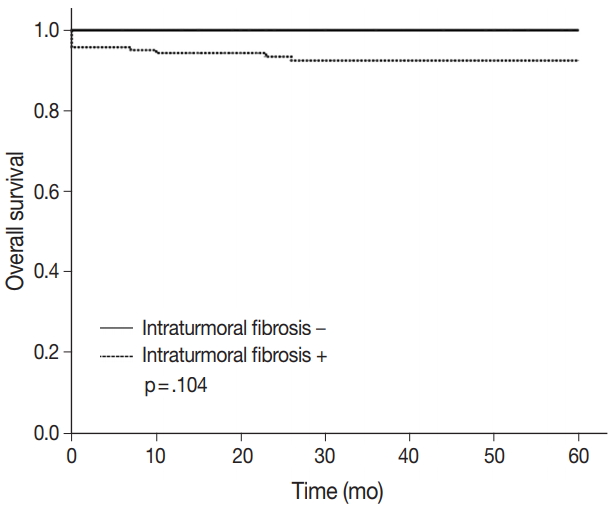
Fig. 1.
Fig. 2.
Fig. 3.
Fig. 4.
| Clinicopathological parameter | No. (%) |
|---|---|
| Age (yr) | |
| Mean ± SD | 59.7 ± 11.8 |
| Range | 27–87 |
| Tumor size (cm) | |
| Average ± SD | 4.48 ± 3.01 |
| Range | 0.5–19 |
| Sex | |
| Male | 139 (68.1) |
| Female | 65 (31.9) |
| Fuhrman nuclear grade | |
| 1 | 12 (5.9) |
| 2 | 118 (57.8) |
| 3 | 55 (27.0) |
| 4 | 19 (9.3) |
| Pathologic stage | |
| 1 | 154 (75.5) |
| 2 | 19 (9.3) |
| 3 | 30 (14.7) |
| 4 | 1 (0.5) |
| Lymphovascular invasion | |
| Absent | 175 (85.8) |
| Present | 29 (14.2) |
| Tumor necrosis | |
| Absent | 169 (82.8) |
| Present | 35 (17.2) |
| Intratumoral inflammation | |
| 0 | 25 (12.3) |
| 1 | 108 (52.9) |
| 2 | 59 (28.9) |
| 3 | 12 (5.9) |
| Intratumoral fibrosis | |
| 0 | 37 (18.1) |
| 1 | 59 (28.9) |
| 2 | 77 (37.7) |
| 3 | 31 (15.2) |
| LOX | |
| 0 | 11 (5.4) |
| 1 | 82 (40.2) |
| 2 | 82 (40.2) |
| 3 | 29 (14.2) |
| Clinicopathological parameter | Intratumoral fibrosis |
p-value | |
|---|---|---|---|
| Absent | Present | ||
| Age (yr) | |||
| < 60 | 21 (20.8) | 80 (79.2) | .330 |
| ≥ 60 | 16 (15.5) | 87 (84.5) | |
| Sex | |||
| Male | 26 (18.7) | 113 (81.3) | .758 |
| Female | 11 (16.9) | 54 (83.1) | |
| Tumor size (cm) | |||
| ≤ 7 | 34 (20.2) | 134 (79.8) | .093 |
| > 7 | 3 (8.3) | 33 (91.7) | |
| Fuhrman nuclear grade | |||
| Low (grade 1 and 2) | 31 (23.8) | 99 (76.2) | .003 |
| High (grade 3 and 4) | 6 (8.1) | 68 (91.9) | |
| Pathologic stage | |||
| Low (stage 1 and 2) | 33 (19.1) | 140 (80.9) | .295 |
| High (stage 3 and 4) | 4 (12.9) | 27 (87.1) | |
| Lymphovascular invasion | |||
| Absent | 36 (20.6) | 139 (79.4) | .027 |
| Present | 1 (3.4) | 28 (96.6) | |
| Tumor necrosis | |||
| Absent | 35 (20.7) | 134 (79.3) | .036 |
| Present | 2 (5.7) | 33 (94.3) | |
| Intratumoral inflammation | |||
| Absent | 25 (18.8) | 108 (81.2) | .738 |
| Present | 12 (16.9) | 59 (83.1) | |
| LOX expression | |||
| Absent | 16 (17.2) | 77 (82.8) | .752 |
| Present | 21 (18.9) | 90 (81.1) | |
| Clinicopathological parameter | Intratumoral fibrosis |
p-value | |
|---|---|---|---|
| Absent | Present | ||
| Age (yr) | |||
| < 60 | 67 (66.3) | 34 (33.7) | .735 |
| ≥ 60 | 66 (64.1) | 37 (35.9) | |
| Sex | |||
| Male | 85 (61.2) | 54 (38.8) | .076 |
| Female | 48 (73.8) | 17 (26.2) | |
| Tumor size (cm) | |||
| ≤ 7 | 120 (71.4) | 48 (28.6) | < .001 |
| > 7 | 13 (36.1) | 23 (63.9) | |
| Fuhrman nuclear grade | |||
| Low (grade 1 and 2) | 102 (78.5) | 28 (21.5) | < .001 |
| High (grade 3 and 4) | 31 (41.9) | 43 (58.1) | |
| Pathologic stage | |||
| Low (stage 1 and 2) | 120 (69.4) | 53 (30.6) | .004 |
| High (stage 3 and 4) | 13 (41.9) | 18 (58.1) | |
| Lymphovascular invasion | |||
| Absent | 124 (70.9) | 51 (29.1) | < .001 |
| Present | 9 (31.0) | 20 (69.0) | |
| Tumor necrosis | |||
| Absent | 124 (73.4) | 45 (26.6) | < .001 |
| Present | 9 (25.7) | 26 (74.3) | |
| Intratumoral fibrosis | |||
| Absent | 25 (67.6) | 12 (32.4) | .738 |
| Present | 108 (64.7) | 59 (35.3) | |
| LOX expression | |||
| Absent | 60 (64.5) | 33 (35.5) | .852 |
| Present | 73 (65.8) | 38 (34.8) | |
| Clinicopathological parameter | LOX expression |
p-value | |
|---|---|---|---|
| Absent | Present | ||
| Age (yr) | |||
| < 60 | 43 (42.6) | 58 (57.4) | .392 |
| ≥ 60 | 50 (48.5) | 53 (51.5) | |
| Sex | |||
| Male | 61 (43.9) | 78 (56.1) | .475 |
| Female | 32 (49.2) | 33 (50.8) | |
| Tumor size (cm) | |||
| ≤ 7 | 77 (45.8) | 91 (54.2) | .879 |
| > 7 | 16 (44.4) | 20 (55.6) | |
| Fuhrman nuclear grade | |||
| Low (grade 1 and 2) | 56 (43.1) | 74 (56.9) | .209 |
| High (grade 3 and 4) | 37 (50.0) | 37 (50.0) | |
| Pathologic stage | |||
| Low (stage 1 and 2) | 77 (44.5) | 96 (55.5) | .295 |
| High (stage 3 and 4) | 16 (51.6) | 15 (48.4) | |
| Lymphovascular invasion | |||
| Absent | 76 (43.4) | 99 (56.6) | .128 |
| Present | 17 (58.6) | 12 (41.4) | |
| Tumor necrosis | |||
| Absent | 80 (47.3) | 89 (52.7) | .270 |
| Present | 13 (37.1) | 22 (62.9) | |
| Intratumoral inflammation | |||
| Absent | 7 (5.3) | 126 (94.7) | .911 |
| Present | 4 (5.6) | 67 (94.4) | |
| Intratumoral fibrosis | |||
| Absent | 16 (43.2) | 21 (56.8) | .752 |
| Present | 77 (46.1) | 90 (53.9) | |
| Clinicopathological parameter | Intratumoral fibrosis size (cm) |
p-value | |
|---|---|---|---|
| ≤ 0.5 | > 0.5 | ||
| Age (yr) | |||
| < 60 | 57 (71.3) | 23 (28.7) | .523 |
| ≥ 60 | 58 (66.7) | 29 (33.3) | |
| Sex | |||
| Male | 82 (72.6) | 31 (27.4) | .135 |
| Female | 33 (61.1) | 21 (38.9) | |
| Tumor size (cm) | |||
| ≤ 7 | 96 (71.6) | 38 (28.4) | .118 |
| > 7 | 19 (57.6) | 14 (42.4) | |
| Fuhrman nuclear grade | |||
| Low (grade 1 and 2) | 76 (76.8) | 23 (23.2) | .007 |
| High (grade 3 and 4) | 39 (57.4) | 29 (42.6) | |
| Pathologic stage | |||
| Low (stage 1 and 2) | 97 (69.3) | 43 (30.7) | .475 |
| High (stage 3 and 4) | 18 (66.7) | 9 (33.3) | |
| Lymphovascular invasion | |||
| Absent | 100 (71.9) | 39 (28.1) | .055 |
| Present | 15 (53.6) | 13 (46.4) | |
| Tumor necrosis | |||
| Absent | 95 (70.9) | 39 (29.1) | .253 |
| Present | 20 (60.6) | 13 (39.4) | |
| Intratumoral inflammaion | |||
| Absent | 78 (72.2) | 30 (27.8) | .205 |
| Present | 37 (62.7) | 22 (37.3) | |
| LOX expression | |||
| Absent | 50 (64.9) | 27 (35.1) | .311 |
| Present | 65 (72.2) | 25 (27.8) | |
SD, standard deviation; LOX, lysyl oxidase.
Values are presented as number (%). LOX, lysyl oxidase. p < .05, p < .01.
Values are presented as number (%). LOX, lysyl oxidase. p < .01, p < .001.
Values are presented as number (%). LOX, lysyl oxidase.
Values are presented as number (%). LOX, lysyl oxidase. p < .01.

 E-submission
E-submission