Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 52(1); 2018 > Article
-
Original Article
Reclassification of Mixed Oligoastrocytic Tumors Using a Genetically Integrated Diagnostic Approach - Seong-Ik Kim1, Yujin Lee1,2, Jae-Kyung Won1, Chul-Kee Park2, Seung Hong Choi3, Sung-Hye Park1,4
-
Journal of Pathology and Translational Medicine 2018;52(1):28-36.
DOI: https://doi.org/10.4132/jptm.2017.09.25
Published online: September 29, 2017
1Department of Pathology, Seoul National University College of Medicine, Seoul, Korea
2Department of Neurosurgery, Seoul National University College of Medicine, Seoul, Korea
3Department of Radiology, Seoul National University College of Medicine, Seoul, Korea
4Department of Neurosicence Institute, Seoul National University College of Medicine, Seoul, Korea
- Corresponding Author Sung-Hye Park, MD Departments of Pathology, Neurosicence Institute, Seoul National University College of Medicine, 103 Daehak-ro, Jongno-gu, Seoul 03080, Korea Tel: +82-2-2072-3090 Fax: +82-2-765-5600 E-mail: shparknp@snu.ac.kr
© 2018 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Mixed gliomas, such as oligoastrocytomas (OA), anaplastic oligoastrocytomas, and glioblastomas (GBMs) with an oligodendroglial component (GBMO) are defined as tumors composed of a mixture of two distinct neoplastic cell types, astrocytic and oligodendroglial. Recently, mutations ATRX and TP53, and codeletion of 1p/19q are shown to be genetic hallmarks of astrocytic and oligodendroglial tumors, respectively. Subsequent molecular analyses of mixed gliomas preferred the reclassification to either oligodendroglioma or astrocytoma. This study was designed to apply genetically integrated diagnostic criteria to mixed gliomas and determine usefulness and prognostic value of new classification in Korean patients.
-
Methods
- Fifty-eight cases of mixed OAs and GBMOs were retrieved from the pathology archives of Seoul National University Hospital from 2004 to 2015. Reclassification was performed according to genetic and immunohistochemical properties. Clinicopathological characteristics of each subgroup were evaluated. Overall survival was assessed and compared between subgroups.
-
Results
- We could reclassify all mixed OAs and GBMOs into either astrocytic or oligodendroglial tumors. Notably, 29 GBMOs could be reclassified into 11 cases of GBM, IDH-mutant, 16 cases of GBM, IDH-wildtype, and two cases of anaplastic oligodendroglioma, IDH mutant. Overall survival was significantly different among these new groups (p<.001). Overall survival and progression-free survival were statistically better in gliomas with IDH mutation, ATRX mutation, no microscopic necrosis, and young patient age (cut off, 45 years old).
-
Conclusions
- Our results strongly suggest that a genetically integrated diagnosis of glioma better reflects prognosis than former morphology-based methods.
- Study population
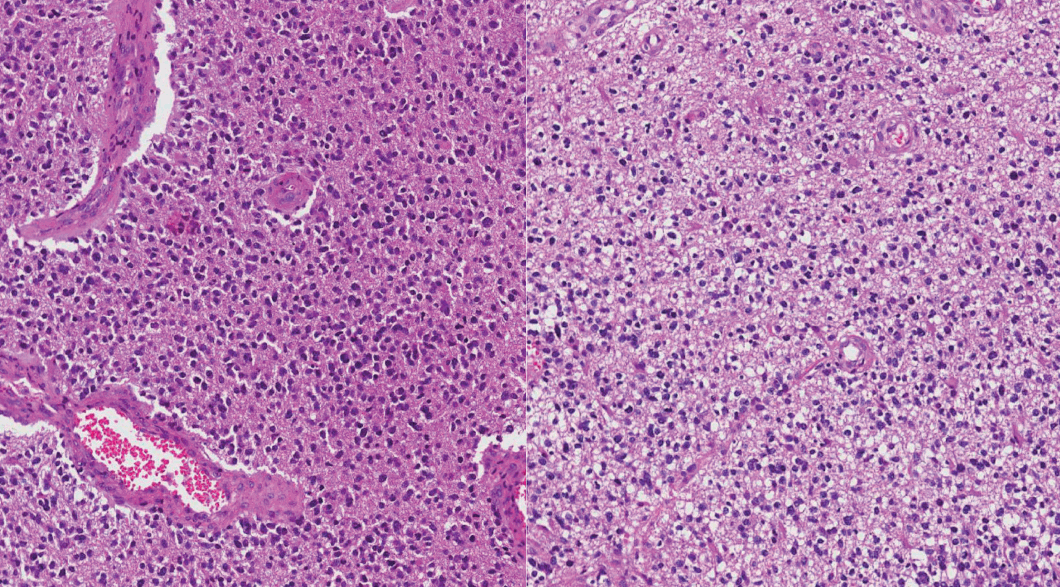
- Formalin-fixed paraffin-embedded (FFPE) tissues from a total of 58 patients were obtained from the archives of the Department of Pathology, Seoul National University Hospital. All patients were Korean (Table 1). Age at first diagnosis was between 14 and 73 years old (median: previously OA, 36 years; previously GBMO, 56 years). All cases were diagnosed between 2004 and 2015. Selected cases had distinct morphological features of mixed astrocytic and oligodendroglial component or mixed GBM and oligodendroglial component. We used a 10% cut-off for the minimum area of oligodendroglial or astrocytic component. Tumors were graded according to the 2007 WHO guidelines. The series contained 15 OAs, 14 AOAs, and 29 GBMOs (Fig. 1). Histopathologic slides were reviewed by two pathologists (S.H. Park, S.I. Kim). Immunohistochemical stainings for IDH1 (clone H09, Dianova, Hamburg, Germany) and ATRX (Sigma-Aldrich, St. Louis, MO, USA) were performed in all 58 cases. If IDH1 immunohistochemistry (IHC) was negative, direct sequencing of IDH1/IDH2 was performed. Fluorescent in situ hybridization (FISH) using Vysis probes (Abbott, Des Plaines, IL, USA) to detect 1p/19q status was done in all 58 cases. Survival plots according to the clinical, histological, and molecular characteristics of tumors were analyzed using Kaplan-Meier (K-M) survival analysis.
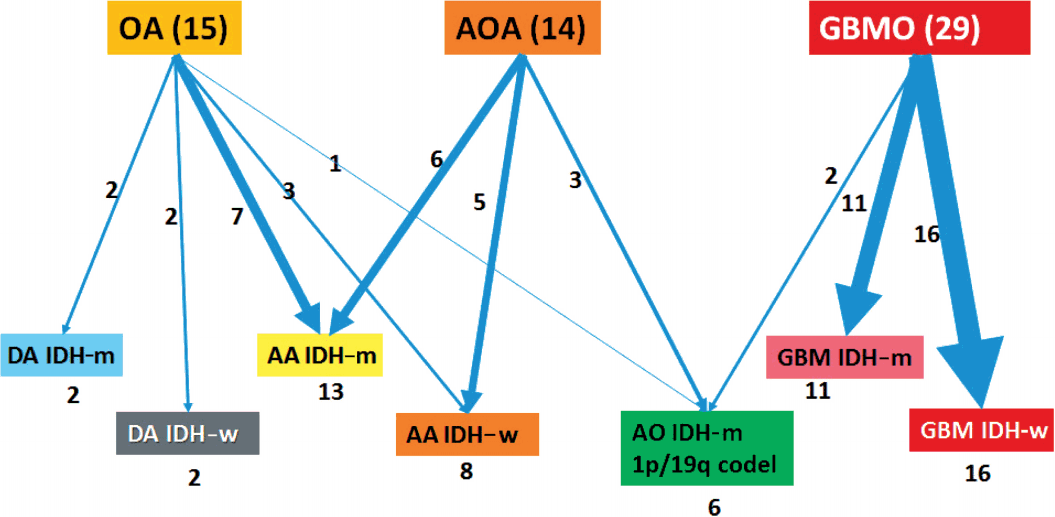
- With the results of our genetic studies, we reclassified tumors based on the diagram by Reuss et al. [13]. If the ATRX IHC was negative, it was classified as astrocytic, independent of its IDH status. Tumors were then subdivided by IDH status based on IHC and direct sequencing. In contrast, all tumors with nuclear ATRX expression were divided by 1p/19q status using FISH. If 1p/19q were non-codeleted, tumors were astrocytic tumors. Then, the tumors were also subdivided by IDH status. If codeletion of 1p/19q and IDH mutation were present, they were defined as oligodendroglial tumors. Next, we graded the tumors according to the new 2016 WHO guidelines. Lastly, 58 cases were reclassified into seven distinct subgroups: 1) DA, IDH-mutant (DA IDH-m), 2) DA, IDH-wildtype (DA IDH-w), 3) AA, IDH-mutant (AA IDH-m), 4) AA, IDH-wildtype (AA IDH-w), 5) AO, IDH-mutant (AO IDH-m), 6) GBM, IDH-mutant (GBM IDH-m), and 7) GBM, IDH-wildtype (GBM IDH-w). We performed further classification of GBM into primary and secondary GBM. Secondary GBM was defined as cases with previous pathology-proven lower grade tumors.Then, we compared the survival plots between reclassified entities using K-M survival analysis.
- This study abided by the world Medical Association Declaration of Helsinki recommendation and was approved by the Institutional Review Board of Seoul National University Hospital (IRB No. 1508-004-690). Written informed consents were obtained.
- Hematoxylin and eosin staining and IHC
- FFPE (10% neutral buffered formalin, routinely processed, and paraffin-embedded) tissue sections (2–3 μm thick) were cut for hematoxylin and eosin staining and IHC. Tissue sections were stained with anti-IDH1 R132H (H09) monoclonal antibody (Dianova) using a 1:100 dilution, anti-ATRX polyclonal antibody HPA001906 (Sigma-Aldrich) using a 1:300 dilution, and anti-p53 monoclonal antibody, DO-7 code M7001 (Dako, Glostrup, Denmark) using a 1:1000 dilution. IHC staining was carried out using a standard avidin–biotin peroxidase method. Tumors were interpreted as positive for p53 expression if ≥ 20% of neoplastic cells showed distinct nuclear staining.
- DNA extraction
- Tumor areas were manually micro-dissected using 6-μm unstained tissue sections made from FFPE tissue. DNA was isolated from the micro-dissected tissue using a DNeasy Blood and Tissue Kit (Qiagen, Valencia, CA, USA) according to the manufacturer’s instructions.
- Polymerase chain reaction amplification and sequencing of IDH1 and IDH2
- Template DNA (1 μL) was added to 100 μL of polymerase chain reaction (PCR) solution (10 μL of 10 × magnesium Taq High-Fidelity [HF] buffer, 10 μL deoxynucleotide triphosphate [dNTP] mixture with 2 mM magnesium, 5 μL of 10 pmol primer [2 ×], 1 μL of magnesium Taq-HF polymerase, and distilled water). The IDH1-Forward (F)/IDH1-Reverse (R) and IDH2-F/IDH2-R primers (Table 2) were used with the following program: 35 cycles of 95℃ for 30 seconds, 55℃ for 30 seconds, and 72℃ for 60 seconds. The product sizes were 130-base pair (bp) (IDH1) and 293-bp (IDH2). Unincorporated PCR primers and dNTPs were removed from the PCR products using a Montage PCR Clean-up Kit (Millipore, Billerica, MA, USA).
- Purified products were sequenced using the same primers. Sequencing was performed using a BigDye Terminator Cycle Sequencing Kit v. 3.1 (Applied Biosystems, Foster City, CA, USA). Sequencing products were resolved on an automated DNA sequencing system, model 3730XL (Applied Biosystems).
- Fluorescence in situ hybridization
- FISH with Vysis probes was used to assess 1p/19q status. Sections (3 μm thick) were first deparaffinized in xylene, incubated with 0.3% pepsin in 10 mM HCl at 37℃ for 10 minutes, and boiled with citrate buffer (pH 6.0) in a microwave. Sections were then incubated in 1 M NaSCN for 35 minutes at 80℃, immersed in the pepsin solution, and fixed in 10% neutral buffered formalin. 1p36/1q25 and 19q13/19p13 labeled locusspecific dual-color probes (Abbott Molecular) were used according to the manufacturer’s protocol. We applied the probe mixture to the slides and incubated them in a humidified atmosphere with HYBrite (Abbott Molecular) at 73℃ for 5 minutes for simultaneous denaturation of the probe and target DNA. Then, we cooled the samples to 37℃ and incubated for 19 hours to hybridize the probe and target DNA. The slides were submerged in 0.4× SSC buffer/0.3% NP-40 for 2 minutes at room temperature, followed by incubation in 2× SSC/0.1% NP-40 for 5 minutes at 73℃.
- Statistical analysis
- OS and progression-free survival (PFS) were estimated using K-M survival analysis and were compared using the log-rank test. All statistical analyses were performed with SPSS ver. 22 (IBM Corp., Armonk, NY, USA); p < .05 was considered significant.
MATERIALS AND METHODS
- Clinicopathological information of patients is provided in Table 1. Our reclassification results are summarized in Table 3 and Fig. 2. Our reclassification analysis showed that the largest fraction of mixed OA was AA IDH-m (50%). The largest part of mixed AOA was also AA IDH-m (42.9%), followed by AA IDH-w (35.7%) and AO (21%). The largest fraction of GBMO was conventional GBM IDH-w (55.2%), followed by GBM IDH-m (37.9%) and only 6.9% AO. Except for one undetermined case of GBM IDH-w, 14 out of 16 GBM IDH-w cases (87.5%) were primary GBM and one case (6.3%) was secondary GBM. In 11 cases of GBM IDH-m, seven cases (63.6%) were primary GBM and four cases (36.4%) were secondary GBM. Therefore, the incidence of primary GBMs (21/27, 77.8%) derived from those previously diagnosed as mixed gliomas was less than the well-known incidence of primary GBM (90%–95%).
- When we carried out K-M survival analysis, two cases of DA IDH-w were excluded; one patient died of suddenly developed progressive hydrocephalus of an unknown cause and the other patient didn’t show up. One case of AO IDH-m was also excluded because the patient died of a worsened general condition from multiple diseases, including pulmonary thromboembolism with basal ganglia and temporal lobe infarction during hospitalization. Recent studies report a very similar incidence of stroke between cancer and non-cancer patients; therefore, it is reasonable to think that stroke was an independent variable for survival in this case, whether the tumor was a causative factor of stroke or not [16].
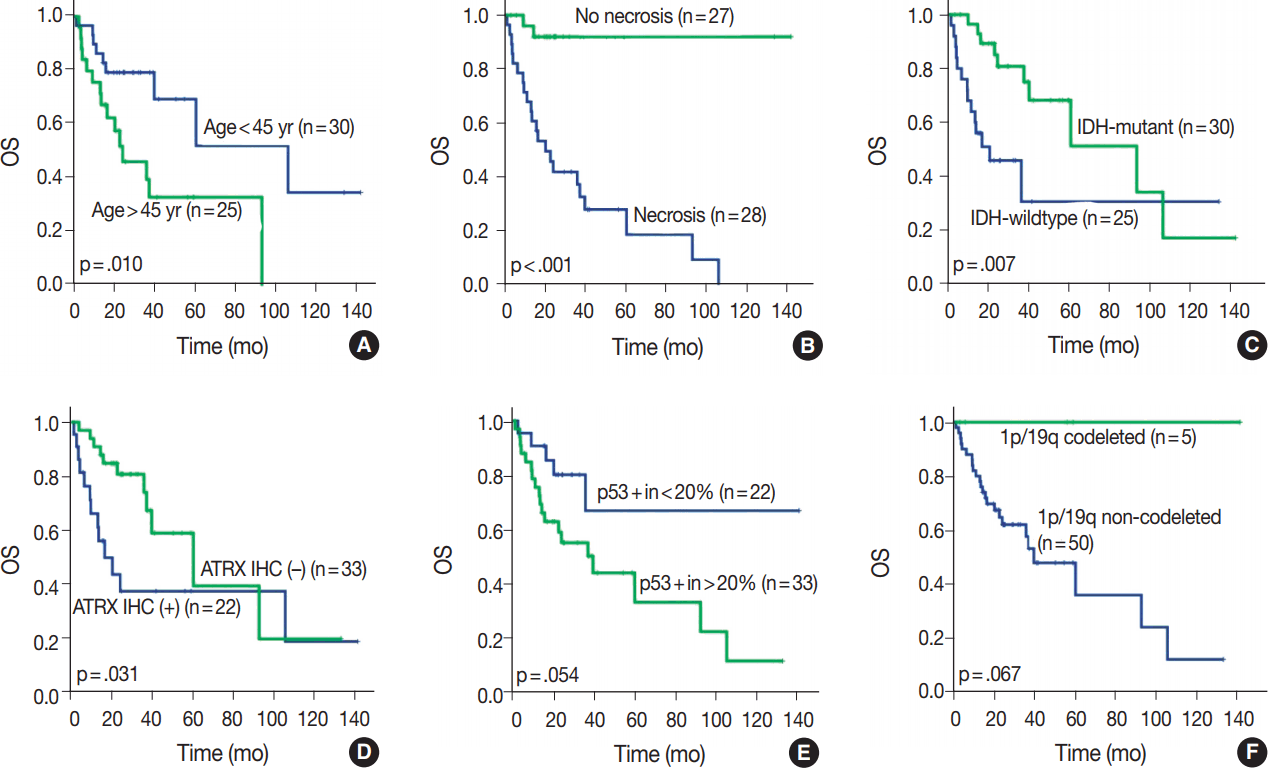
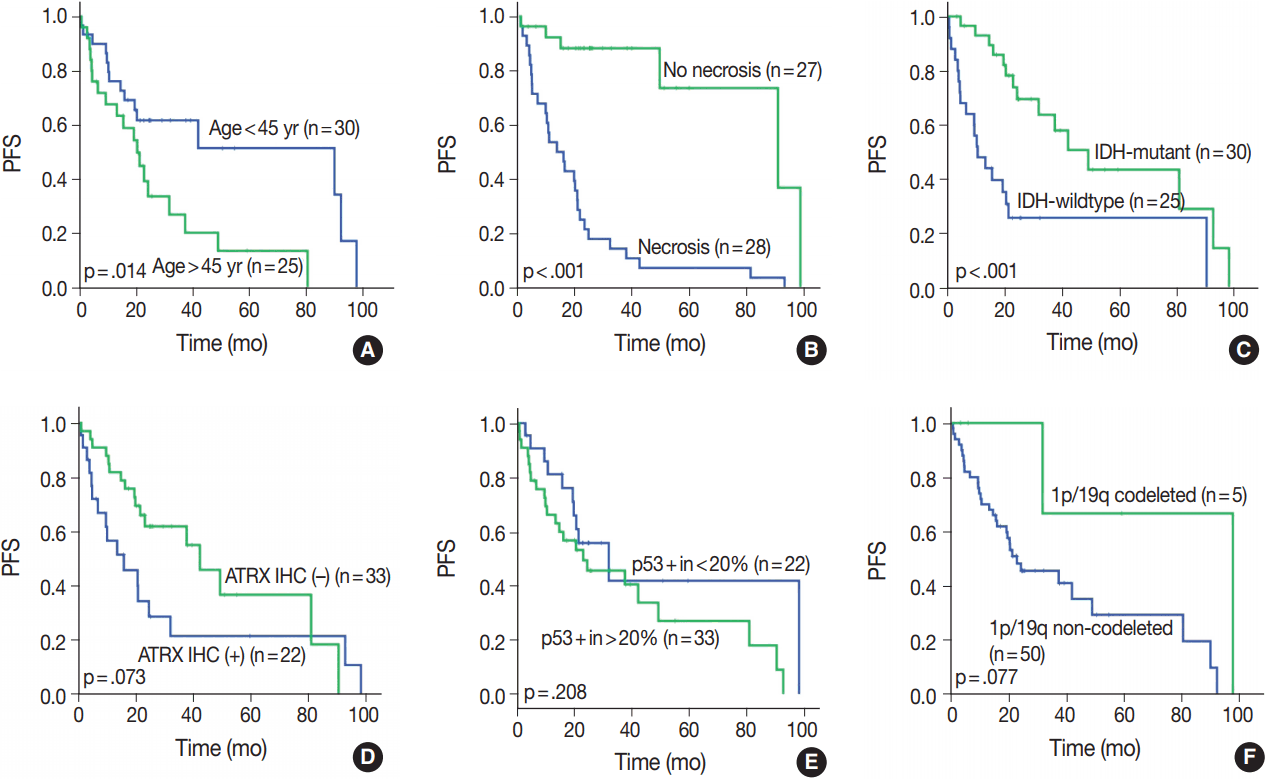
- OS in different clinical, histological, and molecular subgroups has been evaluated (Fig. 3). Prognosis was better in the younger (age < 45 years) (p = .010) and IDH-mutant (p =. 007) subgroups. However, prognosis was worse in subgroups with microscopic necrosis (p < .001) and ATRX-positive IHC (p = .031). Similar results were drawn with PFS (Fig. 4). Other characteristics failed to show statistical significance. The subgroup with the 1p/19q co-deletion had a tendency of better prognosis in terms of OS (p = .067) and PFS (p = .077), despite the lack of statistical significance (Figs. 3F, 4F). Of those compared, all 1p/19q codeleted patients fully survived the study completion. Also, those with p53 overexpression (positive in > 20%) had a tendency for worse prognosis in terms of OS (p = .054) and PFS (p = .208) (Figs. 3E, 4E).
- All these results with demographic characteristics are summarized in Table 4.
- After re-classification of mixed gliomas, we compared survival plots between the reclassified entities using K-M survival analysis. In terms of OS as well as PFS, the genetically integrated diagnostic approach showed clear statistical significance (p < .001) (Fig. 5).
RESULTS
- Mixed gliomas have been previously defined as diffusely infiltrating gliomas composed of a mixture of two distinct neoplastic cell types morphologically resembling the tumor cells in oligodendroglial or astrocytic tumors [9]. However, inter-institutional and inter-observer variations of mixed gliomas have resulted in low diagnostic reproducibility and reliability.
- The ISN-Haarlem’s recommendations for a genetically integrated diagnosis for the classification of diffuse gliomas suggested the removal of mixed glioma entity [11]. We reclassified mixed OA (including GBMO) into either astrocytic or oligodendroglial tumors in agreement with the emphasis by Sahm et al. [12].
- Here, we applied the new criteria to the 58 cases that were previously diagnosed as mixed gliomas. The purpose of our study was to determine the usefulness of genetically integrated classification, true incidence of diffuse gliomas, and prognostic value of a new classification system. Above all, we sought to find out whether there are any intermediate cases or genuine mixed gliomas.
- We primarily used Reuss et al.’s diagnostic criteria [13]; therefore, ATRX IHC and 1p/19q FISH were used to determine whether the tumor was of astrocytic or oligodendroglial lineage.
- Among DA and AA tumors, ATRX-negativity and -positivity were found in 88% and 12%, respectively. Six cases which showed 1p/19q codeletion were also ATRX-positive and IDH1-mutated; therefore, the diagnosis of AO was certain. Among our 27 GBMs, ATRX-positive and -negative cases were 55.6% and 44.4%, respectively. Among ATRX-negative GBMs, 50% (6/12) were secondary, 41.7% (5/12) were primary, and 8.3% (1/12) were unconfirmed. Thus, half of ATRX-negative GBMs were secondary GBM, but among primary GBMs, 27.8% were ATRX-negative.
- Using these criteria, our 58 cases could be classified as either astrocytic or oligodendroglial. None showed mixed genetic features of astrocytic and oligodendroglial. Our results also indicate that the codeletion status of 1p/19q is the most important criteria in the classification of diffuse gliomas into astrocytic or oligodendroglial, and could serve as a useful biomarker in the future diagnosis.
- We evaluated all clinical, histological, and molecular characteristics using K-M survival analysis. OS and PFS were better in gliomas with IDH mutation, ATRX mutation, absence of microscopic necrosis, and young patient age (cut off, 45 years old). Although the impact of ATRX mutation on PFS had no statistical significance with log-rank test (p = .073), the Wilcoxon test and Tarone-Ware test showed statistical significance (p = .010 and p = .016, respectively). This confirms the results of previous studies on the impact of IDH and ATRX on prognosis [17,18]. Because most tumors with microscopic necrosis were diagnosed as either grade IV GBM IDH-m or GBM IDH-w, it is obvious that tumors with no necrosis showed better prognosis. Regarding the patient age, this study also confirms the results of a previous study [1]. Increase in age is associated with more high-grade glial tumors or poorer general conditions; therefore, elderly people could have a poorer prognosis [9]. Among patients with tumors of the same grade, one of the most favorable prognostic factors is young age (cut off, 45 years old) [9]. Similar results can be observed in oligodendroglial tumors [19].
- In this study, 1p/19q status did not achieve statistical significance on OS or PFS. However, excluding one case, of which the cause of death was uncertain, all 1p/19q codeleted patients fully survived the follow-up period. Therefore, the lack of statistical significance might be derived from the short term follow-up period or a small number of patients.
- With regard to both OS and PFS, the genetically integrated diagnostic approach showed statistically significant subgroups, which confirm the results of previous studies carried out by Reuss et al. [13]. Our reclassification revealed that some patients should have received different treatment based on our genetically integrated diagnosis. Two patients initially diagnosed with GBMO were re-diagnosed with AO. One had primarily concurrent chemoradiation therapy and the other had primarily chemotherapy alone. However, according to our hospital protocol, the primary choice for AO is craniotomy and tumor removal with or without adjuvant therapy. Although reclassification itself might produce change in the treatment protocol of gliomas, it can also be useful in the selection of proper therapy for the patients.
- ATRX negativity and p53 positivity directly indicated secondary GBM; however, among these tumors, four cases were primary GBM, which may indicate rapid progression from low grade tumors.
- It is well known that astrocytic tumors have a genetic hallmark of ATRX and TP53 mutation [7,8]. In the genetically integrated diagnostic flow, ATRX mutation proved more useful as TP53 mutation was shown to be poorly detected by IHC [20]. In cases with intact ATRX, a 1p/19q assay should be carried out for designation of oligodendroglial tumor. Since generally extensive p53 staining is regarded as indicative of astrocytoma, p53 IHC might also be helpful in differential diagnosis of those cases. Therefore, we tried to find for a proper cutoff value of p53 positivity for astrocytic designation in our study. The cut-off value of 20% divided the subgroup stringently. However, 20% cut off of p53 had no statistical significance both in OS (p = .054) and PFS (p = .208). In our study, astrocytic tumors had p53 positivity in an average of 42.1%, with a standard deviation of 32.31%. If astrocytic tumors were entirely negative for p53 IHC, those cases should be re-verified by alternative tools, such as next generation sequencing or restriction fragment length polymorphism for detecting loss of function-mutation or methylation of p53. Further studies on staining pattern or correlation with other clinicopathological factors might be helpful.
- To compare prognosis, 55 patients with previously diagnosed mixed GBMO by 2007 WHO classification were classified into six groups, and there was a statistically significant difference (p = .000); however, the number of patients was limited. Thus, if longer follow-up or more prospective studies are ensured in more patients, some prognostic factors that were not statistically significant in current studies may turn out to be statistically significant.
- In conclusion, we successfully reclassified previously diagnosed mixed gliomas into either astrocytic or oligodendroglial tumors with a genetically integrated diagnostic approach. Our study suggests that genetic information, including ATRX and IDH mutation and 1p/19q co-deletion, has powerful prognostic and predictive value. In addition, this information can aid in classifying brain tumors into more distinct categories. Therefore, the integration of phenotypes and genotypes is clearly necessary in the routine practice of brain tumor diagnoses.
DISCUSSION
Acknowledgments
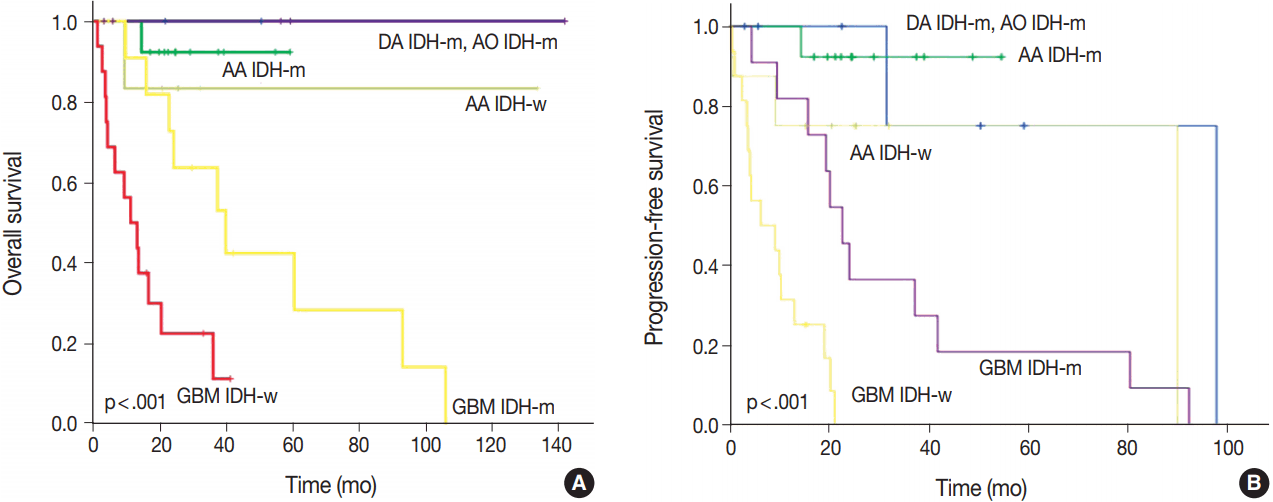
OA, oligoastrocytoma; AOA, anaplastic oligoastrocytoma; GBMO, glioblastoma with oligodendroglioma component; OS, overall survival; PFS, progression-free survival; F, frontal lobe; T, temporal lobe; P, parietal lobe; HIPP, hippocampus; O, occipital lobe; TH, thalamus; CC, corpus callosum; PO, parieto-occipital; FTI, frontotemporoinsular area; FTP, frontotemporoparietal lobe; CCRT, concurrent chemoradiotherapy.
| Primer | Sequence (5’ → 3’) |
|---|---|
| IDH 1-F | ACCAAATGGCACCATACGA |
| IDH 1-R | GCAAAATCACATTATTGCCAAC |
| IDH 2-F | GCTGCAGTGGGACCACTATT |
| IDH 2-R | TGTGGCCTTGTACTGCAGAG |
DA, diffuse astrocytoma; m, mutant; w, wildtype; AA, anaplastic astrocytoma; AO, anaplastic oligodendroglioma; GBM, glioblastoma; OS, overall survival; PFS, progression-free survival; F, frontal lobe; T, temporal lobe; P, parietal lobe; O, occipital lobe; TH, thalamus; HIPP, hippocampus; FTI, frontotemporoinsular area; FTP, frontotemproparietal lobe; CC, corpus callosum; PO, parieto-occipital; CCRT, concurrent chemoradiotherapy.
- 1. Myung JK, Cho HJ, Kim H, et al. Prognosis of glioblastoma with oligodendroglioma component is associated with the IDH1 mutation and MGMT methylation status. Transl Oncol 2014; 7: 712-9. ArticlePubMedPMC
- 2. Jiang H, Ren X, Cui X, et al. 1p/19q codeletion and IDH1/2 mutation identified a subtype of anaplastic oligoastrocytomas with prognosis as favorable as anaplastic oligodendrogliomas. Neuro Oncol 2013; 15: 775-82. ArticlePubMedPMCPDF
- 3. Tortosa A, Viñolas N, Villà S, et al. Prognostic implication of clinical, radiologic, and pathologic features in patients with anaplastic gliomas. Cancer 2003; 97: 1063-71. ArticlePubMedPDF
- 4. Okamoto Y, Di Patre PL, Burkhard C, et al. Population-based study on incidence, survival rates, and genetic alterations of low-grade diffuse astrocytomas and oligodendrogliomas. Acta Neuropathol 2004; 108: 49-56. ArticlePubMedPDF
- 5. Ohgaki H. Contribution of molecular biology to the classification of low-grade diffuse glioma. In: Duffau H, ed. Diffuse low-grade gliomas in adults. London: Springer, 2013; 61-72.
- 6. Kros JM, Gorlia T, Kouwenhoven MC, et al. Panel review of anaplastic oligodendroglioma from European Organization For Research and Treatment of Cancer Trial 26951: assessment of consensus in diagnosis, influence of 1p/19q loss, and correlations with outcome. J Neuropathol Exp Neurol 2007; 66: 545-51. PubMed
- 7. Killela PJ, Pirozzi CJ, Reitman ZJ, et al. The genetic landscape of anaplastic astrocytoma. Oncotarget 2014; 5: 1452-7. ArticlePubMed
- 8. Liu XY, Gerges N, Korshunov A, et al. Frequent ATRX mutations and loss of expression in adult diffuse astrocytic tumors carrying IDH1/IDH2 and TP53 mutations. Acta Neuropathol 2012; 124: 615-25. ArticlePubMedPDF
- 9. Louis DN, Ohgaki H, Wiestler OD, Cavenee WK. WHO classification of tumours of the central nervous system. 4th ed. Lyon: IARC Press, 2016.
- 10. Labussière M, Idbaih A, Wang XW, et al. All the 1p19q codeleted gliomas are mutated on IDH1 or IDH2. Neurology 2010; 74: 1886-90. ArticlePubMed
- 11. Louis DN, Perry A, Burger P, et al. International Society of Neuropathology: Haarlem consensus guidelines for nervous system tumor classification and grading. Brain Pathol 2014; 24: 429-35. PubMedPMC
- 12. Sahm F, Reuss D, Koelsche C, et al. Farewell to oligoastrocytoma: in situ molecular genetics favor classification as either oligodendroglioma or astrocytoma. Acta Neuropathol 2014; 128: 551-9. ArticlePubMedPDF
- 13. Reuss DE, Sahm F, Schrimpf D, et al. ATRX and IDH1-R132H immunohistochemistry with subsequent copy number analysis and IDH sequencing as a basis for an “integrated” diagnostic approach for adult astrocytoma, oligodendroglioma and glioblastoma. Acta Neuropathol 2015; 129: 133-46. ArticlePubMedPDF
- 14. van den Bent MJ, Brandes AA, Taphoorn MJ, et al. Adjuvant procarbazine, lomustine, and vincristine chemotherapy in newly diagnosed anaplastic oligodendroglioma: long-term follow-up of EORTC brain tumor group study 26951. J Clin Oncol 2013; 31: 344-50. ArticlePubMed
- 15. Houillier C, Wang X, Kaloshi G, et al. IDH1 or IDH2 mutations predict longer survival and response to temozolomide in lowgrade gliomas. Neurology 2010; 75: 1560-6. ArticlePubMed
- 16. Grisold W, Oberndorfer S, Struhal W. Stroke and cancer: a review. Acta Neurol Scand 2009; 119: 1-16. ArticlePubMed
- 17. Yan H, Parsons DW, Jin G, et al. IDH1 and IDH2 mutations in gliomas. N Engl J Med 2009; 360: 765-73. PubMedPMC
- 18. Haberler C, Wöhrer A. Clinical neuropathology practice news 2-2014: ATRX, a new candidate biomarker in gliomas. Clin Neuropathol 2014; 33: 108-11. ArticlePubMedPMC
- 19. Love S, Perry A, Ironside J, Budka H. Greenfield's neuropathology. 9th ed. Boca Raton: CRC Press, 2015.
- 20. Louis DN, von Deimling A, Chung RY, et al. Comparative study of p53 gene and protein alterations in human astrocytic tumors. J Neuropathol Exp Neurol 1993; 52: 31-8. ArticlePubMedPDF
REFERENCES
Figure & Data
References
Citations

- SNUH methylation classifier for CNS tumors
Kwanghoon Lee, Jaemin Jeon, Jin Woo Park, Suwan Yu, Jae-Kyung Won, Kwangsoo Kim, Chul-Kee Park, Sung-Hye Park
Clinical Epigenetics.2025;[Epub] CrossRef - Oligodendrogliomas: findings after classifying the same cohort using pre- and post-World Health Organization (WHO) 2021 criteria
Maria Angeles Vaz-Salgado, Juan M Sepulveda, Julie Earl, Jacqueline Gutierrez, Yolanda Ruano, Hector Pian, Diana Cantero, Aurelio Hernández-Lain
Brain Communications.2025;[Epub] CrossRef - The prognostic significance of p16 expression pattern in diffuse gliomas
Jin Woo Park, Jeongwan Kang, Ka Young Lim, Hyunhee Kim, Seong-Ik Kim, Jae Kyung Won, Chul-Kee Park, Sung-Hye Park
Journal of Pathology and Translational Medicine.2021; 55(2): 102. CrossRef - Dynamic susceptibility contrast and diffusion MR imaging identify oligodendroglioma as defined by the 2016 WHO classification for brain tumors: histogram analysis approach
Anna Latysheva, Kyrre Eeg Emblem, Petter Brandal, Einar Osland Vik-Mo, Jens Pahnke, Kjetil Røysland, John K. Hald, Andrés Server
Neuroradiology.2019; 61(5): 545. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
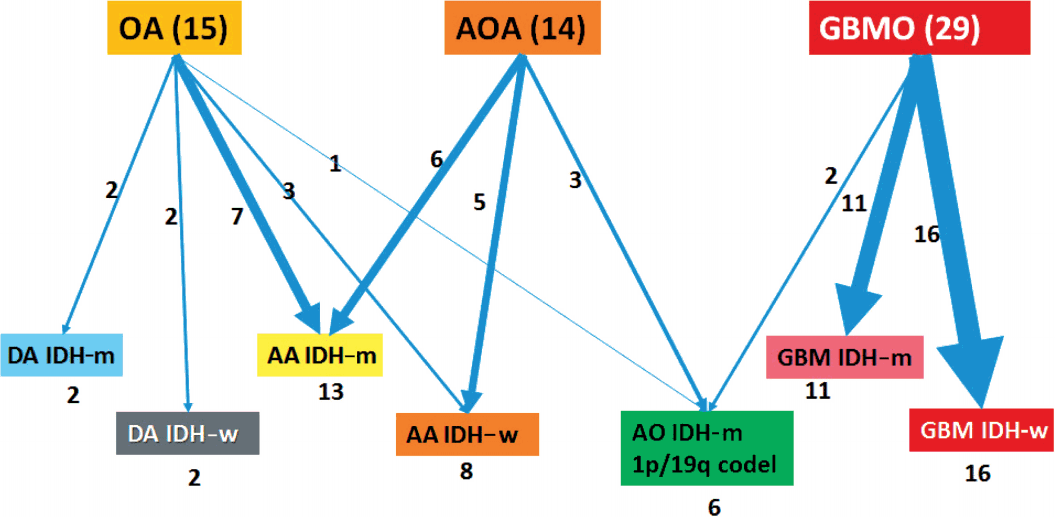
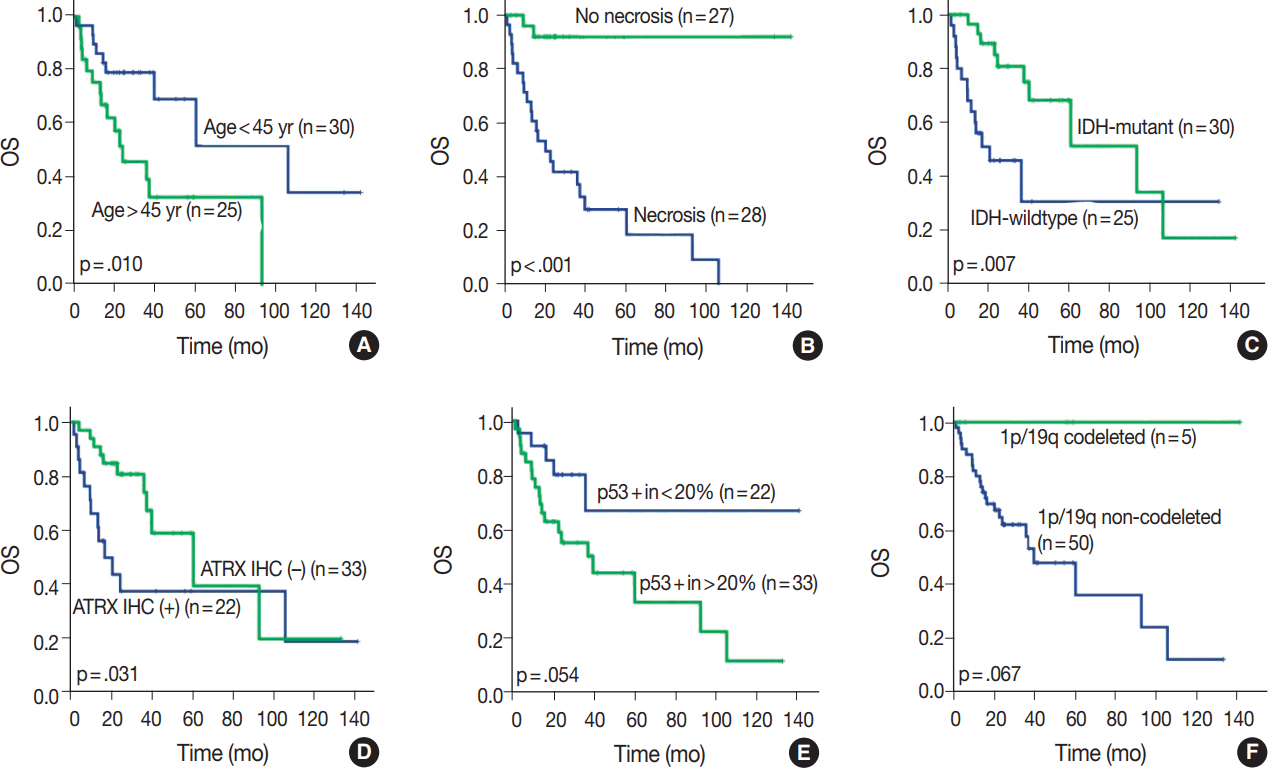
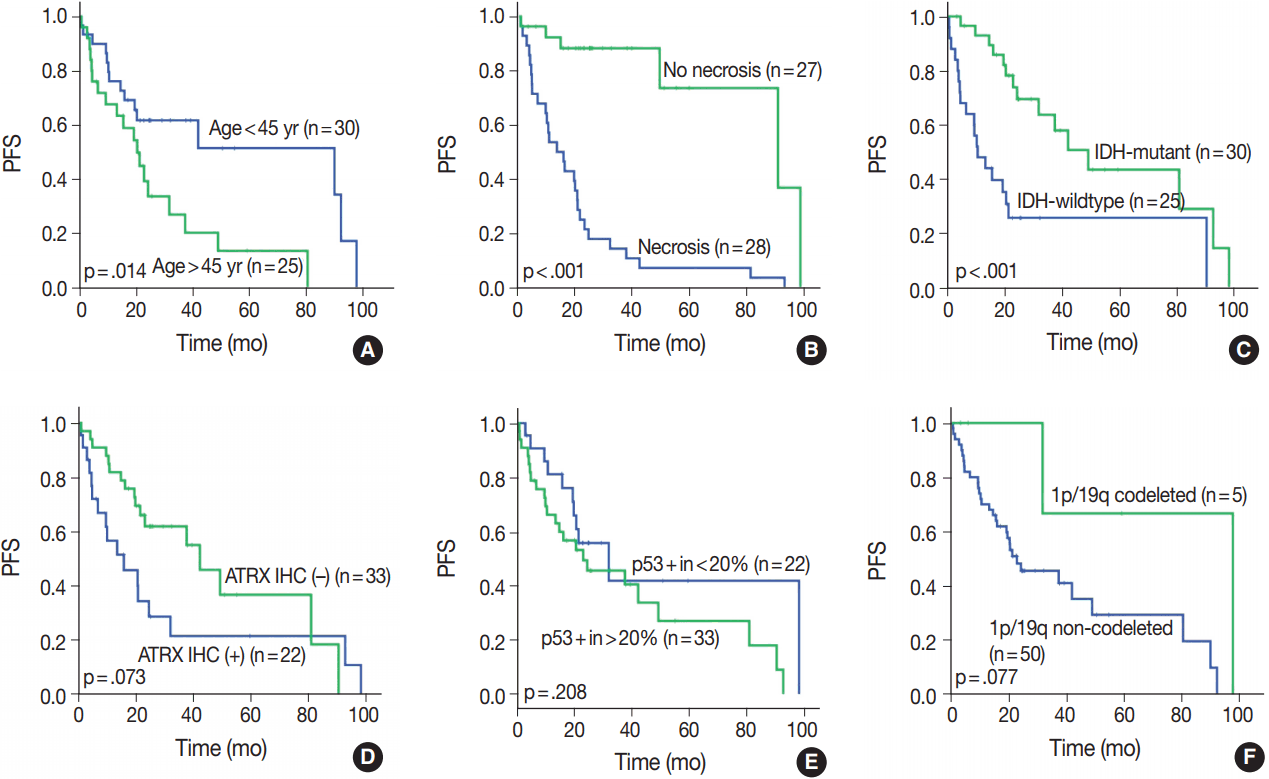
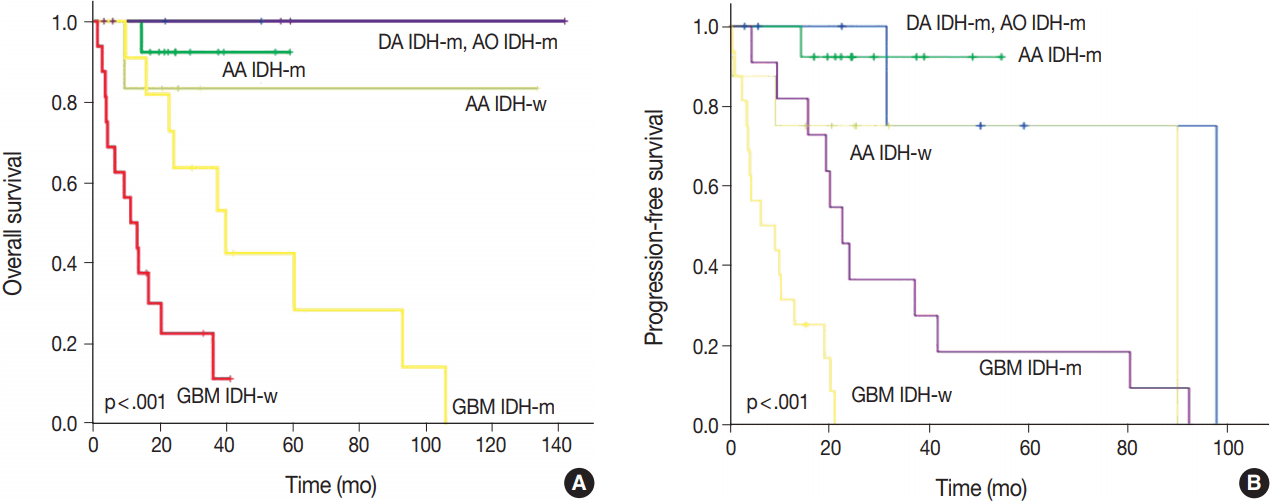
Fig. 1.
Fig. 2.
Fig. 3.
Fig. 4.
Fig. 5.
| Parameter | OA (n = 15) | AOA (n = 14) | GBMO (n = 29) |
|---|---|---|---|
| Age (yr) | |||
| Mean (range) | 40.8 (14–72) | 41.1 (13–64) | 53.5 (31–77) |
| Median | 36 | 35.5 | 56 |
| Sex (male:female) | 11:4 | 5:9 | 22:7 |
| Median OS (mo) | 45.3 | 21.7 | 26.7 |
| Median PFS (mo) | 39.8 | 20.9 | 19.0 |
| Tumor location | F8 T4 P1 PT1 HIPP1 | F6 T5 P1 O1 TH1 | F13 T10 P1 TH1 CC1 PO1 FTI1 FTP1 |
| Treatment | Surgery only | Surgery only 13 | CCRT 21 |
| Others 1 | Others 8 |
| Primer | Sequence (5’ → 3’) |
|---|---|
| IDH 1-F | ACCAAATGGCACCATACGA |
| IDH 1-R | GCAAAATCACATTATTGCCAAC |
| IDH 2-F | GCTGCAGTGGGACCACTATT |
| IDH 2-R | TGTGGCCTTGTACTGCAGAG |
| Original diagnosis (n = 58) | Re-diagnosis (n = 58) |
|
|---|---|---|
| Criteria | No. (%) | |
| OA, grade II (n = 14) | DA IDH-m | 2 (14.3) |
| DA IDH-w | 2 (14.3) | |
| AA IDH-m | 7 (50) | |
| AA IDH-w | 2 (14.3) | |
| AO IDH-m and 1p/19q-codeleted | 1 (7.1) | |
| AOA, grade III (n = 14) | AA IDH-m | 6 (42.9) |
| AA IDH-w | 5 (35.7) | |
| AO IDH-m and 1p/19q-codeleted | 3 (17.6) | |
| GBMO, grade IV (n = 29) | GBM IDH-w | 16 (55.2) |
| GBM IDH-m | 11 (37.9) | |
| AO IDH-m and 1p/19q-codeleted | 2 (6.9) | |
| Parameter | DA IDH-m (n = 2) | DA IDH-w (n = 2) | AA IDH-m (n = 13) | AA IDH-w (n = 8) | AO (n = 6) | GBM IDH-m (n = 11) | GBM IDH-w (n = 16) |
|---|---|---|---|---|---|---|---|
| Age, mean (range, yr) | 37.5 (31–44) | 49.5 (32–67) | 39.3 | 39.9 (14–64) | 52.6 (35–69) | 42.3 (34–58) | 58.4 (31–77) |
| Sex (male:female) | 2:0 | 1:1 | 9:4 | 3:5 | 3:3 | 7:4 | 13:3 |
| ATRX (–), n (%) | 2 (100) | 1 (50) | 12 (92.3) | 7 (87.5) | 0 | 7 (63.6) | 5 (31.3) |
| 1p/19q codel | 0 | 0 | 0 | 0 | 6 | 0 | 0 |
| Primary GBM | 7 | 14 | |||||
| Secondary GBM | 4 | 1 | |||||
| OS (mo) | 36.5 | 19.5 | 29.8 | 34.5 | 45.5 | 43.6 | 14.5 |
| PFS (mo) | 36.0 | 18.9 | 29.0 | 27.3 | 34.0 | 33.4 | 9.1 |
| Tumor location | F1 P1 | F2 | F5 T6 P1 PT1 | F4 T1 O1 TH1 HIPP1 | F4 T2 | F6 T3 FTI1 FTP1 | F5 T7 P1 CC1 PO1 TH1 |
| Treatment | Surgery only | Surgery only | Surgery only | Surgery only 7 | Surgery only 4 | CCRT 10 | CCRT 11 |
| Others 1 | CCRT 1 | Others 1 | Others 5 | ||||
| Others 1 |
OA, oligoastrocytoma; AOA, anaplastic oligoastrocytoma; GBMO, glioblastoma with oligodendroglioma component; OS, overall survival; PFS, progression-free survival; F, frontal lobe; T, temporal lobe; P, parietal lobe; HIPP, hippocampus; O, occipital lobe; TH, thalamus; CC, corpus callosum; PO, parieto-occipital; FTI, frontotemporoinsular area; FTP, frontotemporoparietal lobe; CCRT, concurrent chemoradiotherapy.
F, forward; R, reverse.
OA, oligoastrocytoma; AOA, anaplastic oligoastrocytoma; GBMO, glioblastoma with oligodendroglioma component; DA, diffuse astrocytoma; IDH-m, IDH-mutant; IDH-w, IDH-wildtype; AA, anaplastic astrocytoma; AO, anaplastic oligodendroglioma; GBM, glioblastoma.
DA, diffuse astrocytoma; m, mutant; w, wildtype; AA, anaplastic astrocytoma; AO, anaplastic oligodendroglioma; GBM, glioblastoma; OS, overall survival; PFS, progression-free survival; F, frontal lobe; T, temporal lobe; P, parietal lobe; O, occipital lobe; TH, thalamus; HIPP, hippocampus; FTI, frontotemporoinsular area; FTP, frontotemproparietal lobe; CC, corpus callosum; PO, parieto-occipital; CCRT, concurrent chemoradiotherapy.

 E-submission
E-submission







