Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 55(4); 2021 > Article
-
Review
Standardization of the pathologic diagnosis of appendiceal mucinous neoplasms -
Dong-Wook Kang1
 , Baek-hui Kim2
, Baek-hui Kim2 , Joon Mee Kim,3
, Joon Mee Kim,3 , Jihun Kim4
, Jihun Kim4 , Hee Jin Chang5
, Hee Jin Chang5 , Mee Soo Chang6
, Mee Soo Chang6 , Jin-Hee Sohn7
, Jin-Hee Sohn7 , Mee-Yon Cho8
, Mee-Yon Cho8 , So-Young Jin9
, So-Young Jin9 , Hee Kyung Chang10
, Hee Kyung Chang10 , Hye Seung Han11
, Hye Seung Han11 , Jung Yeon Kim12
, Jung Yeon Kim12 , Hee Sung Kim13
, Hee Sung Kim13 , Do Youn Park14
, Do Youn Park14 , Ha Young Park15
, Ha Young Park15 , So Jeong Lee16
, So Jeong Lee16 , Wonae Lee17
, Wonae Lee17 , Hye Seung Lee18
, Hye Seung Lee18 , Yoo Na Kang19
, Yoo Na Kang19 , Younghee Choi20
, Younghee Choi20 , The Gastrointestinal Pathology Study Group of the Korean Society of Pathologists
, The Gastrointestinal Pathology Study Group of the Korean Society of Pathologists -
Journal of Pathology and Translational Medicine 2021;55(4):247-264.
DOI: https://doi.org/10.4132/jptm.2021.05.28
Published online: July 8, 2021
1Department of Pathology, Chungnam National University Sejong Hospital, Chungnam National University College of Medicine, Daejeon, Korea
2Department of Pathology, Korea University Guro Hospital, Korea University College of Medicine, Seoul, Korea
3Department of Pathology, Inha University School of Medicine, Incheon, Korea
4Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
5Department of Pathology, Research Institute and Hospital, National Cancer Center, Goyang, Korea
6Department of Pathology, Seoul National University Boramae Hospital, Seoul National University College of Medicine, Seoul, Korea
7Department of Pathology, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, Seoul, Korea
8Department of Pathology, Yonsei University Wonju College of Medicine, Wonju, Korea
9Department of Pathology, Soonchunhyang University Seoul Hospital, Seoul, Korea
10Department of Pathology, Kosin University Gospel Hospital, Kosin University College of Medicine, Busan, Korea
11Department of Pathology, Konkuk University School of Medicine, Seoul, Korea
12Department of Pathology, Inje University Sanggye Paik Hospital, Seoul, Korea
13Department of Pathology, Chung-Ang University School of Medicine, Seoul, Korea
14Department of Pathology, St. Maria Pathology, Busan, Korea
15Department of Pathology, Busan Paik Hospital, Inje University College of Medicine, Busan, Korea
16Department of Pathology, Pusan National University Hospital and Pusan National University School of Medicine, Busan, Korea
17Department of Pathology, Dankook University College of Medicine, Cheonan, Korea
18Department of Pathology, Seoul National University College of Medicine, Seoul, Korea
19Department of Forensic Medicine, Kyungpook National University School of Medicine, Daegu, Korea
20Department of Pathology, Hallym University Dongtan Sacred Heart Hospital, Hwaseong, Korea
- Corresponding Author: Joon Mee Kim, MD, PhD, Department of Pathology, Inha University School of Medicine, 27 Inhang-ro, Jung-gu, Incheon 22332, Korea Tel: +82-32-890-3987, Fax: +82-32-890-3464, E-mail: jmkpath@inha.ac.kr
© 2021 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- Abstract
- APPENDICEAL MUCINOUS NEOPLASMS
- LOW-GRADE APPENDICEAL MUCINOUS NEOPLASM
- HIGH-GRADE APPENDICEAL MUCINOUS NEOPLASM
- SERRATED LESIONS
- STAGING OF APPENDICEAL MUCINOUS NEOPLASMS
- APPENDICEAL MUCINOUS ADENOCARCINOMA
- DISSEMINATED PERITONEAL MUCINOUS DISEASE
- OTHER APPENDICEAL EPITHELIAL TUMORS
- CONCLUSION
- Supplementary Materials
- NOTES
- Acknowledgments
- REFERENCES
Abstract
- Although the understanding of appendiceal mucinous neoplasms (AMNs) and their relationship with disseminated peritoneal mucinous disease have advanced, the diagnosis, classification, and treatment of AMNs are still confusing for pathologists and clinicians. The Gastrointestinal Pathology Study Group of the Korean Society of Pathologists (GPSG-KSP) proposed a multicenter study and held a workshop for the “Standardization of the Pathologic Diagnosis of the Appendiceal Mucinous Neoplasm” to overcome the controversy and potential conflicts. The present article is focused on the diagnostic criteria, terminologies, tumor grading, pathologic staging, biologic behavior, treatment, and prognosis of AMNs and disseminated peritoneal mucinous disease. In addition, GPSG-KSP proposes a checklist of standard data elements of appendiceal epithelial neoplasms to standardize pathologic diagnosis. We hope the present article will provide pathologists with updated knowledge on how to handle and diagnose AMNs and disseminated peritoneal mucinous disease.
- Definition and characteristics of appendiceal mucinous neoplasms
- Appendiceal epithelial neoplasms, over 70% of the mucinproducing type, account for most primary appendiceal tumors [1-3]. The classification of appendiceal epithelial tumors of the WHO 5th Digestive System Tumors is listed in Table 1. The WHO 5th Digestive System Tumors defined the AMN as a mucinous epithelial proliferation with extracellular mucin and pushing invasion pattern or pushing tumor margins [1]. Most AMNs develop in middle-aged or elderly patients and present with non-specific symptoms or signs, such as acute abdominal pain [1-5,12]. AMNs can exhibit variable clinical behaviors, ranging from a relatively slow-growing and low-grade tumor, but with considerable risk of recurrence, to a high-grade and aggressive neoplasm that produces eventual peritoneal metastasis [1-7]. AMNs can be divided into LAMN and high-grade appendiceal mucinous neoplasm (HAMN) by cytological grading. The classification, grading, biologic behavior codes, diagnostic criteria, and microscopic features are listed in Table 2.
- Pushing invasion in appendiceal mucinous neoplasms
- Pushing invasion is defined as a tongue-like protrusion, diverticulum- like growing, or broad-front spread of the epithelium into the appendiceal wall [1,3,12]. Pushing invasion can cause the attenuation of lamina propria and muscularis mucosae, and this is not associated with the destructive invasion or desmoplastic infiltration, characteristics of invasive carcinoma [1,3,12,23]. Moreover, pushing invasion through the appendiceal wall presents extra-appendiceal mucin accumulation, leading to diagnostic difficulty or misdiagnosis. Extracellular mucin can lead to the dissection of the wall and rupture with the potential for intraperitoneal dissemination [24,25].
APPENDICEAL MUCINOUS NEOPLASMS
- Definition and histologic findings of low-grade appendiceal mucinous neoplasm
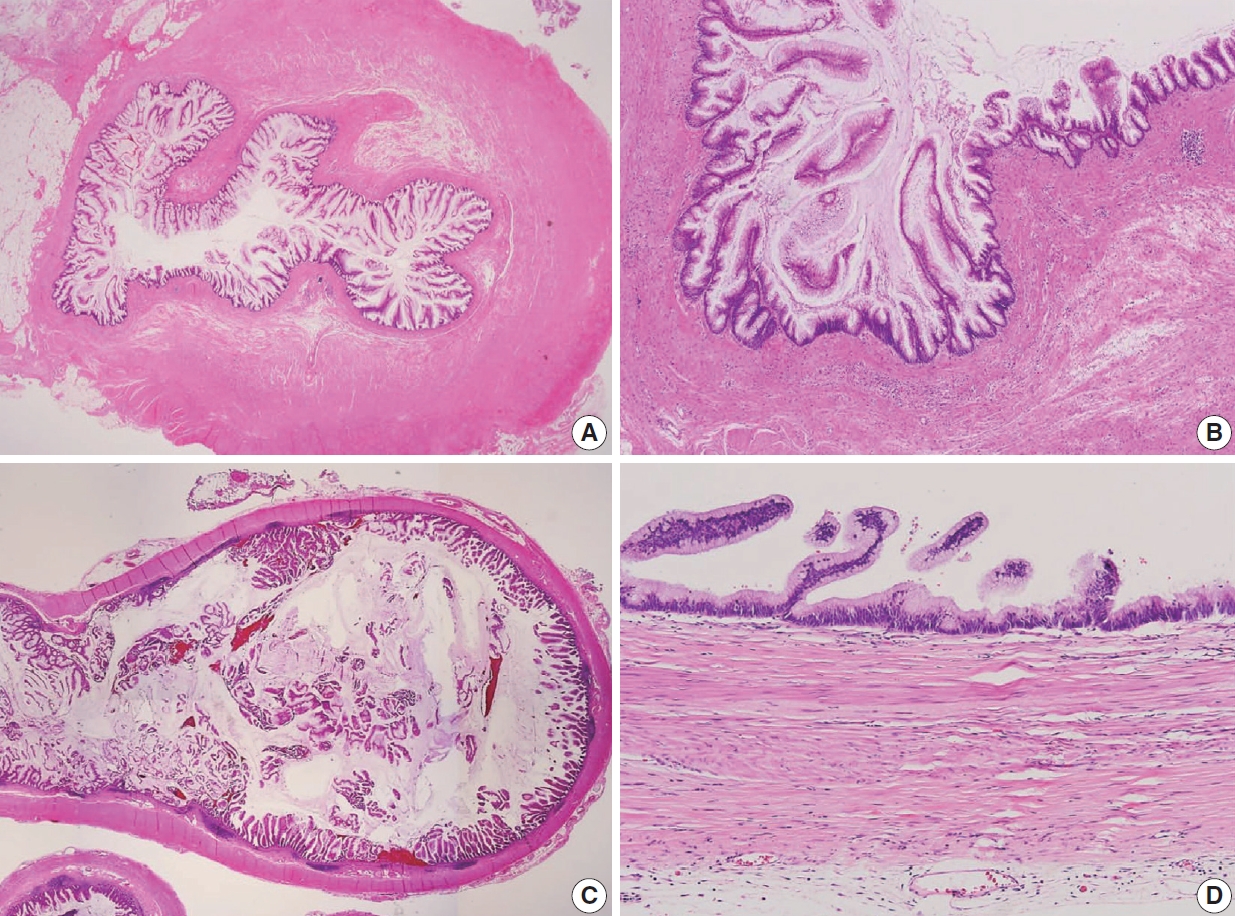
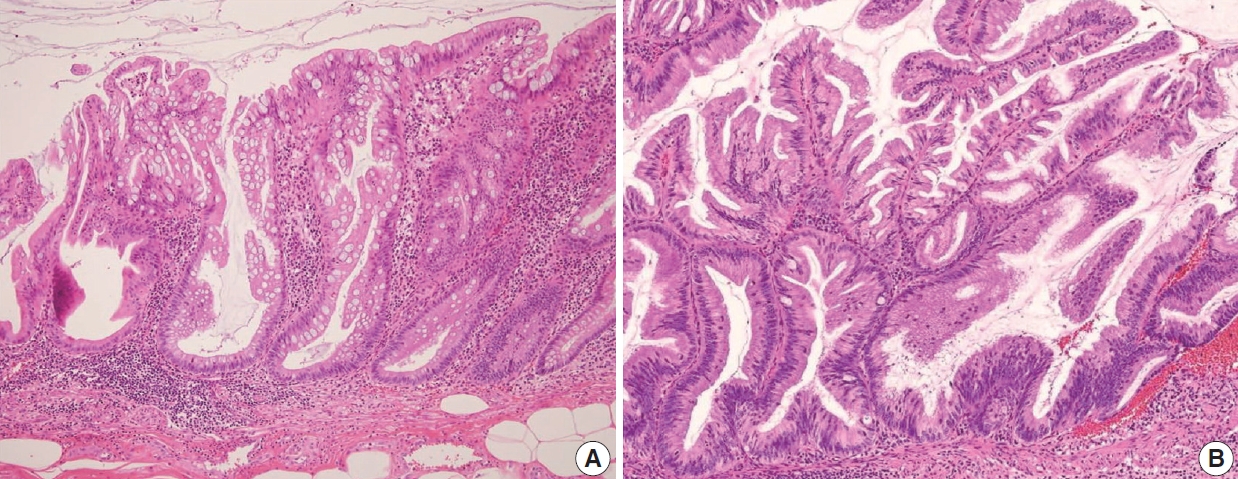
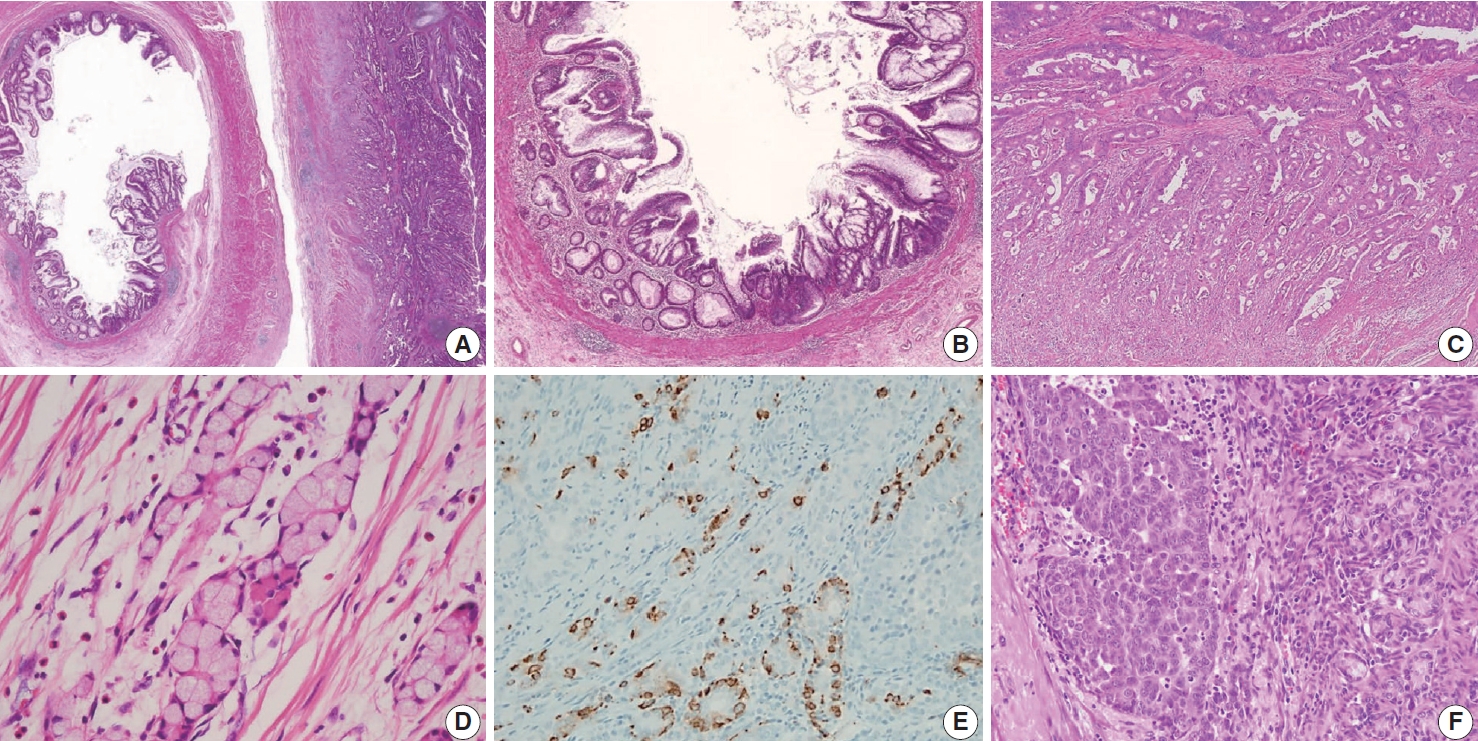
- The AJCC 8th Cancer Staging Manual defined a LAMN as a mucinous neoplasm with low-grade cytology associated with the obliteration of the muscularis mucosae without overt features of the invasion [13]. The lamina propria is frequently effaced, and mucosal lymphoid tissues are decreased or absent. Mucinous cystadenoma and mucocele were histologically used as synonyms for LAMN but are no longer recommended in pathologic reports due to their ambiguous and misleading nature. The essential histologic finding of LAMN is low-grade cytology with pushing invasion; however, there is no destructive invasion in the appendiceal wall. Fig. 1 demonstrates the microscopic features of LAMNs, displaying pushing invasion and broad-front spreading by a mucin-rich epithelium. Notably, when regarding whether pushing invasion is necessary for the diagnosis of LAMN, most of the survey responders diagnosed Fig. 1A, B as LAMN. In typical LAMNs, there is a loss of the normal mucosal architectures, at least focally, such as obliteration of the lamina propria and muscularis mucosa, fibrosis of the submucosa, and atrophy of the lymphoid follicles. Although the epithelium may show a papillary, villous, undulating, or flat architecture, lining epithelial cells are generally arranged in a monolayer (Fig. 1C, D). The microscopic features essential for LAMN proposed by the PSOGI panel are illustrated in Fig. 2. It is not surprising that LAMN with intact muscularis mucosae may develop disseminated peritoneal mucinous disease. For this reason, there is hesitancy in diagnosing a lesion with intact muscularis mucosae as adenoma because adenomas should be limited to benign lesions that have no potential for peritoneal dissemination. Patients with LAMN often present symptoms like acute appendicitis. Typical LAMNs usually have thin fibrotic walls and abundant intraluminal mucin, and less commonly, calcification of the wall. Grossly apparent rupture with mucin extrusion is generally manifested in patients with disseminated peritoneal mucinous disease.
- Mimickers of low-grade appendiceal mucinous neoplasm
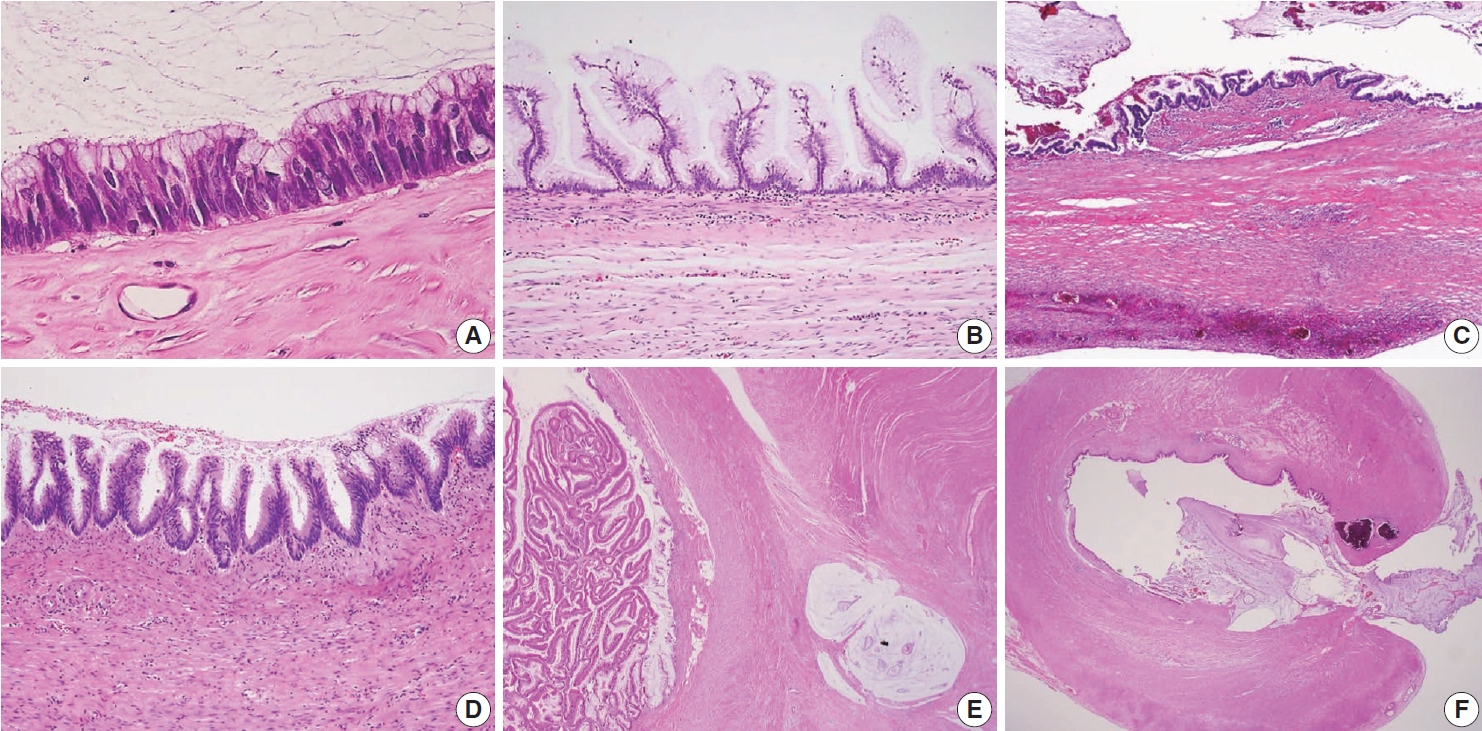
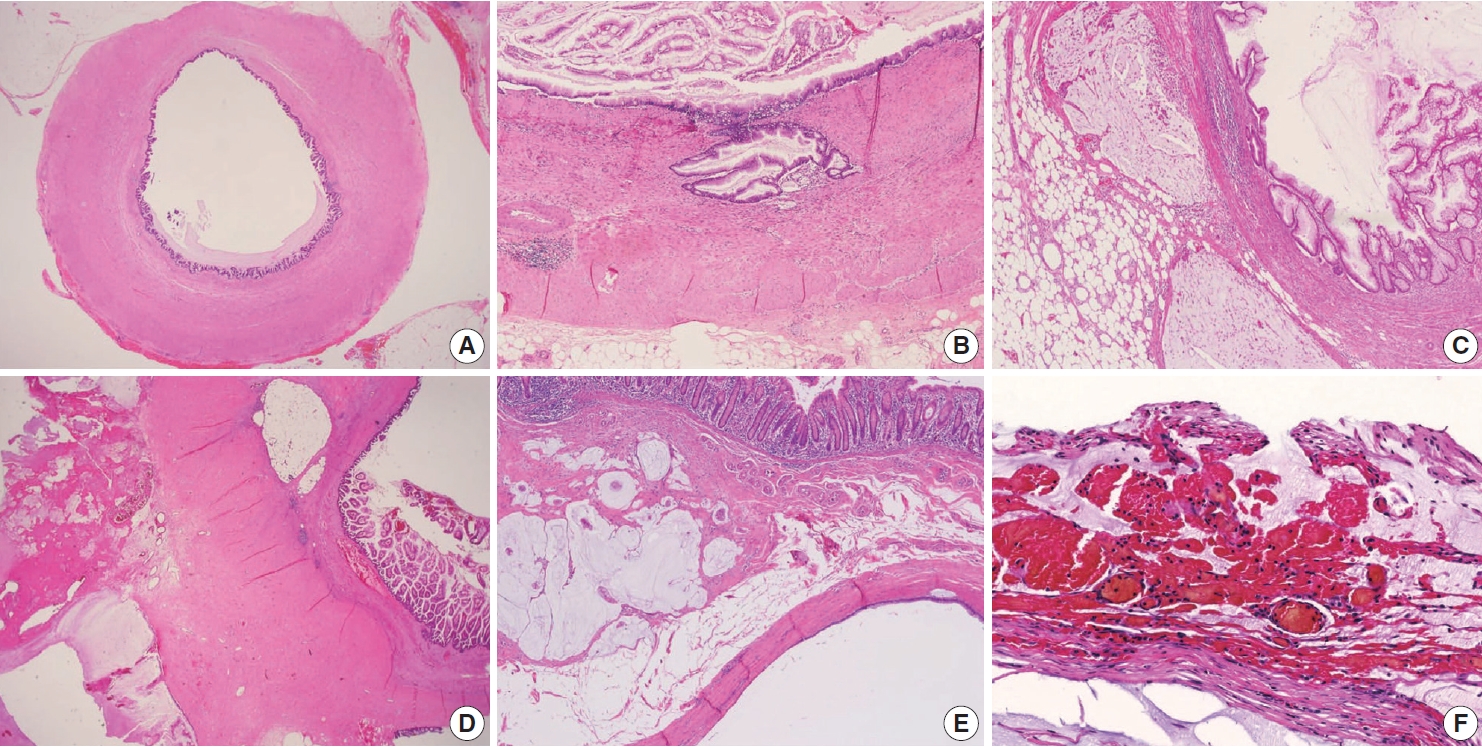
- Because LAMNs show benign-looking cytology, they can mimic several benign lesions associated with mucinous epithelial proliferation. The appendiceal diverticulum is perhaps the most common mimicker of LAMN due to their pushing growing pattern, specifically when related to abundant mucin exposed to the serosal surface. A preserved lamina propria is an indication of the diagnosis of a diverticulum. Careful examination of the entire appendix at lower magnification often leads to identifying a diverticulum. Sometimes, Schwann cell hyperplasia expands the lamina propria, possibly because of the obliteration effects [26,27]. If a histologic examination of the entire appendix fails to reveal a lining epithelium, the possibility of a retention cyst may be considered. Appendiceal retention cysts are rare and mostly small-sized; thus, lesions more than 2 cm in diameter are much more likely to be LAMNs. Appendiceal endometriosis with intestinal metaplasia is uncommon and can resemble a LAMN. The epithelial component acquires an intestinal phenotype characterized by columnar mucin-secreting cells, sometimes with goblet cells. Recognition of the endometrial stroma surrounding the glands and CD10 immunohistochemical stain will lead to a diagnosis. Mucosal hyperplasia can be seen in acute appendicitis. Mucosal hyperplasia tends to be more pronounced toward the luminal side and is frequently observed in areas of intense inflammation. Occasionally, reactive cellular atypia in the acute suppurative appendicitis may approximate the neoplastic atypia in LAMN. Fig. 3 reveals various mimickers of LAMN.
LOW-GRADE APPENDICEAL MUCINOUS NEOPLASM
- Definition and histologic of HAMN
- HAMN was proposed for the lesion with similar architectures of LAMN having high-grade cytologic features but no infiltrative invasion [13]. Fig. 4 demonstrates the histologic findings of HAMN. The microscopic findings include cribriform, loss of nuclear polarity, high-grade cytology (i.e., enlarged, hyperchromatic, pleomorphic nuclei, and atypical mitotic figures), frequent single-cell necrosis and may present sloughing of necrotic cells into the lumen [23]. HAMNs are suggested to reveal an intermediate risk between LAMNs and mucinous adenocarcinomas.
- Biologic behavior code of HAMN
- According to the WHO 5th Digestive System Tumors, the International Classification of Diseases for Oncology, 3rd edition (ICD-O3) behavior codes for LAMN and HAMN were proposed as 8480/1 and 8480/2, respectively (Table 2). The response of the ICD-O3 of the HAMN was varied at the workshop because the questionnaire for the suspected ICD-O3 behavior code of the HAMN was conducted before the publication of the WHO 5th Digestive System Tumors. However, there was a consensus that the behavior code should be 8480/2 to avoid confusion in the following GPSG meeting. Misdraji et al. [7] reported that HAMNs show more aggressive clinical behavior than LAMNs, but the prognosis is still not unveiled because HAMNs are extremely rare. Therefore, nothing of the HAMNs prognosis is known. Yantiss et al. [11] reported that HAMN was more likely to be associated with aggressive clinical course and extra-appendiceal mucin spreading than LAMN. Carr et al. [23] proposed that HAMN should include lesions with high-grade cytologic atypia that is seen only focally, provided it is unequivocal; however, there has been no accepted consensus on the quantification of focal lesions yet. We surveyed the “focal” concept at the workshop, and the responses are shown in Supplementary Data S2.
HIGH-GRADE APPENDICEAL MUCINOUS NEOPLASM
- Classification of appendiceal serrated lesions
- The WHO 5th Digestive System Tumors defined an appendiceal serrated lesion as a mucinous epithelial lesion characterized by a serrated (sawtooth or stellate) architecture of the luminal crypt [1]. Serrated lesions are classified as a hyperplastic polyp, sessile serrated lesion without dysplasia, and serrated lesion with dysplasia or serrated dysplasia [1,14]. Polyps that lack a luminal crypt of serrated architecture and dysplasia are classified as hyperplastic polyps. Sessile serrated lesions are commonly deficient in cytological dysplasia. They often involve mucosa in a diffuse circumferential fashion. Serrated dysplasia refers to conventional adenoma-like dysplasia, traditional serrated adenoma-like dysplasia, and mixed morphological patterns of dysplasia [14]. LAMNs are heterogeneous, with typical LAMN in some areas and serrated lesions in others, raising the possibility that some LAMNs could arise from serrated lesions.
- Pathologic findings and pathogenesis of appendiceal serrated
- Occasionally, LAMNs simulate a serrated lesion and are challenging to diagnose, particularly if the muscularis mucosae is intact. By the diagnostic criteria of the PSOGI classification, a serrated lesion should be confined to the lesion with intact muscularis mucosae, no mucin in the wall or outside, and no expansile growth pattern or pushing invasion [12]. Although appendiceal serrated lesions have similar microscopic features to their colorectal counterparts, they commonly harbor KRAS mutations but lack BRAF mutations, indicating that the serrated pathway in the appendix is likely to be different from those of the colorectum [14,23,28]. However, serrated lesions remain incompletely studied and are not fully understood. The serrated lesions of the appendix are demonstrated in Fig. 5.
SERRATED LESIONS
- AJCC 8th TNM staging
- The AJCC 7th Cancer Staging Manual was not clear on applying the staging criteria of AMNs [29]. However, the AJCC 8th Cancer Staging Manual showed significant changes to the staging criteria of AMNs, particularly for LAMN [13]. In addition, the role of acellular mucin or a mucinous epithelium outside of the appendix is also addressed and included in the staging [1,13]. A summary of the AJCC pT classification of LAMN and prognostic significance from the AJCC 8th Cancer Staging Manual is listed in Table 3. In non-mucinous appendiceal tumors, the pTis category includes high-grade dysplasia, carcinoma in situ, and intramucosal carcinoma [13]. Notably, LAMNs confined to the appendiceal wall are classified as pTis. The staging of pTis reflects the excellent outcome when limited to the appendiceal wall [13]. In most LAMNs, there is no well-preserved mucosal architecture; hence, assessing the involvement of the mucosa and submucosa is impossible, resulting in the inability to apply pT1 designation to the LAMNs. In addition, studies evaluating the outcomes in LAMN have determined that pushing invasion into the appendiceal wall is not associated with tumor recurrence [7,11,30]. Thus, pT2 designation does not apply to LAMN [13].
- Staging of low-grade appendiceal mucinous neoplasm
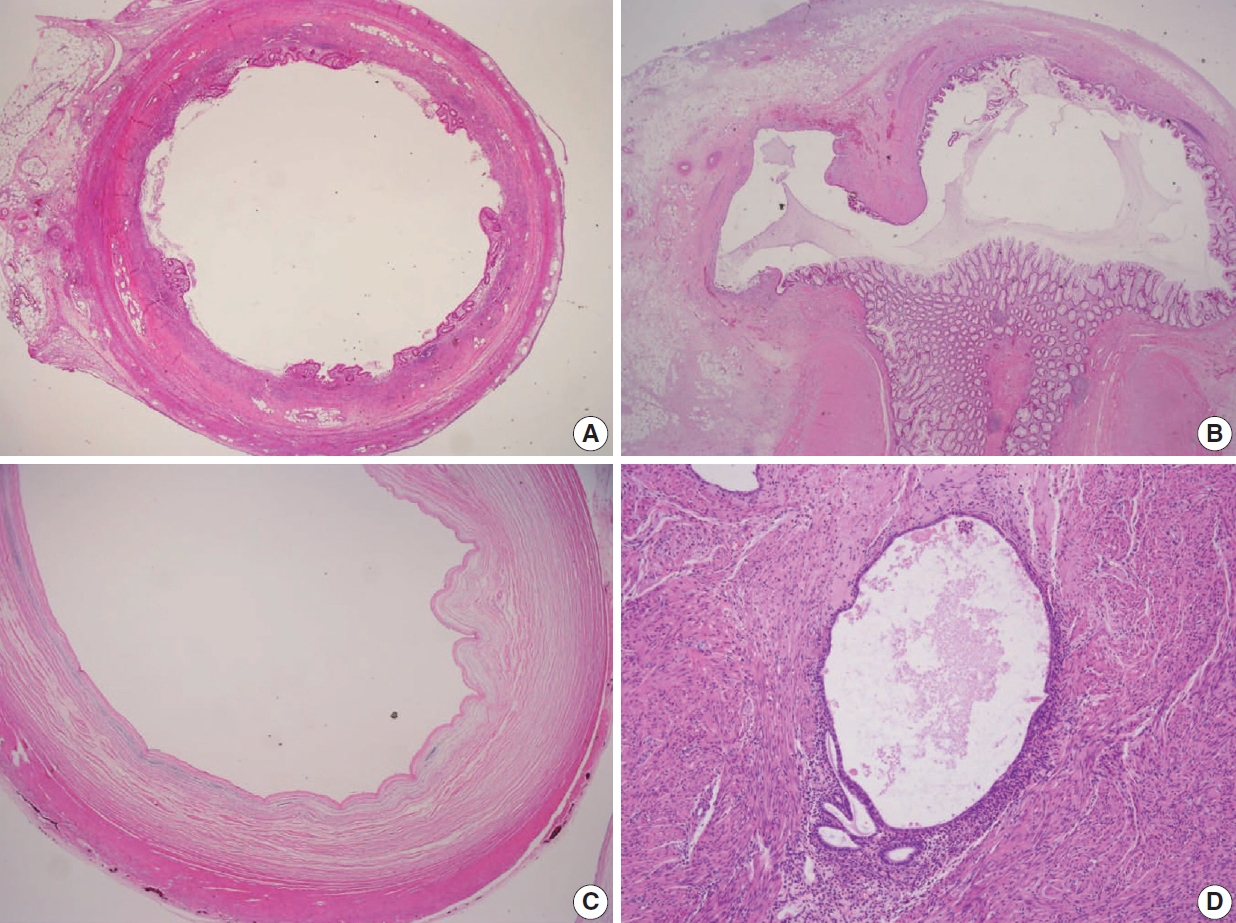
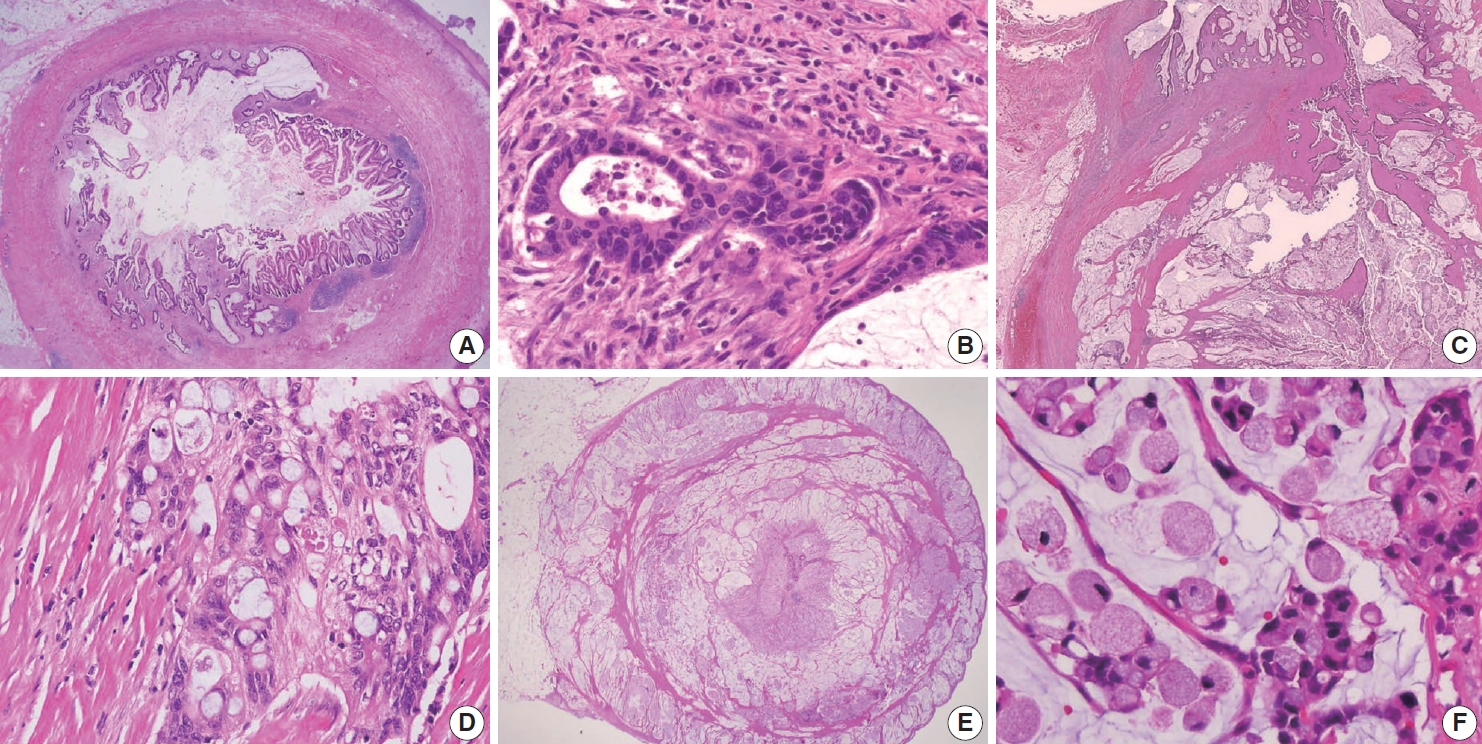
- Various pT stagings of LAMN were presented in the workshop and are illustrated in Fig. 6. In a questionnaire for suspected pTis or other stages, many pathologists were unaware of the new concept of pTis(LAMN). Because mucinous tumors can frequently extend into the muscularis propria by pushing invasion in LAMN, mucin extending into the muscularis propria should be classified as pTis(LAMN) as long as it does not extend into the mesoappendix or serosa (Fig. 6A, B). LAMNs showing either acellular mucin or mucinous epithelium involvement to subserosa or mesoappendix are classified as pT3 (Fig. 6C). Perforation of the appendix by LAMN is associated with a high risk of peritoneal dissemination. Given the risk of disseminated peritoneal mucinous disease, pT4 includes assessing both acellular mucin and the mucinous epithelium and is designated as T4a (penetration of the serosa) (Fig. 6D) and T4b (directly invades adjacent organs or structures) (Fig. 6E). Notably, pT4a does not include luminal or mural spreading into the cecum. Most cases of LAMNs are associated with luminal mucin. This mucin can frequently contaminate the serosa during the gross examination, histologic processing, and even operation. These cases should not be diagnosed as pT4a. Indeed, mucinous deposits on the appendiceal serosa are associated with a granulation tissue-like response and neo-vascularization; numerous small capillaries containing red blood cells are seen coursing through the mucin (Fig. 6F). When the peritoneal dissemination is limited to acellular mucin only, it is classified as M1a. Other metastatic categories are M1b, which refers to metastases confined to the peritoneum only, and M1c which refers to metastases outside the peritoneum, such as pleuropulmonary metastasis.
- Staging of HAMN
- According to the Union for International Cancer Control staging system, LAMN and HAMN are considered as pTis, if confined to the appendiceal wall [1]. However, the AJCC 8th Cancer Staging Manual stated that the HAMN should be staged using the same staging system as invasive mucinous adenocarcinoma [13]. These two different opinions may bring confusion in daily practice. Valasek and Pai [3] proposed that HAMN with pushing invasion into the muscularis propria would be classified as pT2 using the same staging system for invasive adenocarcinoma. However, since there are very few cases of HAMN, the actual application of the staging for HAMNs will be determined after the results of large-scale survival analysis. We surveyed the expected pT stages of the HAMNs at the workshop, and the responses are shown in Supplementary Data S2.
STAGING OF APPENDICEAL MUCINOUS NEOPLASMS
- Definition and histologic findings of appendiceal mucinous adenocarcinoma
- Appendiceal mucinous adenocarcinoma is defined as mucinous neoplasms showing infiltrative invasion comprised of > 50% extracellular mucin [1]. In contrast to the pushing invasion of the LAMN, the infiltrative invasion is confined to destructive stromal invasion into the appendiceal wall. The histologic features of infiltrative invasion include infiltrative tumor cells, which exhibit cribriform, small tubules, or single cells accompanied by mucin within the desmoplastic stroma, and small dissecting mucin containing floating tumor cells but inconspicuous desmoplastic reaction [1,3,12]. The neoplastic epithelium in mucinous adenocarcinoma demonstrates high-grade cytology that may be only seen focally, are characterized by enlarged nuclei, prominent nucleoli, increased mitotic figures, full-thickness stratification, and loss of nuclear polarity, which often extends to the luminal aspect of the epithelial cell.
- Grading system of appendiceal mucinous adenocarcinoma
- The PSOGI consensus panel has classified appendiceal mucinous adenocarcinomas and moderately and poorly differentiated mucinous adenocarcinoma, similar to the grading system of nonmucinous adenocarcinoma in other gastrointestinal tracts [12]. However, the PSOGI consensus classification was not easy to apply to appendiceal mucinous adenocarcinomas. The AJCC 8th Cancer Staging Manual classified the appendiceal mucinous tumors as a three-tier grading system: G1 (well-differentiated), G2 (moderately differentiated), and G3 (poorly differentiated), based on cytologic features, tumor cellularity, and signet-ring components [13].
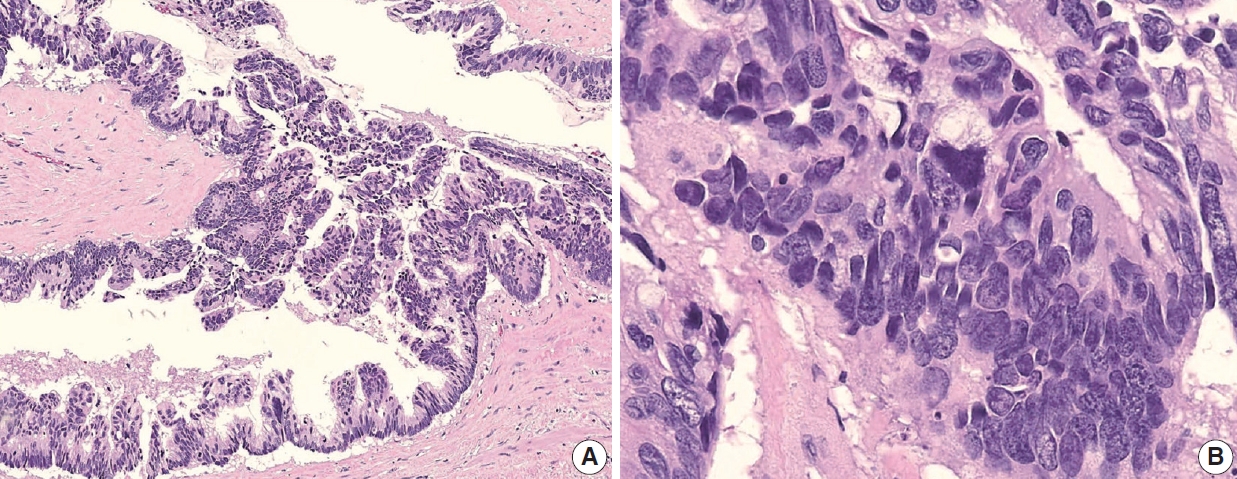
- Appendiceal G1 (well-differentiated) tumors are low-grade cytology, usually lacking infiltrative invasion, and essentially refers to LAMN. Given that mucinous adenocarcinoma is characterized by infiltrative invasion and almost always exhibits at least focal areas of high-grade cytologic features, typical mucinous adenocarcinomas should be classified as either G2 (moderately differentiated) or G3 (poorly differentiated) tumors [3]. Mucinous adenocarcinomas often show complex architecture, such as cribriform and complex papillary structures. The presence of signetring cells is an indication of infiltrative mucinous adenocarcinoma. Poorly differentiated (G3) mucinous adenocarcinoma demonstrates infiltrative invasion, with most having signet-ring cells. Signet-ring cells, characterized by prominent intracytoplasmic mucin displacing the nucleus, may infiltrate single cells or as aggregates, classified as grade G3 (poorly differentiated). Most G3 tumors are almost entirely composed of signet-ring cells, while a minority of cases are composed of mixed signet-ring cells and glandular structures. If cancer comprises ≤ 50% signet-ring cells, the WHO terminology is mucinous adenocarcinoma with signet-ring cells or mucinous adenocarcinoma, poorly differentiated. If the tumor contains > 50% signet-ring cells, the WHO terminology is signet-ring cell carcinoma. The microscopic findings of high-grade (G2 and G3) appendiceal mucinous adenocarcinomas are demonstrated in Fig. 7.
- Most mucinous adenocarcinomas have clinically aggressive behavior, infiltrating through the appendiceal wall (pT4), and frequently metastasize to the abdominal or pelvic peritoneum at diagnosis. After an appendectomy specimen, diagnosis of mucinous adenocarcinoma should result in subsequent right hemicolectomy to evaluate lymph node metastases.
- Ancillary tests of appendiceal mucinous neoplasms
- A limited number of studies have reported that the vast majority of KRAS mutations occurred in G1 and G2 tumors [4,31-35], and most KRAS mutations occur in 50% to 60% of Tis(LAMN) [4]. These data suggest that KRAS mutations are important in tumor initiation but may be less critical for aggressive highgrade tumor progression. GNAS mutations, known as essential in abundant mucin production, are also presented in G1 and G2 tumors but less commonly in G3 tumors [31-35]. The co-mutation of GNAS and KRAS is identified in between 65% to 85% of cases [35,36]. Other minor mutations, including MET, PIK3CA, FAT4, AKT1, SMAD2, JAK3, STK11, and RB1 have been identified [33,37]. However, BRAF mutation, microsatellite instability (MSI), and DNA mismatch repair protein deficiency are rarely seen in LAMN [10,38].
APPENDICEAL MUCINOUS ADENOCARCINOMA
- Pseudomyxoma peritonei should be avoided in pathologic diagnosis
- Pseudomyxoma peritonei, the clinical term for disseminated peritoneal mucinous disease, is a syndrome and applies to a neoplastic condition characterized by the grossly persistent accumulation of mucinous ascites in the peritoneal cavity [12]. The expansion of mucin within the abdominal cavity results from mucus following the normal flow of peritoneal fluid, redistribution of the mucin, and neoplastic cells [23]. Based on clinicopathological and immunohistochemical data, most cases are due to the perforation of AMNs [12,23,24,39,40]. Occasionally, mucinous neoplasms from other organs, including the colon, pancreas, ovary, and urachus, may also present with clinical appearances [12,23,24,39,40]. Immunohistochemical stains of cytokeratin 20 and CDX2 may be helpful in the diagnosis of the appendiceal origin (Supplementary Fig. S1). Clinically, abdominal discomfort, distention, intestinal obstruction, and often omental cake in which the omentum transforms into a firm mass can present within the intraabdominal cavity [4,9,41-43]. Given this, pseudomyxoma peritonei is a clinical term and used mainly by oncologists and radiologists; thus, it should not be used in histopathologic diagnosis [13]. The tendency of tumor cells from the appendix to produce abundant extracellular mucin shows slow infiltration into the peritoneum and underlying tissues with rare lymphovascular invasion relative to the overall tumor bulk [5]. Accurate diagnosis of AMNs and disseminated peritoneal mucinous disease is essential because management may include follow-up or radical treatment such as CRS-HIPEC [23].
- Diagnostic terminology and classification of disseminated peritoneal mucinous disease
- Ronnett et al. [9] classified disseminated peritoneal mucinous disease into three prognostically relevant categories: disseminated peritoneal adenomucinosis (DPAM), peritoneal mucinous carcinomatosis (PMCA), and PMCA with signet-ring cells [44]. However, the classification by Ronnett et al. [44] was not well established, leading to conflicting results. Given recent updates on diagnostic terminology, DPAM and PMCA are no longer recommended in pathology reports. Asare et al. [45] found that histologic grade was an independent predictor of survival in 25,992 patients with appendiceal cancer.
- The PSOGI consensus panel has presented the diagnostic terminology for disseminated peritoneal mucinous disease: lowgrade mucinous carcinoma peritonei, high-grade mucinous carcinoma peritonei, and high-grade mucinous carcinoma peritonei with signet-ring cells are equivalent to the alternative terminology of DPAM, PMCA, and PMCA with signet-ring cells, respectively. The AJCC 8th Cancer Staging Manual has proposed terminologies accompanying grading based on the criteria of Misdraji et al. [13,46,47]. Misdraji [46] used the diagnostic terms directly from the appendiceal neoplasm into the peritoneum: LAMN (G1) with peritoneal involvement, mucinous adenocarcinoma moderately differentiated (G2), and mucinous adenocarcinoma poorly differentiated (G3). In addition, acellular mucin is classified separately. Davison et al. [4] determined that the three-tiered grading is the most important predictive factor of overall survival in patients with stage IV AMNs.
- Grading system of disseminated peritoneal mucinous disease
- Low-grade (G1, well-differentiated) mucinous carcinoma peritonei, synonymous with a LAMN with peritoneal involvement, is defined as a mucinous neoplasm of low-grade cytology involving the peritoneum without infiltrative invasion or desmoplasia. Perineural and lymphovascular invasions are not seen. Typically, the neoplastic epithelial component accounts for less than 20% of the total mucin component. Low-grade (G1, well-differentiated) mucinous carcinoma peritonei almost always arises from a LAMN, so the AJCC 8th Cancer Staging Manual and Misdraji [46] proposed the term of LAMN with peritoneal involvement [13]. High-grade (G2, moderately differentiated) mucinous carcinoma peritonei, a synonym of moderately differentiated mucinous adenocarcinoma, is defined as a disseminated peritoneal mucinous tumor by the presence of high-grade cytology in the absence of signet-ring cells. The high-grade cytology criteria correspond to moderately differentiated (G2) appendiceal mucinous adenocarcinoma or HAMN. These can demonstrate diffuse high-grade cytology or display a mixture of low- and high-grade cytology areas. The cytologic features may be heterogeneous with low-grade areas with high-grade cytology, and this heterogeneity needs generous sampling for histologic evaluation. Cribriform complex growth and infiltrating tubular structures associated with stromal desmoplasia can be seen. Davison et al. [4] defined the high tumor cellularity as > 20% of the mucinous component. High-grade mucinous carcinoma peritonei with signet-ring cells (G3 tumor) shows high-grade cytology and invasive tumors with a signet-ring cell component, equivalent to poorly differentiated mucinous adenocarcinoma. High-grade (G2 and G3) disseminated peritoneal mucinous disease can present destructive infiltrative invasion into the extra-appendiceal spread, peritoneum, or other organs, frequently associated with perineural and lymphovascular invasion. The microscopic findings of a disseminated peritoneal mucinous tumor are presented in Fig. 8.
- Diagnostic difficulty of disseminated peritoneal mucinous disease
- The discordance grade may be presented between appendiceal and peritoneal tumors. The PSOGI consensus panel recommends that peritoneal grading should be used for staging purposes, as intraperitoneal grading is more likely to influence patient prognosis [3]. However, the CAP protocol advocates that more highgrade tumors between the appendix and peritoneum should be assigned to the tumor for staging. These diverse ideas can lead to diagnostic confusion and difficulty; thus, more advanced investigations concerning prognosis are required. Regarding the potential discrepancy between G2 and G3 tumors, Breadly and Carr [40] recommended that more than 10% of the signet-ring cell components should be identified in the diagnosis of G3 tumors. Because degenerating tumor cells or even histiocytes floating within mucin pools without destructive invasion may often exhibit signet-ring cell-like features (Fig. 8D). Sirintrapun et al. [48] reported that isolated signet cells within mucin pools are less prognostically significant than those found in invading tissue. We surveyed the best criterion of a focal lesion with signet-ring cells at the workshop, and the results are shown in Supplementary Data S2.
- The pathologic grade is an independent prognostic factor in patients with stage IV mucinous appendiceal neoplasms. Patients with high-grade (G2 or G3) disseminated peritoneal mucinous tumors have significantly worse survival than patients with low-grade (G1) tumors. Therefore, for high-grade disseminated peritoneal mucinous tumors, systemic chemotherapy followed by the option of CRS-HIPEC is recommended according to the therapeutic response [4,45,49-52].
- Biologic behavior codes of primary and metastatic tumors
- The ICD-O3 code is 8480/6 in secondary disseminated peritoneal mucinous disease regardless of the grading; however, ICD-O3 is 8480/3 in the primary or unknown origin of disseminated peritoneal disease [53]. The histologic grade, behavior codes, diagnostic terminologies, and criteria for acellular mucin and disseminated peritoneal mucinous disease are summarized in Table 4. In addition, the checklist of standard data elements of the appendiceal epithelial tumors, including disseminated peritoneal mucinous disease, is presented in Table 5.
DISSEMINATED PERITONEAL MUCINOUS DISEASE
- Non-mucinous adenocarcinoma and adenoma
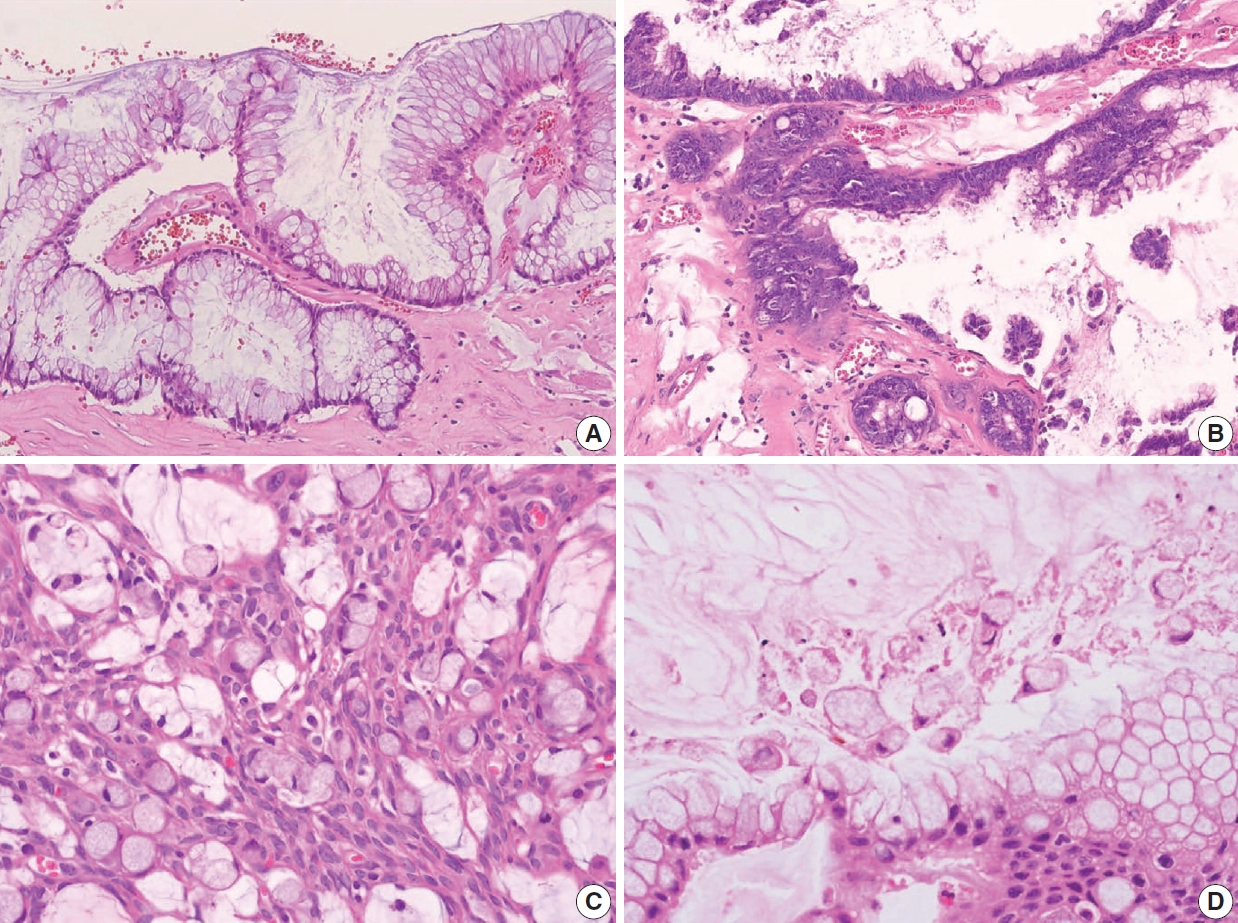
- Non-mucinous adenocarcinoma is less common than mucinous adenocarcinoma [1]. Uemura et al. [54] reported that nonmucinous adenocarcinoma is biologically distinct from the mucinous subtype and shows mostly moderate to poorly differentiated with frequent peritoneal metastasis. The histopathologic findings and tumor grading of non-mucinous adenocarcinoma are the same as conventional colorectal adenocarcinoma [1]. Non-mucinous adenocarcinoma demonstrates a high level of MSI. The frequency of KRAS mutation is similar in both colorectal adenocarcinomas and appendiceal non-mucinous adenocarcinomas, but the molecular pathogenesis is different [38,55,56]. The PSOGI panel prefers to confine “adenoma” to lesions that resemble conventional adenomas of the colorectum limited to the mucosa and intact muscularis mucosae without luminal dilatation or expansile growth pattern [12,23]. Based on this, appendiceal adenomas are uncommon, and many neoplasms that were previously reported as adenomas are now classified as LAMN or serrated lesions. For example, a villous lesion showing serration with conventional dysplasia and intact muscularis mucosae without pushing invasion should be called a serrated dysplasia rather than a villous adenoma. The WHO 5th Digestive System Tumors and CAP protocol recommend that appendiceal adenoma should be restricted to the precursor lesion of non-mucinous adenocarcinoma [1,14,22]. The histologic findings of non-mucinous adenocarcinoma arising in adenoma are presented in Fig. 9A–C.
- Goblet cell adenocarcinoma
- Goblet cell adenocarcinoma can occur almost exclusively in the appendix and is sometimes difficult to differentiate from AMNs and non-mucinous appendiceal tumors. Goblet cell adenocarcinoma, previously called goblet cell carcinoid or mucinous carcinoid, is a rare and aggressive tumor [1]. The tumor components are goblet-like cells and variable numbers of neuroendocrine cells and Paneth-like cells, and it is characteristically arranged as tubules similar to intestinal crypts [1]. The classic low-grade tumor displays tubular or clustered growth and small groups of cohesive goblet cells, whereas the high-grade tumor shows infiltrating tumor cells, convoluted anastomosing tubules, cribriform masses, irregular solid pattern, and large aggregates of goblet cells or signet-ring cells [57]. Nonaka et al. [58] found that neuroendocrine cells are inconsistent and not essential in the tumor component. For this reason, pathologists must find at least a focal lesion of classical low-grade tumor component to diagnose as a goblet cell adenocarcinoma.
- Several classification and grading systems have been described. Tang et al. [59] classified goblet cell adenocarcinomas into three groups based on the histopathologic features: group A (typical goblet cell carcinoid), group B (adenocarcinoma ex-goblet cell carcinoid, signet-ring cell type), and group C (adenocarcinoma ex-goblet cell carcinoid, poorly differentiated type). Recently, the WHO 5th Digestive System Tumors classified goblet cell adenocarcinomas into a three-tiered grading system, based on the proportion of the tumor cells that consists of low-grade and highgrade patterns as follows: grade 1, > 75% of low-grade pattern or < 25% of high-grade pattern; grade 2, 50%–75% of low-grade pattern or 25%–50% of high-grade pattern; and grade 3, < 50% of low-grade pattern or > 50% of the high-grade pattern [1,60]. Nonaka et al. [58] reported that the high-grade component percentage was correlated with cancer-specific survival. The histological and immunohistochemical findings of goblet cell adenocarcinoma are presented in Fig. 9D–F. The pathogenesis remains entirely elusive. It is generally believed that it is derived from the pluripotent stem cells at the crypt base that can undergo dual glandular and neuroendocrine differentiation.
OTHER APPENDICEAL EPITHELIAL TUMORS
- The GPSG-KSP presents a “Standardization of the Pathologic Diagnosis of the Appendiceal Mucinous Neoplasm.” This article focuses on the diagnostic criteria, terminology, grading, staging,biologic behaviors, treatment, and prognosis of AMNs and disseminated peritoneal mucinous disease. In addition, we propose a checklist of standard data elements of the appendiceal epithelial neoplasms. We hope that the present article will lead to the standardization of the pathologic diagnosis of AMNs and disseminated peritoneal mucinous disease and improvement in the communication between pathologists and between pathologists and clinicians.
CONCLUSION
Supplementary Materials
Supplementary Data S1.
Supplementary Data S2.
Supplementary Fig. S1.
Supplementary Information
The Data Supplement is available with this article at https://doi.org/10.4132/jptm.2021.05.28.
Ethics Statement
Not applicable.
Availability of Data and Material
Data sharing not applicable to this article as no datasets were generated or analyzed during the study.
Code Availability
Not applicable.
Author Contributions
Conceptualization: MSC, JMK. Project administration: DWK, YNK, BHK, JYK, JMK, JK, HSK, DYP, HYP, JHS, SJL, WL, HSL, MSC, HKC, HJC, MYC, SYJ, YC, HSH. Supervision: BHK, JMK, JK, MSC, HJC. Writing— original draft: DWK. Writing—review & editing: JMK. Approval of final manuscript: all authors.
Conflicts of Interest
H.S.L., a contributing editor of the Journal of Pathology and Translational Medicine, was not involved in the editorial evaluation or decision to publish this article. All remaining authors have declared no conflicts of interest.
Funding Statement
This research was supported by The Korean Society of Pathologists Grant No. 2018.
Acknowledgments
| Classification | Gradinga | ICD-O3 | KCD-8 | Diagnostic criteria and microscopic features [3,12] | |
|---|---|---|---|---|---|
| Low-grade appendiceal mucinous neoplasm | Low/G1 | 8480/1 | D37.3 | Mucinous neoplasm with low-grade cytology and any of the following (by PSOGI criteria) | |
| Loss of muscularis mucosae | |||||
| Fibrosis of submucosa | |||||
| “Pushing invasion” (expansile or diverticulum-like growth) | |||||
| Dissection of acellular mucin in wall | |||||
| Undulating or flattened epithelial growth | |||||
| Rupture of appendix | |||||
| Mucin and/or cells outside appendix | |||||
| High-grade appendiceal mucinous neoplasm | High/G2 | 8480/2 | D01.7 | Mucinous neoplasm with the architectural features of LAMN and no infiltrative invasion, but with high-grade cytologic atypia | |
| Mucinous adenocarcinoma | High/G2 | 8480/3 | C18.1 | Malignant glandular neoplasm, comprising of > 50% extracellular mucin, characterized by infiltrative invasion. The pattern of infiltrative invasion as follows: Infiltrative glands, or single infiltrative tumor cells associated with extracellular mucin and desmoplastic stroma | |
| Small dissecting mucin pools containing floating nests, glands, or single neoplastic cells | |||||
| Mucinous adenocarcinoma with signet-ring cells | High/G3 | 8480/3 | C18.1 | Mucinous adenocarcinoma, comprising of ≤ 50% signet-ring cells | |
| Signet-ring cell carcinoma | High/G3 | 8490/3 | C18.1 | Adenocarcinoma, comprising up to > 50% signet-ring cells | |
ICD-O3, International Classification of Diseases for Oncology, 3rd edition; KCD-8, Korean Classification of Disease, 8th edition; PSOGI, Peritoneal Surface Oncology Group International; LAMN, low-grade appendiceal mucinous neoplasm.
aGrading divide into low- and high-grade by the two-tier system and G1 to G3 by three-tiered system.
| Lesion | Grade |
Behavior codes |
Terminology |
Criteria | Treatment | ||
|---|---|---|---|---|---|---|---|
| ICD-O3 | KCD-8 | PSOGI/Carr [12] | AJCC/Misdraji [10] | ||||
| Mucin without tumor cells | - | 8480/6 | C78.6 | Acellular mucin | Acellular mucin | Mucin within the peritoneal cavity without neoplastic epithelial cells | CRS-HIPEC |
| Mucinous tumor with low-grade histologic features | G1 | 8480/6 | C78.6 | Low-grade mucinous carcinoma peritonei | LAMN with peritoneal involvement | Scanty (≤ 20% cellularity) | CRS-HIPEC |
| Strips, gland-like or small clusters | |||||||
| Low-grade cytological atypia | |||||||
| Not more than occasional mitosis | |||||||
| Pushing invasion | |||||||
| Mucinous tumor with high-grade histologic features | G2 | 8480/6 | C78.6 | High-grade mucinous carcinoma peritonei | Mucinous adenocarcinoma, moderately differentiated | More cellular (> 20% cellularity) | Systemic chemotherapy ± CRS-HIPEC |
| Cribriform growth | |||||||
| Mixed but mostly high-grade cytological atypia | |||||||
| Numerous mitoses | |||||||
| Destructive infiltrative invasion | |||||||
| Mucinous tumor with signet-ring cells | G3 | 8480/6 | C78.6 | High-grade mucinous carcinoma peritonei with signet-ring cells | Mucinous adenocarcinoma, poorly differentiated | Any lesion with signet-ring cells (degenerating cells within mucin that mimic signet-ring cells should be discounted) | Systemic chemotherapy ± CRS-HIPEC |
Note that ICD-O3 and KCD-8 codes of primary or unknown origin disseminated peritoneal mucinous disease are 8480/3 and C80.0, respectively.
ICD-O3, International Classification of Diseases for Oncology, 3rd edition; KCD-8, Korean Classification of Disease, 8th edition; PSOGI, Peritoneal Surface Oncology Group International; AJCC, American Joint Committee on Cancer; CRS-HIPEC, cytoreductive surgery with hyperthermic intraperitoneal chemotherapy; LAMN, low-grade appendiceal mucinous neoplasm.
| Specimen type | ||
|---|---|---|
| □ Appendectomy | ||
| □ Appendectomy and right colectomy | ||
| □ Other: (specify: ) | ||
| Tumor size | ||
| × × cm | ||
| Tumor site | ||
| □ Proximal half of appendix | ||
| □ Distal half of appendix | ||
| □ Diffusely involving appendix | ||
| □ Other: (specify: ) | ||
| Histologic types (WHO 5th Digestive System Tumors) [1] | ||
| □ Low-grade appendiceal mucinous neoplasm (LAMN) | ||
| □ High-grade appendiceal mucinous neoplasm (HAMN) | ||
| □ Mucinous adenocarcinoma | ||
| □ Non-mucinous adenocarcinoma | ||
| □ Signet-ring cell carcinoma | ||
| □ Goblet cell adenocarcinoma | ||
| □ Large cell neuroendocrine carcinoma | ||
| □ Small cell neuroendocrine carcinoma | ||
| □ Mixed neuroendocrine-non-neuroendocrine carcinoma (MiNEN) | ||
| □ Medullary carcinoma | ||
| □ Adenosquamous carcinoma | ||
| □ Undifferentiated carcinoma | ||
| □ Other histologic type not listed (specify: ) | ||
| Histologic grade | ||
| □ G1: Well differentiated | ||
| □ G2: Moderately differentiated | ||
| □ G3: Poorly differentiated | ||
| □ GX: Cannot be assessed | ||
| □ Other: (specify: ) | ||
| Disseminated peritoneal mucinous disease | ||
| □ Not identified | ||
| □ Present | ||
| □ Low-grade mucinous carcinoma peritonei/LAMN (G1) with peritoneal involvement | ||
| □ High-grade mucinous carcinoma peritonei/Mucinous adenocarcinoma, moderately differentiated (G2) | ||
| □ High-grade mucinous carcinoma peritonei with signet-ring cells / Mucinous adenocarcinoma, poorly differentiated (G3) | ||
| Resection margins including proximal and mesenteric margins | ||
| □ Cannot be assessed | ||
| □ Uninvolved by tumor | ||
| □ Distance of tumor from the closest margin: mm | ||
| □ Involved by tumor | ||
| □ Involved by invasive carcinoma (□ Proximal margin, □ Mesenteric margin) | ||
| □ Involved by appendiceal mucinous neoplasm (□ Proximal margin, □ Mesenteric margin) | ||
| □ Involved by acellular mucin (□ Proximal margin, □ Mesenteric margin) | ||
| Lympho-vascular invasion | ||
| □ Not identified □ Present □ Cannot be determined | ||
| Perineural invasion | ||
| □ Not identified □ Present □ Cannot be determined | ||
| Tumor deposits | ||
| □ Not identified □ Present □ Cannot be determined | ||
| Regional lymph nodes | ||
| □ No lymph nodes submitted or found | ||
| □ Total number of lymph nodes examined: | ||
| □ Number of lymph nodes involved with metastases: | ||
| Pathologic stage classification (pTNM, AJCC 8th edition) [13] | ||
| TNM Descriptors: □ m (multiple) □ r (recurrent) □ y (post-neoadjuvant therapy) | ||
| Primary tumor (pT) | ||
| □ pTX: Primary tumor cannot be assessed | ||
| □ pTX0: No evidence of primary tumor | ||
| □ pTis: Carcinoma in situ (high-grade dysplasia); intraepithelial carcinoma; invasion of lamina propria (intramucosal carcinoma) | ||
| □ pTis(LAMN): LAMN confined to the appendix (defined as involvement by acellular mucin or mucinous epithelium that may extend into the muscularis propria) | ||
| □ pT1: Tumor invades submucosa (does not apply to LAMN) | ||
| □ pT2: Tumor invades muscularis propria (does not apply to LAMN) | ||
| □ pT3: Tumor invades through the muscularis propria into the subserosa or mesoappendix | ||
| □ pT4: Tumor invades the visceral peritoneum, including the acellular mucin or mucinous epithelium involving the serosa of the appendix or mesoappendix, and/or directly invades adjacent organs or structures | ||
| □ pT4a: Tumor invades the visceral peritoneum, including the acellular mucin or mucinous epithelium involving the serosa of appendix or serosa of mesoappendix | ||
| □ pT4b: Tumor directly invades other organs or structures | ||
| Regional lymph nodes (pN) | ||
| □ pNX: Regional lymph nodes cannot be assessed | ||
| □ pN0: No regional nodal metastasis | ||
| □ pN1: One to three regional lymph nodes are positive (tumor in lymph nodes measuring ≥ 0.2 mm), or any number of tumor deposits is present, and all identifiable lymph nodes are negative | ||
| □ pN1a: One regional lymph node is positive | ||
| □ pN1b: Two or three regional lymph nodes are positive | ||
| □ pN1c: No regional lymph node metastasis, but tumor deposits in the subserosa or mesentery | ||
| □ pN2: Metastasis in 4 or more regional nodes | ||
| Distant metastasis (pM) | ||
| □ Not applicable | ||
| □ pM1: Distant metastasis | ||
| □ pM1a: Intraperitoneal acellular mucin only | ||
| □ pM1b: Intraperitoneal metastasis only, including mucinous epithelium | ||
| □ pM1c: Non-peritoneal metastasis | ||
| Additional pathologic findings or pre-existing lesions | ||
| □ None | ||
| □ Other: (specify: ) | ||
| Ancillary studies | ||
| □ Specify: | ||
- 1. Misdraji J, Carr NJ, Pai RK. Appendiceal mucinous neoplasm. In: Who Classification of Tumours Editorial Board, ed. WHO classification of tumours: digestive system tumours. 5th ed. Lyon: International Agency for Research on Cancer, 2019; 135-55.
- 2. Misdraji J. Appendiceal mucinous neoplasms: controversial issues. Arch Pathol Lab Med 2010; 134: 864-70. ArticlePubMedPDF
- 3. Valasek MA, Pai RK. An update on the diagnosis, grading, and staging of appendiceal mucinous neoplasms. Adv Anat Pathol 2018; 25: 38-60. ArticlePubMed
- 4. Davison JM, Choudry HA, Pingpank JF, et al. Clinicopathologic and molecular analysis of disseminated appendiceal mucinous neoplasms: identification of factors predicting survival and proposed criteria for a three-tiered assessment of tumor grade. Mod Pathol 2014; 27: 1521-39. ArticlePubMedPDF
- 5. Pai RK, Longacre TA. Pseudomyxoma peritonei syndrome: classification of appendiceal mucinous tumours. Cancer Treat Res 2007; 134: 71-107. ArticlePubMed
- 6. Ronnett BM, Kurman RJ, Zahn CM, et al. Pseudomyxoma peritonei in women: a clinicopathologic analysis of 30 cases with emphasis on site of origin, prognosis, and relationship to ovarian mucinous tumors of low malignant potential. Hum Pathol 1995; 26: 509-24. ArticlePubMed
- 7. Misdraji J, Yantiss RK, Graeme-Cook FM, Balis UJ, Young RH. Appendiceal mucinous neoplasms: a clinicopathologic analysis of 107 cases. Am J Surg Pathol 2003; 27: 1089-103. PubMed
- 8. Lamps LW, Gray GF Jr, Dilday BR, Washington MK. The coexistence of low-grade mucinous neoplasms of the appendix and appendiceal diverticula: a possible role in the pathogenesis of pseudomyxoma peritonei. Mod Pathol 2000; 13: 495-501. ArticlePubMedPDF
- 9. Ronnett BM, Zahn CM, Kurman RJ, Kass ME, Sugarbaker PH, Shmookler BM. Disseminated peritoneal adenomucinosis and peritoneal mucinous carcinomatosis: a clinicopathologic analysis of 109 cases with emphasis on distinguishing pathologic features, site of origin, prognosis, and relationship to “pseudomyxoma peritonei”. Am J Surg Pathol 1995; 19: 1390-408. PubMed
- 10. Misdraji J, Burgart LJ, Lauwers GY. Defective mismatch repair in the pathogenesis of low-grade appendiceal mucinous neoplasms and adenocarcinomas. Mod Pathol 2004; 17: 1447-54. ArticlePubMedPDF
- 11. Yantiss RK, Shia J, Klimstra DS, Hahn HP, Odze RD, Misdraji J. Prognostic significance of localized extra-appendiceal mucin deposition in appendiceal mucinous neoplasms. Am J Surg Pathol 2009; 33: 248-55. ArticlePubMed
- 12. Carr NJ, Cecil TD, Mohamed F, et al. A consensus for classification and pathologic reporting of pseudomyxoma peritonei and associated appendiceal neoplasia: the results of the Peritoneal Surface Oncology Group International (PSOGI) modified Delphi process. Am J Surg Pathol 2016; 40: 14-26. PubMed
- 13. Amin MB, Edge SB, Greene FL, et al. AJCC cancer staging manual. 8th. New York: Springer, 2017.
- 14. Nagtegaal ID, Odze RD, Klimstra D, et al. The 2019 WHO classification of tumours of the digestive system. Histopathology 2020; 76: 182-8. ArticlePubMedPDF
- 15. Kim WH, Park CK, Kim YB, et al. A standardized pathology report for gastric cancer. Korean J Pathol 2005; 39: 106-13.
- 16. Chang HJ, Park CK, Kim WH, et al. A standardized pathology report for colorectal cancer. Korean J Pathol 2006; 40: 193-203.
- 17. Cho MY, Kang YK, Kim KM, et al. Proposal for creating a guideline for cancer registration of the gastrointestinal tumors (I). Korean J Pathol 2008; 42: 140-50.
- 18. The Gastrointestinal Pathology Study Group of Korean Society of Pathologists, Cho MY, Sohn JH, et al. Proposal for a standardized pathology report of gastroenteropancreatic neuroendocrine tumors: prognostic significance of pathological parameters. Korean J Pathol 2013; 47: 227-37. ArticlePubMedPMC
- 19. Gastrointestinal Pathology Study Group of Korean Society of Pathologists, Cho MY, Kim JM, et al. Current trends of the incidence and pathological diagnosis of gastroenteropancreatic neuroendocrine tumors (GEP-NETs) in Korea 2000-2009: multicenter study. Cancer Res Treat 2012; 44: 157-65. ArticlePubMedPMC
- 20. Kang YK, Kim KM, Sohn T, et al. Clinical practice guideline for accurate diagnosis and effective treatment of gastrointestinal stromal tumor in Korea. J Korean Med Sci 2010; 25: 1543-52. ArticlePubMedPMC
- 21. Jung ES, Kang YK, Cho MY, et al. Update on the proposal for creating a guideline for cancer registration of the gastrointestinal tumors (I-2). Korean J Pathol 2012; 46: 443-53. ArticlePubMedPMC
- 22. Misdraji J, Oliva E, Goldblum JR, Lauwers GY, Compton CC; Members of the Cancer Committee; College of American Pathologists. Protocol for the examination of specimens from patients with invasive carcinomas of the appendix. Arch Pathol Lab Med 2006; 130: 1433-9. ArticlePubMedPDF
- 23. Carr NJ, Bibeau F, Bradley RF, et al. The histopathological classification, diagnosis and differential diagnosis of mucinous appendiceal neoplasms, appendiceal adenocarcinomas and pseudomyxoma peritonei. Histopathology 2017; 71: 847-58. ArticlePubMedPDF
- 24. Carr NJ, Finch J, Ilesley IC, et al. Pathology and prognosis in pseudomyxoma peritonei: a review of 274 cases. J Clin Pathol 2012; 65: 919-23. ArticlePubMed
- 25. Ramaswamy V. Pathology of mucinous appendiceal tumors and pseudomyxoma peritonei. Indian J Surg Oncol 2016; 7: 258-67. ArticlePubMedPMCPDF
- 26. Altieri ML, Piozzi GN, Salvatori P, Mirra M, Piccolo G, Olivari N. Appendiceal diverticulitis, a rare relevant pathology: presentation of a case report and review of the literature. Int J Surg Case Rep 2017; 33: 31-4. ArticlePubMedPMC
- 27. Hsu M, Young RH, Misdraji J. Ruptured appendiceal diverticula mimicking low-grade appendiceal mucinous neoplasms. Am J Surg Pathol 2009; 33: 1515-21. ArticlePubMed
- 28. Hara K, Saito T, Hayashi T, et al. A mutation spectrum that includes GNAS, KRAS and TP53 may be shared by mucinous neoplasms of the appendix. Pathol Res Pract 2015; 211: 657-64. ArticlePubMed
- 29. Milovanov V, Sardi A, Studeman K, Nieroda C, Sittig M, Gushchin V. The 7th edition of the AJCC staging classification correlates with biologic behavior of mucinous appendiceal tumor with peritoneal metastases treated with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy (CRS/HIPEC). Ann Surg Oncol 2016; 23: 1928-33. ArticlePubMedPDF
- 30. Pai RK, Beck AH, Norton JA, Longacre TA. Appendiceal mucinous neoplasms: clinicopathologic study of 116 cases with analysis of factors predicting recurrence. Am J Surg Pathol 2009; 33: 1425-39. PubMed
- 31. Pai RK, Hartman DJ, Gonzalo DH, et al. Serrated lesions of the appendix frequently harbor KRAS mutations and not BRAF mutations indicating a distinctly different serrated neoplastic pathway in the appendix. Hum Pathol 2014; 45: 227-35. ArticlePubMed
- 32. Noguchi R, Yano H, Gohda Y, et al. Molecular profiles of high-grade and low-grade pseudomyxoma peritonei. Cancer Med 2015; 4: 1809-16. ArticlePubMedPMCPDF
- 33. Liu X, Mody K, de Abreu FB, et al. Molecular profiling of appendiceal epithelial tumors using massively parallel sequencing to identify somatic mutations. Clin Chem 2014; 60: 1004-11. ArticlePubMedPDF
- 34. Nummela P, Saarinen L, Thiel A, et al. Genomic profile of pseudomyxoma peritonei analyzed using next-generation sequencing and immunohistochemistry. Int J Cancer 2015; 136: E282-9. ArticlePubMed
- 35. Gleeson EM, Feldman R, Mapow BL, et al. Appendix-derived pseudomyxoma peritonei (PMP): molecular profiling toward treatment of a rare malignancy. Am J Clin Oncol 2018; 41: 777-83. PubMed
- 36. Singhi AD, Davison JM, Choudry HA, et al. GNAS is frequently mutated in both low-grade and high-grade disseminated appendiceal mucinous neoplasms but does not affect survival. Hum Pathol 2014; 45: 1737-43. ArticlePubMed
- 37. Pengelly RJ, Rowaiye B, Pickard K, et al. Analysis of mutation and loss of heterozygosity by whole-exome sequencing yields insights into pseudomyxoma peritonei. J Mol Diagn 2018; 20: 635-42. ArticlePubMed
- 38. Zauber P, Berman E, Marotta S, Sabbath-Solitare M, Bishop T. Kiras gene mutations are invariably present in low-grade mucinous tumors of the vermiform appendix. Scand J Gastroenterol 2011; 46: 869-74. ArticlePubMed
- 39. Bignell M, Carr NJ, Mohamed F. Pathophysiology and classification of pseudomyxoma peritonei. Pleura Peritoneum 2016; 1: 3-13. ArticlePubMedPMC
- 40. Bradley RF, Carr NJ. Pseudomyxoma peritonei: pathology, a historical overview, and proposal for unified nomenclature and updated grading. AJSP Rev Rep 2019; 24: 88-93. Article
- 41. Ihemelandu C, Sugarbaker PH. Clinicopathologic and prognostic features in patients with peritoneal metastasis from mucinous adenocarcinoma, adenocarcinoma with signet ring cells, and adenocarcinoid of the appendix treated with cytoreductive surgery and perioperative intraperitoneal chemotherapy. Ann Surg Oncol 2016; 23: 1474-80. ArticlePubMedPDF
- 42. Morano WF, Gleeson EM, Sullivan SH, et al. Clinicopathological features and management of appendiceal mucoceles: a systematic review. Am Surg 2018; 84: 273-81. ArticlePubMedPDF
- 43. LaFramboise WA, Pai RK, Petrosko P, et al. Discrimination of lowand high-grade appendiceal mucinous neoplasms by targeted sequencing of cancer-related variants. Mod Pathol 2019; 32: 1197-209. ArticlePubMedPDF
- 44. Ronnett BM, Yan H, Kurman RJ, Shmookler BM, Wu L, Sugarbaker PH. Patients with pseudomyxoma peritonei associated with disseminated peritoneal adenomucinosis have a significantly more favorable prognosis than patients with peritoneal mucinous carcinomatosis. Cancer 2001; 92: 85-91. ArticlePubMed
- 45. Asare EA, Compton CC, Hanna NN, et al. The impact of stage, grade, and mucinous histology on the efficacy of systemic chemotherapy in adenocarcinomas of the appendix: analysis of the National Cancer Data Base. Cancer 2016; 122: 213-21. ArticlePubMed
- 46. Misdraji J. Mucinous epithelial neoplasms of the appendix and pseudomyxoma peritonei. Mod Pathol 2015; 28 Suppl 1: S67-79. ArticlePubMedPDF
- 47. Misdraji J, Lauwers GY, Irving JA, Batts KP, Young RH. Appendiceal or cecal endometriosis with intestinal metaplasia: a potential mimic of appendiceal mucinous neoplasms. Am J Surg Pathol 2014; 38: 698-705. PubMed
- 48. Sirintrapun SJ, Blackham AU, Russell G, et al. Significance of signet ring cells in high-grade mucinous adenocarcinoma of the peritoneum from appendiceal origin. Hum Pathol 2014; 45: 1597-604. ArticlePubMedPMC
- 49. Baratti D, Kusamura S, Milione M, Bruno F, Guaglio M, Deraco M. Validation of the recent PSOGI pathological classification of pseudomyxoma peritonei in a single-center series of 265 patients treated by cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Ann Surg Oncol 2018; 25: 404-13. ArticlePubMedPDF
- 50. Narasimhan V, Wilson K, Britto M, et al. Outcomes following cytoreduction and HIPEC for pseudomyxoma peritonei: 10-year experience. J Gastrointest Surg 2020; 24: 899-906. ArticlePubMedPDF
- 51. Shaib WL, Goodman M, Chen Z, et al. Incidence and survival of appendiceal mucinous neoplasms: a SEER analysis. Am J Clin Oncol 2017; 40: 569-73. PubMed
- 52. Kunduz E, Bektasoglu HK, Unver N, Aydogan C, Timocin G, Destek S. Analysis of appendiceal neoplasms on 3544 appendectomy specimens for acute appendicitis: retrospective cohort study of a single institution. Med Sci Monit 2018; 24: 4421-6. ArticlePubMedPMC
- 53. Ball HL. Conducting online surveys. J Hum Lact 2019; 35: 413-7. ArticlePubMedPDF
- 54. Uemura M, Qiao W, Fournier K, et al. Retrospective study of nonmucinous appendiceal adenocarcinomas: role of systemic chemotherapy and cytoreductive surgery. BMC Cancer 2017; 17: 331.ArticlePubMedPMCPDF
- 55. Kabbani W, Houlihan PS, Luthra R, Hamilton SR, Rashid A. Mucinous and nonmucinous appendiceal adenocarcinomas: different clinicopathological features but similar genetic alterations. Mod Pathol 2002; 15: 599-605. ArticlePubMedPDF
- 56. Matson DR, Xu J, Huffman L, et al. KRAS and GNAS co-mutation in metastatic low-grade appendiceal mucinous neoplasm (LAMN) to the ovaries: a practical role for next-generation sequencing. Am J Case Rep 2017; 18: 558-62. ArticlePubMedPMC
- 57. Yozu M, Johncilla ME, Srivastava A, et al. Histologic and outcome study supports reclassifying appendiceal goblet cell carcinoids as goblet cell adenocarcinomas, and grading and staging similarly to colonic adenocarcinomas. Am J Surg Pathol 2018; 42: 898-910. ArticlePubMed
- 58. Nonaka D, Papaxoinis G, Lamarca A, Fulford P, Valle J, Chakrabarty B. A study of appendiceal crypt cell adenocarcinoma (so-called goblet cell carcinoid and its related adenocarcinoma). Hum Pathol 2018; 72: 18-27. ArticlePubMed
- 59. Tang LH, Shia J, Soslow RA, et al. Pathologic classification and clinical behavior of the spectrum of goblet cell carcinoid tumors of the appendix. Am J Surg Pathol 2008; 32: 1429-43. ArticlePubMed
- 60. Taggart MW, Abraham SC, Overman MJ, Mansfield PF, Rashid A. Goblet cell carcinoid tumor, mixed goblet cell carcinoid-adenocarcinoma, and adenocarcinoma of the appendix: comparison of clinicopathologic features and prognosis. Arch Pathol Lab Med 2015; 139: 782-90. ArticlePubMedPDF
REFERENCES
Figure & Data
References
Citations

- Intrasplenic metastasis of appendiceal low-grade mucinous neoplasm – A case report and review of the literature
P. Meister, J. Rawitzer, M. Reschke, H.A. Baba, U. Neumann, M. Kaths
Current Problems in Cancer: Case Reports.2025; 18: 100364. CrossRef - Lower Gastrointestinal Bleeding Secondary to Appendiceal Mucinous Neoplasm: A Report of Two Cases and a Review of the Literature
Jesús Omar Soto Llanes, Samanta Kin Dosal Limón, Ana Jimena Iberri Jaime, Mario Zambrano Lara, Billy Jiménez Bobadilla
Cureus.2024;[Epub] CrossRef - Predicting Survival in Mucinous Adenocarcinoma of the Appendix: Demographics, Disease Presentation, and Treatment Methodology
Paul H. McClelland, Stephanie N. Gregory, Shirley K. Nah, Jonathan M. Hernandez, Jeremy L. Davis, Andrew M. Blakely
Annals of Surgical Oncology.2024; 31(9): 6237. CrossRef - Histoséminaire biopsies péritonéales tumorales. Néoplasies mucineuses appendiculaires
Peggy Dartigues
Annales de Pathologie.2024; 44(4): 274. CrossRef - Histoséminaire biopsies péritonéales tumorales. Cas no 2
Peggy Dartigues
Annales de Pathologie.2024; 44(4): 245. CrossRef - A Case of Low-Grade Appendiceal Mucinous Neoplasm: The Role of Preoperative Imaging and Surgical Technique in Achieving Favorable Outcomes
Daniel A Meza-Martinez, Yeudiel Suro Santos, Samantha J Andrade-Ordoñez, Julio A Palomino-Payan, Brando J Fematt-Rodriguez
Cureus.2024;[Epub] CrossRef - Incidental Appendiceal Mucinous Neoplasm Found During Appendectomy in a 15-Year-Old Patient: A Case Report
Fernando Aguilar-Ruiz, Kevin Joseph Fuentes-Calvo, Sara Fernanda Arechavala-Lopez, Irving Fuentes-Calvo, Luis F Arias-Ruiz
Cureus.2024;[Epub] CrossRef - Uncovering the Hidden Threat: Ileocolic Intussusception in an Adult With Appendicular Tumor
Mrunal Panchal, Shishir Kumar, Khushboo Jha, Kaushik Saha, Abhijit Kundu
Cureus.2024;[Epub] CrossRef - Low-Grade Appendiceal Mucinous Neoplasm vs. Appendiceal Diverticulum: Distinction with Histomorphologic Features
Cevriye Cansiz Ersöz, Siyar Ersöz, Berna Savas, Arzu Ensari
Gastrointestinal Disorders.2024; 6(4): 905. CrossRef - Appendiceal perforation secondary to endometriosis with intestinal metaplasia: A case report
Minghua Wang, Jing Liu, Boxin Hu, Simin Wang, Ping Xie, Ping Li
Experimental and Therapeutic Medicine.2023;[Epub] CrossRef - Primary and secondary tumors of the peritoneum: key imaging features and differential diagnosis with surgical and pathological correlation
Javier Miguez González, Francesc Calaf Forn, Laura Pelegrí Martínez, Pilar Lozano Arranz, Rafael Oliveira Caiafa, Jordi Català Forteza, Lina Maria Palacio Arteaga, Ferrán Losa Gaspà, Isabel Ramos Bernadó, Pedro Barrios Sánchez, Juan Ramón Ayuso Colella
Insights into Imaging.2023;[Epub] CrossRef - Muzinöse Tumoren des Peritoneums
Anne Kristin Fischer, Andrea Tannapfel, Alexander Quaas
Die Chirurgie.2023; 94(10): 823. CrossRef - Landscape of Genetic Mutations in Appendiceal Cancers
Marian Constantin, Cristina Mătanie, Livia Petrescu, Alexandra Bolocan, Octavian Andronic, Coralia Bleotu, Mihaela Magdalena Mitache, Sorin Tudorache, Corneliu Ovidiu Vrancianu
Cancers.2023; 15(14): 3591. CrossRef - Delivery of an Incidental Appendiceal Mucinous Neoplasm
Madison Bowles, Jessica Y Ng, Hajir Nabi
Cureus.2022;[Epub] CrossRef - Unearthing novel fusions as therapeutic targets in solid tumors using targeted RNA sequencing
Sungbin An, Hyun Hee Koh, Eun Sol Chang, Juyoung Choi, Ji-Young Song, Mi-Sook Lee, Yoon-La Choi
Frontiers in Oncology.2022;[Epub] CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
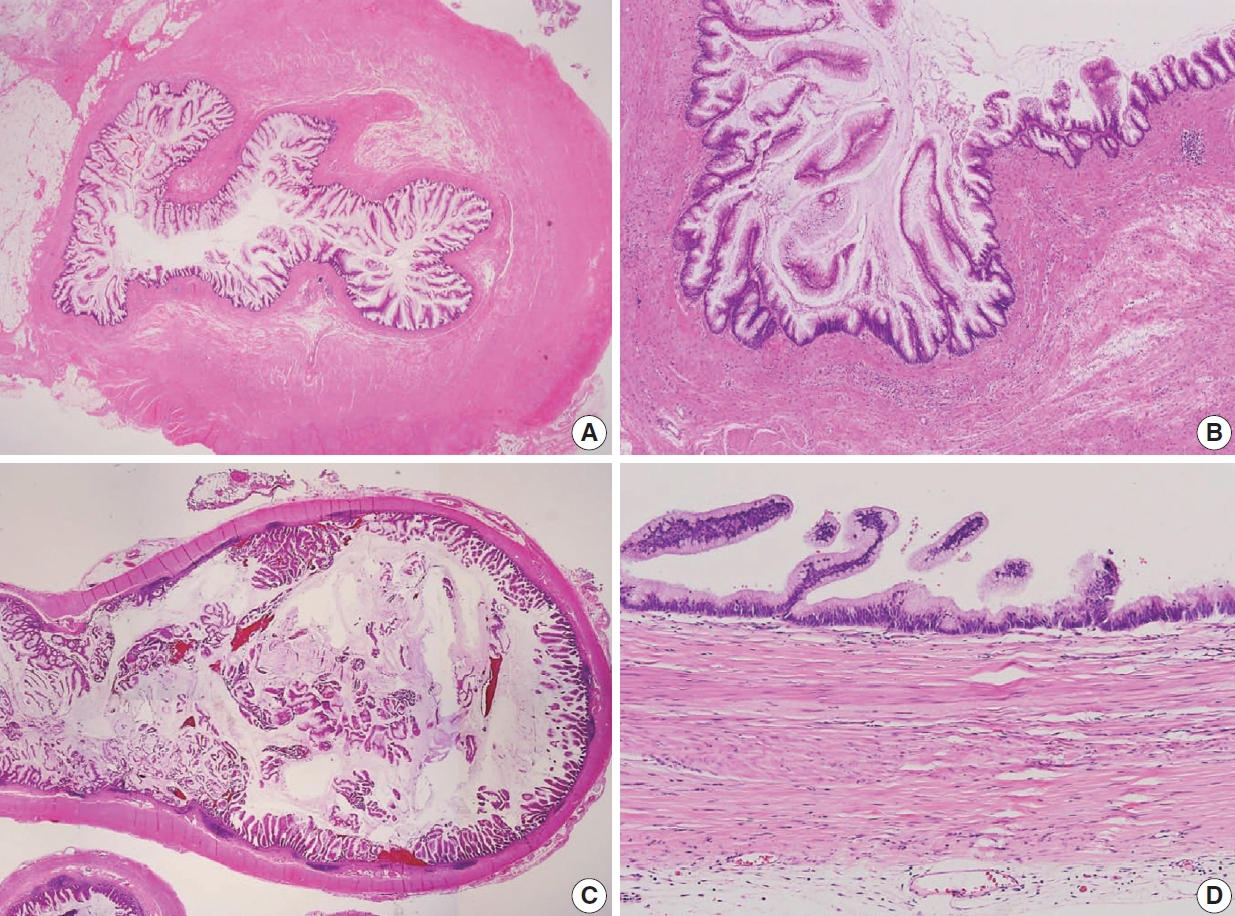
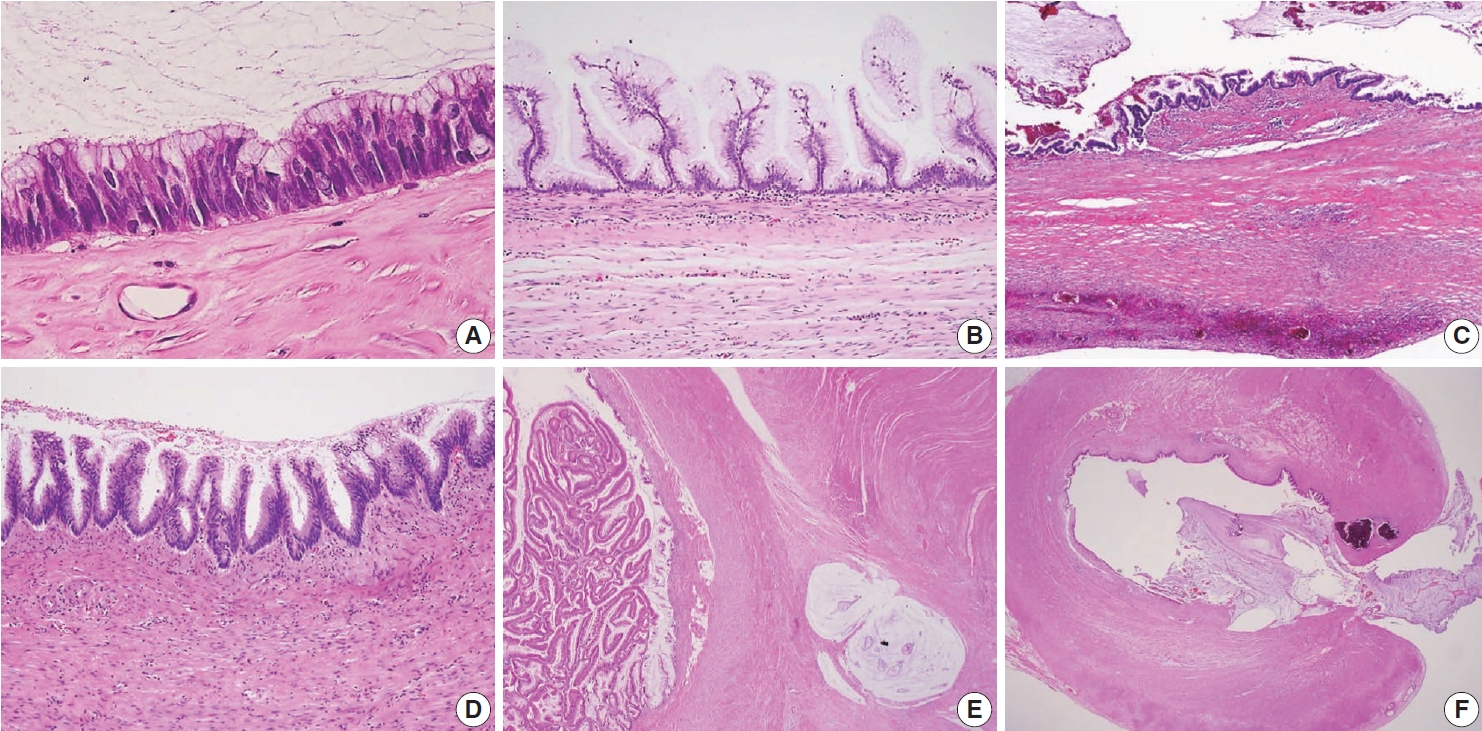
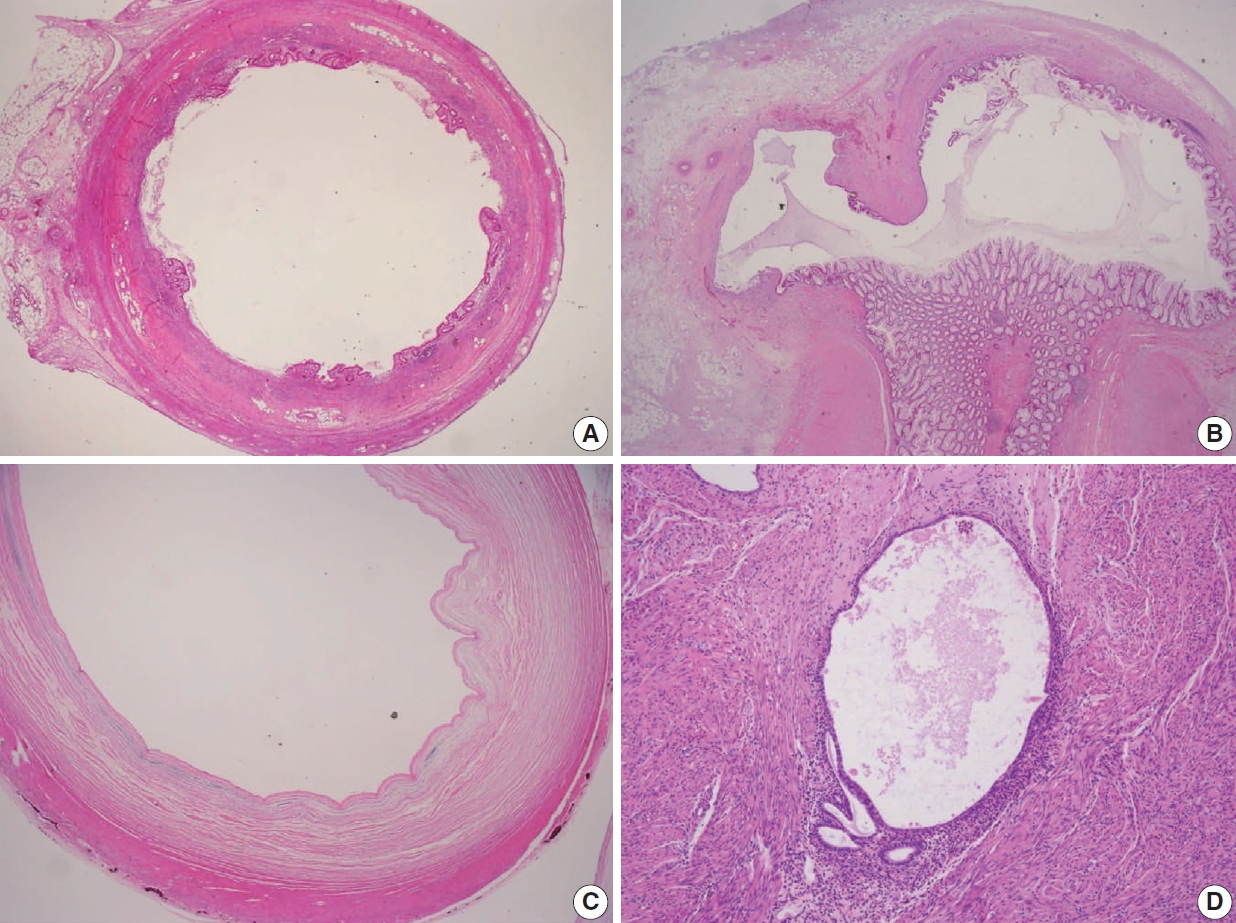
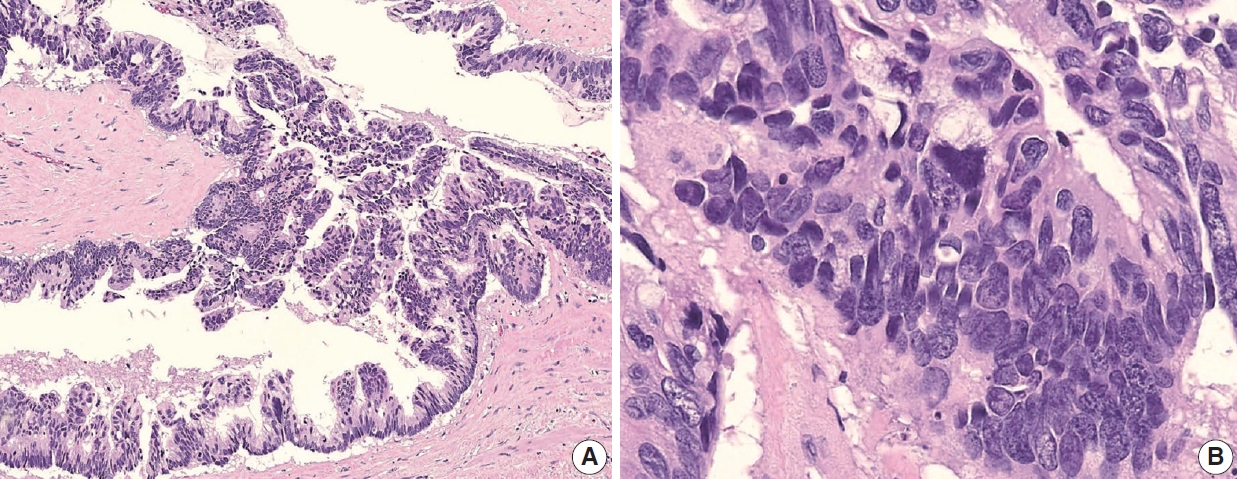
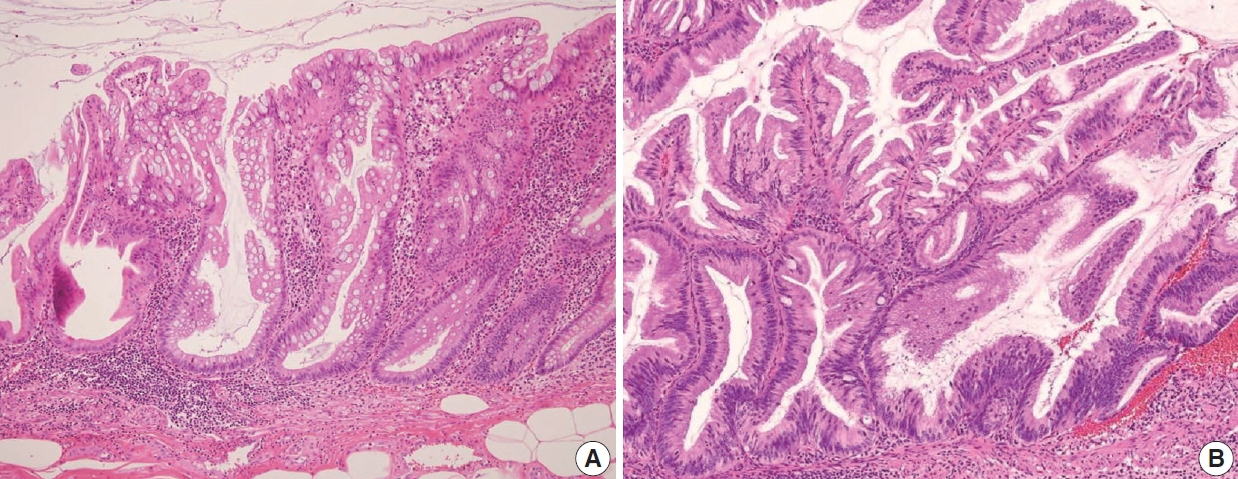
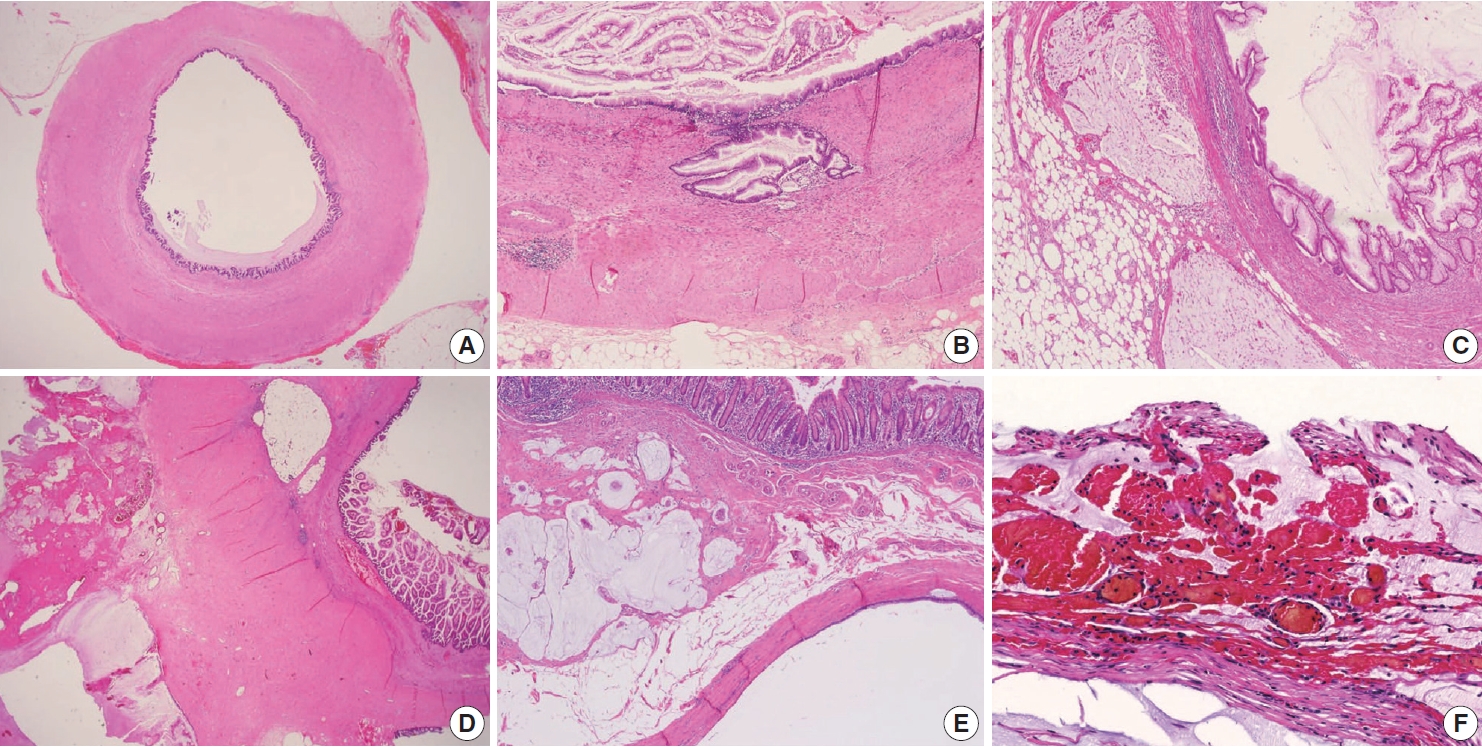
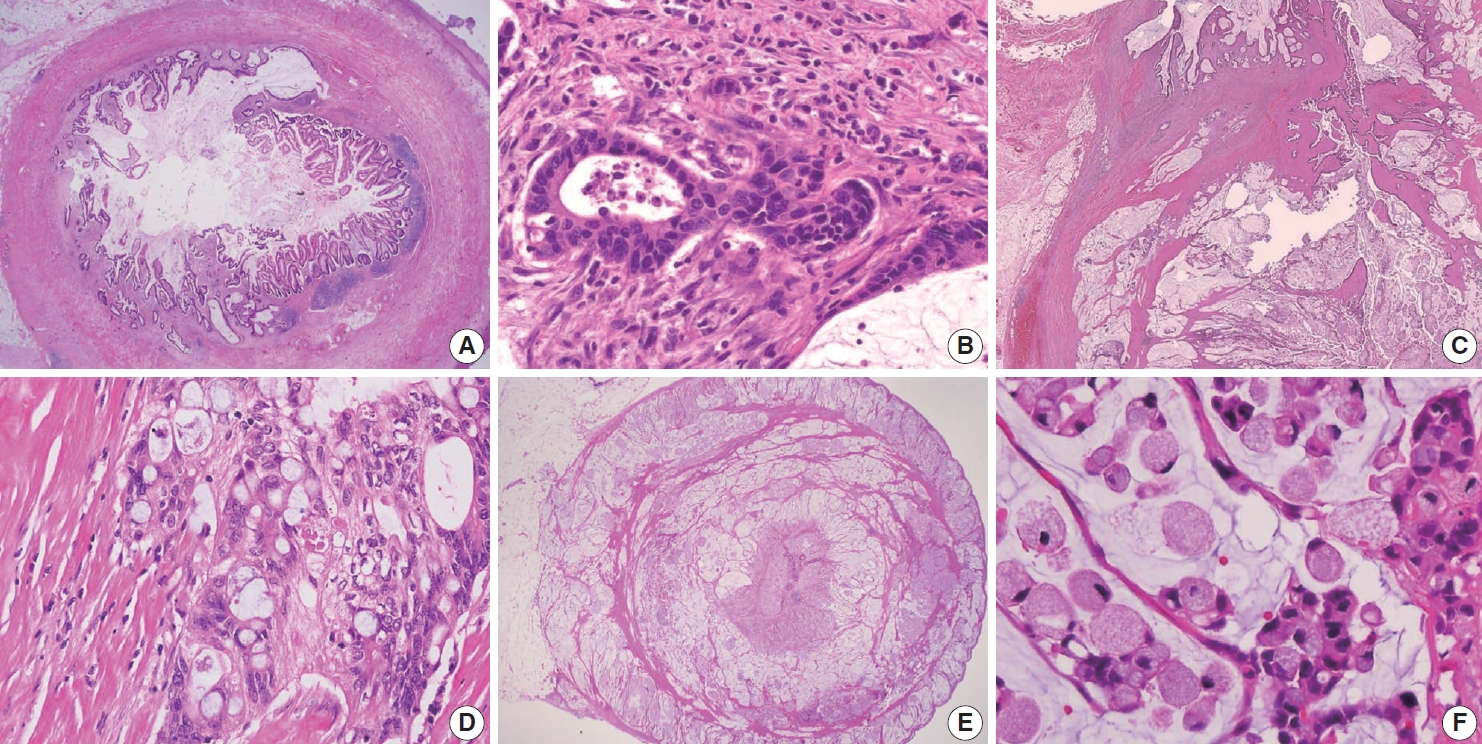
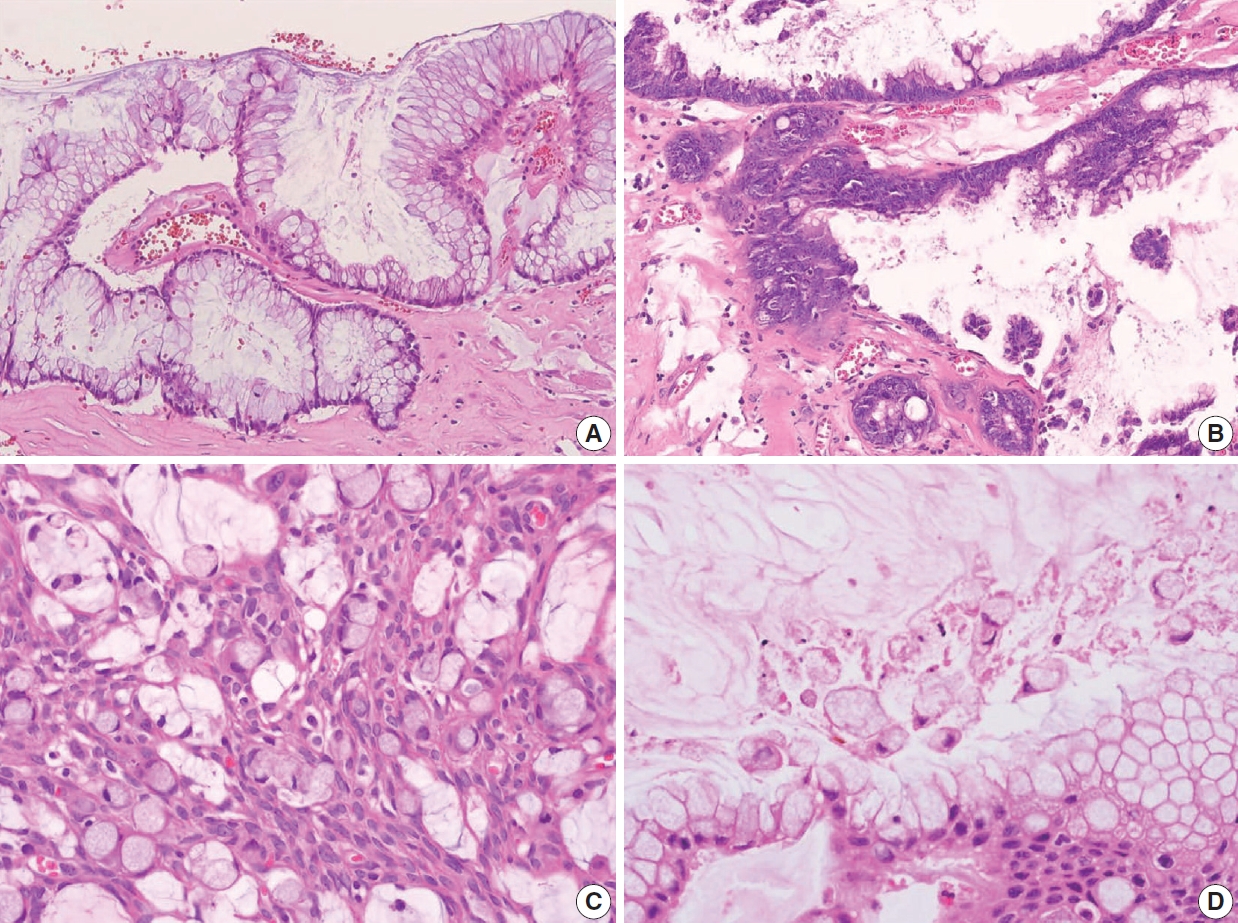
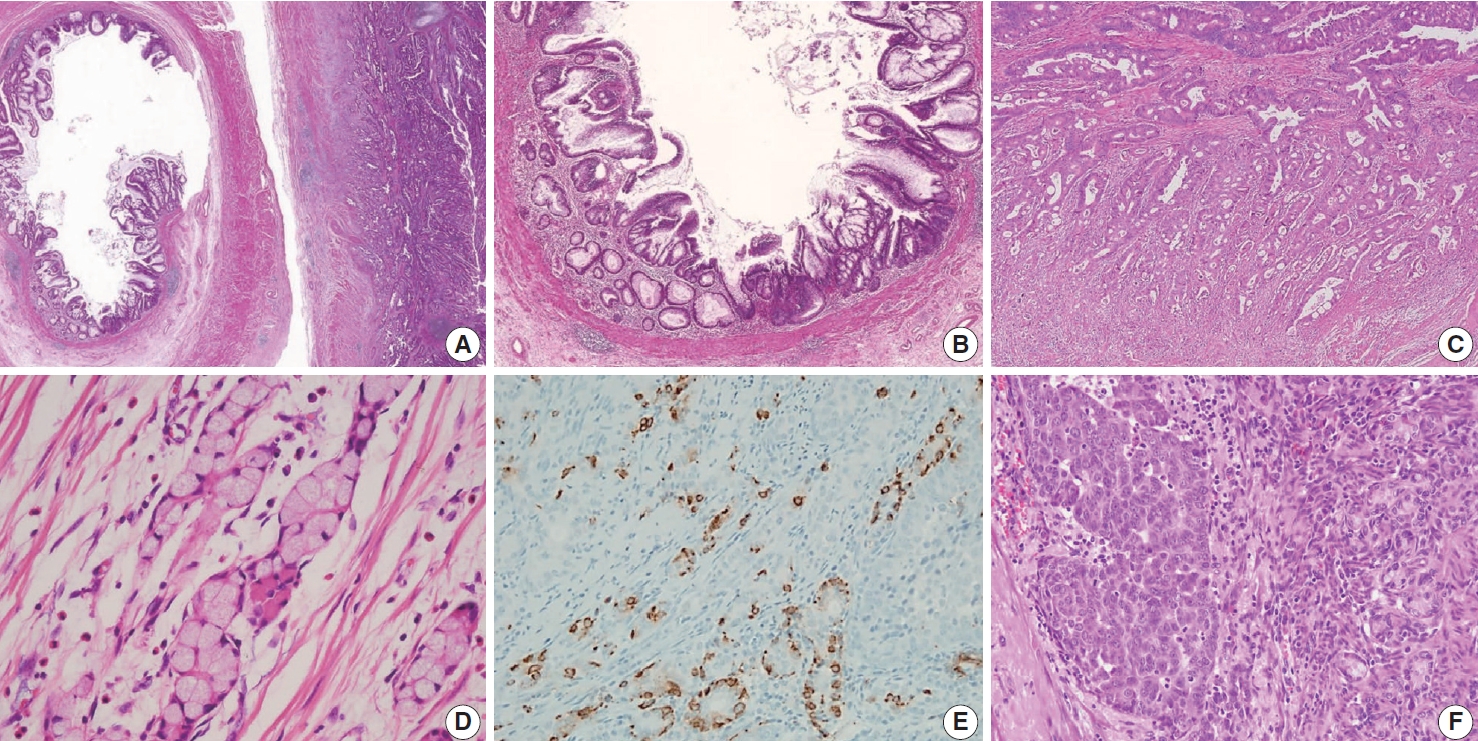
Fig. 1.
Fig. 2.
Fig. 3.
Fig. 4.
Fig. 5.
Fig. 6.
Fig. 7.
Fig. 8.
Fig. 9.
| ICD-O3 | Epithelial tumors | |
|---|---|---|
| - | Hyperplastic polyp | |
| - | Sessile serrated lesion without dysplasia | |
| 8213/0 | Serrated dysplasia, low grade | |
| 8213/2 | Serrated dysplasia, high grade | |
| 8480/1 | Low-grade appendiceal mucinous neoplasm | |
| 8480/2 | High-grade appendiceal mucinous neoplasm | |
| 8140/3 | Adenocarcinoma, NOS | |
| 8480/3 | Mucinous adenocarcinoma (> 50% mucin) | |
| 8490/3 | Signet-ring cell carcinoma (> 50% signet-ring cells) | |
| 8020/3 | Carcinoma, undifferentiated, NOS | |
| 8243/3 | Goblet cell adenocarcinoma | |
| 8240/3 | Neuroendocrine tumor, NOS | |
| 8240/3 | Neuroendocrine tumor, grade 1 | |
| 8249/3 | Neuroendocrine tumor, grade 2 | |
| 8249/3 | Neuroendocrine tumor, grade 3 | |
| 8152/3 | L-cell tumor | |
| 8152/3 | Glucagon-like peptide producing tumor | |
| 8152/3 | PP/PYY-producing tumor | |
| 8241/3 | Enterochromaffin-cell carcinoid | |
| 8241/3 | Serotonin-producing carcinoid | |
| 8246/3 | Neuroendocrine carcinoma | |
| 8013/3 | Large cell neuroendocrine carcinoma | |
| 8041/3 | Small cell neuroendocrine carcinoma | |
| 8154/3 | Mixed neuroendocrine-non-neuroendocrine carcinoma (MiNEN) | |
| Classification | Grading |
ICD-O3 | KCD-8 | Diagnostic criteria and microscopic features [3,12] | |
|---|---|---|---|---|---|
| Low-grade appendiceal mucinous neoplasm | Low/G1 | 8480/1 | D37.3 | Mucinous neoplasm with low-grade cytology and any of the following (by PSOGI criteria) | |
| Loss of muscularis mucosae | |||||
| Fibrosis of submucosa | |||||
| “Pushing invasion” (expansile or diverticulum-like growth) | |||||
| Dissection of acellular mucin in wall | |||||
| Undulating or flattened epithelial growth | |||||
| Rupture of appendix | |||||
| Mucin and/or cells outside appendix | |||||
| High-grade appendiceal mucinous neoplasm | High/G2 | 8480/2 | D01.7 | Mucinous neoplasm with the architectural features of LAMN and no infiltrative invasion, but with high-grade cytologic atypia | |
| Mucinous adenocarcinoma | High/G2 | 8480/3 | C18.1 | Malignant glandular neoplasm, comprising of > 50% extracellular mucin, characterized by infiltrative invasion. The pattern of infiltrative invasion as follows: Infiltrative glands, or single infiltrative tumor cells associated with extracellular mucin and desmoplastic stroma | |
| Small dissecting mucin pools containing floating nests, glands, or single neoplastic cells | |||||
| Mucinous adenocarcinoma with signet-ring cells | High/G3 | 8480/3 | C18.1 | Mucinous adenocarcinoma, comprising of ≤ 50% signet-ring cells | |
| Signet-ring cell carcinoma | High/G3 | 8490/3 | C18.1 | Adenocarcinoma, comprising up to > 50% signet-ring cells | |
| pT stage | Definition and lesions | Prognostic significances |
|---|---|---|
| pTis(LAMN) | LAMN confined to the appendiceal wall after histologic examination of the entire appendix. | pTis(LAMN) has essentially no risk of recurrence. pTis(LAMN) designation requires correlation with the intraoperative findings and an evaluation by the operator. |
| Acellular mucin or mucinous epithelium may interrupt the muscle propria. | ||
| pT3(LAMN) | LAMN with acellular mucin or mucinous epithelium extending into the subserosa or mesoappendix but no involvement of the serosa after histologic examination of the entire appendix. | Unknown risk of peritoneal recurrence. |
| Long-term follow-up for 10 yr is suggested until additional data on recurrence risk becomes available. | ||
| pT4(LAMN) | LAMN invades the visceral peritoneum, including the acellular mucin or mucinous epithelium involving the serosa of the appendix or mesoappendix (pT4a), and/or directly invades adjacent organs or structures (pT4b). | Low risk (acellular mucin) and high risk (mucinous epithelium) of peritoneal recurrence. |
| Long-term follow-up for 10 yr with periodic imaging is required. | ||
| Additional surgery or CRS-HIPEC is uncertain but is used in some centers. |
| Lesion | Grade | Behavior codes |
Terminology |
Criteria | Treatment | ||
|---|---|---|---|---|---|---|---|
| ICD-O3 | KCD-8 | PSOGI/Carr [12] | AJCC/Misdraji [10] | ||||
| Mucin without tumor cells | - | 8480/6 | C78.6 | Acellular mucin | Acellular mucin | Mucin within the peritoneal cavity without neoplastic epithelial cells | CRS-HIPEC |
| Mucinous tumor with low-grade histologic features | G1 | 8480/6 | C78.6 | Low-grade mucinous carcinoma peritonei | LAMN with peritoneal involvement | Scanty (≤ 20% cellularity) | CRS-HIPEC |
| Strips, gland-like or small clusters | |||||||
| Low-grade cytological atypia | |||||||
| Not more than occasional mitosis | |||||||
| Pushing invasion | |||||||
| Mucinous tumor with high-grade histologic features | G2 | 8480/6 | C78.6 | High-grade mucinous carcinoma peritonei | Mucinous adenocarcinoma, moderately differentiated | More cellular (> 20% cellularity) | Systemic chemotherapy ± CRS-HIPEC |
| Cribriform growth | |||||||
| Mixed but mostly high-grade cytological atypia | |||||||
| Numerous mitoses | |||||||
| Destructive infiltrative invasion | |||||||
| Mucinous tumor with signet-ring cells | G3 | 8480/6 | C78.6 | High-grade mucinous carcinoma peritonei with signet-ring cells | Mucinous adenocarcinoma, poorly differentiated | Any lesion with signet-ring cells (degenerating cells within mucin that mimic signet-ring cells should be discounted) | Systemic chemotherapy ± CRS-HIPEC |
| Specimen type | ||
|---|---|---|
| □ Appendectomy | ||
| □ Appendectomy and right colectomy | ||
| □ Other: (specify: ) | ||
| Tumor size | ||
| × × cm | ||
| Tumor site | ||
| □ Proximal half of appendix | ||
| □ Distal half of appendix | ||
| □ Diffusely involving appendix | ||
| □ Other: (specify: ) | ||
| Histologic types (WHO 5th Digestive System Tumors) [1] | ||
| □ Low-grade appendiceal mucinous neoplasm (LAMN) | ||
| □ High-grade appendiceal mucinous neoplasm (HAMN) | ||
| □ Mucinous adenocarcinoma | ||
| □ Non-mucinous adenocarcinoma | ||
| □ Signet-ring cell carcinoma | ||
| □ Goblet cell adenocarcinoma | ||
| □ Large cell neuroendocrine carcinoma | ||
| □ Small cell neuroendocrine carcinoma | ||
| □ Mixed neuroendocrine-non-neuroendocrine carcinoma (MiNEN) | ||
| □ Medullary carcinoma | ||
| □ Adenosquamous carcinoma | ||
| □ Undifferentiated carcinoma | ||
| □ Other histologic type not listed (specify: ) | ||
| Histologic grade | ||
| □ G1: Well differentiated | ||
| □ G2: Moderately differentiated | ||
| □ G3: Poorly differentiated | ||
| □ GX: Cannot be assessed | ||
| □ Other: (specify: ) | ||
| Disseminated peritoneal mucinous disease | ||
| □ Not identified | ||
| □ Present | ||
| □ Low-grade mucinous carcinoma peritonei/LAMN (G1) with peritoneal involvement | ||
| □ High-grade mucinous carcinoma peritonei/Mucinous adenocarcinoma, moderately differentiated (G2) | ||
| □ High-grade mucinous carcinoma peritonei with signet-ring cells / Mucinous adenocarcinoma, poorly differentiated (G3) | ||
| Resection margins including proximal and mesenteric margins | ||
| □ Cannot be assessed | ||
| □ Uninvolved by tumor | ||
| □ Distance of tumor from the closest margin: mm | ||
| □ Involved by tumor | ||
| □ Involved by invasive carcinoma (□ Proximal margin, □ Mesenteric margin) | ||
| □ Involved by appendiceal mucinous neoplasm (□ Proximal margin, □ Mesenteric margin) | ||
| □ Involved by acellular mucin (□ Proximal margin, □ Mesenteric margin) | ||
| Lympho-vascular invasion | ||
| □ Not identified □ Present □ Cannot be determined | ||
| Perineural invasion | ||
| □ Not identified □ Present □ Cannot be determined | ||
| Tumor deposits | ||
| □ Not identified □ Present □ Cannot be determined | ||
| Regional lymph nodes | ||
| □ No lymph nodes submitted or found | ||
| □ Total number of lymph nodes examined: | ||
| □ Number of lymph nodes involved with metastases: | ||
| Pathologic stage classification (pTNM, AJCC 8th edition) [13] | ||
| TNM Descriptors: □ m (multiple) □ r (recurrent) □ y (post-neoadjuvant therapy) | ||
| Primary tumor (pT) | ||
| □ pTX: Primary tumor cannot be assessed | ||
| □ pTX0: No evidence of primary tumor | ||
| □ pTis: Carcinoma in situ (high-grade dysplasia); intraepithelial carcinoma; invasion of lamina propria (intramucosal carcinoma) | ||
| □ pTis(LAMN): LAMN confined to the appendix (defined as involvement by acellular mucin or mucinous epithelium that may extend into the muscularis propria) | ||
| □ pT1: Tumor invades submucosa (does not apply to LAMN) | ||
| □ pT2: Tumor invades muscularis propria (does not apply to LAMN) | ||
| □ pT3: Tumor invades through the muscularis propria into the subserosa or mesoappendix | ||
| □ pT4: Tumor invades the visceral peritoneum, including the acellular mucin or mucinous epithelium involving the serosa of the appendix or mesoappendix, and/or directly invades adjacent organs or structures | ||
| □ pT4a: Tumor invades the visceral peritoneum, including the acellular mucin or mucinous epithelium involving the serosa of appendix or serosa of mesoappendix | ||
| □ pT4b: Tumor directly invades other organs or structures | ||
| Regional lymph nodes (pN) | ||
| □ pNX: Regional lymph nodes cannot be assessed | ||
| □ pN0: No regional nodal metastasis | ||
| □ pN1: One to three regional lymph nodes are positive (tumor in lymph nodes measuring ≥ 0.2 mm), or any number of tumor deposits is present, and all identifiable lymph nodes are negative | ||
| □ pN1a: One regional lymph node is positive | ||
| □ pN1b: Two or three regional lymph nodes are positive | ||
| □ pN1c: No regional lymph node metastasis, but tumor deposits in the subserosa or mesentery | ||
| □ pN2: Metastasis in 4 or more regional nodes | ||
| Distant metastasis (pM) | ||
| □ Not applicable | ||
| □ pM1: Distant metastasis | ||
| □ pM1a: Intraperitoneal acellular mucin only | ||
| □ pM1b: Intraperitoneal metastasis only, including mucinous epithelium | ||
| □ pM1c: Non-peritoneal metastasis | ||
| Additional pathologic findings or pre-existing lesions | ||
| □ None | ||
| □ Other: (specify: ) | ||
| Ancillary studies | ||
| □ Specify: | ||
WHO, World Health Organization; ICD-O3, International Classification of Diseases for Oncology, 3rd edition; NOS, not otherwise specified.
ICD-O3, International Classification of Diseases for Oncology, 3rd edition; KCD-8, Korean Classification of Disease, 8th edition; PSOGI, Peritoneal Surface Oncology Group International; LAMN, low-grade appendiceal mucinous neoplasm. Grading divide into low- and high-grade by the two-tier system and G1 to G3 by three-tiered system.
Note that pT1 and pT2 do not apply to LAMN. LAMN, low-grade appendiceal mucinous neoplasm; AJCC, American Joint Committee on Cancer; CRS-HIPEC, cytoreductive surgery with hyperthermic intraperitoneal chemotherapy.
Note that ICD-O3 and KCD-8 codes of primary or unknown origin disseminated peritoneal mucinous disease are 8480/3 and C80.0, respectively. ICD-O3, International Classification of Diseases for Oncology, 3rd edition; KCD-8, Korean Classification of Disease, 8th edition; PSOGI, Peritoneal Surface Oncology Group International; AJCC, American Joint Committee on Cancer; CRS-HIPEC, cytoreductive surgery with hyperthermic intraperitoneal chemotherapy; LAMN, low-grade appendiceal mucinous neoplasm.

 E-submission
E-submission











