Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 52(5); 2018 > Article
-
Case Study
Abrupt Dyskeratotic and Squamoid Cells in Poorly Differentiated Carcinoma: Case Study of Two Thoracic NUT Midline Carcinomas with Cytohistologic Correlation -
Taebum Lee
 , Sangjoon Choi
, Sangjoon Choi , Joungho Han,
, Joungho Han, , Yoon-La Choi
, Yoon-La Choi , Kyungjong Lee1
, Kyungjong Lee1
-
Journal of Pathology and Translational Medicine 2018;52(5):349-353.
DOI: https://doi.org/10.4132/jptm.2018.07.16
Published online: July 27, 2018
Department of Pathology, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
1Division of Pulmonary and Critical Care Medicine, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
- Corresponding Author Joungho Han, MD Department of Pathology, Samsung Medical Center, Sungkyunkwan University School of Medicine, 81 Irwon-ro, Gangnam-gu, Seoul 06351, Korea Tel: +82-2-3410-2800 Fax: +82-2-3410-0025 E-mail: 'hanjho@skku.edu'
© 2018 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
- Cytologic diagnosis of nuclear protein in testis (NUT) midline carcinoma (NMC) is important due to its aggressive behavior and miserable prognosis. Early diagnosis of NMC can facilitate proper management, and here we report two rare cases of thoracic NMC with cytohistologic correlation. In aspiration cytology, the tumor presented with mixed cohesive clusters and dispersed single cells, diffuse background necrosis and many neutrophils. Most of the tumor cells had scanty cytoplasm and medium-sized irregular nuclei, which had fine to granular nuclear chromatin. Interestingly, a few dyskeratotic cells or squamoid cell clusters were present in each case. Biopsy specimen histology revealed more frequent squamous differentiation, and additional immunohistochemistry tests showed nuclear expression of NUT. Because this tumor has a notorious progression and has been previously underestimated in terms of its prevalence, awareness of characteristic findings and proper ancillary tests should be considered in all suspicious cases.
- Case 1
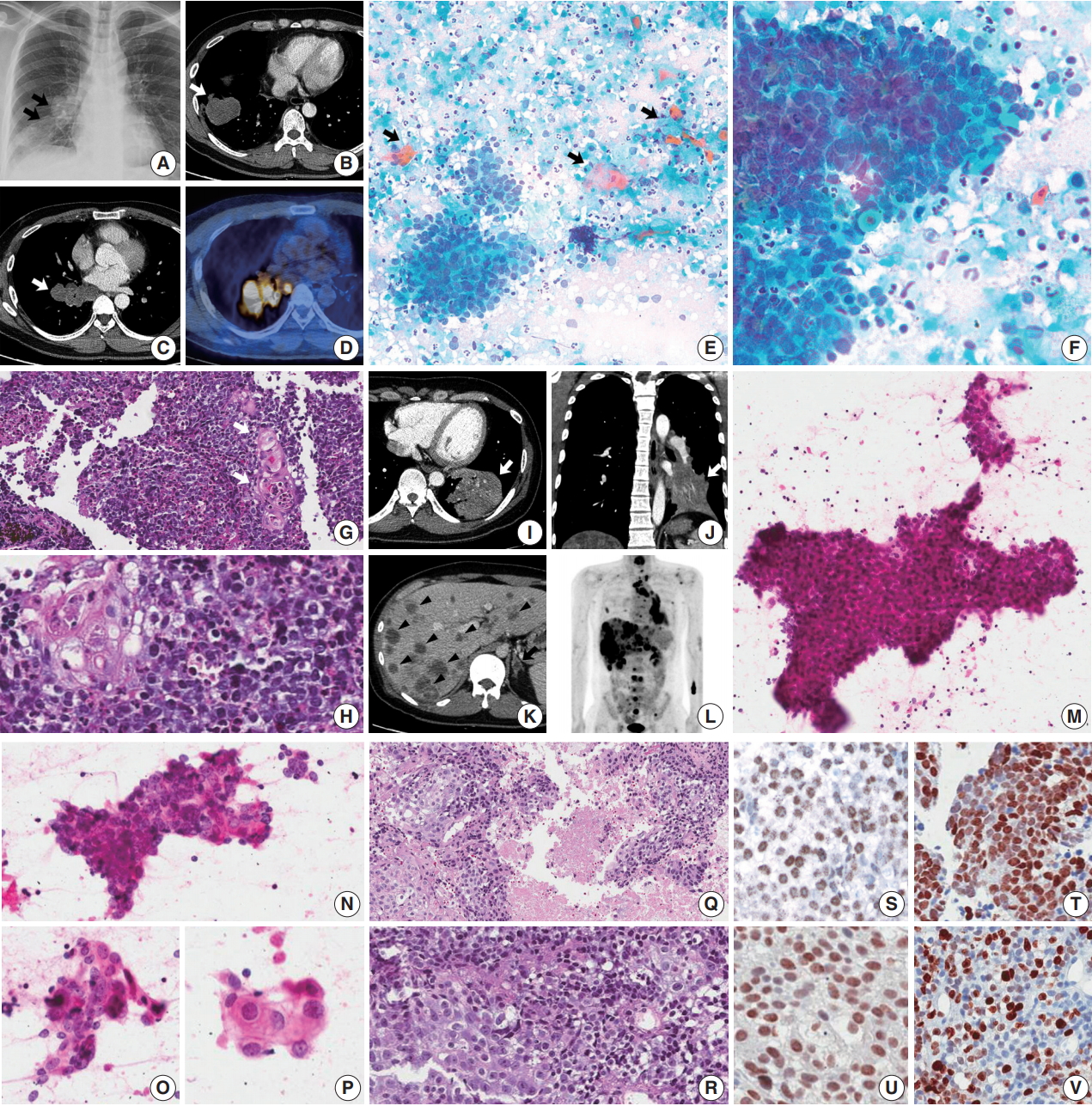
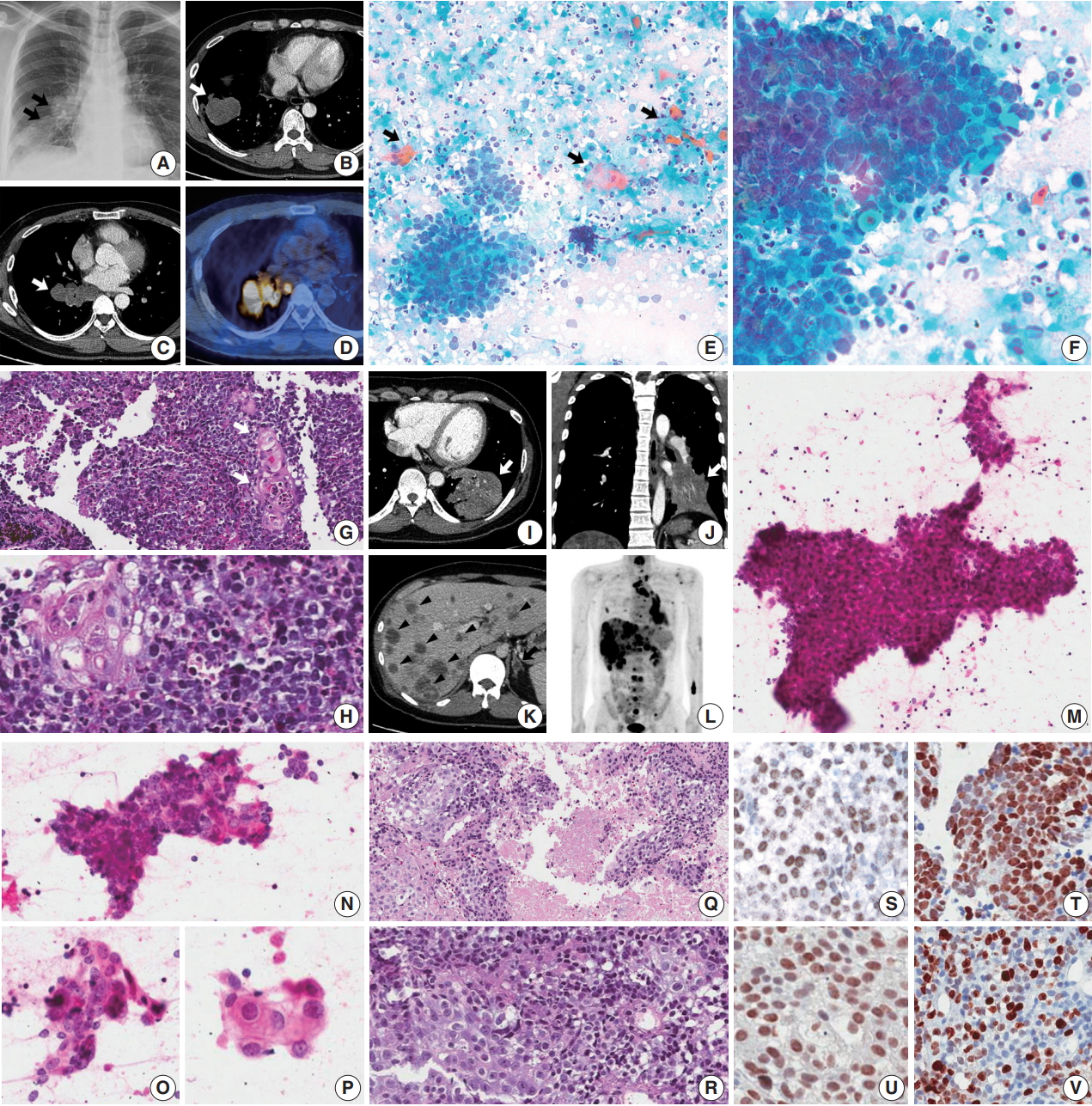
- A 45-year-old male presented with a 3-month history of cough. Chest X-ray and computed tomography (CT) showed a 3.8-cm-sized mass in the right lower lobe and multiple enlarged mediastinal lymph nodes (Fig. 1A–C) in which 18F-fluorodeoxyglucose uptake was detected (Fig. 1D). For diagnosis and nodal staging, endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) and biopsy were performed. Aspirates from both lymph nodes and lung mass were highly cellular. Some tumor cells were cohesive enough to form 3-dimensional clusters, but others were dispersed in single cells (Fig. 1E). Frequent crush artifacts and naked nuclei were easily identified. Tumor cells were poorly to undifferentiated with scanty cytoplasm and irregular nuclear contour. Nuclear chromatin was fine to granular and nucleoli were small but occasionally identified (Fig. 1F). Unexpectedly, a few scattered dyskeratotic cells were noticed (Fig. 1E, F). Many neutrophils and necrosis were present in the background (Fig. 1F). Biopsy of mediastinal lymph nodes showed sheets of metastatic undifferentiated tumor cells with large area of necrosis and peritumoral neutrophilic infiltration (Fig. 1G). Tumor cells had scant and delicate amphophilic cytoplasm. Nuclear chromatin and contour were similar to the findings of aspirate smears. Foci of abrupt squamous differentiation were identified more often than in cytology (Fig. 1H). Under additional immunohistochemistry (IHC) staining, these tumor cells showed a very high proliferation index of up to 70% in Ki-67 (1:200, Dako, Glostrup, Denmark) and were positive for pan-cytokeratin (1:500, Dako), p53 (1:4,000, Thermo Fisher, Rockford, IL, USA), p63 (1:200, Biocare, Concord, CA, USA), and NUT (1:100, C52B1, Cell Signaling Technology, Danvers, MA, USA), and up to 70% of tumor cells expressed programmed death-ligand 1 (PD-L1; RTU, 22C3, Dako) (Fig. 1S, T). Tumor cells were negative for Oct3/4 (1:50, Santa Cruz Biotechnology, Dallas, TX, USA), chromogranin (1:400, Dako), CD99 (1:50, Dako), CD45 (1:1000, Dako), CD56 (1:200, Leica Biosystems, Novocastra, Newcastle upon Tyne, UK), and thyroid transcription factor 1 (TTF-1; 1:50, Dako), which can help rule out the possibilities of other malignancies.
- Case 2
- A previously healthy 32-year-old male presented with a persistent cough for 3 months, blood-tinged sputum and chest pain. On chest CT, a 5.5-cm-sized consolidative lung mass accompanied by enlarged mediastinal lymph nodes was identified at the left lower lobe (Fig. 1I, J). Abdomen-pelvis CT showed multiple necrotic masses scattered throughout both lobes of the liver and bilateral adrenal glands (Fig. 1K), and positron emission tomography highlighted multiple hypermetabolic bony lesions in the whole spine, bilateral ribs, pelvic bones, sternum and both scapulae (Fig. 1L). Cytology of lymph node aspirates from EBUS-TBNA showed dispersed and clustered tumor cells in the background of necrosis (Fig. 1M). Some tumor cells had large vesicular nuclei with open chromatin, prominent nucleoli, scanty cytoplasm and a high nuclear to cytoplasmic (N/C) ratio. Naked nuclei were frequently identified in scattered individual cells. However, others exhibited more abundant amphophilic to eosinophilic cytoplasm and granular nuclear chromatin with a lower N/C ratio, which could be considered squamous differentiation (Fig. 1N–P). Biopsy of the mediastinal lymph nodes and liver showed metastatic carcinoma in a necrotic background composed of two morphologically different tumor cell components: poorly differentiated cells with hyperchromatic nuclei and scanty cytoplasm, and squamoid cells with less hyperchromatic nuclei, prominent nucleoli and ample cytoplasm (Fig. 1Q, R). By IHC, tumor cells were positive for p63, p53, and NUT, negative for TTF-1 and CD56, and had a relatively low Ki-67 proliferation index of approximately 25% (Fig. 1U, V). PD-L1 IHC test revealed low expression of about 1% of the tumor.
CASE REPORT
- Diagnosis of NMC in cytology is challenging. Cytologic findings are characterized by high cellularity with medium-sized poorly to undifferentiated cells. Cohesiveness of these tumor cells is variable [2] and while clusters of tumor cells are usually patternless, some can be arranged in a pseudoglandular pattern in focal areas [4]. Round to oval nuclei have irregular contours, but their size is relatively uniform and monotonous rather than pleomorphic. Nuclear chromatin is not coarse or condensed but fine to granular and vesicular. Nucleoli are usually single, small and prominent [5]. Scant cytoplasm is amphophilic to eosinophilic and nuclear molding is frequent. The absence of cytoplasm makes nuclei naked and squeezing artifacts are common, especially in smears rather than liquid-based preparations. Mitosis is frequent, but atypical mitosis is rare. Necrotic debris, apoptotic bodies and nuclear dust with many neutrophils fill the background of the tumor cells. Previous reports have described difficulty in finding squamous differentiation in cytology specimens [2,5], but tumor cell clusters with a squamoid appearance or a few dyskeratotic cells were easily noticed in our cases. This finding can be variable, from subtle to very distinct with obvious keratinization.
- The cytogenetic rearrangement of this tumor is unique and characteristic. Two-thirds of such tumors have chromosomal translocation t(15;19), which forms the BRD4-NUT fusion oncogene, and subsets have BRD3-NUT and other NUT-variant rearrangement [1]. In recent studies, knockdown of BRD-NUT induces rapid squamous differentiation with a halt in proliferation in patient-derived NMC cells [6,7]. This result strongly suggests the role of BRD-NUT as a fusion protein that blocks differentiation, and is compatible with its characteristic cytologic and histologic findings. The tumor histology is usually made of sheets of undifferentiated cells. Nuclear size and shape, chromatin pattern, nucleoli and cytoplasm are similar to the cytology findings. Frequent areas of necrosis are common, and diffuse infiltrations of neutrophils among tumor cells are easily identified. Despite the simple and uniform chromosomal abnormality, some tumor cells might be out of influence from the cytogenetic alteration. Accordingly, there may be focal areas of abrupt squamous differentiation that are more commonly identified than in cytology specimens.
- Differential diagnosis of NMC in cytology includes small cell carcinoma (SCC), basaloid squamous cell carcinoma (BSqCC), and small cell variant of squamous cell carcinoma (SCVSqCC). The cytology of SCC shows predominantly single cells, but has some cell clustering with nuclear molding exhibiting fine, open chromatin, and was inconspicuous to small nucleoli [8,9]. Scant cytoplasm, artifactually crushed and naked nuclei, and background necrosis are common, and all of these findings mimic NMC. However, keratinized squamous cells are rare, even if some aspirate smears have bronchial epithelial cells with squamous metaplasia.
- BSqCC and SCVSqCC are more problematic diagnoses because both have small cells with some dyskeratosis and squamous differentiation. The cytology of BSqCC has small to medium-sized nuclei with fine chromatin, small to inconspicuous nucleoli, scant cytoplasm and nuclear molding with necrosis [9]. SCVSqCC shows similar small cells but slightly more abundant, bluish cytoplasm, with vesicular nuclei and prominent nucleoli [10]. Both basaloid and SCVSqCC have areas of necrosis, squamous differentiation and dyskeratosis, which is often noticed in NMC but usually composed of predominantly cohesive cell clusters rather than dispersed single cells [9]. It is important to note that all these tumors have similar cytologic findings and aggressive behaviors but require different management.
- In IHC, NMC tumor cells are positive for pan-cytokeratin, p53, p63 and NUT, and usually have a high but variable proliferation index by Ki-67. It is hard to differentiate NMC from possible mimickers without diagnostic markers, which are not routinely screened. If the cytomorphology is suspicious for NMC, a FISH assay and IHC test should be performed in all cases due to the underestimation of its prevalence [1]. A previous study examined NUT IHC expression using a monoclonal antibody in 114 cases of poorly differentiated carcinoma or unclassified malignancy in the mediastinum, and 3.5% of cases showed nuclear expression with a median age of 50 [11]. Further studies are warranted in other anatomic sites, regardless of age.
- Currently, conventional cytotoxic chemotherapy and radiation therapy are not effective in treating NMC. Unlike other usual carcinomas, this tumor has a typical chromosomal alteration and genetic stability. Novel targeted therapy using histone deacetylase inhibitors and BET inhibitors has been developed lately based on the idea that BRD-NUT fusion proteins specifically bind to acetylated histones [1]. These drugs are now in several clinical trials (ClinicalTrials.gov identifiers: NCT01587703, NCT01987362, NCT02711137, and NCT02307240). Not only doctors, but also patients, continue to struggle with NMC at this moment. To treat this disease properly, all pathologists should pay attention to this underestimated entity.
DISCUSSION
-
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Notes
Acknowledgments
- 1. French CA. Pathogenesis of NUT midline carcinoma. Annu Rev Pathol 2012; 7: 247–65. ArticlePubMed
- 2. Stelow EB. A review of NUT midline carcinoma. Head Neck Pathol 2011; 5: 31–5. ArticlePubMedPMC
- 3. Park HS, Bae YS, Yoon SO, et al. Usefulness of nuclear protein in testis (NUT) immunohistochemistry in the cytodiagnosis of NUT midline carcinoma: a brief case report. Korean J Pathol 2014; 48: 335–8. ArticlePubMedPMC
- 4. Policarpio-Nicolas ML, de Leon EM, Jagirdar J. Cytologic findings of NUT midline carcinoma in the hilum of the lung. Diagn Cytopathol 2015; 43: 739–42. ArticlePubMed
- 5. Bishop JA, French CA, Ali SZ. Cytopathologic features of NUT midline carcinoma: a series of 26 specimens from 13 patients. Cancer Cytopathol 2016; 124: 901–8. ArticlePubMed
- 6. French CA, Ramirez CL, Kolmakova J, et al. BRD-NUT oncoproteins: a family of closely related nuclear proteins that block epithelial differentiation and maintain the growth of carcinoma cells. Oncogene 2008; 27: 2237–42. ArticlePubMedPDF
- 7. Schwartz BE, Hofer MD, Lemieux ME, et al. Differentiation of NUT midline carcinoma by epigenomic reprogramming. Cancer Res 2011; 71: 2686–96. ArticlePubMedPMC
- 8. Sturgis CD, Nassar DL, D'Antonio JA, Raab SS. Cytologic features useful for distinguishing small cell from non-small cell carcinoma in bronchial brush and wash specimens. Am J Clin Pathol 2000; 114: 197–202. ArticlePubMedPDF
- 9. Marks RA, Cramer HM, Wu HH. Fine-needle aspiration cytology of basaloid squamous cell carcinoma and small cell carcinoma-a comparison study. Diagn Cytopathol 2013; 41: 81–4. ArticlePubMed
- 10. Gray W, Kocjan G. Diagnostic cytopathology. Philadelphia: Elsevier, 2010; 68–9. 3rd.
- 11. Evans AG, French CA, Cameron MJ, et al. Pathologic characteristics of NUT midline carcinoma arising in the mediastinum. Am J Surg Pathol 2012; 36: 1222–7. ArticlePubMedPMC
References
Figure & Data
References
Citations

- Diagnostic significance and cytological features of NUT carcinoma by EBUS‐FNA, a case report and literature review
Yaping Ju, Miriam Velazquez, Andy Sherrod, Tiannan Wang
Cytopathology.2024;[Epub] CrossRef - BRD3‐NUTM1‐expressing NUT carcinoma of lung on endobronchial ultrasound‐guided transbronchial needle aspiration cytology, a diagnostic pitfall
Sameer Chhetri Aryal, Shereen Zia, Shannon Rodgers, Yulei Shen, Kyle Perry, Lisi Yuan
Diagnostic Cytopathology.2022;[Epub] CrossRef - Nuclear protein of the testis midline carcinoma of the thorax
Ayae Saiki, Keita Sakamoto, Yuan Bee, Takehiro Izumo
Japanese Journal of Clinical Oncology.2022; 52(6): 531. CrossRef - Approach to Mediastinal Fine Needle Aspiration Cytology
Zaibo Li, Huihong Xu, Fang Fan
Advances in Anatomic Pathology.2022; 29(6): 337. CrossRef - Diagnosis, Treatment and Prognosis of Primary Pulmonary NUT Carcinoma: A Literature Review
Jiaqian Yuan, Zhili Xu, Yong Guo
Current Oncology.2022; 29(10): 6807. CrossRef - Case report: Immunovirotherapy as a novel add-on treatment in a patient with thoracic NUT carcinoma
Linus D. Kloker, Branko Calukovic, Katrin Benzler, Alexander Golf, Sebastian Böhm, Sven Günther, Marius Horger, Simone Haas, Susanne Berchtold, Julia Beil, Mary E. Carter, Tina Ganzenmueller, Stephan Singer, Abbas Agaimy, Robert Stöhr, Arndt Hartmann, Tho
Frontiers in Oncology.2022;[Epub] CrossRef - Cytomorphology of primary pulmonary NUT carcinoma in different cytology preparations
Rimlee Dutta, Aruna Nambirajan, Saurabh Mittal, Sinchita Roy‐Chowdhuri, Deepali Jain
Cancer Cytopathology.2021; 129(1): 53. CrossRef - Update on genetically defined lung neoplasms: NUT carcinoma and thoracic SMARCA4-deficient undifferentiated tumors
Kyriakos Chatzopoulos, Jennifer M. Boland
Virchows Archiv.2021; 478(1): 21. CrossRef - Immunotherapy and Targeting the Tumor Microenvironment: Current Place and New Insights in Primary Pulmonary NUT Carcinoma
Xiang Li, Hui Shi, Wei Zhang, Chong Bai, Miaoxia He, Na Ta, Haidong Huang, Yunye Ning, Chen Fang, Hao Qin, Yuchao Dong
Frontiers in Oncology.2021;[Epub] CrossRef - Prevalence of NUT carcinoma in head and neck: Analysis of 362 cases with literature review
Taebum Lee, Junhun Cho, Chung‐Hwan Baek, Young‐Ik Son, Han‐Sin Jeong, Man Ki Chung, Sang Duk Hong, Yong Chan Ahn, Dong Ryul Oh, Jae Myoung Noh, Keunchil Park, Myung‐Ju Ahn, Hyung‐Jin Kim, Yi Kyung Kim, Young Hyeh Ko
Head & Neck.2020; 42(5): 924. CrossRef - Lung nuclear protein in testis carcinoma in an elderly Korean woman: A case report with cytohistological analysis
Hwa Jin Cho, Hyun‐Kyung Lee
Thoracic Cancer.2020; 11(6): 1724. CrossRef - Clinicopathological characteristics of primary lung nuclear protein in testis carcinoma: A single‐institute experience of 10 cases
Yoon Ah Cho, Yoon‐La Choi, Inwoo Hwang, Kyungjong Lee, Jong Ho Cho, Joungho Han
Thoracic Cancer.2020; 11(11): 3205. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure

 E-submission
E-submission