Articles
- Page Path
- HOME > J Pathol Transl Med > Volume 48(6); 2014 > Article
-
Brief Case Report
Combined Squamous Cell Carcinoma and Follicular Carcinoma of the Thyroid - Da Hye Son, Jong-Lyel Roh1, Kyung-Ja Cho,
-
Korean Journal of Pathology 2014;48(6):418-422.
DOI: https://doi.org/10.4132/KoreanJPathol.2014.48.6.418
Published online: December 31, 2014
Departments of Pathology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
1Departments of Otorhinolaryngology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- Corresponding Author: Kyung-Ja Cho, M.D. Department of Pathology, Asan Medical Center, University of Ulsan College of Medicine, 88 Olympic-ro 43-gil, Songpa-gu, Seoul 138-736, Korea Tel: +82-2-3010-4545, Fax: +82-2-472-7898, E-mail: kjc@amc.seoul.kr
• Received: March 26, 2014 • Revised: May 29, 2014 • Accepted: June 2, 2014
© 2014 The Korean Society of Pathologists/The Korean Society for Cytopathology
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
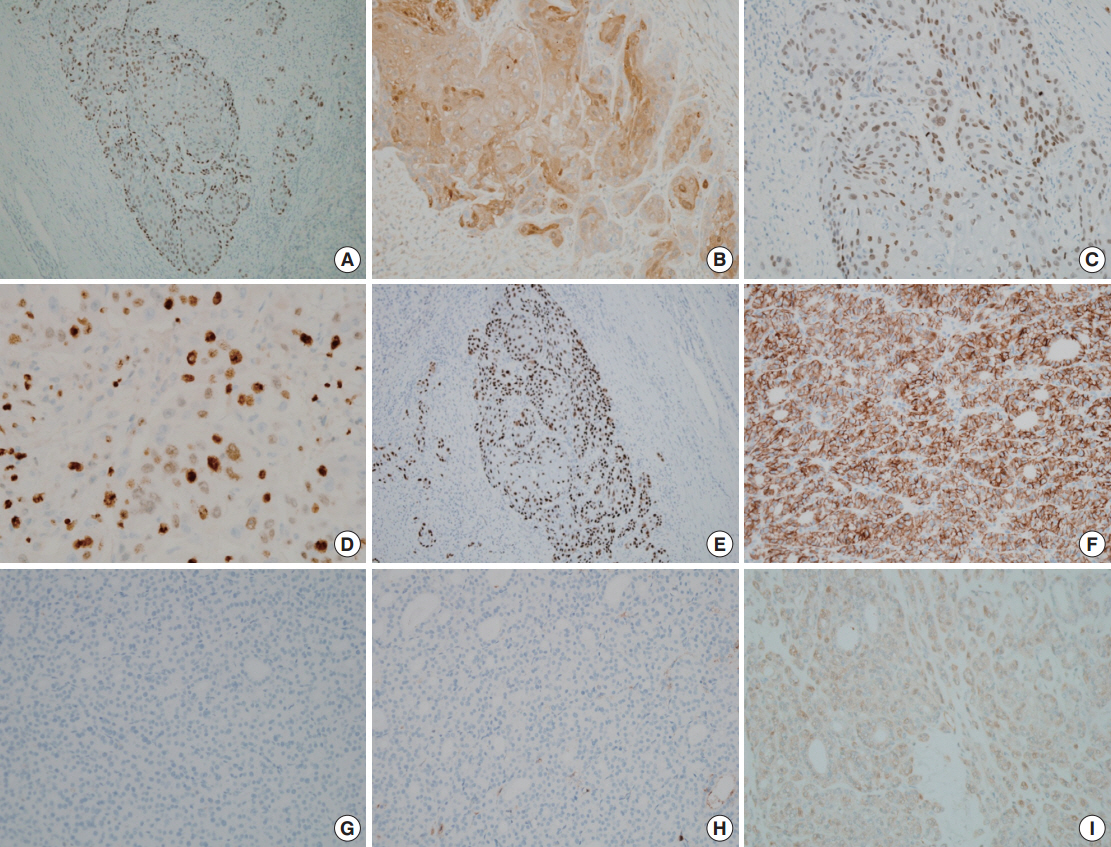
- A 69-year-old man visited Asan Medical Center for dysphagia and a gradually growing huge anterior neck mass of 10 years duration. He had no clinical symptoms related to thyroid dysfunction and had been repeatedly treated at another institution with radiofrequency ablation for his cystic neck lesion. Fine needle aspiration cytology at that time showed squamous cells and many inflammatory cells, leading to a diagnosis of branchial cleft cyst. Neck computed tomography on this occasion showed a 13×11 cm cystic lesion with irregularly thick walls involving the left lobe of the thyroid gland; the lesion also contained a strongly enhanced solid portion, septation, and calcification (Fig. 1A). Fusion whole body positron emission tomography fluorine-18 fluorodeoxyglucose was performed, showing abnormal uptake only by the neck mass (Fig. 1B). The esophagus, stomach, and larynx were unremarkable on esophagogastroduodenoscopy. The patient underwent total thyroidectomy with neck lymph node dissection. Macroscopically, a huge mass measuring 14×13×8 cm had replaced the entire left lobe of his thyroid gland, with the mass also involving the right lobe. Multiple cystic spaces measuring up to 8.2 cm in the greatest dimension were present in the central portion of the mass and were filled with dirty necrotic fluid. The solid part of the mass was well demarcated, round in shape and had tan, firm, and smooth cut surfaces. Although the mass abutted the thyroid capsule, there was no extension to the surrounding soft tissues or other organs (Fig. 1C). The tumor consisted histologically of two well-delineated components. The center contained a cystic lesion lined by atypical squamous epithelium. The squamous cells showed unequivocal pleomorphism and extensive keratinization, resulting in a diagnosis of squamous cell carcinoma with cystic degeneration (Fig. 2A). The peripheral part of the mass was composed of a solid growth of thyroid follicles. The two components were in direct contact, but they did not show areas of transition (Fig. 2B). The follicular lesion was cellular and displayed microfollicular and trabecular growth patterns. The follicle cells contained abundant eosinophilic cytoplasm and mildly atypical round nuclei with smooth contour. Small nucleoli were frequently seen, whereas mitotic figures were rare (Fig. 2C). The peripheral boundary of the follicular lesion was surrounded by a thick fibrous capsule, and showed a generally expansile, but partly invasive, growth pattern (Fig. 2D). Immunohistochemical examination showed that the squamous cell carcinoma was positive for p63, thyroid transcription factor 1 (TTF-1), and p53 and had a 20% Ki-67 labeling index (Fig. 3A-E). The follicular carcinoma component was diffusely positive for galectin-3 (Fig. 3I), TTF-1, and CD56 (Fig. 3F) and negative for p53, cytokeratin 19 (Fig. 3G), and HBME-1 (Fig. 3H), and the Ki-67 labeling index was 2%. Of the 18 dissected cervical lymph nodes, none was found to contain a metastatic tumor. The patient was lost to follow up 1 month later, but Korean National Health Insurance data show that he survived for 15 months after surgery.
CASE REPORT
- We describe a patient with a combined squamous cell carcinoma and follicular carcinoma of the thyroid. Because one of these components was observed on aspiration cytology and the other by core needle biopsy, a preoperative diagnosis could not be determined. The resected tumor also required detailed consideration. Differential diagnoses included anaplastic transformation from a follicular carcinoma, metastatic squamous cell carcinoma to a follicular carcinoma, follicular carcinoma with squamous differentiation, and primary thyroid squamous cell carcinoma intermingling with follicular carcinoma.
- Patients with anaplastic carcinoma usually present with a rapidly growing neck mass and have a median survival of 6 months [6]. Histologically, anaplastic carcinomas tend to show widely invasive growth, extensive tumor necrosis, marked nuclear pleomorphism, and high mitotic activity. When anaplastic carcinomas show squamoid differentiation, squamoid cells are usually mixed with spindle or giant cells [6]. Our patient had a slow growing neck mass and mild to moderate cellular atypia without giant cells, spindle cells or atypical mitoses. Moreover, keratinizing squamous cell nests were in contact with the follicular carcinoma without transitional areas.
- Metastasis to the thyroid gland is rare, with fewer than 2% of thyroid tumors being metastatic squamous cell carcinomas to the thyroid gland [7]. Metastases to the thyroid gland generally develop within two years of identification of the primary site [8]. Our patient showed no evidence of squamous cell carcinoma of non-thyroidal origin. Squamous metaplasia has been reported from both benign and malignant thyroid lesions, including Hashimoto’s thyroiditis, follicular adenoma, and papillary thyroid carcinoma [3,9], but there are no reports of squamous metaplasia transforming into squamous cell carcinoma in thyroid tissue. In many cases, the de novo appearance of primary squamous cell carcinoma from follicular epithelium without prior metaplastic change is considered likely [10]. Our patient showed no evidence of squamous metaplasia, and the border between the two components was abrupt. Overall features of the case led to a diagnosis of PSCCT combined with follicular carcinoma.
- PSCCT is reported to have an aggressive clinical course, similar to that of anaplastic carcinoma. However, the two prior case reports of mixed squamous cell carcinoma and follicular carcinoma of the thyroid showed less aggressive courses. One report described a 56-year-old man with a hoarse voice and a rapidly growing mass over 3 months on the right side of the thyroid gland. The resected 3-cm thyroid mass was found to be a well differentiated squamous cell carcinoma intermingled with follicular carcinoma. Metastatic follicular carcinomas were found in the left upper lung and T7 spine at that time and 7 years later, respectively, but the patient survived more than 8 years after surgery [5]. The other patient was a 67-year-old man with a 6 months history of progressive enlarging anterior neck mass. A right lobectomy was performed, yielding an 8-cm mass. Histologic examination showed a poorly differentiated squamous cell carcinoma mixed with follicular carcinoma. Despite the development of a metastatic follicular carcinoma in the right iliac crest 3 years after surgery, the patient survived more than 6 years after surgery [2]. The different biologic behaviors displayed by PSCCT and mixed squamous cell carcinoma and follicular carcinoma are not fully understood, but they could be related to the presence of follicular carcinoma as a barrier. It is noteworthy that the metastatic diseases in both cases involved follicular carcinoma alone. Our patient could have undergone a similar clinical course but died 15 months after surgery of unknown cause.
- In summary, we have described a patient with mixed squamous cell carcinoma and follicular carcinoma of the thyroid gland, including a discussion of its histogenesis and clinical differences from PSCCT.
DISCUSSION
Fig. 1.Neck computed tomography with enhancement, showing a 13×11-cm cystic lesion with irregular thick walls involving the left lobe of the thyroid gland (A). (B) Fusion whole body positron emission tomography (fluorine-18 fluorodeoxyglucose) showing abnormal uptake only in the left anterior neck mass. (C) Replacement of the thyroid gland by a large solid mass with central cystic changes (arrow, solid part consisting of follicular carcinoma; arrowhead, cyst wall lined by squamous cell carcinoma).
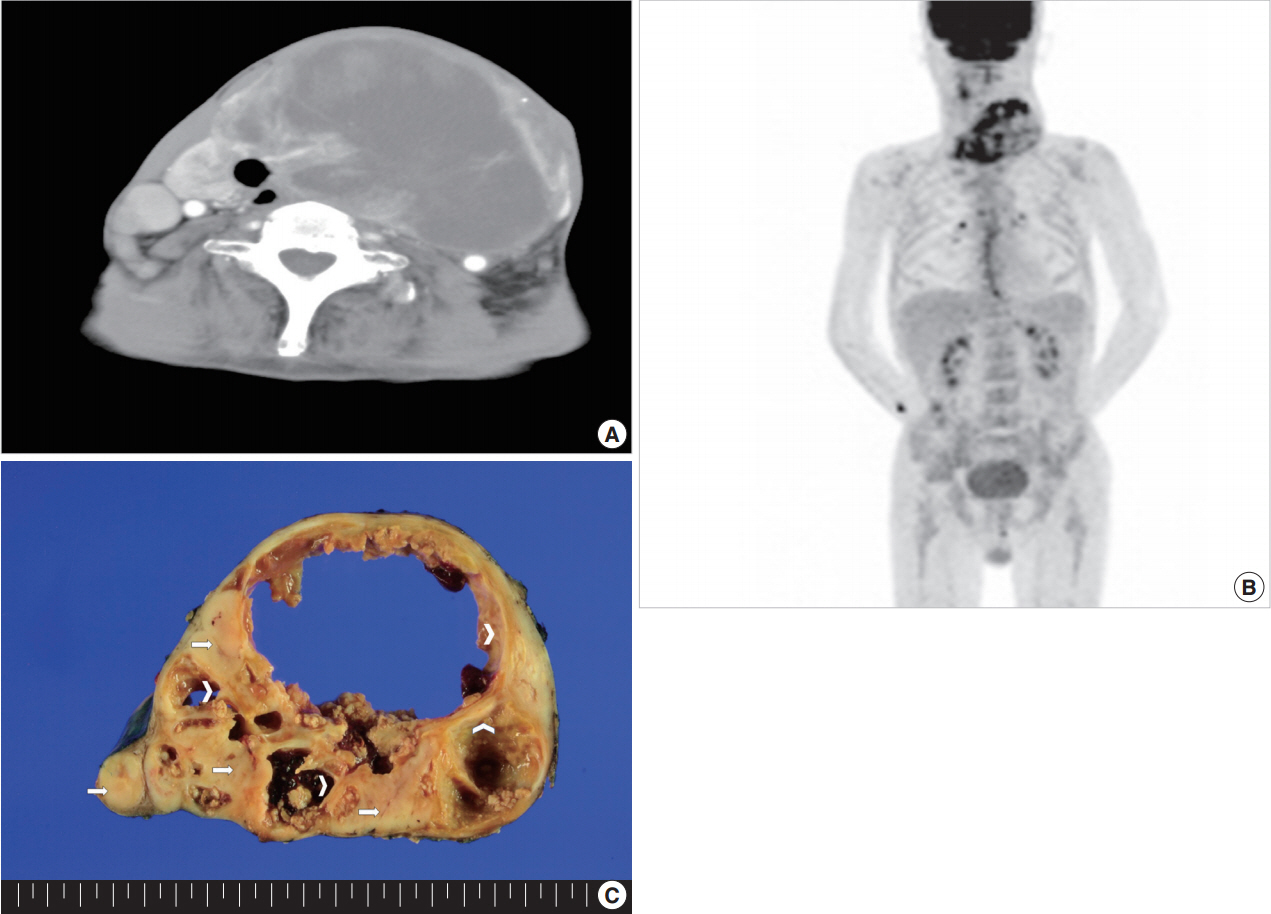

Fig. 2.Histologic examination of the mass, showing it is composed of a cystic zone lined by a well-differentiated squamous cell carcinoma (A) and a solid zone consisting of an invasive follicular carcinoma (C, D). (B) The squamous cell carcinoma nests are in direct contact with the follicular carcinoma, but the two elements are well distinguished.
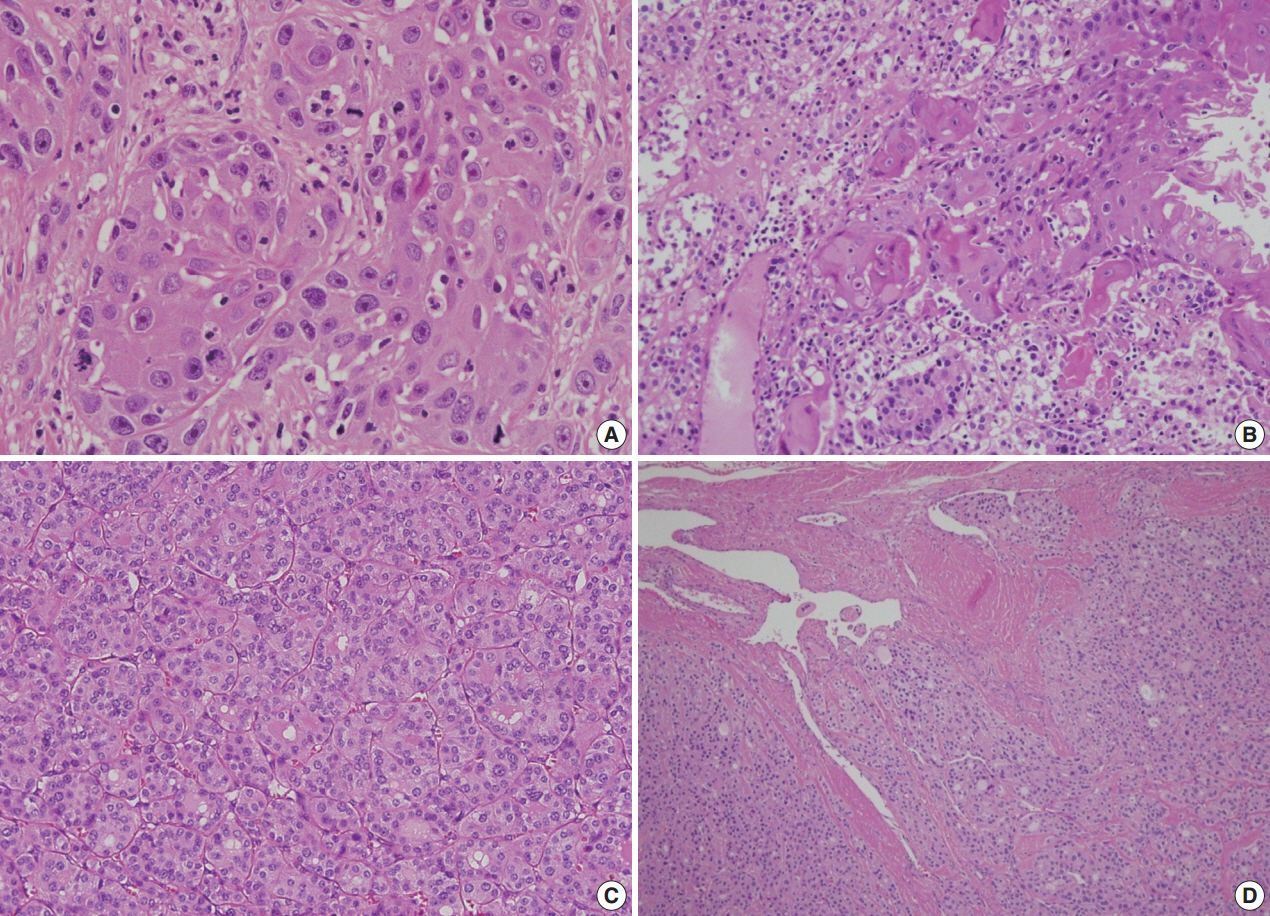

Fig. 3.Immunohistochemical staining of portions of the mass. The squamous cell carcinoma is positive for p63 (A), galectin-3 (B), thyroid transcription factor 1 (C), and p53 (E), and had a Ki-67 labeling index of 20% (D). The follicular carcinoma is diffuse positive for CD56 (F) and galectin-3 (I) and negative for cytokeratin 19 (G) and HBME-1 (H).

- 1. Goldman RL. Primary squamous cell carcinoma of the thyroid gland: report of a case and review of the literature. Am Surg 1964; 30: 247-52. PubMed
- 2. Mercante G, Marchesi A, Covello R, Dainese L, Spriano G. Mixed squamous cell carcinoma and follicular carcinoma of the thyroid gland. Auris Nasus Larynx 2012; 39: 310-3. ArticlePubMed
- 3. Sanchez-Sosa S, Rios-Luna NP, Tamayo Bdel R, Simpson K, Albores-Saavedra J. Primary squamous cell carcinoma of the thyroid arising in Hashimoto’s thyroiditis in an adolescent. Pediatr Dev Pathol 2006; 9: 496-500. ArticlePubMedPDF
- 4. Simpson WJ, Carruthers J. Squamous cell carcinoma of the thyroid gland. Am J Surg 1988; 156: 44-6. ArticlePubMed
- 5. Jung TS, Oh YL, Min YK, et al. A patient with primary squamous cell carcinoma of the thyroid intermingled with follicular thyroid carcinoma that remains alive more than 8 years after diagnosis. Korean J Intern Med 2006; 21: 73-8. ArticlePubMedPMC
- 6. Giuffrida D, Gharib H. Anaplastic thyroid carcinoma: current diagnosis and treatment. Ann Oncol 2000; 11: 1083-9. ArticlePubMed
- 7. Rausch T, Benhattar J, Sutter M, Andrejevic-Blant S. Thyroid carcinoma with papillary and squamous features: report of a case with histogenetic considerations. Pathol Res Pract 2010; 206: 263-9. ArticlePubMed
- 8. Chen H, Nicol TL, Udelsman R. Clinically significant, isolated metastatic disease to the thyroid gland. World J Surg 1999; 23: 177-80. ArticlePubMedPDF
- 9. Albores-Saavedra J, Wu J. The many faces and mimics of papillary thyroid carcinoma. Endocr Pathol 2006; 17: 1-18. ArticlePubMed
- 10. Sahoo M, Bal CS, Bhatnagar D. Primary squamous-cell carcinoma of the thyroid gland: new evidence in support of follicular epithelial cell origin. Diagn Cytopathol 2002; 27: 227-31. ArticlePubMed
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- Efficacy of combined immunotherapy and targeted therapy in overcoming barriers to postoperative recurrence in squamous subtype anaplastic thyroid carcinoma with abscess: a case report and literature review
Shuyun Jiang, Xiaowu Wang, Zhijun Ma
Frontiers in Oncology.2025;[Epub] CrossRef - State of Knowledge About Thyroid Cancers in the Era of COVID-19—A Narrative Review
Agnieszka Bronowicka-Szydełko, Maciej Rabczyński, Ilias Dumas, Żanna Fiodorenko-Dumas, Beata Wojtczak, Łukasz Kotyra, Irena Kustrzeba-Wójcicka, Łukasz Lewandowski, Beata Ponikowska, Aleksandra Kuzan, Joanna Kluz, Andrzej Gamian, Katarzyna Madziarska
Biomedicines.2024; 12(12): 2829. CrossRef - Primary and metastatic squamous cell carcinoma of the thyroid gland: Two case reports
Xing Zhao, Pengyu Hao, Jiangbei Tian, Jirun Sun, Dawei Chen, Zhehui Cui, Libo Xin, Yanmin Song, Gang Zhang
Open Life Sciences.2022; 17(1): 1148. CrossRef - Aggressive Thyroid Gland Carcinoma: A Case Series
JP Dworkin-Valenti
Archives of Otolaryngology and Rhinology.2017; : 129. CrossRef
 PubReader
PubReader ePub Link
ePub Link-
 Cite this Article
Cite this Article
- Cite this Article
-
- Close
- Download Citation
- Close
- Figure
Combined Squamous Cell Carcinoma and Follicular Carcinoma of the Thyroid
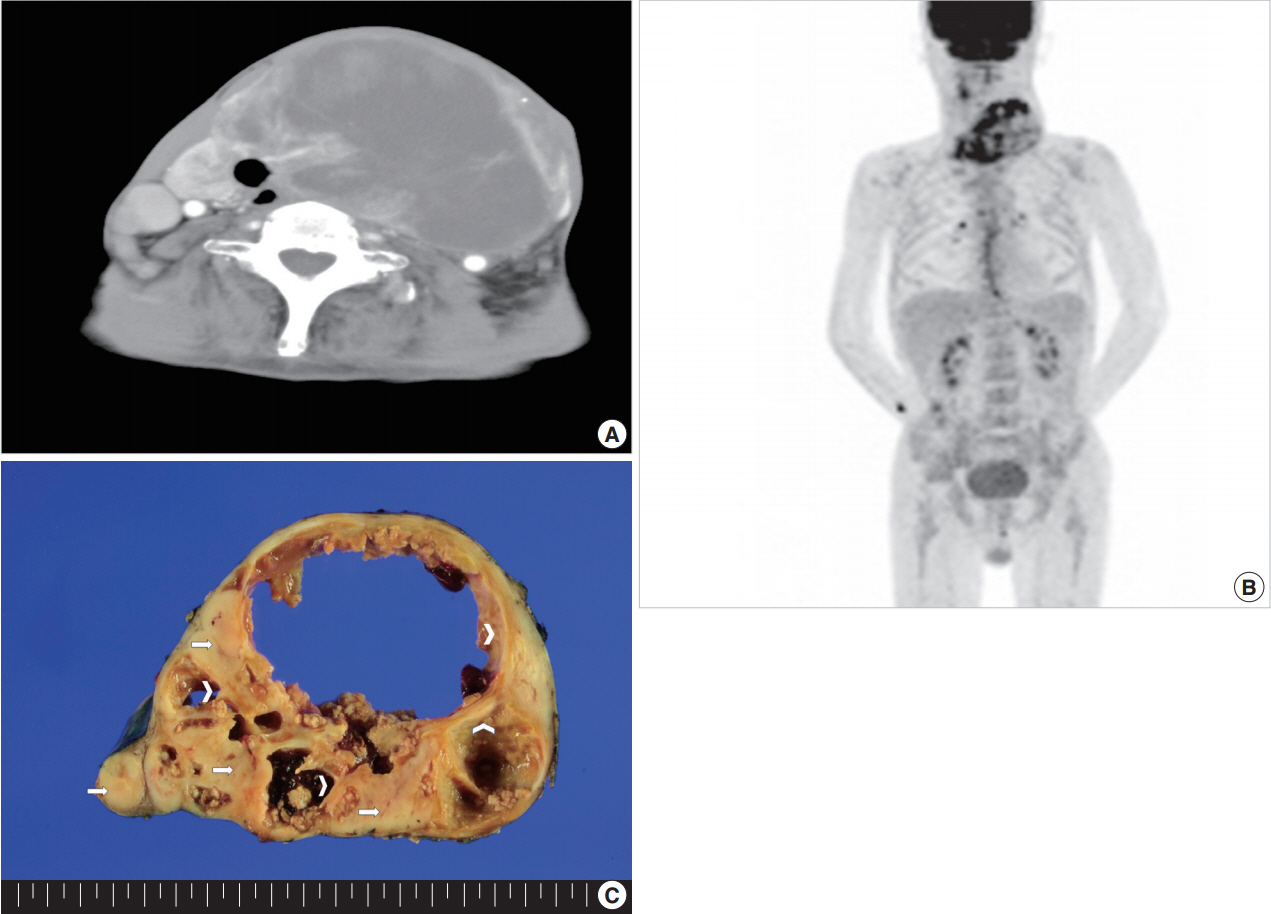
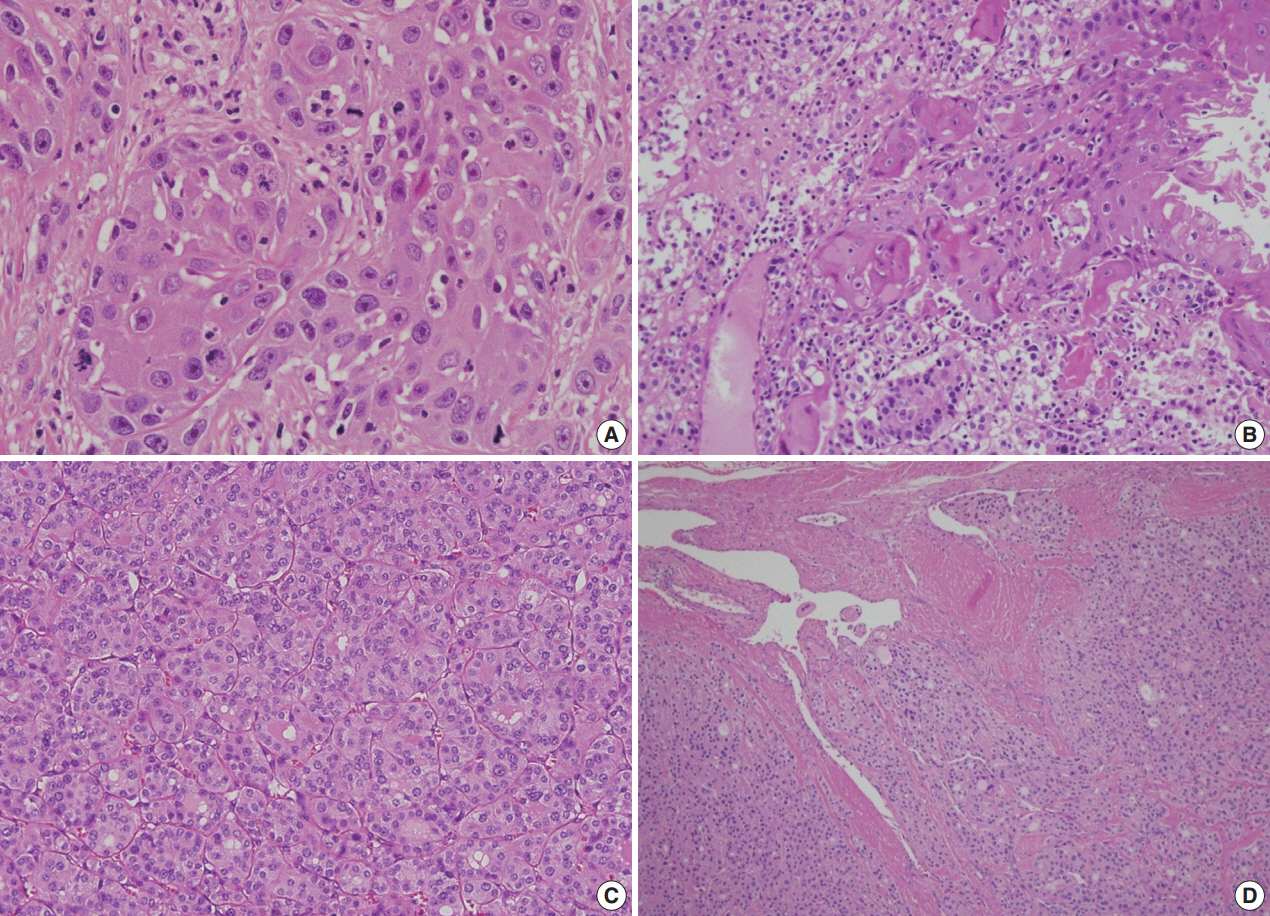
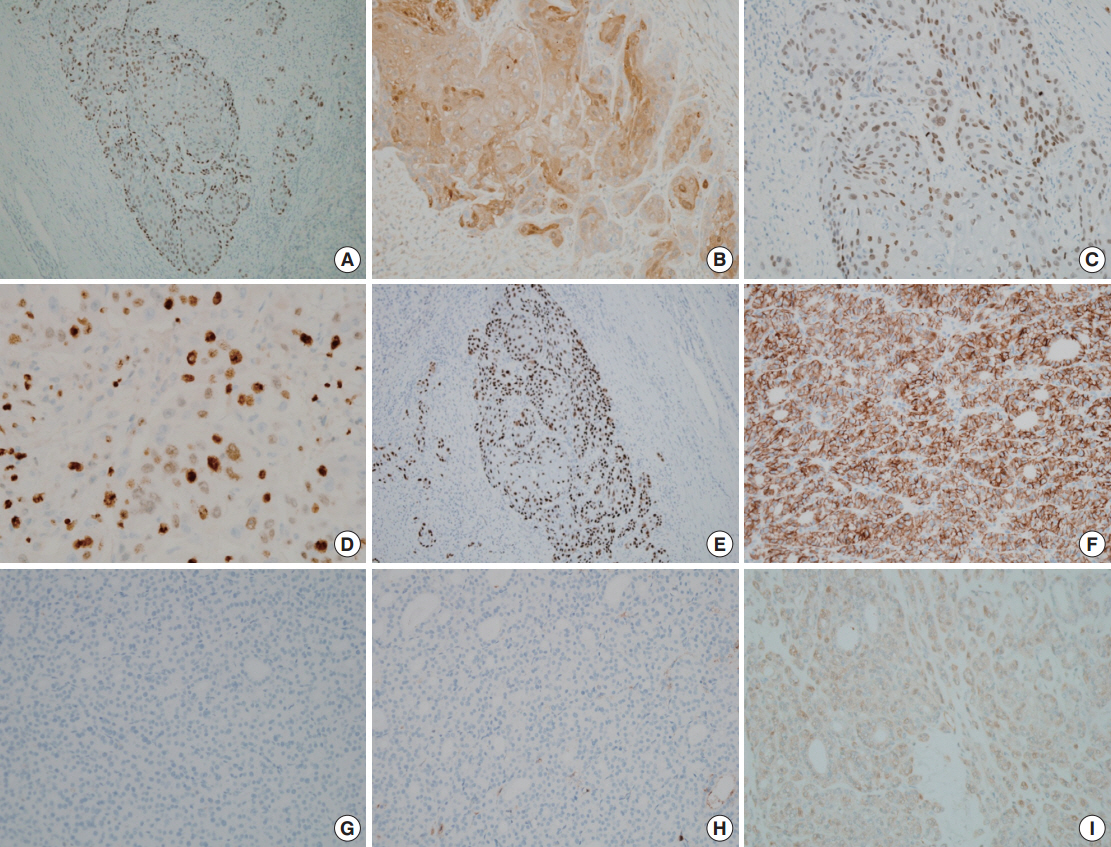
Fig. 1. Neck computed tomography with enhancement, showing a 13×11-cm cystic lesion with irregular thick walls involving the left lobe of the thyroid gland (A). (B) Fusion whole body positron emission tomography (fluorine-18 fluorodeoxyglucose) showing abnormal uptake only in the left anterior neck mass. (C) Replacement of the thyroid gland by a large solid mass with central cystic changes (arrow, solid part consisting of follicular carcinoma; arrowhead, cyst wall lined by squamous cell carcinoma).
Fig. 2. Histologic examination of the mass, showing it is composed of a cystic zone lined by a well-differentiated squamous cell carcinoma (A) and a solid zone consisting of an invasive follicular carcinoma (C, D). (B) The squamous cell carcinoma nests are in direct contact with the follicular carcinoma, but the two elements are well distinguished.
Fig. 3. Immunohistochemical staining of portions of the mass. The squamous cell carcinoma is positive for p63 (A), galectin-3 (B), thyroid transcription factor 1 (C), and p53 (E), and had a Ki-67 labeling index of 20% (D). The follicular carcinoma is diffuse positive for CD56 (F) and galectin-3 (I) and negative for cytokeratin 19 (G) and HBME-1 (H).
Fig. 1.
Fig. 2.
Fig. 3.
Combined Squamous Cell Carcinoma and Follicular Carcinoma of the Thyroid

 E-submission
E-submission





