Search
- Page Path
- HOME > Search
Original Articles
- Progastrin, annexin A2, and tumor-associated macrophages in gastric adenocarcinoma
- Konstantinos Christofidis, Rodanthi Fioretzaki, Stylianos Mavropoulos Papoudas, Nikolaos Charalampakis, Nikolaos Kavantzas, Dimitrios Schizas, Stratigoula Sakellariou
- J Pathol Transl Med. 2026;60(2):263-279. Published online March 10, 2026
- DOI: https://doi.org/10.4132/jptm.2025.12.20
- 1,221 View
- 121 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material - Background
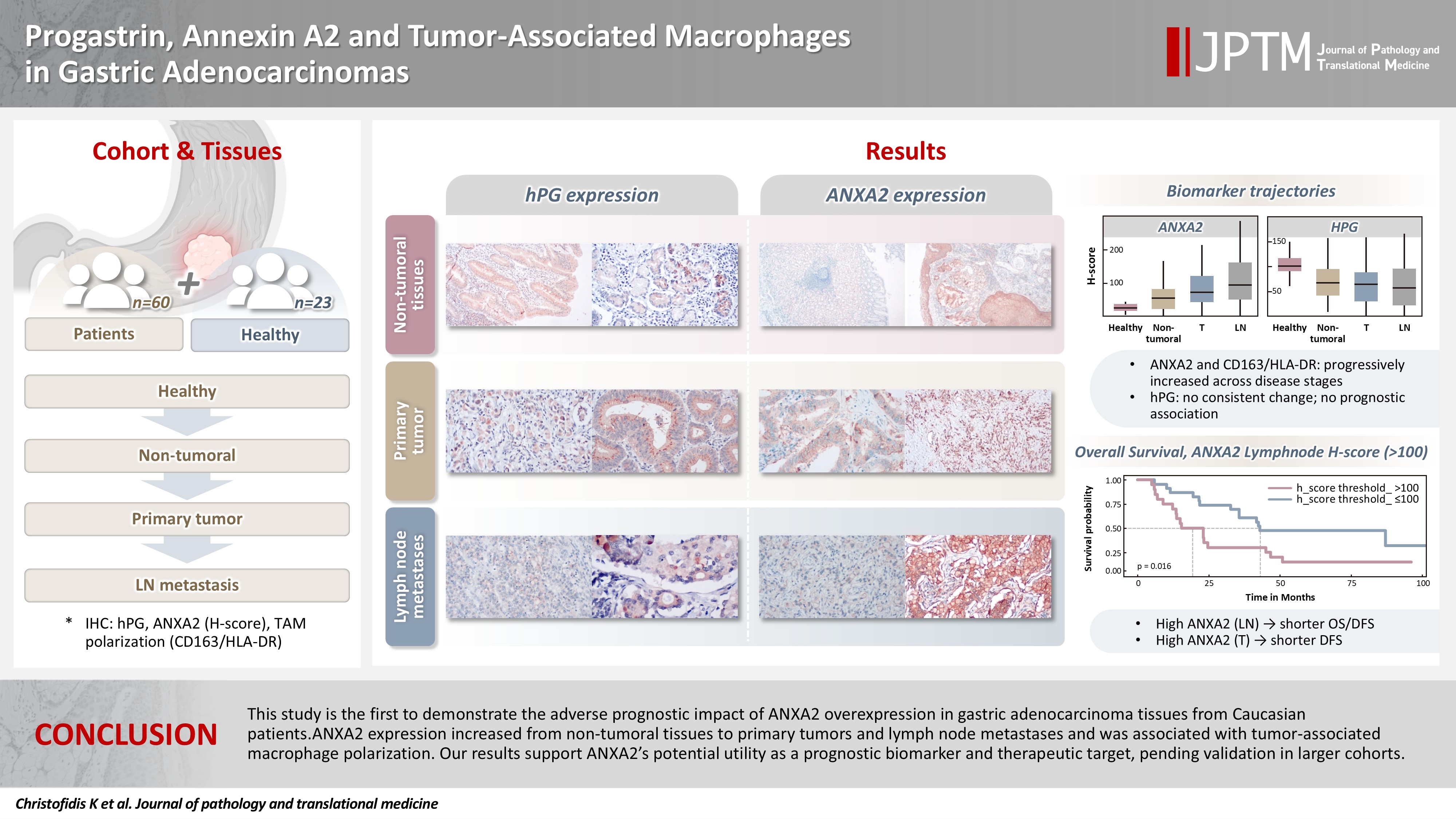
Gastric adenocarcinoma is a major cause of cancer mortality worldwide, and reliable biomarkers remain insufficient. This study investigates the immunohistochemical expression of progastrin (hPG) and annexin A2 (ANXA2) and the polarization of tumor-associated macrophages in gastric adenocarcinoma to explore their potential prognostic and biological significance. Methods: A retrospective analysis was conducted on formalin-fixed, paraffin-embedded tissue samples from 60 patients with gastric adenocarcinoma (primary tumors, lymph node metastases, and non-tumoral gastric mucosa) and gastric biopsies from 23 healthy controls. The expression of hPG and ANXA2 was quantified using the H-score, and the CD163/human leukocyte antigen–DR (HLA-DR) ratio was used to represent macrophage polarization (M2/M1). Statistical analyses included non-parametric tests, Spearman correlations, Kaplan-Meier survival curves, and Cox proportional-hazards models. Results: ANXA2 expression was significantly elevated in cancer cells from primary tumors and lymph node metastases, compared with the non-tumoral gastric mucosa tissues and gastric mucosa tissues from healthy controls. ANXA2 expression increased with the tumor grade. High ANXA2 levels were associated with shorter overall and disease-free survival, but they did not have independent prognostic value. Although hPG expression correlated positively with ANXA2, it showed no significant prognostic association. The CD163/HLA-DR ratio increased with tumor progression and negatively correlated with ANXA2, but it did not influence survival outcomes. Conclusions: This study is the first to demonstrate the adverse prognostic impact of ANXA2 overexpression in gastric adenocarcinoma tissues from Caucasian patients. Our results suggest that ANXA2 might have utility as a prognostic biomarker and therapeutic target, if further large-scale studies validate and expand our findings.
- Paricalcitol prevents MAPK pathway activation and inflammation in adriamycin-induced kidney injury in rats
- Amanda Lima Deluque, Lucas Ferreira de Almeida, Beatriz Magalhães Oliveira, Cláudia Silva Souza, Ana Lívia Dias Maciel, Heloísa Della Coletta Francescato, Cleonice Giovanini, Roberto Silva Costa, Terezila Machado Coimbra
- J Pathol Transl Med. 2024;58(5):219-228. Published online August 27, 2024
- DOI: https://doi.org/10.4132/jptm.2024.07.12
- 4,646 View
- 224 Download
- 2 Web of Science
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF - Background
Activation of the mitogen-activated protein kinase (MAPK) pathway induces uncontrolled cell proliferation in response to inflammatory stimuli. Adriamycin (ADR)-induced nephropathy (ADRN) in rats triggers MAPK activation and pro-inflammatory mechanisms by increasing cytokine secretion, similar to chronic kidney disease (CKD). Activation of the vitamin D receptor (VDR) plays a crucial role in suppressing the expression of inflammatory markers in the kidney and may contribute to reducing cellular proliferation. This study evaluated the effect of pre-treatment with paricalcitol on ADRN in renal inflammation mechanisms.
Methods
Male Sprague-Dawley rats were implanted with an osmotic minipump containing activated vitamin D (paricalcitol, Zemplar, 6 ng/day) or vehicle (NaCl 0.9%). Two days after implantation, ADR (Fauldoxo, 3.5 mg/kg) or vehicle (NaCl 0.9%) was injected. The rats were divided into four experimental groups: control, n = 6; paricalcitol, n = 6; ADR, n = 7 and, ADR + paricalcitol, n = 7.
Results
VDR activation was demonstrated by increased CYP24A1 in renal tissue. Paricalcitol prevented macrophage infiltration in the glomeruli, cortex, and outer medulla, prevented secretion of tumor necrosis factor-α, and interleukin-1β, increased arginase I and decreased arginase II tissue expressions, effects associated with attenuation of MAPK pathways, increased zonula occludens-1, and reduced cell proliferation associated with proliferating cell nuclear antigen expression. Paricalcitol treatment decreased the stromal cell-derived factor 1α/chemokine C-X-C receptor type 4/β-catenin pathway.
Conclusions
Paricalcitol plays a renoprotective role by modulating renal inflammation and cell proliferation. These results highlight potential targets for treating CKD. -
Citations
Citations to this article as recorded by- Attenuating amiodarone-induced lung toxicity by the vitamin D receptor activator paricalcitol in rats: targeting TLR4/NF-κB/HIF-1α and TGF-β/Smad signaling pathways
Aamal G. El-Waseif, Mahmoud Elshal, Dalia H. El-Kashef, Nashwa M. Abu-Elsaad
Naunyn-Schmiedeberg's Archives of Pharmacology.2026; 399(3): 4067. CrossRef - The gut-kidney axis in chronic kidney disease: a vicious cycle of microbial dysbiosis and uremic toxin accumulation
Ruohan Guan, Jiaxuan Peng, Qianxi Gu, Jiayi Xu, Jiaxin Zhang, Hamdalatu Adamu, Qiao Yang, Lu Wang, Gang Cao
Frontiers in Immunology.2026;[Epub] CrossRef - Perirenal fat differs in patients with chronic kidney disease receiving different vitamin D-based treatments: a preliminary study
Ana Checa-Ros, Antonella Locascio, Owahabanun-Joshua Okojie, Pablo Abellán-Galiana, Luis D’Marco
BMC Nephrology.2025;[Epub] CrossRef
- Attenuating amiodarone-induced lung toxicity by the vitamin D receptor activator paricalcitol in rats: targeting TLR4/NF-κB/HIF-1α and TGF-β/Smad signaling pathways
- Expression of Surfactant-D Protein and TNF-alpha in the Interaction of Pneumocystis Carinii and Alveolar Macrophages in Pneumocystis Carinii Pneumonia.
- Kun Young Kwon, Kwan Kyu Park, Chang Kwon Park, Young June Jeon, Eun Sook Chang
- Korean J Pathol. 1999;33(9):684-694.
- 1,961 View
- 13 Download
-
 Abstract
Abstract
 PDF
PDF - Alveolar macrophages participate in the host defense against P. carinii, but the mechanisms in degradation and clearance of the organism from lung has not been well established. We observed the transmission and scanning electron microscopic features and evaluated the expression of TNF-alpha and Surfactant-D in the interaction of P. carinii with alveolar macrophages. Expression of TNF-alpha and Surfactant-D in the experimentally induced P. carinii pneumonia in rat was examined by immunohistochemistry and immunoelectron microscopy. Electron microscopically, the alveolar macrophages phagocytized trophozoites and cysts of P. carinii micro-organisms. Immunohistochemically TNF-alpha was strongly expressed in the cytoplasms of alveolar macrophages. Postembedding immunogold labeling for Surfactant-D protein was expressed on the pellicles of trophozoites and cysts, P. carinii micro-organisms in the cytoplasms of macrophages, free floating surfactant materials and multilamellar bodies of type II epithelial cells. We conclude that alveolar macrophages interacted with P. carinii micro-organisms respond with increased expression of TNF-alpha. TNF-alpha may bind to P. carinii and exert a direct toxic effect upon the micro-organisms. Surfactant-D protein may augment binding of P. carinii to the alveolar macrophages and enhance the clearance of the micro-organisms.

 E-submission
E-submission


 First
First Prev
Prev



