Nodular Fasciitis of the Parotid Gland, Masquerading as Pleomorphic Adenoma
Article information
Abstract
It is difficult to distinguish nodular fasciitis (NF) from other neoplasm of the parotid gland, especially pleomorphic adenoma (PA) by fine needle aspiration cytology. A 39-year-old female noticed a mass in the parotid region. The aspirate material showed cohesive parts composed of the cells that had oval or spindle-shaped nuclei and relatively abundant cytoplasm and some cells with plasmacytoid features. The background substance was fibromyxoid. PA was diagnosed based on the cytologic findings. Subsequently, parotidectomy was performed and NF was diagnosed based on histologic and immunohistochemical findings. NF in the parotid region is rare and may be misdiagnosed as other benign or malignant tumors of the parotid gland. The clinical history of rapid growth and the presence of mitoses and inflammatory cells help to distinguish NF from PA. In addition, immunohistochemical stains for smooth muscle actin and CD68 are useful to confirm the diagnosis of NF.
Nodular fasciitis (NF) is a pseudosarcomatous, self-limiting reactive process composed of fibroblasts and myofibroblasts. NF is most common in young adults between 20 and 40 years of age and its preferential site is the upper extremities, followed by the trunk, especially the chest wall and back. NF in the head and neck is next in frequency and the head and neck is the most common site in infants and children. Most cases of NF measure less than 5 cm, but a few are up to 10 cm in diameter. They typically arise at the subcutaneous or fascial level, but intramuscular location and involvement of organs such as the parotid gland may be seen.
Because NF is rare in the parotid gland and has many cytological similarities to other benign or malignant tumors, cytological misinterpretation is common[1,2]. In particular, NF may be similar to pleomorphic adenoma (PA) in cytological features and is occasionally misdiagnosed as PA[3]. Therefore, we report a case which was diagnosed as PA in the parotid gland based on cytologic findings but was finally diagnosed as NF based on histopathologic findings.
CASE REPORT
The patient was a 39-year-old female who had a mass in the right parotid region. The mass was found 2 months previously and had subsequently showed a gradual increase in size. No history of trauma or injury was identified. On physical examination, a firm, mobile, well-circumscribed mass was palpable in the subcutaneous tissue of the right cheek. No nerve paralysis or paresthesia was identified. At the same time, the patient was diagnosed with papillary carcinoma in the right thyroid gland by fine needle aspiration cytology (FNAC). Before the patient underwent a right hemithyroidectomy, ultrasonography and FNAC were performed. Ultrasonography showed a 2.3-cm-sized irregular shaped mass in the right parotid gland with ill-defined margins. Subsequently, computed tomography scan showed a 1.9×1.3×1.2-cm-sized, heterogeneously enhancing lobulated mass (Fig. 1A). Laboratory examination data were within normal limits. FNAC was performed for the mass of the right parotid region.
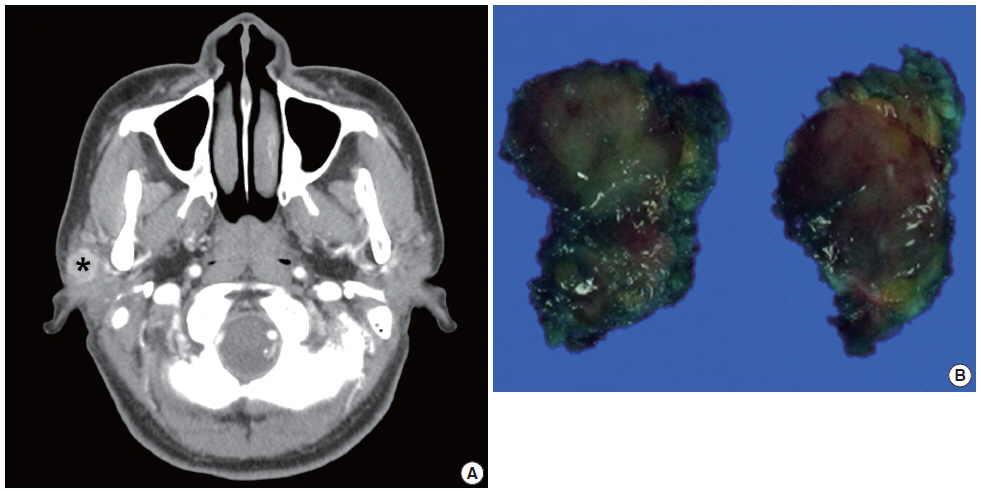
Computed tomography image and gross finding of the parotid mass. (A) Computed tomography scan shows a 1.9×1.3×1.2-cm-sized, heterogeneously enhancing lobulated mass (*). (B) The excised tumor is relatively well demarcated, but not encapsulated. The cut surface of the mass is solid and gray white.
Cytology
Papanicolaou staining from the aspirate showed moderately increased cellularity and was composed of isolated and cohesive cells with loose fibrous stroma. In cohesive parts, the cells consisted of short bundles or fascicles that did not form a characteristic pattern of arrangement. Most of the cells had oval or spindle-shaped nuclei and relatively abundant cytoplasm with rather indistinct cytoplasmic outline. Some cells had plasmacytoid features. Cellular pleomorphism was minimal. Inflammatory cells were also diffusely scattered (Fig. 2). Neither necrosis nor brisk mitotic activity was seen. Giemsa staining revealed that the mucoid-like stromal materials were metachromatic, which was reminiscent of the fibromyxoid matrix that is commonly seen in PA. Therefore, PA was initially diagnosed based on the cytologic findings.
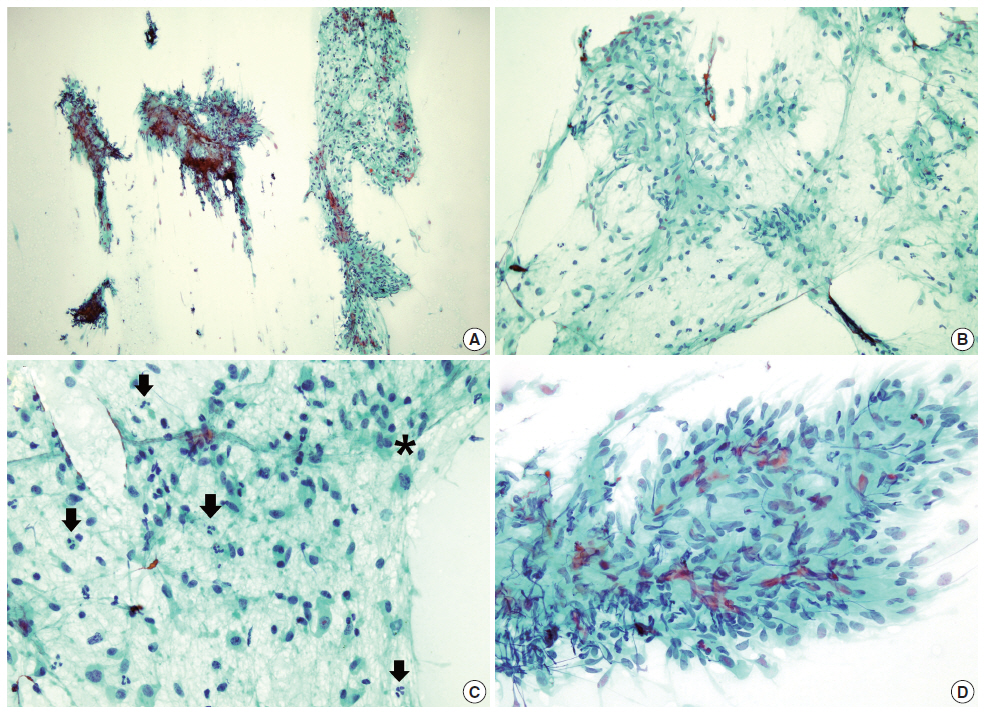
The cytologic findings of the parotid mass. (A) On low power, the aspirate shows moderately increased cellular areas and rather fibrous stroma. (B) The cells in the loose fibrocellular part have oval or spindle-shaped nuclei with abundant cytoplasm and are arranged in short loose bundles with myxoid background. (C) Some cells seem to have plasmacytoid features and many inflammatory cells are present (arrows). One mitotic cell is also found (*). (D) The cells in some areas consist of tight fascicles (A–D, Papanicolaou stain).
Histology
After FNAC diagnosis of the mass, superficial parotidectomy was performed for treatment and histological confirmation. The removed specimen was 2.3 g in weight and 2.4×1.8×1.2 cm in dimensions. On section, the tumor was well demarcated, but not encapsulated. The cut surface of the mass was solid and gray white (Fig. 1B).
Histologically, the mass was predominantly composed of relatively uniform spindle cells which showed moderate cellularity. These spindle-shaped cells were arranged in short fascicles arranged in haphazard pattern. Focally, a storiform growth pattern was also noted. The tumor cells had centrally located round to oval nuclei with finely granular chromatin and one or two inconspicuous nucleoli. The stromal part was predominantly collagenous with focal myxoid areas. Glands or squamous structures were not noted. Some inflammatory cells including lymphocytes, neutrophils, and histiocytes were found. Many small thin-walled vessels and extravasated erythrocytes were scattered diffusely. Regarding mitotic count, about 8 per 10 high power fields were found, but no atypical mitotic figure was identified (Fig. 3A–C). There was no necrosis. Combined with these histologic findings, NF was diagnosed. Additionally, immunohistochemical staining was performed. The spindle cells showed positivity to vimentin (1:1,000, V9, Zymed, South San Francisco, CA, USA), smooth muscle actin (1A4, 1:400, Dako, Glostrup, Denmark), but negativity to S-100 (1:1,000, polyclonal antibody, Novocastra, Newcastle Upon Tyne, UK), desmin (1:200, D33, Dako), CD34 (1:400, QBEnd-10, Dako), and pan-cytokeratin (1:500, AE1&AE3, Labvision, Fremont, CA, USA) (Fig. 3D–F). The results of immunohistochemical staining supported the diagnosis of NF
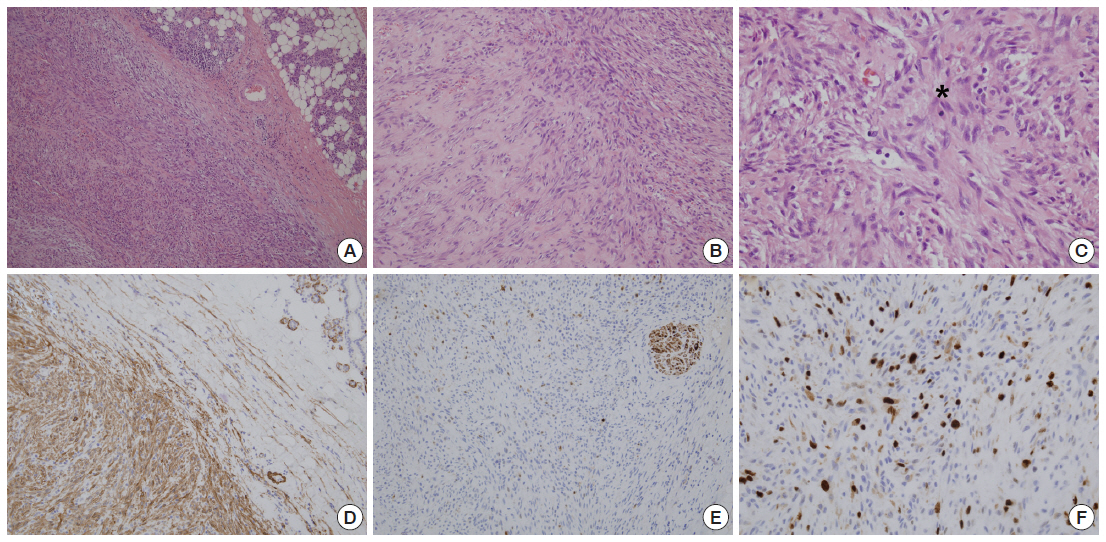
The histologic and immunohistochemical findings of the parotid mass. (A) The tumor is composed of relatively uniform spindle-shaped cells arranged in short fascicles. (B) The stroma focally shows loose myxoid features and diffusely scattered extravasated erythrocytes are also noted. (C) A mitotic cell (*) is present. The spindle cells are positive for alpha-smooth muscle actin (D), and negative for S-100 (E). (F) Ki-67 nuclear labeling cells are mildly increased.
DISCUSSION
Soft-tissue tumors or lesions of the head and neck are very rare compared with the far more common epithelial tumors that arise in these anatomic locations. Among those lesions, NF arising in the parotid gland is especially exceptional, so it is essentially impossible to give any estimate of their relative frequency.
Most cases of NF spontaneously resolve in 1–16 weeks after diagnosis based on FNAC findings[4]. It is thought that NF is triggered by a local injury or local inflammatory episode. However, only a small number of patients are confirmed to have histories of trauma[5]. In our patient, no trauma history was present.
Until now, there have been few reports about the cytologic features of NF arising in a salivary gland such as the parotid, and there are no definite criteria that aid pathologists in diagnosing NF of the gland based on cytology alone. According to the reports which have been published so far, the general cytology of NF shows aspirate material characterized by smears of low to moderate cellularity with a varying degree of myxoid background. The cells, which are composed of mainly fibrillary spindle cells, some plasmacytoid cells and ganglion-like cells, lie singly and in small clusters. Cytoplasm is present in moderate amounts and has a wispy flowing appearance. Nuclei are bland with an open chromatin pattern and small but distinct nucleoli. The cells do not show bona fide cytologic atypia or atypical mitoses.
The cytologic features of NF are often similar to many benign or malignant neoplasms arising in the parotid area, especially PA. PA is the most common salivary neoplasm and is 10 times more common in the parotid gland than in the submandibular gland. Therefore, if FNAC is performed from the tumor of the parotid area and the aspirated material shows many spindle shaped cells or plasmacytoid cells with a myxoid background, the cytologists who examine the sample may suspect at first glance that the tumor is PA, instead of NF. In comparison with NF, the typical cytology of PA consists of small round or cuboidal epithelial cells and occasional spindled or plasmacytoid mesenchymal cells with fibrillary, myxochondroid stromal substance. The tumor can also show a varied cytomorphologic appearance and a varied proportion between epithelial and mesenchymal components. However, because FNAC itself represents only a small portion of the tumor, the aspirated material of the mass can reveal a different proportion of cell components and limited cytologic features each time, compared with those of typical PA.
Some literature and case reports suggest criteria to distinguish NF from PA in cytology. If the aspirate material is composed of plasmacytoid cells on the whole, this more strongly PA. The presence of chondromyxoid matrix also helps to diagnose PA rather than NF. On the contrary, NF may be suspected when inflammatory cells or mitoses are seen in the aspirate materials[6]. In the present case, some plasmacytoid cells were seen, but their presence was not enough to consider PA. Although no ganglion-like cells or multinucleated cells were present, we found that inflammatory cells were frequently scattered in all slides and one mitotic cell was also present when reviewing our cytology slides (Fig. 2C). Otherwise, ductal or sheet-like epithelial structures which may be present in FNAC of PA were not noted in this case. The clinical course may also be important. In particular, rapid growth of the lesion is helpful for suggesting NF. Typically, it presents as a rapid-forming mass that is recognized in 1–2 weeks and it is out of ordinary for the lesion to be present for more than 3 months before surgery[7]. Immunohistochemistry can provide evidence to support the diagnosis of NF. Kong and Cha[8] reported that an immunohistochemical stain for smooth muscle actin showed cytoplasmic reactivity in the spindle cells, but immunohistochemical stain for CD68 can be also helpful when smooth muscle actin is negative, because of both the fibrohistiocytic and myofibroblastic characters of the cells. Therefore, if there is a clinical history of rapid tumor growth, NF can be suggested in the differential diagnosis, and pathologists subsequently can make an effort to find mitotic cells, multinucleated cells, ganglion-like cells or inflammatory cells such as neutrophils or lymphocytes. It may be helpful to perform immunohistochemical staining for alpha-smooth muscle actin and CD68 in case of the presence of these cytologic features suggesting NF.
In some cases, spindle cell-dominant NF may be difficult to cytologically distinguish from fibromatosis. However, the latter lacks a myxoid background, actual mitotic activity, and inflammation. Immunohistochemically, β-catenin may be helpful in the differential diagnosis, because nuclear positive staining for β-catenin is the specific finding in fibromatosis, whereas it is absent in NF. A benign peripheral nerve sheath tumor such as neurofibroma may also be included in the list of differential diagnoses. Neurofibroma commonly consists of a slender wavy spindle cell population of low to moderate cellularity which may be accompanied by a loose mucoid background. However, NF shows short fibrillary spindle cells often arranged in a whorllike pattern. In confusing cases, immunohistochemical stain for S-100 protein, using cell block or liquid-based preparation samples, is very helpful[9]. Among seriously misinterpreted cases of NF, a majority were misdiagnosed as low-grade sarcoma from frequent mitoses, cellular spindle cell population, and low-grade nuclear atypia[9], leading to unnecessary extensive surgical resection. Taking a careful look at the FNAC smear, however, NF can be distinguished by its more loosely arranged short bundles of fibroblasts, abundant isolated cells, prominent mucoid matrix, mild cytologic atypia, absence of atypical mitoses, and no necrotic background.
In conclusion, the cytologic diagnosis of NF in the parotid region may be difficult to distinguish from other neoplasms, especially PA, because of its extreme rarity and cytologic similarity. However, it is crucial to precisely diagnose NF since it is a self-limiting reactive process treated by conservative surgery, or even follow-up, whereas PA should be treated with superficial parotidectomy[10]. Therefore, careful evaluation for smears with spindle cell components is required to avoid a misdiagnosis of spindle cell-dominant lesion arising in the parotid region.
Notes
No potential conflict of interest relevant to this article was reported.
Acknowledgements
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2011-0012351).