Primary Myxoid Leiomyoma of the Liver
Article information
Abstract
Herein, we report a case of primary myxoid leiomyoma of the liver. A 60-year-old woman complained of upper abdominal fullness. Computed tomography showed a solid tumor (8 cm) in the liver. The patient underwent right hepatectomy and histological findings from the resected specimen revealed scattered bland spindle cells in a background of exuberant myxoid material. The tumor cells were immunoreactive for smooth muscle actin and desmin. No other lesions were found elsewhere in the body. Thus, the tumor was diagnosed as a primary myxoid leiomyoma of the liver.
Primary leiomyoma of the abdominal organs is common in the uterus and gastrointestinal tract; however, it rarely originates in the liver, and very few cases have been reported.1-9 Moreover, a primary "myxoid" leiomyoma of the liver is extremely rare. Indeed, we are only aware of one case: a 41-year-old woman who presented with a right abdominal mass, reported by by Yoon et al. in 1998.1 This report describes another case of primary myxoid leiomyoma arising from the right lobe of the liver, in a 60-year-old woman.
CASE REPORT
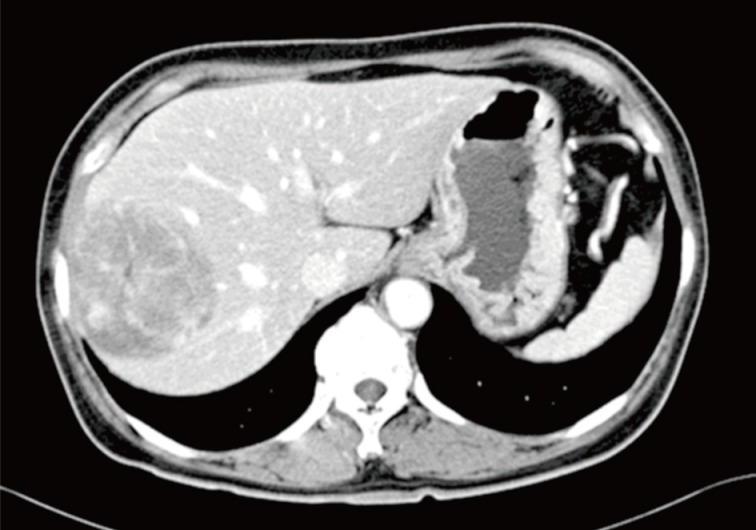
A 60-year-old woman complained of upper abdominal fullness. She was taking hypertension and diabetes medications and had undergone a thyroid lobectomy for thyroid papillary carcinoma 4 years earlier. She was discovered to have a low-echogenic mass in the right lobe of the liver on an ultrasound scan. Computed tomography of the abdomen revealed an 8-cm enhancing mass in segment 7 and 8 of the liver (Fig. 1). Liver function tests were unremarkable. Blood tumor markers, including carcinoembryonic antigen, α-fetoprotein, and carbohydrate antigen 19-9 were within normal ranges. The patient was not a hepatitis B carrier and her human immunodeficiency virus serology was nonreactive.
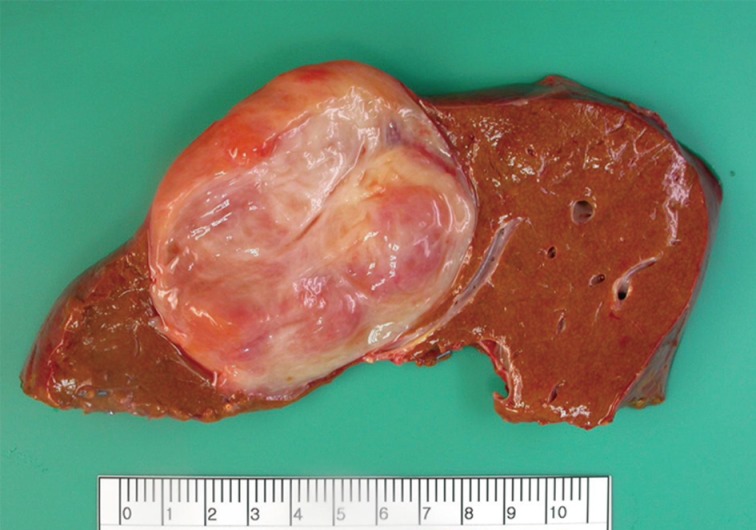
Results from a further magnetic resonance study suggested metastatic carcinoma from the thyroid. The gastrointestinal and gynecologic tracts were examined and no other lesions were detected. The patient underwent a right hepatectomy to remove the tumor. Grossly, there was an 8×5×4.5 cm, well-demarcated, unencapsulated and soft mass in the liver. The cut surface was yellow-to-tan, myxoid, and rubbery (Fig. 2). Histologically, the tumor was uniformly hypocellular and consisted of scattered, oval-to-spindle tumor cells and variable sized vessels. The stroma was myxoid and edematous (Fig. 3A). The spindled tumor cells had slightly eosinophilic cytoplasm. The nuclei of the spindle cells were cigar-shaped, blunt-ended, and showed vesicular chromatin. The nucleoli were inconspicuous. No cellular atypia or mitosis was observed (Fig. 3B). Inflammatory cells, especially mast cell and lymphocytes, were easily found.
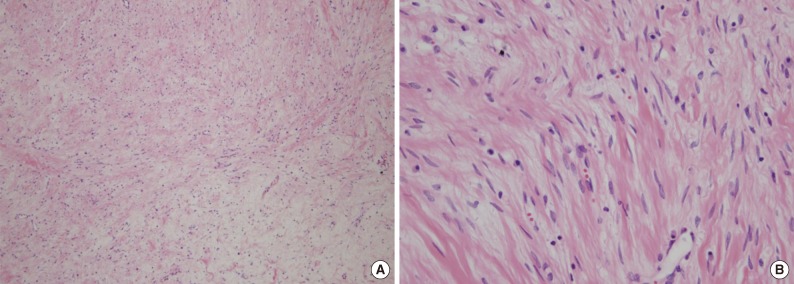
Histological features of myxoid leiomyoma. (A) Spindle tumor cells are scattered in a prominent myxoid stroma. (B) The spindle cells have an eosinophilic cytoplasm with bland, cigar-shaped nuclei.
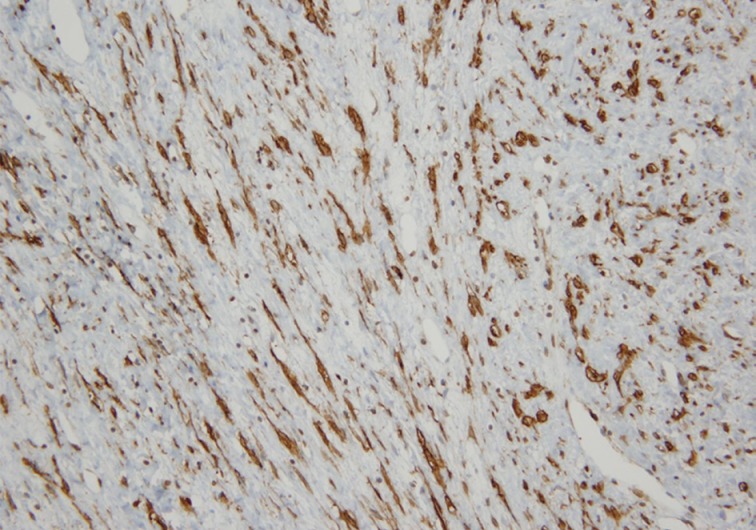
Immunohistochemically, the spindle cells stained positively for smooth muscle actin (SMA) (Fig. 4) and desmin, but were negative for anaplastic lymphoma kinase (ALK), S-100 protein, human melanoma black 45 (HMB45), c-kit, CD21, CD31, and CD34. Only a few tumor cells were labeled with ki-67. Estrogen receptor and progesterone receptor studies demonstrated negative staining. Thus, the histomorphological and immunophenotype findings were consistent with myxoid leiomyoma of the liver. The patient's postoperative course was uneventful and she has had no sign of recurrence to date.
DISCUSSION
Primary hepatic leiomyoma is very rare.1-9 In 1980, Hawkins et al.2 proposed some criteria to consider leiomyoma of the liver as a primary tumor. Namely, the tumor must be composed of smooth muscle cells, and must exclude the presence of a primitive smooth muscle tumor in the genitourinary tract and gastrointestinal areas. Radiographic studies, preoperative, and intraoperative pelvic examinations failed to reveal any other smooth muscle tumor in the present case.
The origin of hepatic leiomyomas is unclear. One site of origin may be smooth muscle cells in the walls of intrahepatic vessels. Key and Rao10 described a case of multiple smooth muscle tumors of the liver. Because typical cavernous hemangiomas were also present in the liver and the smooth muscle had merged with adjacent portal veins, the authors diagnosed as leiomyomatous hemangiomas of the liver. Others have suggested that the leiomyoma could be derived from the hepatic perisinusoidal cells (Ito cells), which are peculiar cells involved in myofibroblastic differentiation.1 The smooth muscle cells of the bile duct and the subcapsular mesenchymal cells of the liver may also be candidates.1,11
There are some cases of hepatic leiomyoma related to immunodeficiency, although it has also been described in immunocompetent patients. The reason for the association of primary hepatic leiomyomas in immunocompromised patients is unknown. Ha et al.12 suggested that immunocompromised hosts have an impaired immune surveillance mechanism, allowing tumors to develop unchecked, and Prevot et al.13 proposed that a weak immune system predisposes one to various viral infection, such as Epstein-Barr virus (EBV), that are associated with the pathogenesis of neoplasm. There have been reported cases of hepatic leiomyomas in acquired immunodeficiency syndrome and transplantation patients that have shown EBV positivity in the tumor.13-15 However, it also occurs in immunocompetent patients, as in our case.
Leiomyoma can undergo various types of degeneration, including hyaline, myxoid and cystic degeneration, and dystrophic calcification. Myxoid leiomyomas are a subtype of degenerated leiomyoma that show abundant acellular material, rich in acid mucin.16 This material is composed of acid mucopolysaccharides and glycoproteins. Some investigators believe that the gelatinous matrix is caused by cellular degeneration, with shrinkage of individual cells and an increase in interstitial myxoid material.17 Others assume that this material results from active secretion by tumor cells.18
To our knowledge, only one other case of primary myxoid leiomyoma of the liver has reported.1 As in the present case, the previous case involved a single mass in an immunocompetent woman. However, the two cases differ in several ways. Grossly, the tumor in our case was solid but the previous case showed central cystic degeneration with intracystic polypoid excrescences. Microscopically, the tumor in our case was diffusely hypocellular and showed abundant myxoid material throughout, making its diagnosis difficult. In contrast, the previous tumor showed densely arranged, long fascicles of spindle cells which are characteristic features of leiomyomas, in some areas.
The differential diagnosis includes myxoid tumors that occur in the liver, and more specifically, mesenchymal hamartoma, embryonal sarcoma, inflammatory myofibroblastic tumor, neurogenic tumors, and angiomyolipoma. Mesenchymal hamartoma is usually diagnosed in the first 2 years of life and nearly always by the age of 5. It consists of a disorganized arrangement of primitive mesenchyme, bile ducts, and hepatic parenchyme. The mesenchymal element consists of a mixture of spindle cells and collagen in a loose, myxoid stroma. This tumor is distinguished from myxoid leiomyoma by age of onset and the existence of disorganized bile ducts and hepatic parenchyme. In embryonal sarcoma, the tumor cells are stellate or spindle-shaped and may be compactly or loosely arranged in an abundant myxoid matrix. However, the tumor cells in embryonal sarcoma often have irregular, hyperchromatic nuclei, frequent mitosis and often bizarre, giant cells. Inflammatory myofibroblastic tumors are histologically distinctive lesions that occur primarily in the viscera and soft tissue of children and young adults. Although original descriptions of this lesion focused on its occurrence in the lung, inflammatory myofibroblastic tumors have been described in virtually every anatomic location. Because this tumor may be composed of cytologically bland spindle- or stellate-shape cells that are loosely arranged in a myxoid stroma, and scattered inflammatory cells, the differentiation from myxoid leiomyoma may pose a problem. Unlike inflammatory myofibroblastic tumors, however, the nuclei of leiomyoma are cigar-shaped and the tumor cells are ALK-negative. Neurogenic tumor, like schwannomas and neurofibromas, may closely resemble a myxoid leiomyoma, but they are consistently positive for S-100 protein. Some angiomyolipomas entirely consist of smooth muscle cells; however, these smooth muscle cells are positive for HMB45, as well as SMA.
Herein, we describe an extremely rare, primary myxoid leiomyoma of the liver, in which the myxoid changes were so exuberant that it required differentiation from other hepatic myxoid tumors.
Notes
No potential conflict of interest relevant to this article was reported.