SIRT1 Expression Is Associated with Good Prognosis in Colorectal Cancer
Article information
Abstract
Background
Silent mating type information regulation 2 homolog 1 (SIRT1), an NAD+-dependent deacetylase, might act as a tumor promoter by inhibiting p53, but may also as a tumor suppressor by inhibiting several oncogenes such as β-catenin and survivin. Deleted in breast cancer 1 (DBC1) is known as a negative regulator of SIRT1.
Methods
Immunohistochemical expressions of SIRT1, DBC1, β-catenin, surviving, and p53 were evaluated using 2 mm tumor cores from 349 colorectal cancer patients for tissue microarray.
Results
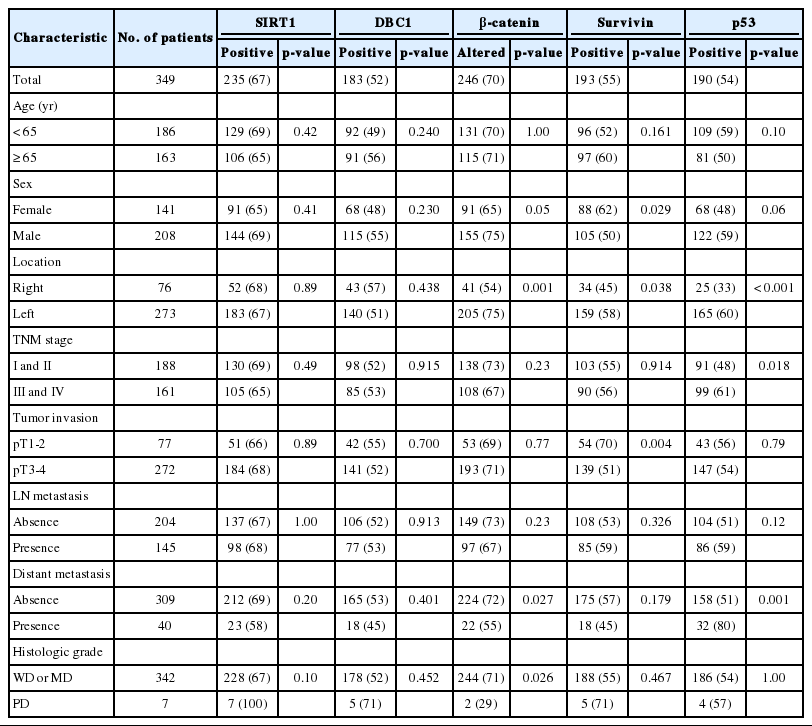
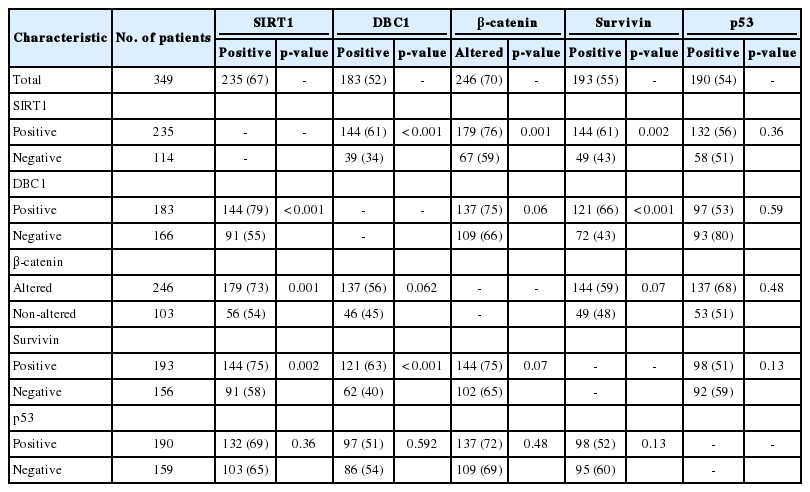
Overexpression of SIRT1, DBC1, survivin, and p53 was seen in 235 (67%), 183 (52%), 193 (55%), and 190 (54%) patients, respectively. Altered expression of β-catenin was identified in 246 (70%) patients. On univariate analysis, overexpression of SIRT1 (p=0.029) and altered expression of β-catenin (p=0.008) were significantly associated with longer overall survival. Expression of SIRT1 was significantly related to DBC1 (p=0.001), β-catenin (p=0.001), and survivin (p=0.002), but not with p53. On multivariate analysis, age, tumor stage, differentiation, and expression of SIRT1 were independent prognostic factors significantly associated with overall survival.
Conclusions
SIRT1 overexpression is a good prognostic factor for colorectal cancer, and SIRT1 may interact with β-catenin and survivin rather than p53.
Silent mating type information regulation 2 homolog 1 (SIRT1) is one of the mammalian homologue of silent information regulator 2 (Sir2) which is a nicotinamide adenine dinucleotide (NAD+)-dependent histone deacetylase that belongs to the class III histone deacetlylases.1 SIRT1 deacetylates not only histones but also many non-histone proteins which are involved in adaptation to calorie restriction, cell growth, apoptosis, cell senescence, and tumorigenesis.2 Recent reports have revealed that SIRT1 expression is increased in various human malignant tumors such as colon and breast cancer.3,4 However, the role of SIRT1 in malignant tumors is controversial. SIRT1 might act as a tumor promoter by inhibiting tumor suppressor genes such as p53,5 but SIRT1 might also act as a tumor suppressor by repressing several oncogenes or oncoproteins such as β-catenin and survivin.6,7 Thus, SIRT1 can serve as a tumor promoter or tumor suppressor, depending on the oncogenic pathways specific to particular tumors.
Colorectal cancer is one of the most common malignancies in the world. However, there are few studies revealing the clinical relevance of the expression of SIRT1 and related markers in colorectal cancer with human tissue.8,9 Although recent studies have revealed that SIRT1 plays an important role in colorectal tumorigenesis, the exact role of SIRT1 in colorectal tumorigenesis is still controversial.6,10 Some studies showed conflicting results regarding the relationship between SIRT1 and clinical features in several tumors depending on which proteins interacted with SIRT1.11,12
Deleted in breast cancer 1 (DBC1) was found to be homozygously deleted in human chromosome 8p21 in breast cancer.13 DBC1 enhances p53-mediated apoptosis through specific inhibition of deacetylase activity of SIRT.14 Recent studies have demonstrated that DBC1 expression is associated with poor prognosis in gastric and breast carcinoma.12,15 A previous study revealed that, in colorectal cancer, DBC1 overexpression is related to poor overall survival and poor prognostic indicators such as high tumor node metastasis (TNM) stage, poor histologic grade and presence of lymph node metastasis.16
β-catenin is a key regulator of Wnt signaling and is also known as an important regulator in cancer development. β-catenin is considered to play various roles in cadherin-mediated cell-to-cell adhesion, Wnt signaling transduction, and carcinogenesis.17 The altered expression of β-catenin and its relation to tumorigenesis have been investigated in several studies. One study reported that SIRT1 promotes cytoplasmic localization of β-catenin in colon cancer.6 Another study reported that altered expression of β-catenin was independently associated with poor prognosis in colon cancer.18 The diverse functions of β-catenin in tumorigenesis are thought to be associated with its altered expression. SIRT1 may be an important regulator of β-catenin in colorectal tumorigenesis.
Survivin, a member of the chromosomal passenger protein complex, is a small molecular weight protein that is an inhibitor of apoptosis, and is overexpressed in various cancers.19 SIRT1 is thought to inhibit survivin expression by deacetylation of histone H3 within nucleosomes at the survivin promoter.7 Previous studies have reported that survivin expression is impaired by loss of p53 function, which is often observed in cancer cells.20
In this study, we examined the expression of SIRT1 and related proteins such as DBC1, β-catenin, survivin, and p53, and their relationship to clinicopathologic features and prognosis. Through these results, we attempted to reveal the role of SIRT1 in colorectal cancer patients and their prognosis, as well as which proteins interact with SIRT1.
MATERIALS AND METHODS
Patients and samples
Three hundred forty nine patients who were diagnosed with colorectal cancer at the Korea University Guro Hospital from January 2002 to December 2009, who had surgical procedure such as right hemicolectomy, left hemicolectomy, sigmoidectomy, anterior resection, and abdominoperineal resection and had no prior history of neo-adjuvant chemotherapy were included in this study. Clinicopathologic data such as sex, age, and distant metastasis were collected from medical records. All of the cases were reviewed and classified according to the criteria of the World Health Organization (WHO) classification.21 Pathologic staging was reviewed based on the TNM staging system of the American Joint Committee on Cancer.22 The patients were grouped according to age, sex, location, TNM stage, tumor invasion, presence of lymph node metastasis, presence of distant metastasis, and histologic grade. The mean follow-up period was 55.3 months. The median age was 63 years (range, 27 to 88 years). This study obtained approval from the institutional review board of Korea University Guro Hospital (KUGGR-2011-015).
Tissue preparation and immunohistochemical staining
Hematoxylin and eosin-stained glass slides of selected patients were reviewed for construction of tissue microarray (TMA). Representative portions of tumor and normal mucosa were marked for TMA construction. One 2.0 mm core of tumor per case and one 2.0 mm core of representatively sampled normal mucosa were arrayed. TMA blocks were cut into 4 µm slices for immunohistochemical staining. A standard streptavidin-biotin peroxidase complex method was used. After deparaffinization and rehydration, slides were heated in a microwave oven for 15 minutes in 10 mM citrate buffer (pH 6.0) and treated with 3% hydrogen peroxide for 20 minutes. We used the Bond-maX autostainer (Leica, Wetzlar, Germany). The following antibodies were used: SIRT1 (1:50, clone H-300, Santa Cruz Biotechnology, Santa Cruz, CA, USA), DBC1 (1:100, polyclonal, Abcam, Cambridge, MA, USA), β-catenin (1:500, clone β-catenin-1, Dako, Carpinteria, CA, USA), survivin (1:400, polyclonal, Novus, Littleton, MA, USA), and p53 (1:500, clone DO-7, Novocastra, Newcastle upon Tyne, UK). Each case was evaluated by estimating the percentages and intensity of tumor cells showing a nuclear staining pattern. Immunostaning for SIRT1 was considered positive if 30% or more of the tumor cells were stained with an antibody, referring to other previous studies using the same antibody.12,15 Immunostaining for DBC1 was considered negative if more than 30% of tumor cells showed loss of expression, because DBC1 was expressed in all of the normal mucosa with nuclear staining pattern and at least moderate intensity; the cutoff value of 30% was chosen because DBC1 is known as negative regulator of SIRT1, so it was adjusted to the cutoff value of SIRT1. Survivin was also expressed in normal colonic mucosa with moderate intensity and is known to be inhibited by SIRT1. In the same manner, immunostaining for survivin was considered negative if more than 30% of tumor cells showed loss of expression. Immunostaining for β-catenin was considered positive if the tumor cells showed cytoplasmic and/or nuclear expression, so called altered expression. Immunostaining for p53 was considered positive if more than 10% of tumor cells were stained with the antibody, according to previous guidelines.23
Statistical analysis
All statistical analyses were performed using IBM SPSS ver. 20 (IBM, Armonk, NY, USA). The end point of interest was overall survival. The endpoint of follow-up was the date of final contact or the date of death through July 2011. Correlations between immunohistochemical expression and the clinicopathological characteristics were analyzed by Pearson's χ2 tests. Overall survival was calculated as the time from the date of the surgery to the date of death or final contact. Univariate and multivariate analyses for overall survival were performed using the Cox proportional hazard model. In all statistical analyses, p<0.05 was considered statistically significant.
RESULTS
Expression of SIRT1 and related proteins and correlations with clinicopathologic characteristics
The expression of SIRT1 and DBC1 in colorectal cancer was observed in 235 (67%) and 183 (52%) patients with nuclear staining pattern, respectively. All of the sampled normal mucosa showed weak SIRT1 expression with nuclear staining. DBC1 was expressed in all of the normal mucosa with moderate intensity (Fig. 1). SIRT1 and DBC1 were not significantly related to clinicopathological features such as age, sex, location, stage, invasion depth of tumor, lymph node metastasis, distant metastasis, or histologic grade (Table 1).
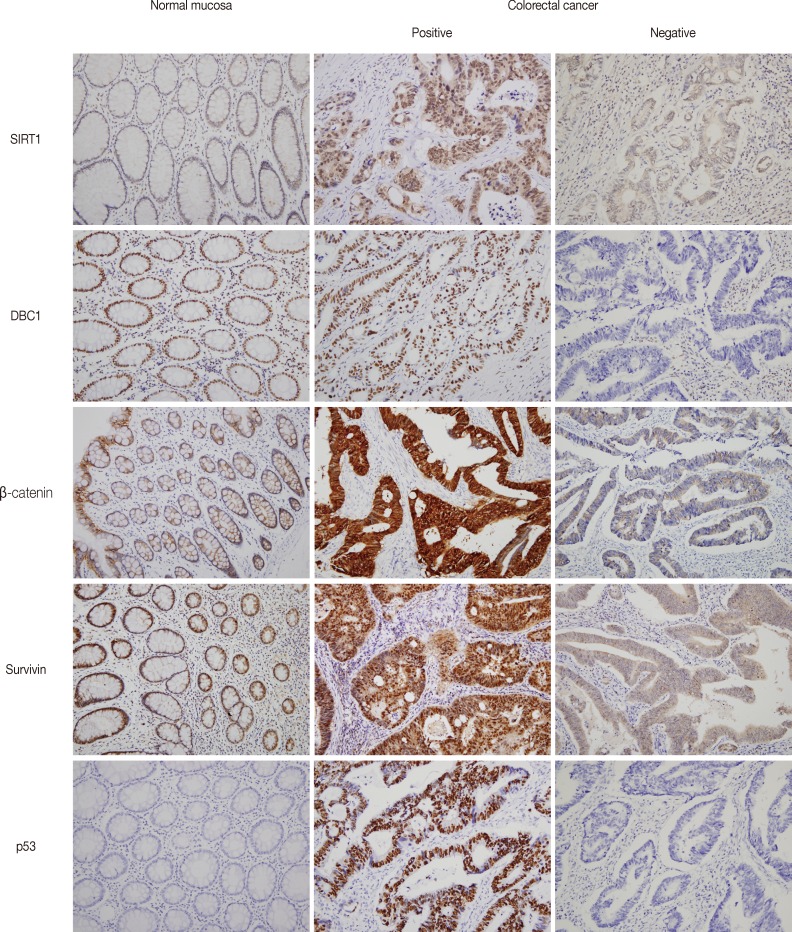
Immunohistochemical expression of silent mating type information regulation 2 homolog 1 (SIRT1), deleted in breast cancer 1 (DBC1), β-catenin, survivin, and p53 in normal colorectal mucosa and colorectal cancer. Normal colorectal mucosa shows SIRT1 expression with weak nuclear staining. DBC1 and survivin are expressed in all of the normal mucosa with moderate to strong intensity. β-catenin shows membranous staining pattern in normal colorectal mucosa. p53 is not expressed in normal mucosa (left panel). Representative figures of positive and negative expression of SIRT, DBC1, β-catenin, survivin, and p53 in adenocarcinoma are shown (middle and right panel, respectively).
All of the sampled normal mucosa showed membranous expression of β-catenin with weak or moderate intensity. The nuclear and/or cytoplasmic expression of β-catenin was observed in 246 (70%) patients (Fig. 1). The altered expression of β-catenin was correlated with left-located tumor (p=0.001), absence of distant metastasis (p=0.027), and non-poorly differentiation (p=0.026) (Table 1).
Survivin was expressed in all of the normal mucosa with moderate or strong intensity with nuclear staining pattern. The loss of expression of survivin was observed in 156 (45%) patients (Fig. 1). The loss of nuclear expression of survivin was correlated with higher T stage (p=0.004) (Table 1). Survivin also showed cytoplasmic expression in 70 (20%) patients. Cytoplasmic expression of survivin was not related to overall survival or clinicopathological features such as age, sex, location, stage, invasion depth of tumor, lymph node metastasis, distant metastasis, or histologic grade.
Expression of p53 was observed in 190 (54%) patients. Tumor cells were stained with nuclear staining pattern of strong intensity. Normal sampled mucosa showed no expression of p53 (Fig. 1). Expression of p53 was positively correlated with TNM stage (p=0.018) and distant metastasis (p=0.001) (Table 1).
Expression of SIRT1 and related proteins with their correlation
The expression of SIRT1 was significantly related to the expression of DBC1 (p<0.001), β-catenin (p=0.001), and survivin (p=0.002). The expression of DBC1 was significantly related to the expression of β-catenin (p=0.006) and survivin (p<0.001). β-catenin was not related to DBC1 or survivin. p53 was not related to any other proteins evaluated in this study. Expression of SIRT1 and related proteins and their correlations are summarized in Table 2.
Survival analysis
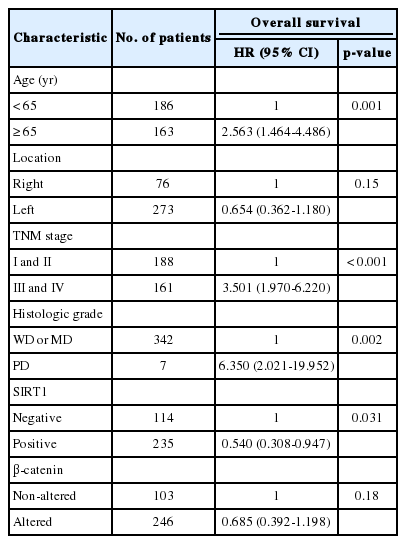
For univariate analysis of overall survival, a Cox proportional hazard model was used (Table 3). Among analyzed clinicopathologic features, old age (≥65) (p=0.005), high TNM stage (stages III and IV) (p<0.001), left-side tumor (p=0.034), invasion of more than the muscularis propria (p=0.024), presence of lymph node metastasis (p=0.001), presence of distant metastasis (p<0.001), and poorly differentiated histologic grade (p=0.001) were significantly associated with shorter overall survival. The overexpression of SIRT1 and altered expression of β-catenin showed significant association with better overall survival (p=0.029 and p=0.008, respectively). However, the expression of DBC1 (p=0.221), survivin (p=0.537), and p53 (p=0.218) was not associated with overall survival. Sex (p=0.101) was also not associated with overall survival.
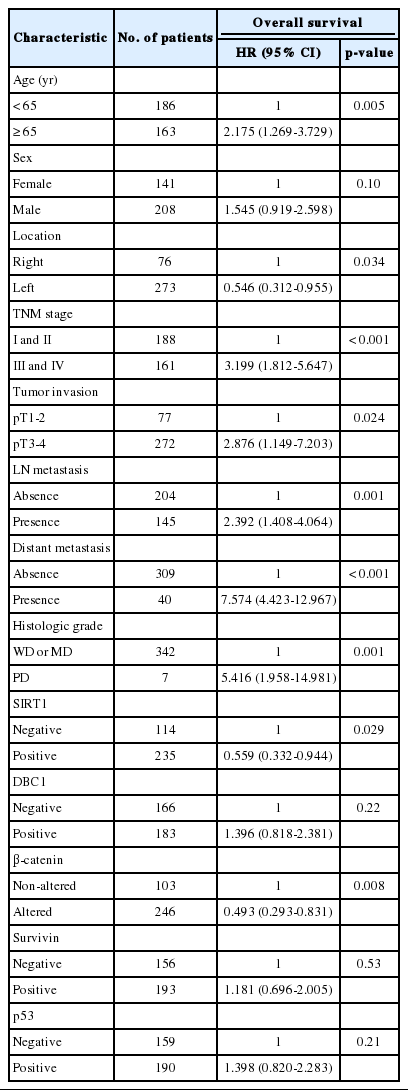
Clinicopathologic characteristics and their overall survival by univariate Cox proportional hazards regression analysis
Multivariate analysis was performed (Table 4). The factors considered in the analysis were age, tumor location, tumor stage, histologic grade, and the expression of SIRT1 and β-catenin. On multivariate analysis, age, tumor stage, histologic grade, and SIRT1 expression were independent prognostic factors significantly associated with overall survival.
DISCUSSION
The present study examined the immunohistochemical expressions of SIRT1, DBC1, β-catenin, survivin, and p53 in human colorectal cancer and their relationships with clinicopathologic factors and prognostic significance. Only a few studies that have examined expression of SIRT1 and its relationship to prognosis in colorectal cancer.8,9 Our study is the first to examine expression of SIRT1 and its relationship to expression of other related markers, DBC1, β-catenin, survivin, and p53 in colorectal cancer with human tissue.
As mentioned above, SIRT1 might act as a tumor promoter by inhibiting tumor suppressor genes and might also act as a tumor suppressor by repressing several oncogenes.5-7 A number of recent studies have revealed that SIRT1 expression was increased in many human malignant tumors.3,4 However, the role of SIRT1 in human malignant tumors is controversial. Some previous studies have reported that SIRT1 overexpression was associated with shorter overall survival or poor prognostic indicators in breast and gastric carcinoma.12,15 In contrast to these studies, one study reported that SIRT1 expression was gradually decreased as tumor progression and was associated with better overall survival in colorectal adenocarcinoma.8 Our study also revealed that expression of SIRT1 was significantly related to better overall survival. Taken together with these reports, our results suggest that SIRT1 expression is related to better prognosis in colorectal cancer and that SIRT1 acts differently depending on the specific organ or type of tumor involved.
Our study examined the expression of DBC1 and its relationship to prognosis in colorectal cancer. A recent study revealed that DBC1 overexpression was related to poor prognosis in colorectal cancer.16 However, our study failed to determine the relationship between the expression of DBC1 and prognosis in colorectal cancer. DBC1 is known as negative regulator of SIRT1.14 The present study found that the expressions of SIRT1 and DBC1 were positively correlated with each other (p<0.001). We could surmise that this result might be due to a close functional relationship between SIRT1 and DBC1 in carcinogenesis. However, further functional study will be needed to investigate the relationship between SIRT1 and DBC1.
β-catenin is a key regulator of Wnt signaling and is also known as an important regulator in cancer development.17 The adenomatous polyposis coli (APC) protein negatively regulates the Wnt signaling pathway.24 The APC gene mutation results in nuclear accumulation of β-catenin which becomes phosphorylated and is eventually degraded via the APC-dependent ubiquitin-proteosome pathway.25 In our study, normal colonic mucosa showed membranous staining of β-catenin. The altered expression of β-catenin was negatively correlated with the presence of distant metastasis and poorly differentiated histologic grade. The altered expression of β-catenin was related to better overall survival. This is a conflicting result to previous study which reported that altered expression of β-catenin was independently associated with poor prognosis in colon cancer.18 Therefore, the relationship between β-catenin expression and prognosis remains controversial, and further studies on Wnt/β-catenin and target gene activity and protein expression are needed.
Survivin is a protein that is an inhibitor of apoptosis, and is overexpressed in various cancers.19 In our study, normal colonic mucosa showed expression of survivin with nuclear staining pattern. Some studies have reported that altered expression of survivin was related to poor prognosis in several tumors.26,27 Another study reported that cytoplasmic expression of survivin was associated with a poor prognosis, but nuclear overexpression was related to a better prognosis.28 Our study also showed that the loss of nuclear expression of survivin was related to advanced invasion depth. Cytoplasmic expression of survivin was not associated with prognostic indicators; however, loss of nuclear expression of survivin may be related to poor prognosis in colorectal cancer.
p53 is well known as a key regulator of cell cycle progression and apoptosis. SIRT1 is considered to inhibit p53-mediated apoptosis through deacetylation of p53.5 However, there was no relationship between the expression of SIRT1 and p53 in our study and a previous study.11
Overexpression of SIRT1 was related to better overall survival. If SIRT1 predominantly acts like a tumor suppressor, it might have less effect on p53 in colorectal cancer. The finding that SIRT1 expression was correlated with altered expression of β-catenin and survivin which are considered to be oncoproteins, supports the hypothesis that SIRT1 predominantly acts as a tumor suppressor and might have less effect on p53 in colorectal cancer. Through multivariate analysis, we revealed that SIRT1 was an independent prognostic indicator for overall survival but β-catenin was not. This may also be due to the strong correlation between SIRT1 and β-catenin expression. However, to ensure our hypothesis, further functional study is needed.
In summary, expression of SIRT1 is associated with better overall survival in colorectal cancer patients. Although the exact role of SIRT1 in colorectal cancer is not clear, nor whether it is a tumor suppressor or promoter, the results of our study suggest that the expression of SIRT1 is related to better prognosis and predominantly acts like tumor suppressor in colorectal cancer patients.
Notes
No potential conflict of interest relevant to this article was reported.