In-house Manual Construction of High-Density and High-Quality Tissue Microarrays by Using Homemade Recipient Agarose-Paraffin Blocks
Article information
Abstract
Background
Self-made tissue punches can be effectively used to punch holes in blank recipient paraffin blocks and extract tissue cores from the donor paraffin blocks for the low-cost construction of tissue microarrays (TMAs). However, variable degrees of section distortion and loss of the tissue cores can occurs during cutting of the TMAs, posing technical problems for in-house manual construction of high-density TMAs. We aimed to update the method for in-house manual TMA construction to improve the quality of high-density TMAs.
Methods
Blocks of agarose gel were subjected to the standard tissue processing and embedding procedure to prepare recipient agarose-paraffin blocks. The self-made tissue punches and recipient agarose-paraffin blocks were used to construct TMAs, which were completely melted and re-embedded in paraffin to make finished TMA blocks.
Results
The donor tissue cores were completely integrated into the surrounding paraffin of the recipient blocks. This method enabled us to construct high-density TMAs with significantly less section distortion or loss of tissue cores during microtomy.
Conclusions
Simple and inexpensive construction of high-density and high-quality TMAs can be warranted by using paraffinized agarose gels as recipient blocks.
The tissue microarray (TMA) is an essential research tool for high-throughput in situ analysis of potential biomarker expression, in which a very large number of diagnostic samples can be analyzed simultaneously on a single paraffin block to limit run-to-run variability.1-4 Construction of TMAs usually involves the use of expensive commercially available microarray instruments such as automated or semi-automated tissue microarrayers. The cost for commercially available manual TMA devices and/or consumables such as pre-made recipient blocks can also be a barrier to some researchers in small institutions with limited funds.
Alternatively, self-made tissue punches and homemade recipient paraffin blocks can be effectively used for the construction of high-density and high-quality TMAs which can be employed in high-throughput screening and validation for analysis of in situ biomarker expression in diagnostic tissue samples.5-9 Section distortion and loss of tissue cores can occur to various degrees during cutting of TMA blocks and mounting of TMA sections on glass slides.5,8,10 These are critical but inevitable technical obstacles to using self-made tissue punches and conventional recipient paraffin blocks for in-house manual construction of high-density TMAs. We also experienced these problems when we first used self-made tissue punches made of cannula-piercing needles, disposable skin biopsy punches and cannula of bone marrow biopsy needle kits to construct large-format TMAs.11 The problems are mainly caused by incomplete integration of the tissue cores from donor blocks into the paraffin of the recipient block.
To prevent incomplete or folded TMA sections during cutting and mounting of the TMA sections for subsequent procedures, the tissue cores and the paraffin in the TMAs should be homogenized. This can be accomplished by enduring complete melting of the TMAs and re-embedding them in paraffin. Song et al.12 introduced a one-step complete melting technique to assure total integration of the tissue cores into the recipient paraffin block. Using recipient blocks made of paraffinized agarose gels, construction of high-quality TMA blocks is possible without limiting on the number or diameter of the tissue cores.13,14 In this study, we updated the methods for the in-house manual construction of high-density TMAs at low cost by using recipient agarose-paraffin blocks.
MATERIALS AND METHODS
Preparation of recipient blocks made of paraffinized agarose gel
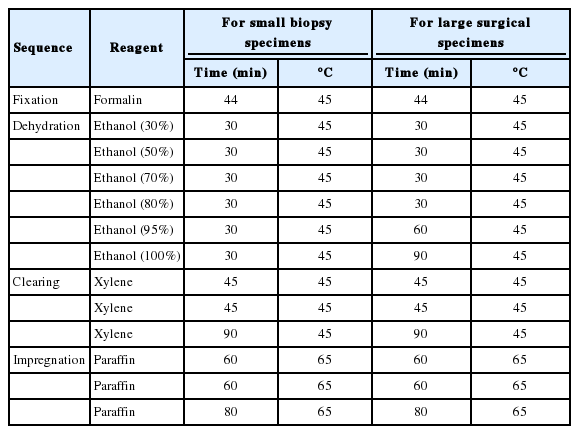
Agarose (Agarose Type I-A, Low EEO, Sigma-Aldrich, St. Louis, MO, USA) melted in distilled water at 0.5 to 5% (w/v) was poured into paraffin embedding molds and allowed to cool slowly at room temperature to make agarose gels 2-5 mm in thickness. The agarose gels were removed from the molds and redundant gel edges were trimmed to fit into the conventional plastic tissue cassette. Several agarose gels were placed in the automatic tissue processor (Premium Tissue Processing Leica PELORIS II, Leica Biosystems Melbourne, Pty Ltd., Sydney, NSW, Australia) and processed following procedures as in other clinical pathological samples: the gels and tissue specimens were fixed with 10% formalin, dehydrated through graded ethyl alcohol, cleared by xylene, and impregnated with paraffin (Table 1). The paraffinized agarose gels were embedded in conventional paraffin to make agarose-paraffin blocks.
Array construction using homemade tissue punches and recipient blocks
The agarose-paraffin blocks were trimmed using a microtome to expose the surface and prepare them for use as blank recipient blocks. Redundant tissue of normal tonsil, liver, pancreas, spleen, and colon was used to make donor paraffin blocks. Donor blocks made of redundant tissues of malignant melanoma were also used to allow easy orientation of the tissue arrays. The self-made tissue punches that we made previously were used to bore holes in the recipient blocks and extract tissue cores from donor blocks.11 For the construction of the TMAs with a 2 mm core diameter, an array of holes was bored in the recipient block using a recipient punch made of a modified bone marrow biopsy needle, which was guided by a sheet of grid paper attached to the surface of the blank recipient block with double-faced adhesive. Subsequently, a donor punch made of a disposable skin biopsy punch 2 mm in diameter was used to punch out tissue cores from the donor block and implant them into the pre-made holes in the recipient block (Fig. 1). For the construction of TMAs with a 1 mm core diameter, a recipient punch made of a modified 18-gauge needle was used to create a hole at the desired start position in the recipient block. Subsequently, a donor punch made of a modified 16-gauge needle was used to extract a tissue core from the donor block and implant it into a pre-made hole in the recipient block (Fig. 2). Then, a fresh recipient hole was punched with minimal gap between each core. Upon completion of the tissue arrays in the recipient agarose-paraffin block, a clean microscope slide was placed on the TMA face to apply firm yet gentle pressure to flatten any protrusions from the surface. The resulting blocks were placed in liquid paraffin in a conventional tissue embedding machine. When the paraffin of the recipient blocks was completely melted, TMAs were re-embedded in paraffin blocks.

Manual construction of a tissue microarray (TMA) with a 2 mm core diameter. (A) A sheet of grid paper is attached to the surface of the blank recipient block made of a paraffinized agarose gel with a double-faced adhesive. (B) A recipient punch made of a modified bone marrow biopsy needle is used to bore an array of holes in the recipient block. (C) After boring the holes, the grid paper is removed. Subsequently, a donor punch made of a disposable skin biopsy punch 2 mm in diameter is used to punch out tissue cores from the donor block and implant them into the pre-punched holes in the recipient block (D). Upon completion of the tissue array (E), the initial TMA is detached from the tissue cassette (F), a misplaced core is replaced with new one (G). The TMA is placed in the embedding mold to be re-melted (H) and re-embedded in paraffin (I).

Manual construction of a tissue microarray (TMA) with a 1 mm core diameter. (A, B) Tissue punches made of a modified 18 (*)- and 16 (**)-gauge needle are used to create holes in the recipient agarose-paraffin block, punch donor tissue cores and implant them into the pre-made holes. Once the TMA is completed (C), the TMA is detached from the cassette (D, E) and placed in the embedding mold in order to be re-melted and re-embedded in paraffin (F-H). The TMA block is trimmed (I) and cut for hematoxylin and eosin (J) and immunohistochemical staining (K, cytokeratin; L, CD20).
Hematoxylin and eosin and immunohistochemical staining
The finished TMA blocks were cut in 4 µm sections and routinely processed for hematoxylin and eosin (H&E) stain and immunohistochemical stains for cytokeratin (1:400, MNF116, pepsin antigen retrieval, Dako, Carpinteria, CA, USA), CD20 (1:500, H1 [FB1], heat-induced antigen retrieval, BD Pharmingen, San Diego, CA, USA), and CD138 (1:50, clone 5F7, heat-induced antigen retrieval, Neomarkers, Cheshir, UK). An automated immunohistochemical stainer (Ventana BenchMark XT, Ventana Medical Systems Inc., Tucson, AZ, USA) was used for immunohistochemistry. The Ventana i VEIW DAB detection kit was used to visualize in situ expression of the antigen. A digital slide scanner (VM600, Motic, Xiamen, China) was used to acquire the entire image of the H&E stained- and immunostained slides.
Preparation of pre-made ready-to-use recipient blocks using an electric hand drill and X-Y table
In order to make high-density recipient blocks with more precisely aligned holes, we used methods previously described by Vogel et al.15-18 Briefly, after trimming with a microtome to expose the surfaces of the agarose gels, holes in 1 and 2 mm in diameter were drilled in the blank recipient blocks as previously described.11 A microtome blade was used to detach the predrilled recipient blocks from the tissue cassette and the paraffin encasing the block.
RESULTS
Impregnation of agarose gels with paraffin using a conventional tissue processor
When the agarose gels were processed using the routine tissue processing protocol optimized for large surgical or biopsy tissue specimens in our histology laboratory, most of the paraffinized agarose gels retained their original shape and thickness. The greatest success in obtaining optimally paraffinized agarose gels resulted when agarose gels made on Friday and reserved in formalin solution along with other surgical pathological samples for the weekend were placed in the tissue processor for overnight processing on Monday. At that time, tissue processor reagents (ethyl alcohol and xylene) were replaced with new reagents, and the total number of surgical specimens needed to be processed was minimal. The resulting agarose gels appeared translucent with optimal consistency for easy handling when kept on a hot plate or in melted paraffin. However, the quality of paraffinized agarose gel cannot be always assured independently of the concentrations or thickness of the gels. Sometimes, the paraffinized agarose gels were variably distorted, especially when the tissue processing reagents were not fresh or when the gels were processed with a protocol optimized for small biopsy samples in which the time for dehydration and clearing steps was decreased by 90 min (Table 1). Although concentration (0.5 to 5%) or thickness (2 to 5 mm) of the agarose gels did not affect the quality of the paraffinized agarose gels (data not shown), 4 mm-thick 2% agarose gels were easier to manipulate for the preparation of the recipient blocks.
Construction of TMA blocks using recipient blocks made of paraffinized agarose gels
When the recipient blocks were trimmed to expose the surfaces of the paraffinized agarose gels, they appeared homogenously pearly white with similar consistency to embedding paraffin. The holes in the blocks were easily punched out in a fairly well-aligned array pattern by self-made tissue punches without damaging the paraffinized agarose gels. Complete homogenization between the tissue cores and the paraffin in the recipient blocks was assured by completely melting the initial TMAs and re-embedding them in paraffin to ready the finished TMA block for cutting. This procedure did not cause any distortion of the tissue array during the TMA construction. The TMAs were more easily cut without section distortion or loss of tissue cores during the cutting of the TMA blocks for H&E and immunohistochemical staining. We were able to construct 2 mm TMAs with 80 cores in 10×8 grid (Fig. 1) and 1 mm TMAs with 220 cores in a 20×11 grid (Fig. 2). Microscopic examination of the H&E-stained TMA sections revealed a thin layer of transparent agarose gel retained outside the tissue core on the glass slide. When the TMA sections were immunohistochemically stained for cytokeratin using protease for epitope retrieval, the agarose gel was also found to be retained on the slide. However, the agarose gel retained outside the tissue did not obscure the histological architecture of TMA sections nor cause any unusual immunostaining problem. Furthermore, heat-induced epitope retrieval used in immunohistochemical staining for CD20 and CD138 completely melted the agarose gel retained in the TMA sections.
Pre-made ready-to-use recipient agarose-paraffin block
It was easy to bore arrays of holes with precise spacing in the recipient agarose-paraffin blocks using a mini electric hand drill with drill bits (1 and 2 mm in diameter) installed on a drill stand that was assembled with an X-Y precision microcompound table. Production of pre-made and ready-to-use recipient blocks had well-aligned holes to receive the tissue cores was easy by removing the tissue cassettes and redundant paraffin encasing predrilled recipient blocks: 1 mm×192 holes in a 16×12 grid and 2 mm×108 holes in a 12×9 grid, respectively (Fig. 3).

Production of pre-made ready-to-use recipient agarose-paraffin blocks. The recipient block is trimmed to expose the surface of the paraffinized agarose gel (A) and an array of holes with precise spacing is drilled using a mini electric hand drill (B). When the tissue cassette and paraffin encasing the pre-punched or pre-drilled recipient blocks are removed, they can be used as pre-made and ready-to-use recipient blocks (C, 2 mm×108 holes in a 12×9 grid; D, 1 mm×192 holes in a 16×12 grid), which can be used to array tissue cores at a high-density using a self-made donor punch (E, F). (G, H) Complete homogenization between the tissue cores and the recipient block is assured by the re-melting and re-embedding procedure.
DISCUSSION
Research in pathology has been revolutionized by advances in the TMA technique that has enabled high-throughput in situ analysis of biomarker expression in archived pathological specimens.1-4 In order to maximize the speed of data acquisition and minimize experimental variables, constructing TMAs of high-density and high-quality, which usually involves using an automated or semi-automated TMA builder machine, especially when resources and valuable diagnostic tissue samples are limited, is a prequisite.19,20 Because of the high cost of these machines, the construction of TMAs is usually performed using a commercially available manual tissue arrayer. The basic prototype is Manual Tissue Arrayer MTA-1 (Beecher Instruments, Sun Prairie, WI, USA) that uses two separate core needles for punching the donor and recipient blocks and a micrometer-precise coordinate system for tissue assembly on a single recipient block.10 The core needles are sophisticatedly designed so that tissue cores can perfectly fit the corresponding holes in the recipient block. When this type of commercialized manual arrayer is used, high-quality and high-density TMAs can be assured by tempering the TMA blocks with heating and cooling cycles which stabilize the tissue array and improve adherence of the tissue cores to paraffin of the recipient blocks.
Many researchers, especially in small institutions, with limited funds are still deterred from using such a simple but essential instrument needed for high-throughput in situ analysis. They can resort to simple and inexpensive alternative methods using self-made tissue punches and recipient paraffin blocks.7-10 However, these alternative low-cost methods have their own intrinsic drawbacks: the distortion of the tissue array during TMA construction and loss of tissue cores during the cutting of the TMA blocks.9,15 The most important cause of these problems is incomplete bonding between the tissue cores and wall of the holes in the recipient block because the self-made recipient punches cannot bore holes that perfectly fit the diameter of the donor tissue cores extracted by corresponding donor punches. Even though distortion of the TMA sections and/or loss of tissue cores during microtomy can be minimized by tempering the recipient blocks to facilitate bonding between tissue cores and wall of the holes in the recipient block,8,15 the tempering procedure itself cannot always ensure complete fusion between tissue cores and the recipient block. Furthermore, a cycle of repeated tempering procedures can distort the tissue array, displacing some tissue cores to the edge of the TMA block.
In order to overcome these problems, we updated the method a previously described methods for the in-house manual construction of high-density TMAs.11 The updated method involves incorporation of agarose gels in the recipient blocks. To the best of our knowledge, Song et al.12 were the first to introduce the method to prepare recipient array blocks made of paraffinized agarose gel. Yan et al.13 also proposed using recipient agarose-paraffin blocks for TMA construction and used a quite prolonged tissue processing cycle of longer than 28 hours to impregnate 2% (w/v) agarose gels with conventional paraffin. Conversely, the method we used was quite simple, and the additional cost was negligible. In this study, we placed several agarose gels in the empty space of the tissue-processing machine that was always occupied by routine clinical samples including surgical or biopsy specimens of the patients. The gels were dehydrated through a graded series of alcohol, cleared with xylene and impregnated with paraffin at the same time and in the same manner as the clinical samples, which took approximately 10 to 12 hours. Then, the paraffinized gels were embedded in paraffin in the same way as for clinical tissue samples. No additional tissue processing machine or device was needed to make paraffinized agarose gels, which were used as the recipient blocks for in-house manual construction TMAs. This updated method allowed for a significant increase in both the capacity and quality of TMAs without additional cost. We successfully constructed 2 mm TMAs with 80 cores in a 10×8 grid and 1 mm TMAs with 220 cores in a 20×11 grid in a single block, respectively.
Most of the paraffinized agarose gels retained their original size and shape. As reported by Vogel14 who also proposed using recipient agarose-paraffin blocks for TMA construction, we occasionally experienced failure in the optimal paraffinization of the agarose gels, resulting in distorted gels not suitable for use as a recipient block. This was not associated with concentration (0.5 to 5%) or thickness (2 to 5 mm) of the agarose gels used in the present study. Although the exact cause of this problem remains unclear, we speculate that some of the agarose gels placed in the processing machine could be suboptimally dehydrated due to daily variations in the reagent concentration, especially alcohol: the variations in the total amount of clinical tissue samples processed daily by the machine may affect the reagent concentration.
When conventional recipient paraffin blocks are used for manual construction of TMAs, cycles of heating and cooling should be applied to the TMA blocks before subsequent cutting for histological sections in order to stabilize and enhance the adhesion between the tissue cores and the paraffin of the recipient blocks. This procedure of tempering is essential to prevent loss of tissue cores during the cutting of the TMA blocks. However, when high-density TMAs are constructed manually using self-made recipient/donor punches, ensuring complete fusion between tissue cores and paraffin in the recipient blocks is very difficult. This can cause partial/complete loss or folding of TMA sections during microtomy, resulting in unnecessary consumption and wasting of valuable TMAs followed by loss of tissue cores on deeper sections, especially the case when the surface of the deeper levels are cut for additional studies. Conversely, construction of TMAs using recipient agarose-paraffin blocks allows one-step complete melting of the TMAs, rendering full incorporation of tissue cores into the paraffin of the recipient blocks. The resulting TMA blocks can be easily cut like any ordinary paraffin-embedded tissue blocks. By preventing in advance the distortion of the paraffin ribbon which occurs during sectioning of the TMA blocks at a substantially deeper level, this technique can save valuable TMAs especially when the original donor blocks are thin (Table 2).

Overview of in-house manual construction of TMAs using self-made recipient blocks and recipient/donor punches
When the conventional paraffin blocks are used as recipient blocks for TMA construction, removing sub-optimally or erroneously arrayed tissue cores to replace them with new ones is very difficult. Conversely, the TMA construction using recipient agarose-paraffin blocks requires no additional steps for stabilization, thus saving time and labor. In addition, the recipient blocks with a completed tissue array can be easily detached from the tissue cassette and embedding paraffin, so that any misplaced or sub-optimal tissue cores in the initial TMA can be replaced without causing damage in the original tissue array. The capacity of the recipient blocks can be significantly maximized by using a mini electric hand drill and a mini X-Y precision table installed on a drill stand.15 When the tissue cassette and paraffin encasing the pre-punched or pre-drilled recipient blocks are removed, they can be used as pre-made and ready-to-use recipient blocks with a higher capacity than the commercially available blocks.
In conclusion, we have modified the method for in-house manual construction of TMAs be less costly. The self-made tissue punches and recipient blocks made of paraffinized agarose gel can be effectively used for the construction of high-density and high-quality TMAs, which can be employed in the high-throughput in situ analysis of biomarker expression in the archived pathological samples.
Acknowledgments
This work was supported by Inha University Research Grant. The authors wish to thank Yu Hwan Lim, Kyung Hwan Jang, Yong Hoon Lee, Young Min Lee, and Seok Joon Hong in the histology lab for their technical support and advice in the construction of the TMA blocks and Kyung Shin Kim and Eun Sook Kim in the immunopathology section for their excellent immunohistochemistry.
Notes
No potential conflict of interest relevant to this article was reported.