Primary Pulmonary Myxoid Liposarcoma with Translocation t(12;16)(q13;p11) in a Young Female Patient: A Brief Case Report
Article information
Abstract
Primary liposarcoma of the lung is an extremely rare disease. To date, only 14 cases have been reported in the literature. We experienced a case of myxoid liposarcoma of the lung treated by surgery. The tumor was well-defined, solid, lobulated mass measuring 3.5×2 cm, involving the bronchus of the left lower lobe. Microscopically, myxoid liposarcoma was identified. The fluorescence in situ hybridization confirmed the presence of a reciprocal translocation involving DNA damage-inducible transcript 3 (DDIT3) and fused in sarcoma (FUS) genes. The patient is still alive with no recurrence or metastasis at the time of writing this report (on 20 months postoperatively). To our knowledge, this is the first cytogenetic case report of pulmonary myxoid liposarcoma.
Primary liposarcoma of the lung is an extremely rare disease. To date, only 14 cases have been reported in the literature.1-7 In addition, only two cases of myxoid subtype have been presented in the English literature.4,7 We experienced a case of myxoid liposarcoma of the lung treated by surgery. Here, we describe the characteristic cytogenentic features of the lesion with a review of literatures.
CASE REPORT
A 24-year-old woman was referred to Dong-A University Medical Center following the detection of a well-defined mass in the left lower lobe (LLL) of the lung on chest X-ray during a routine health check. On medical and family history, the patient had no notable findings other than a 6-month history of an intermittent dry cough. On chest computed tomography (CT) scan, the patient had a lobulated, well-enhancing mass with an endobronchial protrusion in the LLL. On positron emission tomography-CT scans, the patient had no notable findings of other organs. The patient underwent a bronchoscopy, but a diagnosis could not be made on biopsy. A left lower lobectomy with regional lymph node dissection was performed. The tumor was a well-defined, solid, lobulated mass measuring 3.5×2 cm, involving the bronchus of the LLL (Fig. 1A). Microscopically, the lesion was a multinodular mass composed of bland fusiform or round cells in a myxoid background with a delicate plexiform capillary vascular network. The tumor cells showed a lipoblastic differentiation of multivacuolar and univacuolar cells (Fig. 1B). Partly, there were sheets of primitive round cells with a high nuclear/cytoplasmic ratio. On immunohistochemistry, the tumor expressed vimentin and S-100 protein. Nineteen dissected regional lymph nodes were negative for metastasis. The fluorescence in situ hybridization specific for the t(12;16)(q13; p11) using DNA damage-inducible transcript 3 (DDIT3) and fused in sarcoma (FUS) break-apart probes (Vysis/Abbot Molecular, Downers Grove, IL, USA) was successfully performed, which confirmed the presence of a reciprocal translocation involving DDIT3 and FUS genes (Fig. 2). The patient is still alive with no recurrence or metastasis at the time of writing this report (on 20 months postoperatively).
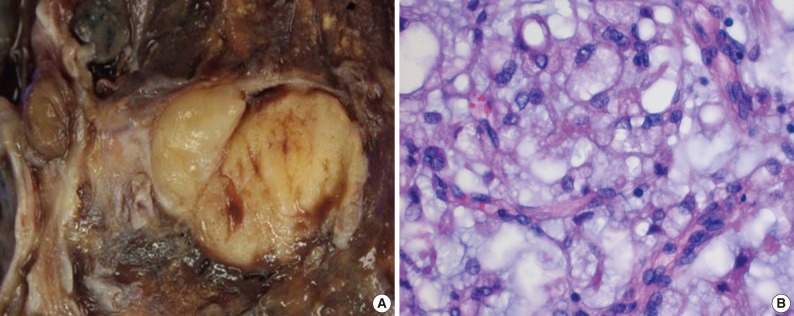
The resected lung shows a well-defined, solid mass with a yellowish gelatinous appearance devoid of necrosis, involving the bronchus of the left lower lobe (A). Microscopically, the mass is composed of tumor cells showing lipoblastic differentiation in a prominent myxoid stroma with arborizing capillaries (B).

Representative photomicrographs from the bicolor fluorescence in situ hybridization (FISH) analysis. Break-apart FISH probes for DNA damage-inducible transcript 3 (DDIT3) (12q13) (A), and for fused in sarcoma (FUS) (16p11) (B) show intact signal and translocation within a single nucleus, respectively. This indicates that the patient most likely has a reciprocal translocation of the DDIT3 and FUS genes.
DISCUSSION
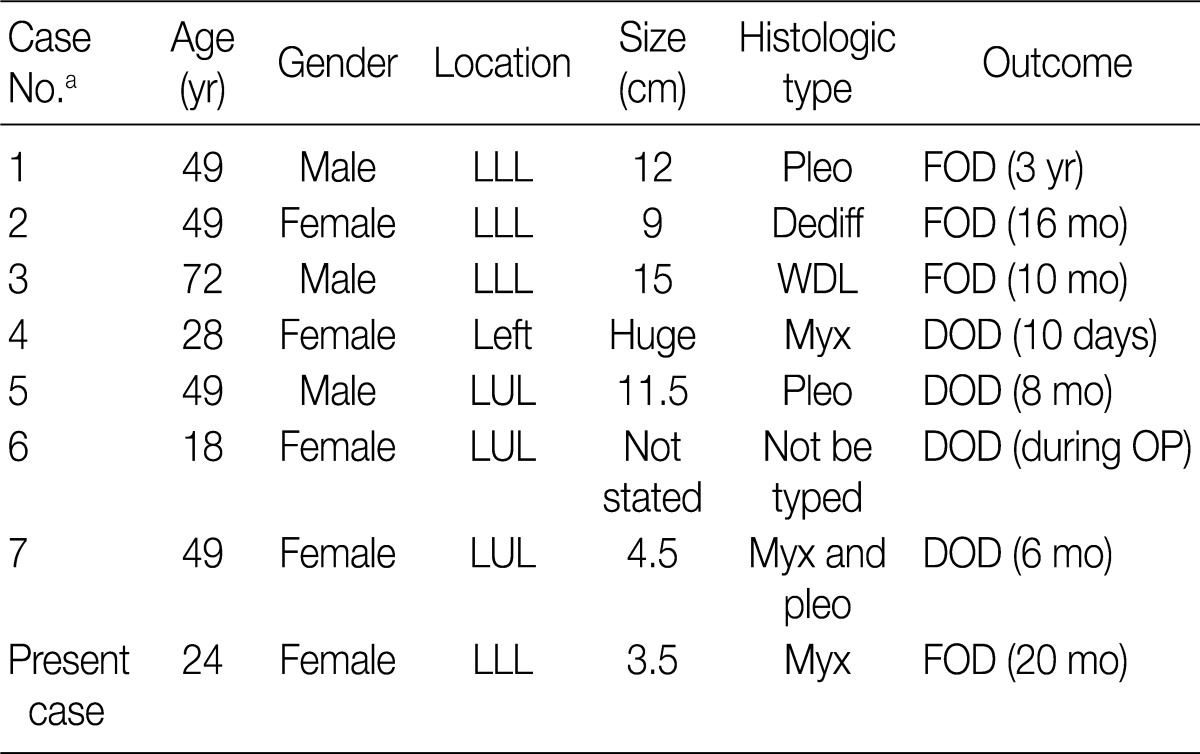
To date, a total of 14 cases of primary liposarcoma of the lung have been reported in the literature. But only seven articles were available when the PubMed and KoreaMed searches were conducted for English- and Korean-language articles.1-7 Clinicopathologic features reported in the literature are summarized in Table 1. The patient in our case report is the second youngest one that has been reported and presented to have the smallest tumor mass (3.5 cm). Of note, all the tumors have been reported to involve the left lung. Our case supports the view that a wide surgical excision may be effective in treating primary liposarcoma of the lung. But no specific conclusions can be drawn due to the limited number of reported cases.
Nearly all the myxoid/round cell liposarcomas are characterized by a reciprocal translocation between the chromosomes 12 and 16, t(12;16)(q13;p11). This translocation leads to the generation of a DDIT3/FUS hybrid protein, which has the ability to cause deregulation of other target genes, to interfere with terminal differentiation of various cells and to down-regulate programmed cell death. It appears to play a direct role in oncogenesis of myxoid liposarcoma.8 However, there is no correlation between DDIT3/FUS fusion type and grade or disease-specific survival.8 In our case, there was a reciprocal translocation involving DDIT3 and FUS. This suggests a central role of these fusion transcripts in the pathogenesis of pulmonary myxoid liposarcoma like the same tumor occurring in other sites.
To our knowledge, this is the first cytogenetic case report of pulmonary myxoid liposarcoma.
Acknowledgments
This work was supported by the Dong-A University research fund. We would like to thank Mr. Sang Soo Park for his technical assistant with the fluorescence in situ hybridization (FISH) study and Ms. Boram Seo for her artwork.
Notes
No potential conflict of interest relevant to this article was reported.