Correlation of TTF-1 immunoexpression and EGFR mutation spectrum in non–small cell lung carcinoma
Article information
Abstract
Background
Thyroid transcription factor (TTF-1) is a diagnostic marker expressed in 75%–85% of primary lung adenocarcinomas (ACs). Activating mutations in the tyrosine kinase domain of the epidermal growth factor receptor (EGFR) gene is the most common targetable driver alteration in lung AC. Previous studies have shown a positive correlation between TTF-1 and EGFR mutation status. We aimed to determine the predictive value of TTF-1 immunoexpression for underlying EGFR mutation status in a large Indian cohort.
Methods
This retrospective designed study was conducted with medical record data from 2011 to 2020. All cases of primary lung AC and non–small cell lung carcinoma not otherwise specified (NSCLC, NOS) with known TTF-1 expression diagnosed by immunohistochemistry using 8G7G3/1 antibodies and EGFR mutation status diagnosed by quantitative polymerase chain reaction were retrieved, reviewed, and theresults were analyzed.
Results
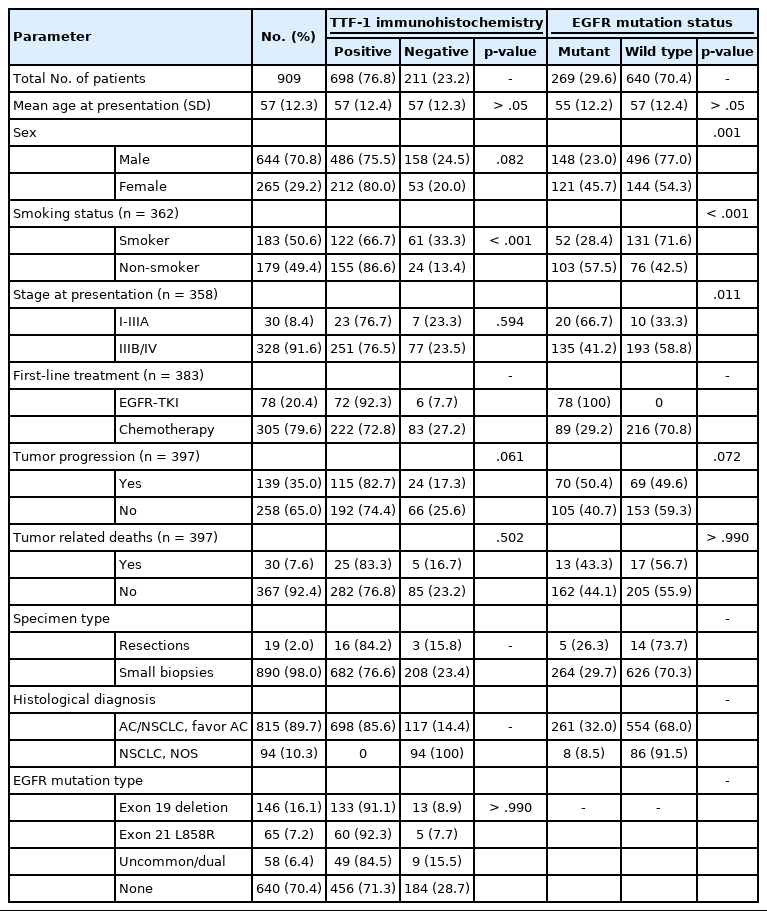
Among 909 patient samples diagnosed as lung AC and NSCLC, NOS, TTF-1 was positive in 76.8% cases (698/909) and EGFR mutations were detected in 29.6% (269/909). A strong positive correlation was present between TTF-1 positivity and EGFR mutation status (odds ratio, 3.61; p < .001), with TTF-1 positivity showing high sensitivity (90%) and negative predictive value (87%) for EGFR mutation. TTF-1 immunoexpression did not show significant correlation with uncommon/dual EGFR mutations (odds ratio, 1.69; p = .098). EGFR–tyrosine kinase inhibitor therapy was significantly superior to chemotherapy among EGFR mutant cases irrespective of TTF-1 status; however, no significant differences among survival outcomes were observed.
Conclusions
Our study confirms a strong positive correlation between TTF-1 expression and common EGFR mutations (exon 19 deletion and exon 21 L858R) in advanced lung AC with significantly high negative predictive value of TTF-1 for EGFR mutations.
Thyroid transcription factor 1 (TTF-1) is a regulatory transcription factor for maintaining peripheral lung function and is required for normal lung morphogenesis [1]. It is expressed normally in type II pneumocytes and non-ciliated bronchiolar epithelial cells, and this expression pattern is retained in 75%–85% of primary lung adenocarcinomas (ACs) [2,3]. TTF-1 positivity is highly specific to lung AC among all non-neuroendocrine neoplasms of the lung and is used widely as an immunohistochemical (IHC) marker for diagnosis of AC of lung origin and in subtyping of non–small cell lung carcinoma (NSCLC) not otherwise specified (NOS) wherein TTF-1 positivity favors diagnosis of lung AC over that of squamous cell carcinoma [4].
AC is the most common histological subtype of lung carcinoma, both worldwide and in India, with an increasing trend in incidence [5,6]. With discovery of mutations in the epidermal growth factor receptor (EGFR) gene in up to one-third of all ACs and the high response rates of these patients to EGFR tyrosine kinase inhibitor (EGFR-TKI) therapy, the latter is the first-line treatment of choice in patients with advanced non-squamous NSCLC harboring EGFR mutations. In accordance, any lung carcinoma sample diagnosed as AC, or NSCLC, NOS on histology with or without IHC should be subjected to molecular testing (MT) for targetable alterations in EGFR, anaplastic lymphoma kinase (ALK), or c-ros oncogene 1 (ROS1) genes at minimum [7].
One of the major constraints in diagnosis and clinical management of lung AC is the non-availability of adequate tumor tissue for MT. With nearly 70% of all lung cancer patients presenting with advanced stage, small biopsies/cytology represent the major source of tumor tissue, and successful MT on such small samples requires triage of material by informed pathologists and trained technicians [8]. Further, in a developing country such as India, the majority of laboratories do not have facilities for MT, and the samples usually are deferred to a higher medical center, resulting in delay and difficulty in timely treatment decisions.
Unlike MT, IHC is a well-established technique practiced even in small laboratories, and IHC using the recommended TTF-1 8G7G3/1 mouse monoclonal antibody [9] is available in most laboratories. Limited studies have analyzed the correlation between TTF-1 expression and EGFR mutations in lung AC and have reported a positive correlation between the two. Further, like EGFR mutant ACs, TTF-1 positivity has been shown to be associated with longer overall survival (OS), especially in nonmetastatic NSCLC [10-12]. This retrospective study was undertaken to assess the correlation of TTF-1 IHC with EGFR mutation status to understand whether TTF-1 expression status can be used as a guide for prioritizing EGFR MT and/or for treatment decisions in resource-limited settings.
MATERIALS AND METHODS
Case selection
The current study was of a retrospective design spanning over duration of 9 years (January 2011 to February 2020), approved by the institutional ethics committee (IECPG-480/29.08.2016).
All primary lung AC patients with available medical record data pertaining to both TTF-1 and EGFR mutation testing on surgical pathology samples were selected for this study. The present study cohort included patients from previously published data pertaining to EGFR mutation rate [13-15]. Histopathological slides were reviewed, and diagnosis was reconfirmed in accordance with WHO classification of tumours of the lung, pleura, thymus and heart (2015) [16]. Detailed histopathological examination and subtyping of all cases were performed. Clinicopathological data including age, sex, smoking status, specimen type, site of biopsy, IHC result of TTF-1, and EGFR mutation status were recorded from the histopathology requisition forms.
Immunohistochemistry for TTF-1
IHC for TTF-1 was performed on 4-μm-thick, formalin-fixed, paraffin-embedded tissue sections as part of diagnostic work-up during routine clinical practice. Heat-induced epitope retrieval was conducted in citrate buffer at pH 6, followed by a 3-hour incubation period with TTF-1 antibody (1:400, mouse monoclonal antibody, clone 8G7G3/1, Bio-SB, Santa Barbara, CA, USA). A universal-labeled streptavidin biotin kit was used as a detection system. Nuclear staining of weak or strong intensity in ≥ 5% of tumor cells was considered positive for TTF-1 expression [4]. For every case, negative and positive internal controls (normal lung tissue) or external controls (absence of normal lung parenchyma) were used.
EGFR mutation analysis
Real-time polymerase chain reaction–based EGFR mutation testing was conducted using the EGFR RGQ PCR Kit (Therascreen, Cat No. 870111, Qiagen, Hilden, Germany) to detect 29 hotspot mutations in EGFR exons 18–21.
Statistical analysis
Data was analyzed using GraphPad Prism ver. 8 (GraphPad Software Inc., San Diego, CA, USA). Categorical variables were tabulated as frequency and percentage. The chi-square or Fisher exact tests were used to analyze the categorical variables between TTF-1 expression and EGFR mutations. Patient age was represented in mean values with standard deviations. A p < .05 was considered statistically significant.
RESULTS
A total of 909 patient samples were included in the study (Table 1). The mean age of the cohort was 57 years, with a male preponderance (male:female ratio of 2.4:1). Sample types were tumor resections (n = 19) and small biopsies (n = 890), the latter also including lymph node excision biopsies and Tru-Cut biopsies from metastatic sites. TTF-1 was positive in 76.8% cases (698/909) (Fig. 1) and was significantly more prevalent among non-smokers (p < .001); however, there was no difference in prevalence among sex or stage-based groups. EGFR mutations were detected in 29.6% of cases (269/909). EGFR mutations were significantly more frequent among NSCLC occurring in females (46% vs. 23% in males, p = .001), non-smokers (58% vs. 28% in smokers, p < .001), and in early stages of presentation (67% vs. 41% in late stages, p = .011). However, only a small proportion of our cases was diagnosed at earlier stages. The most common mutation detected was exon 19 deletion (146/269, 54.3%), followed by exon 21 mutation encoding the L858R amino acid change (exon 21 L858R; 65/269, 24.2%). Other less common mutations observed were in exon 21 L861Q (2/269, 0.7%), exon 20 insertion (4/269, 1.5%), exon 20 T790M (7/269, 2.6%), exon 20 S768I (3/269, 1.1%), and exon 18 G719X (3/269, 1.1%). Dual mutations involving multiple exons were detected in 39/269 cases (14.5%), the majority of which encoded the T790M amino acid change (17/39, 43.6%) in combination with other sensitive mutations. The rate of TTF-1 expression was lower among uncommon/dual mutations (84%, 49/58) compared to exon 19 deletion (91%, 133/146) and exon 21 L858R (92%, 60/65), although this did not reach statistical significance (Table 1).
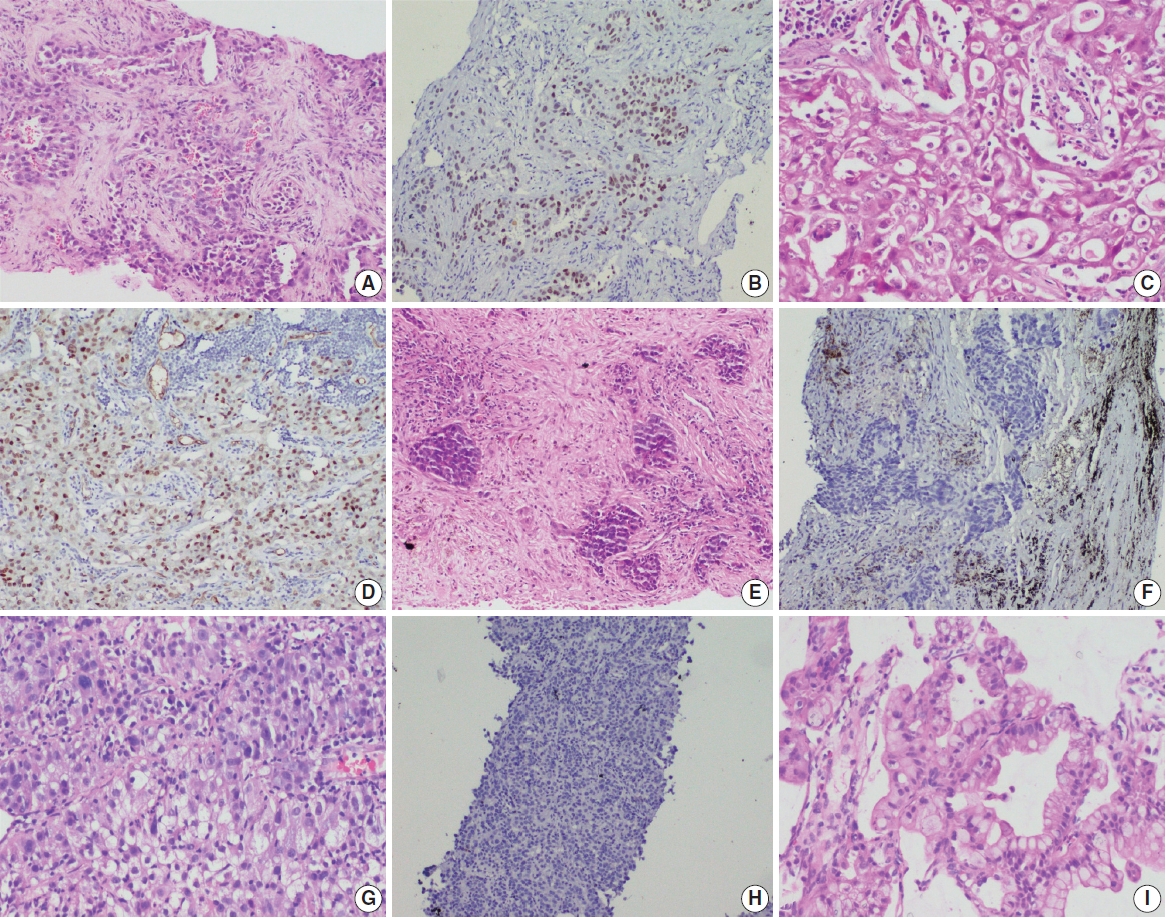
Photomicrographs of non–small cell lung carcinoma (NSCLC). Epidermal growth factor receptor (EGFR) mutant lung adenocarcinoma with acinar and solid patterns (A) showing nuclear staining for thyroid transcription factor 1 (TTF-1) in tumor cells (B). EGFR wild type NSCLC with solid architecture (C) immunopositive for TTF-1 (D). Examples of EGFR mutant NSCLC not otherwise specified (NOS) (E) and EGFR wild type NSCLC, NOS (G) that are negative for TTF-1 (F, H); EGFR mutant invasive mucinous adenocarcinoma (I).
Correlation between TTF-1 immunopositivity and EGFR mutation
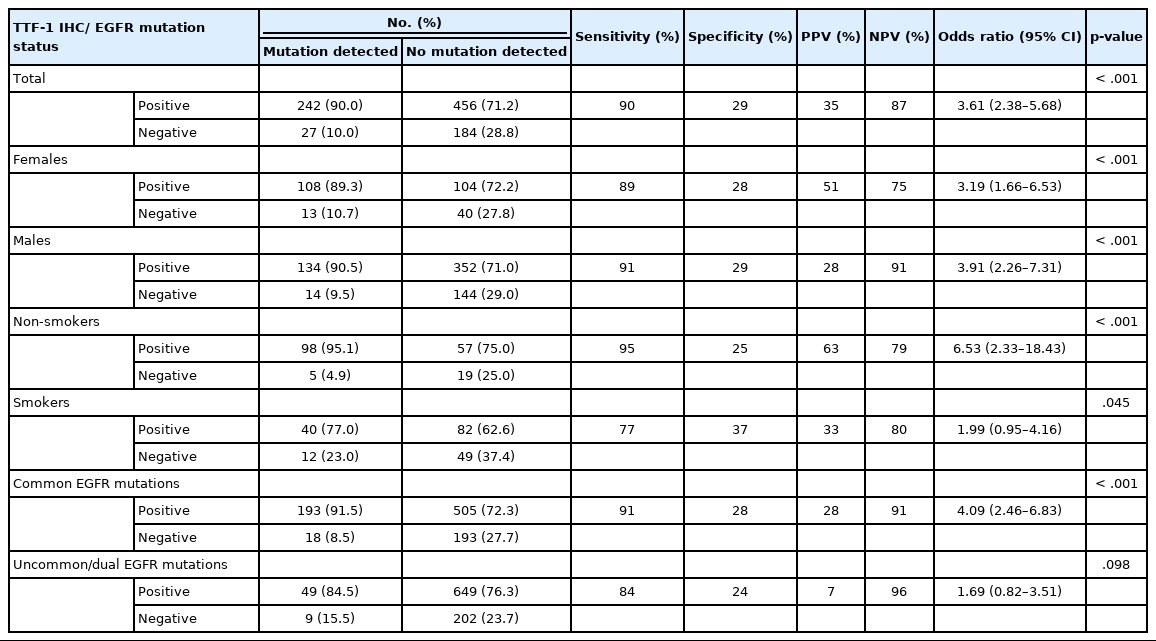
mutation Ninety percent of all EGFR mutant NSCLCs were TTF-1 positive, with TTF-1 positivity showing high sensitivity (90%) and negative predictive value (NPV; 87%) for EGFR mutations. However, only 30% of all TTF-1–positive ACs harbored EGFR mutations, showing low specificity (29%) and poor positive predictive value (PPV; 35%) (Table 2). Considering that the prevalence of EGFR mutations varies among clinicopathological subsets, in particular with sex and smoking status, we analyzed the correlation between TTF-1 and EGFR mutations in these subgroups separately. As expected, the PPV of TTF-1 positivity for EGFR mutations was highest among females (51%) and nonsmokers (63%), which are the two clinical groups that show a higher prevalence of EGFR mutations. The lowest prevalence of EGFR mutations was among males (28%) and smokers (33%). Among individual types of mutations, PPV was higher with common EGFR mutations (both exon 19 deletion and exon 21 L858R combined at 28%) than with uncommon/dual mutations (7%). The overall NPV of TTF-1 for EGFR mutations was 87%, was increased to 91% in males and for common EGFR mutations, and was highest for uncommon/dual mutations (96%).
We also calculated the diagnostic odds ratio (OR) within the entire cohort and within distinct clinicopathological subsets. OR denotes the ratio of the odds of TTF-1 being positive in an EGFR mutant tumor relative to the odds of TTF-1 being positive in an EGFR wild type tumor in this study and is independent of prevalence of EGFR mutations in the group under study. Overall, we found that TTF-1 was 3.6 times more likely to be positive in EGFR mutant tumors compared to EGFR wild type (OR, 3.61), with strong statistical strength (p < .001). This positive correlation between TTF-1 and EGFR mutation status remained significant within all individual clinicopathological subsets except when only considering uncommon/dual EGFR mutations (Table 2, Fig. 2). The highest OR was observed in non-smokers (OR, 6.53) and among those with common EGFR mutations (OR, 4.09), with no significant difference in OR between the two common mutations, exon 19 deletion (OR, 3.59; 95% confidence interval [CI], 2.06 to 6.79; p < .001) and exon 21 L858R (OR, 3.88; 95% CI, 1.69 to 11.20; p = .001).
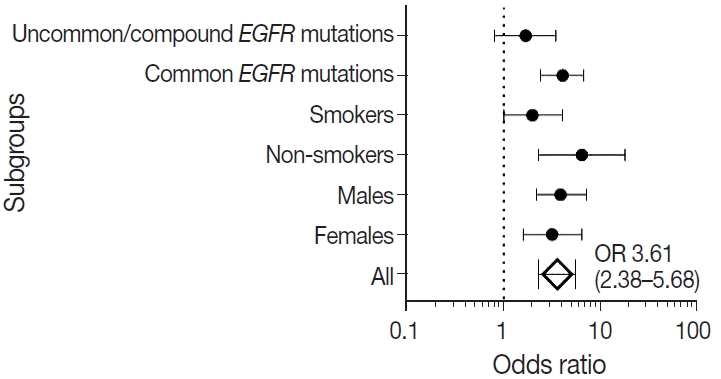
Forest plot for correlation between thyroid transcription factor 1 and epidermal growth factor receptor (EGFR) mutation status in clinicopathological subgroups. Subgroup-specific odds ratios (ORs) are denoted by black dots (line width corresponding to 95% confidence intervals). Combined OR estimate for all patients is represented by a black diamond, where the width corresponds to 95% confidence intervals.
Histopathological subtyping based on TTF-1 and EGFR mutation status
TTF-1–positive EGFR-positive cases (n = 242)
Most of these cases were classified as AC (n = 235). Other rare subtypes included one case each of invasive mucinous AC and sarcomatoid carcinoma and five cases of adenosquamous carcinoma. The spectrum of EGFR mutations is shown in Fig. 3.
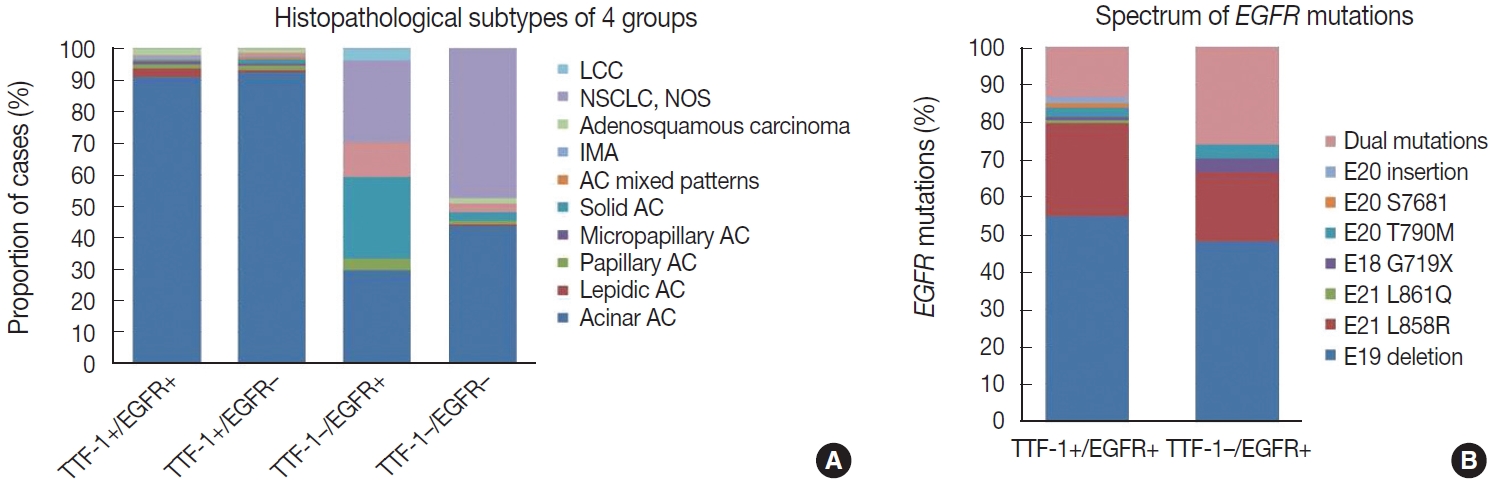
Bar diagram showing histopathological subtyping of four groups based on thyroid transcription factor 1 (TTF-1) and epidermal growth factor receptor (EGFR) mutation status (A) and spectrum of EGFR mutations (B) in these groups. AC, adenocarcinoma; IMA, invasive mucinous adenocarcinoma; LCC, large cell carcinoma; NSCLC, non small cell lung carcinoma; NOS, not otherwise specified.
TTF-1–negative EGFR-positive cases (n = 27)
case as large cell carcinoma. Among the remaining cases, 16 cases classified as AC. EGFR mutations detected in these cases are shown in Fig. 3. Among dual mutations, five were E20 T790M in combination with other activating mutations [exon 19 deletion (4) and exon 18 G719X (1)] and two were combination of exon 19 deletion with exon 21 L858R.
TTF-1–positive EGFR-negative cases (n = 456)
The majority of this group showed AC (n = 435) with predominant patterns, as shown in Fig. 3. The following subtypes were identified: three cases of sarcomatoid carcinoma, four cases of invasive mucinous AC, and six cases of adenosquamous carcinoma. A few rare variants were noted (signet cell morphology in six, cribriform architecture in one, and hepatoid in one case).
TTF-1–negative EGFR-negative cases (n = 184)
This group showed AC in 88 cases (Fig. 3), invasive mucinous AC in five cases, and adenosquamous carcinoma in three cases. One case showed AC with signet ring cell differentiation. A total of 87 cases with solid architecture and without evidence of glandular differentiation were classified as NSCLC, NOS.
Treatment details and survival analysis
Treatment details were available for 383 patients, where 305 patients received platinum-based chemotherapy and 78 patients received EGFR-TKI therapy as first-line treatment. A total of 29.2% of patients who received chemotherapy had EGFR mutated NSCLC, where treatment decisions were made before availability of EGFR mutation status results. Response to firstline therapy as per the Response Evaluation Criteria in Solid Tumors (RECIST) was available for 249 patients. Irrespective of TTF-1 and EGFR status, EGFR-TKI treatment resulted in superior response rates (71.7%, 38/53) compared to chemotherapy (37.2%, 73/196) (OR, 3.47; 95% CI, 1.84 to 6.53; 0.48, p < .001). For treatment response and survival analysis, patients were stratified into four groups based on TTF-1 expression and EGFR mutations: TTF-1+/EGFR+, TTF-1+/EGFR–, TTF-1–/EGFR+, and TTF-1–/EGFR–. Response to treatment in these four groups is depicted in Table 3. Among EGFR wild type cases, patients receiving chemotherapy and harboring TTF-1–positive tumors responded better than patients with TTF-1–negative tumors. However, this difference did not reach statistical significance (OR, 1.32; 95% CI, 0.57 to 3.03; p = .542).
Response to treatment with first-line EGFR-TKI treatment versus chemotherapy in the four patient groups
Survival data was available for 397 patients. The median duration of follow-up was 5.3 months. Overall, 35.01% of the patients experienced tumor progression during the course of therapy, and 7.6% of patients died. Patients with EGFR mutation and TTF-1 expression showed the best overall one-year survival rate (90.6%), while those who were double negative showed the shortest (86.2%), although these differences did not reach statistical significance. Notably, the OS rates among patients with EGFR wild type tumors positive for TTF-1 (86.7%) and negative for TTF-1 (86.2%) were very similar. On the other hand, progression- free survival (PFS) was more homogenous and the PFS durations and hazard ratios for progression were comparable among the four groups.
DISCUSSION
Many observational studies over the years have reported strong positive correlation between TTF-1 expression and EGFR mutation status [17-22]. From a meta-analysis of 9,764 patients, Kim et al. [23] reported an OR of 5 between TTF-1 and EGFR mutations, and the OR was similar among European (OR, 4) and Asian (OR, 4) cohorts. The only previous study from India on a smaller South Indian cohort of 85 patients with advanced NSCLC reported an OR of 15 between TTF-1 and EGFR mutations [19]. In our single-institutional North Indian cohort of 909 patients, we found a similar strong correlation between TTF-1 and EGFR mutations (OR, 3.61) comparable to the results observed previously [17-23].
While EGFR mutations are more frequent in females and nonsmokers [13,20,24], TTF-1 expression in NSCLC has shown inconsistent correlation with clinical parameters [21,25]. In the present study comprising predominantly of advanced stage nonsquamous NSCLC diagnosed on small biopsies, TTF-1 expression was seen in approximately 77% of cases and was significantly more common among non-smokers but lacked association with age or sex, while EGFR mutations were detected in about 30% of all cases and significantly more frequent among females, non-smokers, and at early clinical stages. The frequency of EGFR mutation is similar to those reported in the Indian literature [13].
The correlation between TTF-1 and EGFR mutations in the present study remained significant irrespective of sex or smoking habit, indicating that the positive association is not merely due to increased prevalence of EGFR mutations in some demographic groups. Kim et al. [23] reported higher OR among females (OR, 4.87) compared to males (OR, 3.34), while Zhao et al. [21] reported lack of significant association between TTF-1 and EGFR mutations in males. In the present study, we observed comparable results between males and females, with ORs slightly higher among males (OR, 3.91) than females (OR, 3.19). Similarly, although non-smokers showed the highest ORs (6.53), there was significant positive correlation of TTF-1-EGFR among smokers as well, albeit with a lower OR of 1.99, as has been observed previously [21]. The degree of expression of TTF-1 (i.e., in > 50% of tumor cells or in 10%–50% of tumor cells) also does not appear to show significant difference in positive association with EGFR mutations, especially the exon 21 mutation [25]. In our study, we used a cutoff of > 5% tumor nuclei staining for positivity.
Morphological analysis of the TTF-1–negative cases harboring EGFR mutations showed histological evidence of AC in 70.4% of cases. On further subtyping of these cases, the association of EGFR mutations with predominant subtyping of the cases with TTF-1 negativity was akin to the published literature [26]. Acinar predominant AC showed higher propensity for EGFR mutations. A few cases of mucinous AC, which were mostly TTF-1 negative, also were positive for EGFR mutations.
The strength of correlation between TTF-1 and EGFR appears to vary according to type of mutation. While initial studies found significant association with exon 21 mutations and not with exon 19 deletion [21,25], others have found significant association of TTF-1 expression with both of these common mutations [23, 27,28]. We found strong association with both types of TKI-sensitive activating mutations, with an OR slightly higher for exon 19 deletion (OR, 4.63) than for exon 21 L858R (OR, 3.16). Interestingly, as has been observed in one previous study [27], we did not find significant correlation of TTF-1 with uncommon/ dual mutations (OR, 1.69; p > .05).
The reported literature also states that more than 90% of EGFR mutated lung ACs are positive for TTF-1 [20-22,29]. In the present study, TTF-1 positivity showed 90% sensitivity for EGFR mutations, and the sensitivity was higher among males and non-smokers. Considering that TTF-1 positivity is not specific for EGFR mutations, with only 34.6% of all TTF-1 positive tumors harboring EGFR mutations, the NPV rather than the PPV becomes the important criterion in our assessment of the value of TTF-1 IHC as a screening tool for EGFR mutations. We found that TTF-1 IHC shows NPV of 87% for EGFR mutations, comparable to the NPVs of 88%–97% reported in published cohorts [17,19,23,30-32]. NPV varies with prevalence of EGFR mutations and sensitivity of TTF-1 IHC [33] and is likely to be higher in populations with lower prevalence of EGFR mutations, e.g., Western cohorts [30,31], and with the use of more sensitive TTF-1 IHC assays such as the SPT24 antibody clone (vs. 8G7G3/1) [9]. In line with these results, the highest NPVs in the present study were observed among males and among patients with uncommon/dual mutations.
EGFR mutation–specific antibodies have high specificity (94%–100%) and sensitivity (60%–100%) with high PPV and NPV for EGFR mutations compared to TTF-1 IHC. These antibodies have shown high concordance rate compared to the gold standard molecular techniques [34]. However, its interpretation is highly variable due to differences in criteria for positivity, and its practical application is limited as these antibodies are meant to detect the two most common mutations (exon 19 deletion and exon 21 L858R), with significant numbers of false positive and false negative cases [34]. A molecular-based assay is needed to confirm EGFR mutation status, and screening of EGFR mutations by IHC seems not to be applicable [35]. TTF-1 is a diagnostic marker that can be used as a screening tool for EGFR mutations, but it cannot be replaced by EGFR mutation-specific antibodies.
The high NPV of TTF-1 for EGFR mutations has led some to suggest that patients with negative TTF-1 can be started on chemotherapy without MT in time- and resource-constrained settings [31]. We found, however, that approximately 13% of TTF-1–negative tumors harbored EGFR mutations, 50% of which were TKI-sensitive exon 19 deletion and exon 21 L858R mutations. Thus, until greater clarity is achieved on the tumor response rates of TTF-1–negative/EGFR mutant tumors for EGFR-TKI vs. chemotherapy, excluding patients for MT based on TTF-1 negativity should be discouraged.
Activating EGFR mutations are established as predictive biomarkers for TKI responsiveness in NSCLC and are known to confer a favorable prognostic effect on recurrence-free survival and OS irrespective of treatment arms [36,37]. Similarly, TTF- 1 expression has been reported as a favorable prognostic biomarker in NSCLCs [11,12,38-40]. Transcriptional activation of TTF-1 has been shown to be necessary for EGFR downstream signaling in EGFR mutant tumors [41], and TTF-1 has been suggested to correlate with EGFR oncogene addiction in these tumors [28]. Tumors showing dual positivity for EGFR mutations and TTF-1 IHC have shown the best outcomes, while those negative for both carried the worst outcomes [12,21,28]. TTF-1 positive/EGFR wild type tumors and TTF-1 negative/ EGFR mutant tumors show intermediate outcomes, with the former showing longer progression-free and OS than the latter in some studies [12,21,28]. In the present study, limited by availability of patient follow-up data, one-year OS was highest among TTF-1–positive/EGFR mutant cases and lowest for dual negative cases. TTF-1 positivity did not appear to alter the oneyear OS or PFS among EGFR wild type tumors, as shown in previous studies [12,21,28]. The improved survival of TTF-1–positive/ EGFR wild type cases can be attributed in part to the presence of other prognostically favorable alterations such as ALK or BRAF [42]; however, these alterations were not analyzed in our study.
TTF-1 positivity has been correlated with better response rates to chemotherapy [28,39,43] and EGFR-TKI [39], including some EGFR wild type tumors [28,38]. With regard to chemotherapy, we did observe slightly better response rates in TTF- 1–positive/EGFR wild type cases compared to TTF-1–negative/ EGFR wild type cases. However, with regard to EGFR-TKI, we found no differences in response rates between TTF-1–positive and TTF-1–negative EGFR mutant cases. None of our EGFR wild type cases received TKI treatment; hence, we could not analyze TKI response rates based on TTF-1 expression status. Nevertheless, our observations lack statistical significance, and observational trials including more patients are necessary to confirm or refute previous observations.
Conclusion
In agreement with previous studies, we found a strong positive correlation between TTF-1 expression and common EGFR mutations (exon 19 deletion and exon 21 L858R) in advanced lung AC. The strength of correlation was highest among nonsmokers and lowest among those with uncommon/dual mutations. Despite a high NPV of TTF-1 IHC for EGFR mutations in the present study and previous studies, a better understanding of TTF-1–mediated modulation of treatment responses and its underlying biological mechanisms in EGFR mutant ACs is necessary before considering implementation of TTF-1 IHC as a surrogate for EGFR mutations.
Notes
Ethics Statement
Approval from the Institute Ethics Committee (IECPG-480/29.08.2016), All India Institute of Medical Sciences, was obtained to conduct this retrospective study on archival patient samples. Thus, the informed consent was waived due to the retrospective nature of this study.
Availability of Data and Material
The datasets generated or analyzed during the study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
Author Contributions
Conceptualization: DJ. Data curation: TN, VS, AN, PSM, AM, DJ. Formal analysis: TN, VS, AN, DJ. Methodology: TN, VS, AN, DJ. Writing—original draft: TN. Writing—review & editing: TN, VS, AN, PSM, AM, DJ. Approval of final manuscript: all authors.
Conflicts of Interest
D.J., a contributing editor of the Journal of Pathology and Translational Medicine, was not involved in the editorial evaluation or decision to publish this article. All remaining authors have declared no conflicts of interest.
Funding Statement
This work was supported by Department of Biotechnology (grant number N-1611) and The Lady Tata Memorial Trust (grant number N-1590).