Incidental serrated lesions of the appendix: analysis of 2,137 appendectomy specimens
Article information
Abstract
Background
Serrated lesions of the appendix are rare, often incidental findings in routine appendectomy specimens. Their true frequency, histopathologic spectrum, and anatomic distribution remain incompletely characterized, partly due to variability in sampling practices.
Methods
We retrospectively reviewed 2,137 appendectomy specimens (2015–2025) from a single tertiary pathology center. Cases with histologically confirmed serrated lesions were reexamined, classified as hyperplastic polyp (HP) or sessile serrated lesion/polyp (SSL/P), and assessed for clinicopathologic parameters including lesion size, location, and associated pathologies. Nonparametric tests were used, with statistical significance defined as p < .05.
Results
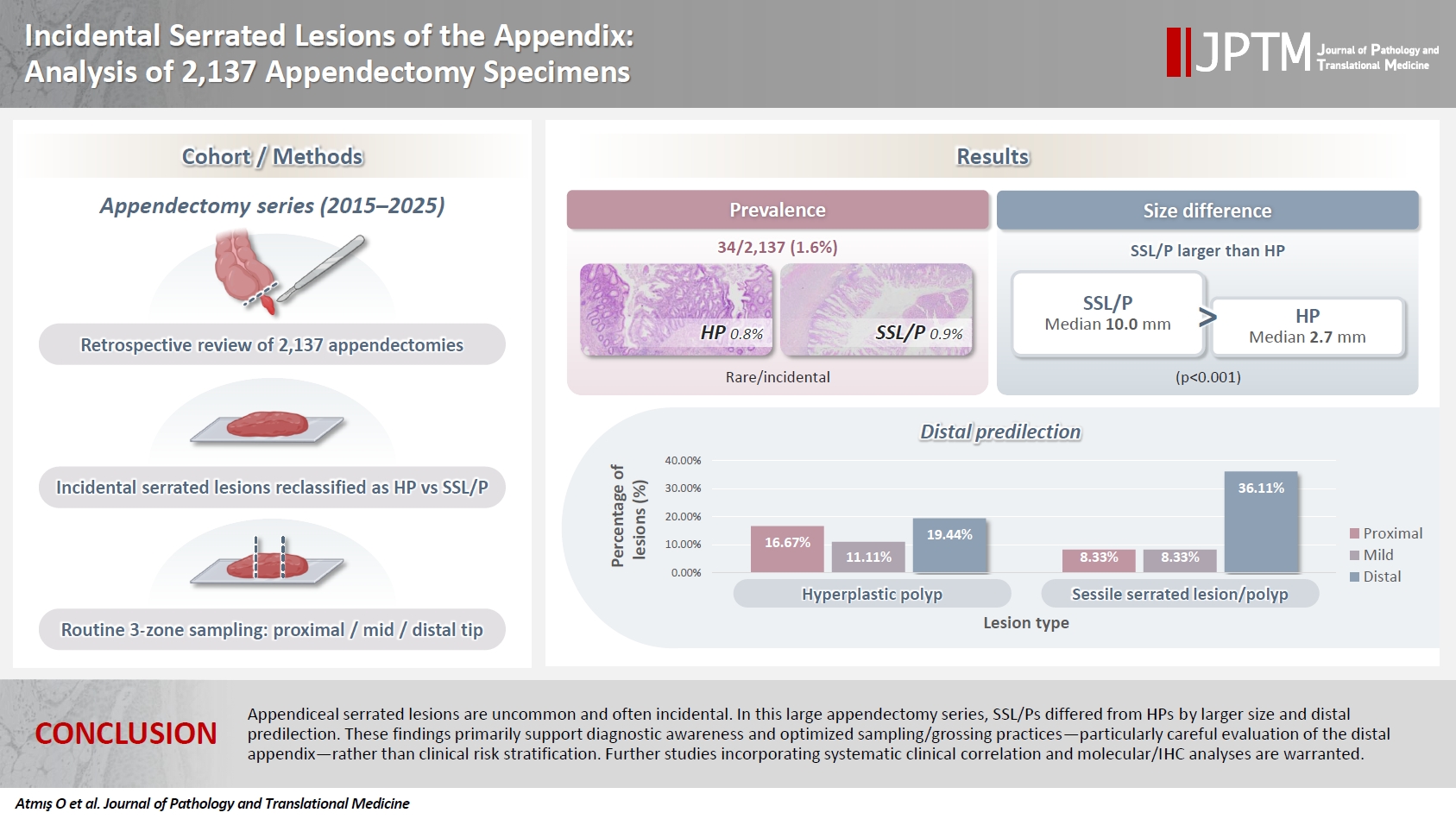
Serrated lesions were identified in 34 cases (1.6%) with 36 serrated lesions, comprising 17 HPs (0.8%) and 19 SSL/Ps (0.9%). SSL/Ps were significantly larger than HPs (median 10.0 vs. 2.7 mm, p < .001) and were more frequently located in the distal appendix (68.4% vs. 33.3%, p = .045, one-tailed Fisher’s exact test). No dysplasia or traditional serrated adenoma was detected. Acute appendicitis was present in 88% of cases, and associated neoplasms in 9%.
Conclusions
Appendiceal serrated lesions are uncommon and often incidental. In this large appendectomy series, SSL/Ps differed from HPs by larger size and distal predilection. These findings primarily support diagnostic awareness and optimized sampling/grossing practices—particularly careful evaluation of the distal appendix—rather than clinical risk stratification. Further studies incorporating systematic clinical correlation and molecular/immunohistochemistry analyses are warranted.
INTRODUCTION
The appendix is one of the most frequently excised gastrointestinal organs due to the clinical suspicion of acute appendicitis, and in a subset of cases, unexpected or incidental lesions are identified during histopathological examination [1]. Recognition of these incidental findings in appendectomy specimens has gained increasing importance in surgical pathology practice [2]. Among these lesions, the serrated polyp family—including hyperplastic polyp (HP), sessile serrated lesion/polyp (SSL/P), and traditional serrated adenoma (TSA)—is classified according to colorectal terminology in most reports of appendiceal serrated lesions [3,4].
Although serrated lesions of the appendix share many morphologic similarities with their colorectal counterparts, certain unique features arise due to the appendix’s distinct anatomy and sampling characteristics. For example, a Turkish series reported circumferential involvement in 63.9% of HPs and 74.3% of SSL/Ps [5]. Most of these lesions cannot be detected clinically or radiologically, and diagnosis is usually established during routine histopathological evaluation of the appendectomy specimen [6].
The reported incidence of serrated lesions varies markedly depending on the sampling strategy. In one study, the incidence was 9.3% when the entire appendix was entirely submitted for histologic evaluation, compared with 1.8% in partially sampled specimens [7]. In a Turkish cohort of 960 appendectomy specimens, 71 serrated polyps (7.39%) were identified—36 (50.7%) HPs, 33 (46.5%) SSL/Ps, and two (2.8%) TSAs [5].
Several studies have provided insights into the distribution and size of these lesions. In one series, 45% of 40 serrated lesions were located at the distal tip of the appendix, and 87.5% exhibited a local continuous distribution. Notably, 42.5% of lesions measured ≤6 mm in length, suggesting that appendiceal serrated lesions are typically small and can be easily overlooked both macroscopically and microscopically [7]. Previous studies have shown that extensive and complete sampling of appendectomy specimens increases the detection rate of incidental lesions [8]; however, there is no established consensus regarding the optimal number of blocks required for adequate routine sampling.
From a molecular perspective, KRAS mutations are more frequently observed than BRAF mutations in appendiceal serrated lesions, supporting the notion that the serrated neoplastic pathway in the appendix may differ from that of the colon [2,4,9].
In summary, the identification of serrated lesions—many of which may be missed in routine practice—depends on the sampling protocol, the level of histologic scrutiny, and the representativeness of the examined tissue. Investigating their true prevalence and clinicopathologic features is therefore warranted. In particular, defining pathological parameters such as anatomic distribution and lesion size may provide valuable contributions to the existing literature.
The present study aims to provide a descriptive analysis of the incidence, histopathologic spectrum, macroscopic/grossing-related sampling characteristics, and anatomic distribution of incidentally detected appendiceal serrated lesions in appendectomy specimens (2015–2025), with a particular focus on the practical utility of a routine zonal sampling approach.
MATERIALS AND METHODS
Study design and cases
This retrospective descriptive study included 2,137 appendectomy specimens evaluated between 2015 and 2025 at the Department of Pathology, Manisa Celal Bayar University Faculty of Medicine. The pathology database was reviewed to identify incidentally detected serrated lesions among appendectomy specimens performed for suspected appendicitis or other indications.
Patient data—including age, sex, diagnosis, presence of associated neoplasms, and macroscopic findings of the appendix—were retrieved from pathology reports. All slides of cases with serrated lesions were re-examined, diagnoses were confirmed, and necessary measurements were performed.
Histopathological examination and sampling protocol
In our department, appendectomy specimens are routinely sampled in three anatomical zones as follows: block 1, proximal surgical margin (en face); block 2, mid-appendiceal segment (2–3 tissue sections); block 3, distal tip of the appendix.
In cases where no definite evidence of acute appendicitis was observed macroscopically or when a suspicious lesion was noted, additional sampling was performed to increase the number of tissue blocks. Serial sections were obtained when necessary to ensure complete morphologic evaluation of the lesion. In cases with suspected neoplasia, additional blocks were taken, and if required, the entire appendix was submitted for histologic examination.
Lesion characterization and measurements
All slides were re-reviewed to confirm the diagnosis and to classify the lesions as HP or SSL/P according to the criteria of the WHO Classification of Digestive System Tumours, 5th Edition (2019) [10].
For each case, the largest diameter of the lesion was measured microscopically using a calibrated micrometer and recorded in millimeters. The anatomic location (zone) of each lesion was determined based on the block in which it was identified—proximal, mid, or distal. In cases where a lesion was present in multiple blocks, the most proximal block was designated as the primary site.
Since additional resampling was performed in some cases and the total number of tissue pieces in the standard three-block protocol varied, the total number of sampled tissue fragments was also recorded for each case.
Statistical analysis
All statistical analyses were performed using IBM SPSS Statistics ver. 25.0 (IBM Corp., Armonk, NY, USA). The normality of data distribution was evaluated using the Kolmogorov-Smirnov and Shapiro–Wilk tests. Descriptive statistics were presented as mean ± standard deviation, median (minimum–maximum), and percentage (%) values. Categorical variables were compared using the chi-square test or Fisher’s exact test, while continuous variables were compared using the Mann-Whitney U test or Kruskal-Wallis test as appropriate.
For the analysis of lesion location, a one-tailed Fisher’s exact test was applied to test the directional hypothesis that SSL/Ps would show distal predominance, as suggested by previous reports. A p-value < .05 was considered statistically significant.
RESULTS
Among 2,137 appendectomy specimens examined during the study period, 34 cases (1.6%) contained serrated lesions—17 HP (0.8%) and 19 SSL/P (0.9%). One patient had three HPs, while the remaining patients had a single lesion. For case-based analyses, only the most proximal lesion was considered to represent that case, in order to avoid clustering effects. Lesion-based analyses (n = 36 lesions in total) were performed separately where appropriate. No TSA or dysplasia was identified. Table 1 summarizes the main clinical and histologic findings. Representative histopathologic features are shown in Fig. 1.
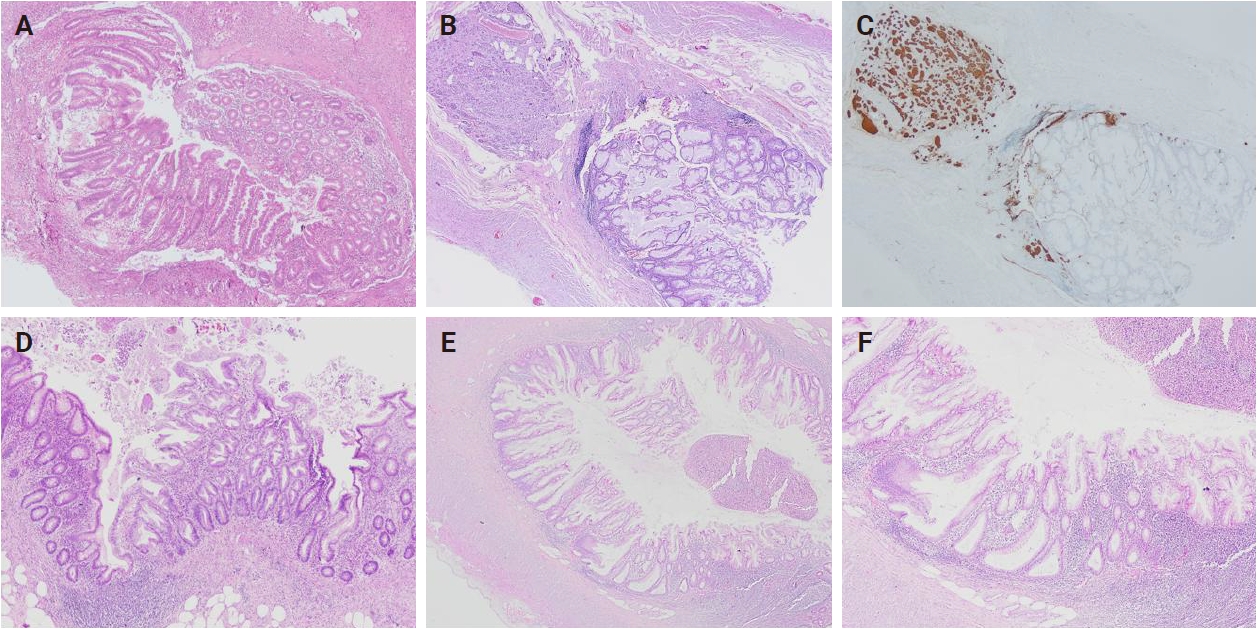
Representative histopathologic features of appendiceal serrated lesions, including a case coexisting with a neuroendocrine tumor. (A) Proximally located hyperplastic polyp. (B) Distal-type neuroendocrine tumor adjacent to a sessile serrated lesion/polyp (SSL/P). (C) Synaptophysin immunoreactivity in the neuroendocrine tumor. (D) Distally located hyperplastic polyp. (E) Distally located SSL/P. (F) The same SSL/P showing dilated, asymmetric crypt bases.
The patients included 16 men and 18 women (mean age, 52.6 ± 18.4 years). A total of 36 serrated lesions were recorded across these 34 cases. Acute appendicitis was present in most specimens (88.2%), and periappendicitis in 61.8%. Diverticula were noted in 20.6%, and associated neoplasms—two grade I neuroendocrine tumors and one metastatic adenocarcinoma—were identified in 8.8% of cases.
Colonoscopy and upper gastrointestinal endoscopy data were available in three patients (one with SSL/P and two with HP). One patient with an appendiceal HP had multiple tubular adenomas and right-sided colonic adenocarcinoma, along with a gastric hyperplastic polyp. No colorectal or gastric polyps were detected in the remaining two patients. In addition, one patient with appendiceal SSL/P without available colonoscopy data was diagnosed with left-sided colonic adenocarcinoma.
Lesions were predominantly located in the distal appendix (55.6%), followed by the proximal (25.0%) and mid segments (19.4%) (Table 1). The mean lesion diameter was 6.5 ± 5.0 mm (median, 4.0 mm; range, 1.5 to 17.9 mm). No significant differences were found between HP and SSL/P groups with respect to age, appendix length, or diameter (p > .05). However, SSL/Ps were significantly larger than HPs in case-based analysis (p < .001) (Table 2, Fig. 2A). Lesion-based analysis yielded similar results, with SSL/Ps showing significantly larger sizes than HPs (p < .001) and a tendency for distal predominance (p = .242). The number of sampled tissue fragments was slightly higher in SSL/Ps, showing a near-significant trend (p = .079).
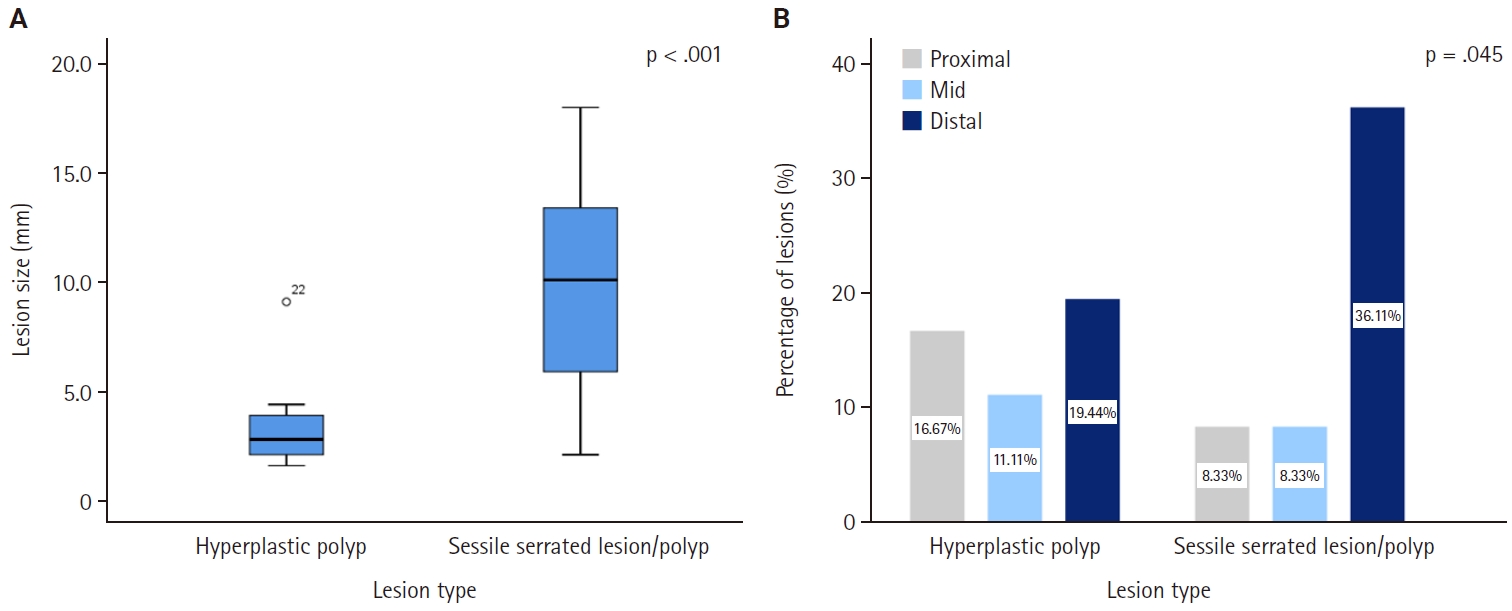
Comparison of lesion size and anatomic distribution between hyperplastic polyps (HP) and sessile serrated lesions/polyps (SSL/P). (A) Boxplot showing lesion size by lesion type. SSL/Ps were significantly larger than HPs (p < .001, Mann-Whitney U test). (B) Bar chart illustrating the anatomic distribution of HP and SSL/P within the appendix. SSL/Ps were significantly more frequent in the distal segment (p = .045, Fisher’s exact test, one-tailed).
Clinicopathologic variables—including sex, presence of acute appendicitis, periappendicitis, diverticulum, and perforation—did not differ significantly between the two groups (all p > .05). Although associated neoplasms were more frequent among SSL/Ps (15.8%) than HPs (0%), this difference did not reach statistical significance (p = .238). These comparative data are detailed in Table 3.
SSL/Ps demonstrated a distal predilection (68.4% vs. 33.3% in HPs), which was statistically supported by a one-tailed Fisher’s exact test (p = .045) and a linear-trend analysis (p = .047) (Fig. 2B). Lesion size did not vary significantly by anatomic zone (p = .520, Kruskal-Wallis test).
DISCUSSION
This single-center study evaluated the prevalence and histopathologic characteristics of incidentally detected serrated lesions (HP and SSL/P) in appendectomy specimens. The observed rate of 1.6% aligns with previously reported frequencies of incidental appendiceal neoplasms (1.5%–2.1%) [11,12], indicating methodological comparability with prior series. Differences in reported prevalence across institutions are largely attributable to sampling strategy: complete submission of the entire appendix substantially increases detection compared with partial sampling (9.3% vs. 1.8%) [7], and intensified block submission similarly improves the yield of otherwise rare or subtle lesions [8].
Morphologically, SSL/Ps slightly outnumbered HPs (52.8% vs. 47.2%), were significantly larger, and showed a distal predilection in our cohort. This distal tendency is in line with previous studies describing a tip-dominant distribution of appendiceal serrated lesions, particularly for SSL/Ps [3,5,7]. Because many lesions are small (≤6 mm in fully submitted specimens), limited or non-representative sectioning may miss focal serrated changes, underscoring the need for careful microscopic scrutiny and appropriate block selection, particularly in the distal segment [7,8].
Although colonoscopy was not routinely performed, endoscopic data were available only in a very small subset of patients; therefore, the present cohort does not allow any inference regarding synchronous or metachronous colorectal lesions. Accordingly, any potential clinical association should be explored in future studies with systematic colonoscopic correlation.
From a molecular standpoint, appendiceal serrated lesions preferentially harbor KRAS rather than BRAF mutations, suggesting divergence from the colonic serrated pathway [9]. Broader profiling further supports the dominant role of KRAS across appendiceal serrated and mucinous neoplasia, suggesting site-specific biological features, as reported in prior studies, with potential diagnostic relevance [13,14]. However, molecular or immunohistochemical analyses were not performed in the present study, and the molecular characteristics discussed herein are based on previously published data. Accordingly, molecular considerations in this study are intended to provide contextual background rather than direct evidence.
Historically, the taxonomy of appendiceal epithelial proliferations was heterogeneous. The adaptation of colorectal serrated classifications to the appendix—since the seminal works of Longacre and Fenoglio-Preiser [15], Williams et al. [16], and Carr et al. [17]—and the demonstration of outcome associations have shaped contemporary diagnostic and reporting practices. Clinically, SSL/Ps may occasionally present with acute appendicitis and, rarely, with complications such as pyogenic liver abscesses, implying potential mechanical effects (e.g., luminal obstruction) even for small, focal lesions [18]. In parallel, growing interest in non-operative (antibiotic-based) management of appendicitis raises concerns about delayed recognition of incidental tumors, a consideration supported by our detection rate [19].
Current recommendations emphasize complete submission in suspicious or neoplastic settings; however, no universal consensus exists regarding a fixed number of blocks for routine appendectomies. Institutional variation in sampling intensity likely contributes to incidence variability across studies [7,8]. The distribution of HP versus SSL/P and the spectrum of concomitant lesions in our series (e.g., neuroendocrine tumors, metastasis) broadly mirror prior reports, including a Turkish series with a 7.39% serrated polyp rate, near-equal HP/SSL/P proportions, and frequent circumferential involvement [5].
In summary, although uncommon, appendiceal serrated lesions show distinct subtype-specific features: SSL/Ps are significantly larger than HPs and tend to occur distally. These observations reinforce the importance of vigilant morphologic assessment and adequate sampling—particularly of the distal portion—for accurate detection and classification. Recognition and accurate classification of these lesions may provide a framework for future studies addressing whether appendiceal serrated neoplasia follows a molecular trajectory distinct from the colorectal serrated pathway, as suggested by prior literature.
Our study has several limitations. The retrospective design, the absence of routine colonoscopic evaluation in patients with incidentally detected appendiceal serrated lesions, and the lack of molecular or immunohistochemical analyses represent major constraints. These limitations preclude meaningful clinicopathologic correlation and restrict the interpretation of our findings primarily to diagnostic and sampling-related implications. Because the majority of specimens were obtained for acute appendicitis, the low number of advanced appendiceal neoplasms may also reflect inherent selection bias. Importantly, given its retrospective and primarily descriptive design, the study was not intended to establish biological mechanisms or clinically meaningful risk estimates.
In conclusion, incidentally detected serrated lesions of the appendix are uncommon but may be underrecognized in routine practice. In our series, the prevalence was 1.6%, with SSL/Ps being significantly larger and showing a distal predilection compared with HPs. No dysplasia or TSA was observed. These findings primarily reinforce diagnostic awareness and the importance of adequate sampling and careful microscopic evaluation—particularly of the distal appendix—for the detection and classification of incidental serrated lesions. In the absence of systematic clinical correlation and molecular/immunohistochemical validation, our results should be interpreted as descriptive observations with practical implications for routine pathology practice. Prospective studies integrating standardized submission protocols with systematic clinical correlation and molecular/immunohistochemical analyses are warranted.
Notes
Ethics Statement
All procedures performed in the current study were approved by the Ethics Committee of Manisa Celal Bayar University Faculty of Medicine (approval number: 20.478.486-3609, date: 03.12.2025) in accordance with the 1964 Helsinki declaration and its later amendments. Formal written informed consent was not required, with a waiver granted by the appropriate Institutional Review Board (IRB) of Manisa Celal Bayar University Faculty of Medicine.
Availability of Data and Material
The datasets generated or analyzed during the study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
Author Contributions
Conceptualization: ÖA, SA. Methodology: ÖA, AT. Data curation: EDK, FSP. Formal analysis: ÖA, AT. Investigation: ÖA, HSM. Supervision: AT, SA. Writing—original draft: ÖA. Writing—review & editing: ÖA, HSM. Approval of final manuscript: all authors.
Conflicts of Interest
The authors declare that they have no potential conflicts of interest.
Funding Statement
No funding to declare.