Clinicopathological profile of high-grade differentiated thyroid carcinoma in Indonesian tertiary hospital
Article information
Abstract
Background
High-grade differentiated thyroid carcinoma (HGDTC) is a recently recognized entity in the 2022 World Health Organization classification, representing a more aggressive subtype of differentiated thyroid carcinoma. Previously, high-grade features such as increased mitotic activity and tumor necrosis were often overlooked, despite being important independent prognostic factors. Although rare, HGDTC carries significant diagnostic, prognostic, and therapeutic implications. Data remain limited in Indonesia.
Methods
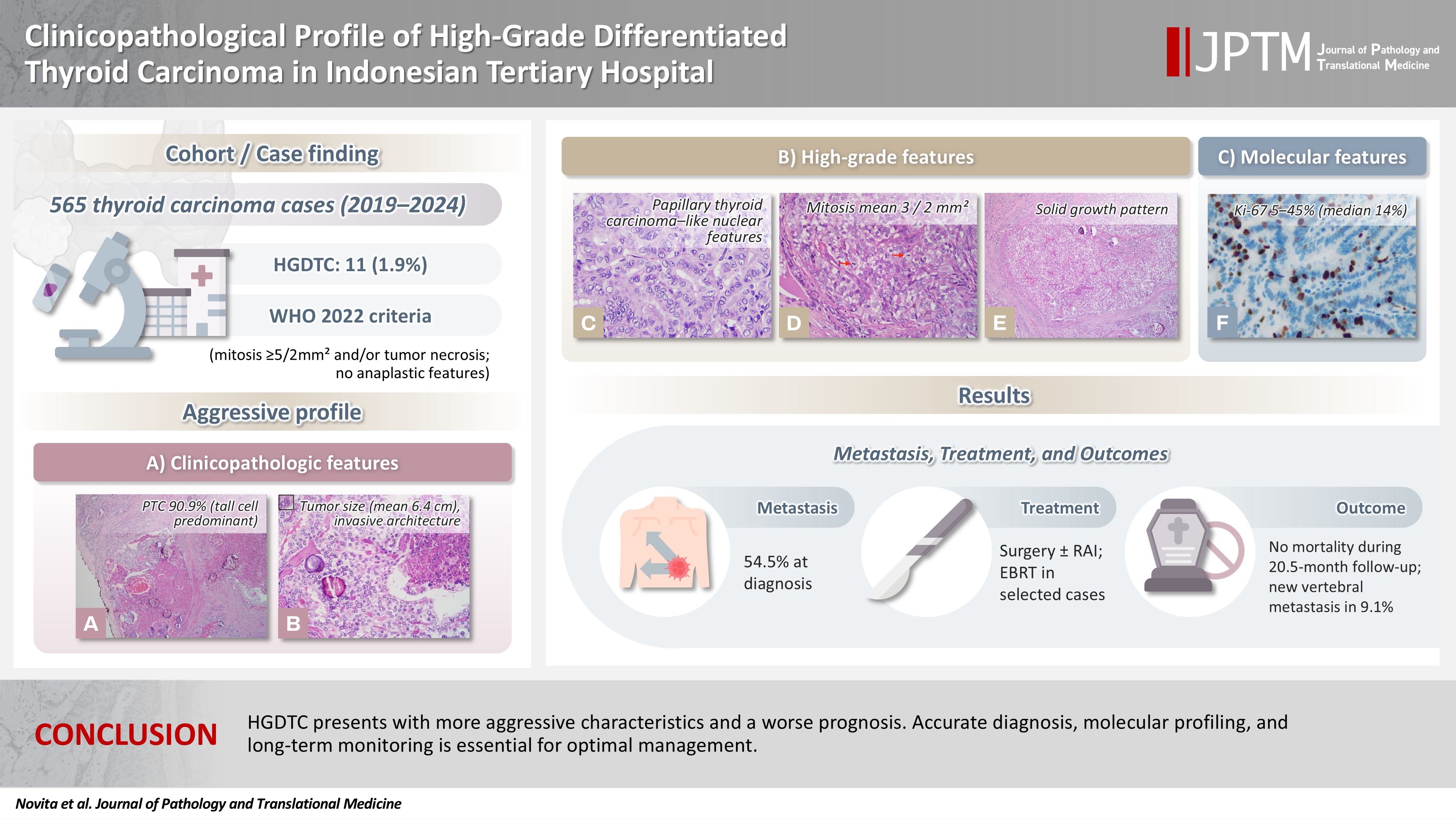
This retrospective descriptive study reviewed 565 thyroid carcinoma cases diagnosed at Cipto Mangunkusumo Hospital from 2019 to 2024. Eleven cases (1.9%) met HGDTC criteria. Clinicopathological characteristics, histologic subtypes, Ki-67 proliferation index, molecular alterations, treatment modalities, and clinical outcomes were analyzed.
Results
Patients had a mean age of 54.6 years, with a female-to-male ratio of 2.7:1. Papillary thyroid carcinoma was the main type (90.9%), with the tall cell subtype predominating. Mean tumor size was 6.4 cm. Lymphatic invasion, vascular invasion, and extrathyroidal extension were present in 54.5%, 18.2%, and 45.5% of cases, respectively. All tumors showed necrosis. Mean mitotic count was 3 per 2 mm². The Ki-67 index ranged from 5% to 45% (median, 14%). BRAFV600E and TERT promoter mutations were detected in 18.2% and 36.4% of cases, respectively, with co-mutations in 18.2%. Six cases (54.5%) had metastases at time of diagnosis. During a mean follow-up of 20.5 months, one patient (9.1%) developed new vertebral metastases and all patients (100%) remained alive.
Conclusions
HGDTC presents with more aggressive characteristics and a worse prognosis. Accurate diagnosis, molecular profiling, and long-term monitoring are essential for optimal management.
INTRODUCTION
Thyroid carcinoma is one of the most prevalent endocrine malignancies worldwide. According to the Global Cancer Observatory (GLOBOCAN), it ranked seventh in global cancer incidence in 2022, with approximately 821,000 new cases reported [1]. The majority of thyroid carcinomas are classified as differentiated thyroid carcinoma (DTC), which typically exhibits indolent behavior and is associated with a favorable prognosis. However, DTCs that display high-grade features, such as tumor necrosis or an elevated mitotic index, tend to follow a more aggressive clinical course. These cases are linked to a significantly higher risk of extrathyroidal extension and distant metastases compared to DTCs without such features [2]. The 5-year overall survival rate among patients with high-grade features ranges from 50% to 70%, with recurrences most commonly occurring within the first 3 years. Therefore, these features should not be overlooked [3].
The term high-grade differentiated thyroid carcinoma (HGDTC) refers to this specific subset of tumors. In 2022, the World Health Organization (WHO) formally introduced HGDTC as a distinct diagnostic category. It is defined as a follicular cell–derived thyroid carcinoma with a follicular, papillary, or solid growth pattern; invasive architecture; nuclear features resembling either follicular or papillary neoplasms; a mitotic rate of at least five per 2 mm2; and/or the presence of tumor necrosis, without any features of anaplastic transformation [4,5]. The inclusion of HGDTC in the classification of thyroid malignancies provides pathologists with more precise criteria to identify aggressive lesions that do not meet the Turin criteria for poorly differentiated thyroid carcinoma (PDTC) or the diagnostic threshold for anaplastic carcinoma [2]. Recognition of this entity is clinically important, as it enables more accurate patient triage. HGDTC is associated with more aggressive biological behavior and poorer prognosis than both conventional DTC and PDTC [3]. As such, all patients diagnosed with HGDTC should receive close clinical monitoring [2,5].
Only a limited number of studies have revisited previous DTC diagnoses to identify cases that fulfill the new criteria for HGDTC. In Indonesia, data on HGDTC are lacking. The present study was conducted to investigate the incidence and clinicopathologic profile of HGDTC diagnosed at the Department of Anatomical Pathology, Faculty of Medicine, Universitas Indonesia/Dr. Cipto Mangunkusumo Hospital. The findings are expected to support pathologists in recognizing and diagnosing this entity accurately and to assist clinicians in making informed decisions regarding surveillance and treatment strategies.
MATERIALS AND METHODS
Case selection and histopathologic review
A total of 565 thyroid carcinoma cases diagnosed at our institution between 2019 and 2024 and meeting the inclusion criteria were reviewed. Of these, 516 cases (91.3%) were papillary thyroid carcinomas (PTC), five cases (0.9%) were follicular thyroid carcinomas (FTC), 15 cases (2.6%) were oncocytic carcinomas (OCA), 10 cases (1.8%) were medullary thyroid carcinomas, four cases (0.7%) were PDTC, and 15 cases (2.7%) were anaplastic thyroid carcinomas. Following histopathological re-evaluation, only 11 cases (1.9%) fulfilled the diagnostic criteria for HGDTC.
All thyroid carcinoma cases diagnosed at our institution from 2019 to 2024 were independently reviewed by the primary investigator and confirmed by board-certified endocrine pathologists to ensure diagnostic concordance. Cases were reclassified as HGDTC when they exhibited follicular, papillary, or solid growth patterns with invasive behavior; follicular neoplasm-like or papillary-like nuclear features; and/or oncocytic morphology, with a mitotic rate of ≥5 per 2 mm² and/or tumor necrosis in the absence of anaplastic features.
Tumor necrosis referred to geographic or focal necrotic areas showing a sharply abrupt transition from viable to nonviable tumor without intervening granulation tissue, fibrosis, or hyalinization. Necrotic regions displayed cellular debris, cytoplasmic dissolution, nuclear fragmentation with scattered karyorrhectic bodies, irregular chromatin distribution, ghost cell outlines, and apoptotic remnants. Pre-existing vascular structures were often preserved with a peripheral rim of viable tumor cells. Ischemic or infarct-type necrosis attributable to prior fine-needle aspiration (FNA) or core needle biopsy (CNB) typically accompanied by granulation tissue, fibrosis, hemorrhage, histiocytic infiltration, cholesterol clefts, and absence of karyorrhexis—was excluded.
For each case meeting HGDTC criteria, the following clinicopathologic data were retrieved: sex, age, tumor size, tumor focality, laterality, histologic subtype, Ki-67 proliferation index, solid/trabecular (ST) growth pattern, mutational status, lymphatic, vascular, and perineural invasion, extrathyroidal extension, surgical margin status, tumor necrosis, mitotic count, presence of metastases, American Joint Committee on Cancer (AJCC) 8th edition stage, FNA and CNB results, treatment modalities, and follow-up information.
Immunohistochemical analysis
Immunohistochemical staining for Ki-67 was performed using an anti-Ki-67 (SP6) rabbit monoclonal primary antibody (Ventana Medical Systems, Inc., Tucson, AZ, USA). Tumor areas with the highest Ki-67 expression (hot spots) were identified. Photomicrographs were captured from ten high-power fields (40× objective), and 1,000 tumor cells were manually counted using QuPath software to calculate the percentage of positively stained nuclei.
Molecular analysis
Targeted mutation testing was performed using polymerase chain reaction (PCR) followed by Sanger sequencing to evaluate alterations in BRAF (codon 600), NRAS (codon 61), HRAS (codon 61), TP53 (codons 208, 220, 241, 277), PIK3CA (codon 1047), PTEN (codon 130), and TERT promoter hotspots (C228T, C250T). Genomic DNA was extracted from 5-µm sections of formalin-fixed paraffin-embedded (FFPE) tumor tissue using the QIAamp DNA FFPE Tissue Kit (Qiagen, Valencia, CA, USA) according to the manufacturer’s instructions, and DNA purity and concentration were assessed using a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA). PCR amplification was carried out using KOD One PCR Master Mix (Toyobo, Osaka, Japan), and sequencing was performed with the BigDye Terminator v3.1 Cycle Sequencing Kit on an ABI 3730xl Genetic Analyzer (Applied Biosystems, Carlsbad, CA, USA). Resulting chromatograms were aligned with reference sequences to determine mutational status. The DNA primers of each gene are included in Supplementary Table S1.
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics ver. 24 (IBM Corp., Armonk, NY, USA). Descriptive statistics were used to summarize clinicopathologic features. Frequencies and percentages were calculated for categorical variables, and means, medians, and ranges were reported for continuous variables as appropriate. Comparative analyses to distinguish clinicopathological features between HGDTC cases with an ST pattern and those without an ST pattern were conducted using Fisher’s exact test. A p-value <0.05 was considered statistically significant.
RESULTS
A total of 565 thyroid carcinoma cases were diagnosed at our institution between 2019 and 2024. Following slide re-evaluation, 11 cases (1.9%) were found to fulfill the diagnostic criteria for HGDTC as defined by the 2022 WHO classification. The clinicopathologic profiles of these patients are summarized in Tables 1 and 2.
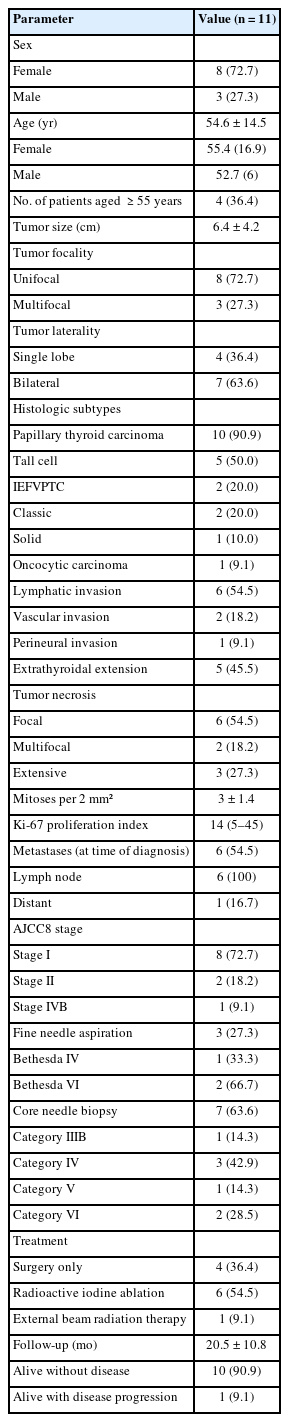
Among the 11 HGDTC patients, eight were female and three were male, with a mean age of 54.6 (standard deviation [SD], 14.5) years. Four patients (36.4%) were ≥55 years old. Tumor sizes varied, with mean size of 6.4 (SD, 4.2) cm. Among all cases, eight cases (72.7%) were unifocal, and three (27.3%) were multifocal. Seven cases (63.6%) involved both thyroid lobes, and four (36.4%) were confined to a single lobe. Ten cases (90.9%) were originally diagnosed as PTC, five tall cell subtypes (50.0%), two follicular subtypes (20.0%), two classic subtypes (20.0%), and one solid subtype (10.0%). The two follicular-patterned cases in our series were identified as invasive encapsulated follicular variant papillary thyroid carcinoma (IEFVPTC). One case (9.1%) was initially diagnosed as OCA. Tall cell components exceeded 30% in most of the subtype cases.
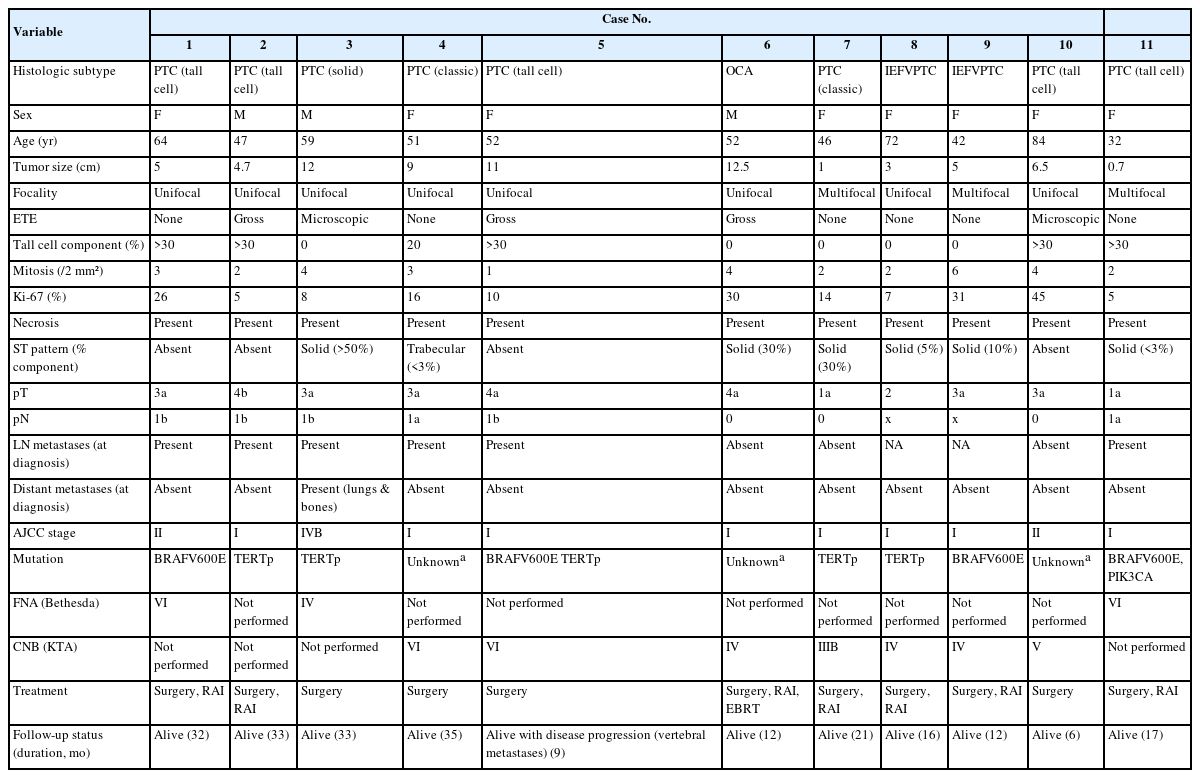
Lymphatic invasion was observed in six cases (54.5%), vascular invasion in two cases (18.2%), and perineural invasion in one case (9.1%). Extrathyroidal extension was present in five cases (45.5%). All tumors exhibited necrosis: six (54.5%) focal, two (18.2%) multifocal, and three (27.3%) extensive. Mitotic figures ranged from 1 to 6 per 2 mm² with mean of 3 (SD, 1.4) per 2 mm² and no significant inter-slide variation across three separate hotspots. The Ki-67 proliferation index ranged from 5% to 45% (median, 14%). Five cases had an index between 5% and 10%, while six cases had an index greater than 10% (Fig. 1).
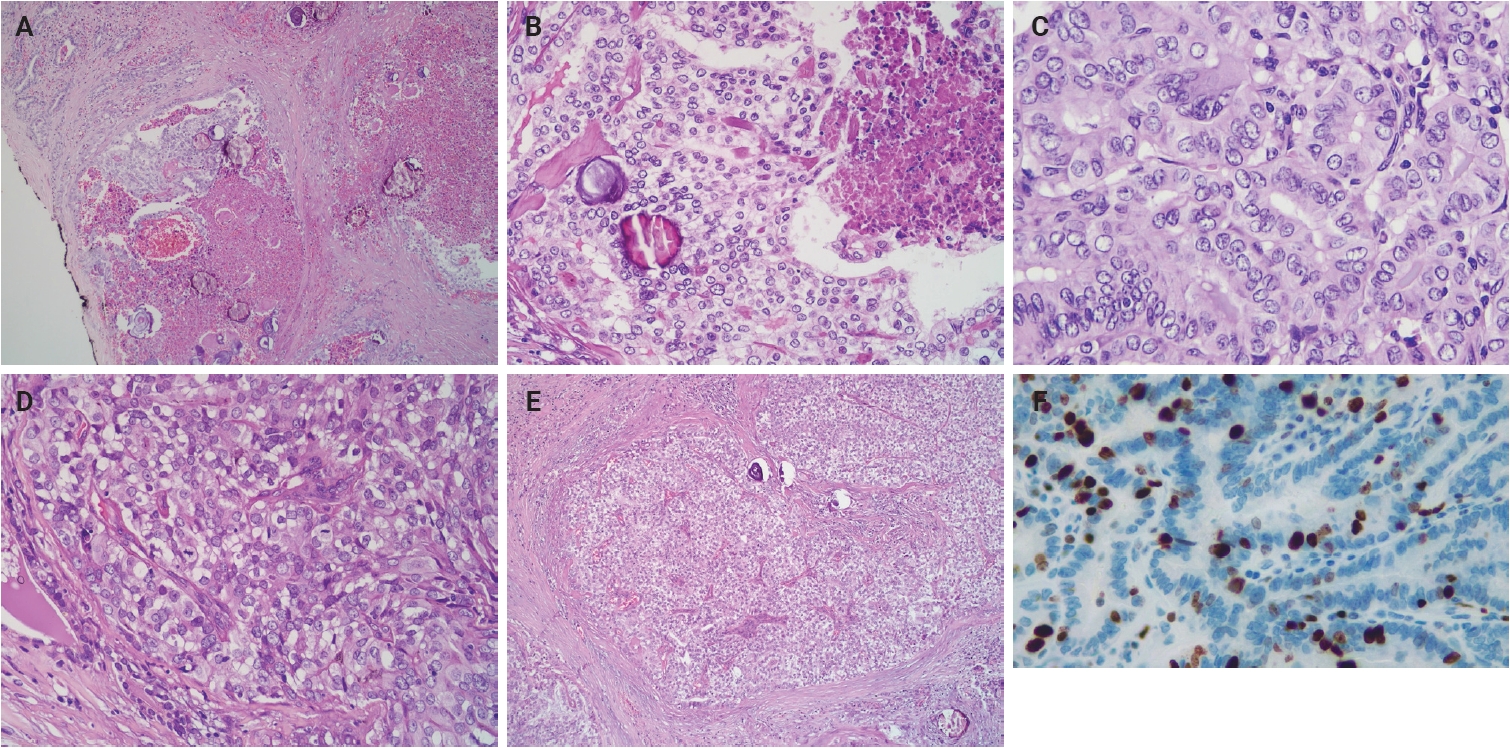
High-grade differentiated thyroid carcinoma. (A) High-grade papillary thyroid carcinoma (classic subtype) shows tumor necrosis. (B) Area of tumor necrosis adjacent to viable tumor cells. (C) Papillary thyroid carcinoma-like nuclear features. (D) Mitoses (arrows). (E) Solid pattern. (F) The Ki-67 immunostaining demonstrates a proliferation index of 45%.
ST growth pattern was identified in seven cases: six (85.7%) with solid growth patterns and one (14.3%) with trabecular growth. However, none of these cases fulfilled the Turin criteria for PDTC, as the ST areas retained classic PTC-type nuclear features rather than the convoluted nuclei required for a diagnosis of PDTC. No significant differences in clinicopathological profiles or disease outcome were observed between HGDTC cases with an ST pattern and those without an ST pattern.
Six patients (54.5%) had evidence of metastases at the time of diagnosis: all of them (100%) showed lymph node involvement, of whom one (16.7%) had concurrent distant metastases to the lungs and bones. All cases had molecular investigation: two cases with BRAFV600E mutations, four cases with TERTp mutations, two cases with multiple mutations (BRAFV600E and TERTp (C228T), BRAFV600E and PIK3CA), while the remaining cases were negative for mutations in BRAFV600E, NRAS, HRAS, TP53, PIK3CA, PTEN, and TERTp.
According to the AJCC 8th edition staging system, eight cases (72.7%) were classified as stage I, two (18.2%) as stage II, and one (9.1%) as stage IVB. FNA cytology was performed in three patients (27.3%): one was categorized as Bethesda IV and the others as Bethesda VI. CNB had been conducted before thyroidectomy in seven patients (63.6%), with results showing one case in category IIIB, three in category IV, one in category V, and two in category VI, based on the Korean Thyroid Association (KTA) classification [6].
Regarding treatment, four patients underwent surgery alone, including lobectomy or total thyroidectomy. Six patients received combined therapy with radioactive iodine (RAI), and one patient received additional external beam radiotherapy (EBRT). The mean follow-up period was 20.5 months (SD, 10.8), during which all patients remained alive. Among the surgery-only group, one patient remained alive with lungs and bones metastases at initial presentation (33 months), one developed vertebral bone metastases nine months postoperatively, and two were alive without evidence of disease (mean follow-up, 20.5 months). Of the six patients who received RAI, all remained disease-free (mean follow-up, 21.8 months). One patient treated with surgery, RAI, and EBRT also remained disease-free (12 months).
DISCUSSION
HGDTC is a newly recognized entity included in the 2022 WHO classification. It is defined as a follicular cell-derived tumor exhibiting follicular, papillary, or solid growth patterns with invasive behavior, follicular or papillary-like nuclear features, a mitotic rate of ≥5 per 2 mm2, and/or tumor necrosis, in the absence of anaplastic features [2,3,5]. This classification provides pathologists with clearer guidelines for identifying aggressive tumors that do not meet the Turin criteria for PDTC or the criteria for anaplastic thyroid carcinoma. Prior to its recognition as a distinct entity, DTCs with high-grade or aggressive features were often overlooked. However, features such as increased mitotic activity, with or without tumor necrosis, are now understood to be independent prognostic factors. DTC with such features, now termed HGDTC, is associated with worse prognosis than conventional DTC and a significantly higher risk of extrathyroidal extension and distant metastases [7-9].
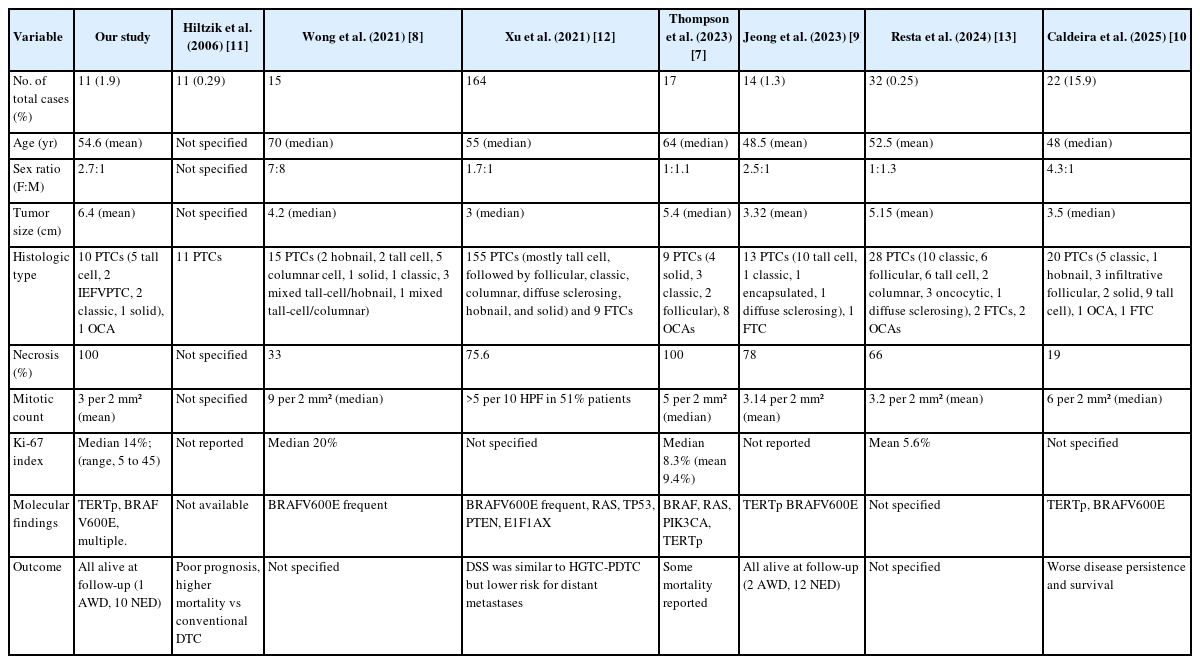
Our study characterizes the incidence and clinicopathological features of HGDTC, whose data remain limited in Indonesia. Of 565 thyroid carcinoma cases diagnosed in our institution, 11 (1.9%) met the criteria for HGDTC, a proportion comparable to that reported in a cohort involving 1,069 PTC and FTC cases (1.3%) [8]. The prevalence of HGDTC varies considerably across studies (0.25%–15.9%) (Table 3) [7-13], largely due to heterogeneity in study populations. A recent meta-analysis reported a 7.2% prevalence among unselected DTC patients, while a study focusing on advanced thyroid carcinoma cases observed a rate of 15.9% [10,14].
Although earlier reports identified a peak incidence of HGDTC and PDTC in patients aged 55–65 years, more recent studies, including the present study, have observed a younger age distribution [3,7,8,12]. In our cohort, the mean patient age was 54.6 years, with most cases occurring in individuals under 55, consistent with findings from more recent series [9,10,13]. These variations may reflect differences in population demographics, genetic backgrounds, environmental exposures, or healthcare access across regions and study settings. A similar observation is also reported for sex distribution across various studies (Table 3).
The mean tumor size in our study was 6.4 (SD, 4.2) cm, slightly larger than displayed in prior studies [7,8,13]. This may support the observation that HGDTC often presents with relatively large tumors (>3 cm), possibly reflecting its more aggressive biological behavior compared to classic DTC. Despite this size-associated trend, the 2022 WHO classification does not mandate a minimum tumor size for defining HGDTC. In principle, high-grade morphology can be diagnosed even in very small lesions (<1 cm). However, the biological and clinical interpretation of such “micro-HGDTCs” requires prudence. While these lesions demonstrate unequivocal high-grade morphology, the prognostic implications of sub-centimeter high-grade tumors remain insufficiently characterized and may not parallel those of conventionally sized counterparts. In the present study, we included one case with a sub-centimeter HGDTC (0.7 cm) because it fully met the WHO diagnostic criteria from a methodological standpoint. This tumor was multifocal, demonstrated tall-cell morphology, and presented with lymph-node metastases. Further studies are required to determine the true clinical relevance of this specific setting.
While limited studies have documented multifocality and laterality status in HGDTC, our study corroborates the findings of Thompson [7], who reported that HGDTC tumors most commonly present as a unifocal lesion, though in some cases it may involve both lobes (bilateral involvement). This pattern may reflect the aggressive behavior of HGDTC, where rapid proliferation results in a dominant tumor mass with limited time for the development of additional distinct foci [12,15,16].
Histologic subtypes observed in our cohort were primarily PTC, including tall cell, IEFVPTC, solid, and classic subtypes, along with one case of OCA. These findings are broadly consistent with those reported in prior studies (Table 3). Most series demonstrate a predominance of aggressive PTC subtypes such as tall cell, hobnail, and columnar cell, though the presence of OCA and FTC has also been variably reported [7-10,12,13]. Although our study did not identify any cases of the diffuse sclerosing subtype of PTC, prior studies have reported a series of HGDTC cases exhibiting this subtype [9,12,13,16,17].
Tumor necrosis and mitotic activity are key diagnostic features of HGDTC. All tumors in our study exhibited necrosis, with 54.5% focal, 18.2% multifocal, and 27.3% extensive necrosis. The mean mitotic count was 3 per 2 mm² (SD, 1.4), which is slightly lower than the counts in other studies [7-9]. This discrepancy may reflect the intrinsic heterogeneity of HGDTC. Although studies have shown that aggressive driver mutations such as TERTp and TP53 favor rapid tumor cell proliferation and extensive necrosis, our cohort has found that tumors with those mutations can have extensive necrosis even with low mitotic count. This phenomenon can be explained by some alternative mechanisms. Tumor cells experience metabolic stress caused by hypoxia and nutrient deprivation from insufficient vascularization, leading to increased oxidative stress and cell death pathways. Release of pro-inflammatory cytokines (e.g., tumor necrosis factor-alpha) by immune cells can also induce necrosis through inflammation-related metabolic disruption and a direct cytotoxic effect.
Apart from intrinsic tumor biology, technical factors may also influence mitotic assessment. Mitoses tend to be focal and unevenly distributed, the extent of tissue sampled may result in a different estimation of mitotic number [18-20]. Mitotic figures may also be difficult to distinguish from apoptotic bodies or other morphologic mimics on hematoxylin and eosin (H&E) sections, which may contribute to underestimation. This issue is illustrated by the observation that three of eleven tumors in our cohort had markedly elevated Ki-67 indices (≥30%) despite few identifiable mitoses (range, 4 to 6 mitoses per 2 mm2). These findings highlight the need for more standardized approaches to mitotic evaluation in HGDTC and support the potential role of ancillary techniques such as digital image analysis or phospho-histone H3 (PHH3) immunostaining for improving mitotic detection.
High-grade necrosis in HGDTC is characterized by geographic or focal necrosis with abrupt borders surrounded by viable tumor cells without stromal changes. Associated features include cellular debris, cytoplasmic degeneration, karyorrhexis, irregular chromatin distribution, ghost cells, and apoptotic bodies. It is essential to distinguish tumor necrosis from infarct-type necrosis, which may result from FNA or CNB and is associated with fibrosis, granulation tissue, histiocytes, hemosiderin, calcification, and cholesterol clefts [2,21].
ST growth pattern was observed in seven cases, including six showing solid growth and one with trabecular architecture. Despite the presence of these patterns, none of the cases met the Turin criteria for PDTC due to retention of PTC-type nuclear features. Similarly, Tondi Resta et al. [13] also identified poorly differentiated features (solid and trabecular patterns) in five of 32 HGDTC cases. In the present study, no significant differences in clinicopathological profiles or outcomes were observed between HGDTC cases with an ST pattern and those without an ST pattern. This may be attributable to the small sample size. Hitherto, the prognostic significance of ST patterns remains unclear in the current literature of HGDTC.
Six patients (54.5%) presented with metastases at diagnosis, all of them found with lymph node involvement (100%) and one patient with concurring distant metastases to the lungs and bones (16.7%). One patient (9.1%) developed vertebral metastases 9 months after diagnosis. These findings are consistent with previous studies and reflect the aggressive clinical behavior of HGDTC [7,12].
Lymphatic invasion was present in 54.5% of cases, vascular invasion in 18.2%, perineural in 9.1%, and extrathyroidal extension in 45.5%. These findings are consistent with prior reports, which noted high rates of invasion and extrathyroidal extension in HGDTC [9,13]. Although lymphatic and vascular invasion are not part of the diagnostic criteria for HGDTC, their presence is linked to aggressive tumor behavior and higher recurrence rates, underscoring their importance in assessing tumor aggressiveness [22,23].
AJCC 8th edition staging classified eight cases (72.7%) as stage I, two (18.2%) as stage II, and one (9.1%) as stage IVB. Similar staging distributions were highlighted by Jeong et al. [9] who found most patients in stage I and II, likely due to the younger age profile of the cohort. Although Thompson [7] reported that 64% of cases occurred in patients ≥55 years, most were staged as AJCC stage I–II, with only a few reaching stages III or IV. As with other thyroid carcinomas, a higher AJCC stage is associated with worse prognosis [13].
The Ki-67 proliferation index varied across studies, with our median value of 14% (range, 5% to 45%). While some studies did not report or specify Ki-67 values, those that did reported medians ranging from 8.3% to 20%, and means from 5.6% to 9.4% (Table 3). Our findings, however, are consistent with WHO’s suggested range (10%–30%) [5]. Despite evidence linking high Ki-67 to metastatic potential, the prognostic significance remains debated, as Thompson [7] observed no clear difference in metastatic risk between low (<5%) and high (>15%) Ki-67 groups.
All cases had molecular investigation; two cases (18.2%) with BRAFV600E mutations, four cases (36.4%) with TERTp mutations, and two cases (18.2%) with multiple mutations (one with BRAFV600E and TERTp (C228T) mutation, the other with BRAFV600E and PIK3CA mutation). The remaining cases showed no detectable mutations in BRAFV600E, NRAS, HRAS, TP53, PIK3CA, PTEN, or the TERTp. Further investigation is warranted since the remaining three cases may harbor genetic alterations not assessed in this study. Cracolici and Cipriani [2] and Wong et al. [8] suggested that BRAFV600E mutations are more frequently associated with HGDTC. In this series, the molecular profiles align with prior reports across the different tumor types. HGDTC commonly showed driver mutations such as RAS, BRAF, PIK3CA, PPARγ, PTEN, CDKN2C, ARID1A, and PIK3R1, while TERTp mutations were typically observed as a later genetic event [7].
FNA was performed in three patients (27.3%), among these two (66.7%) were classified as Bethesda VI and one (33.3%) as Bethesda IV. Similar results were discovered by Tondi Resta et al. [13] (43.8% Bethesda VI and 31.3% Bethesda IV), reflecting the predominance of PTC subtypes in HGDTC [12,24]. That being said, cytologic diagnosis of HGDTC remains challenging. The presence of DTC components may obscure high-grade features and evaluation of necrosis along with mitotic activity is limited on cytologic preparations. FNA has low sensitivity for detecting high-grade features. Necrosis may appear nonspecific and mitotic figures are rarely seen. Even when present, they can be difficult to distinguish from apoptotic or degenerative cells. Additionally, mitotic index cannot be reliably quantified in cytology specimens due to a lack of standardized field size [24-26].
CNB was conducted in seven (63.6%) cases prior to thyroidectomy, with one (14.3%) case categorized as KTA IIIB, three (42.9%) as KTA IV, one (14.3%) as KTA V, and two (28.5%) as KTA VI. No prior studies have included CNB classification as a variable, and discrepancies between CNB and final histopathology were observed. These may be due to inadequate sampling or poor representation of nuclear features in follicular cells, where nuclei may appear smaller and hyperchromatic, limiting detection of PTC features [27]. In daily practice, HGDTC is rarely encountered and difficult to diagnose using small CNB specimens. Harahap et al. [27] reported only 3 HGDTC cases among 338 thyroid nodules sampled by CNB between July 2022 and July 2024 in our institution.
In terms of treatment, four patients underwent surgery alone, six received combined RAI therapy, and one with additional EBRT. All of our patients were still alive at follow-up. Thompson [7] also reported a mortality rate of three out of 17 HGDTC cases, with a median survival of 12.1 months following diagnosis [7,9,12].
There are several limitations to our study. The follow-up duration was relatively short, even though some of the reviewed cases dated back to 2019. A proper assessment of disease-specific survival and overall survival generally requires a minimum follow-up period of 5 years. In addition, the limited number of genes examined may have failed to reveal the underlying molecular alterations in some cases. Another limitation relates to mitotic quantification. Although all slides were re-reviewed and the mitotic counts remained largely consistent, some degree of measurement imprecision is still possible when relying solely on H&E sections. Our institution lacks PHH3 immunostaining, a technique that improves the accuracy of counting cell division (mitoses). This absence may have reduced the precision of our proliferative assessment.
This study provides a comprehensive overview of our institutional experience with HGDTC. We reported 11 cases of HGDTC diagnosed between 2019 and 2024. The mean patient age was 54.6 years, which is similar to those in previous studies. Most cases of HGDTC are dominated by women, with a female-to-male ratio of 2.7:1. The cases in our cohort were consistent with the characteristics described by the WHO, including relatively large tumor size (mean, 6.4 cm), a high frequency of extrathyroidal extension, and the presence of metastases at initial presentation. Six cases (54.5%) had metastases at diagnosis; all involved lymph nodes (100%), and one case (16.7%) showed distant organ metastasis. Most cases (90.9%) were PTC, with the tall cell subtype being the most frequent, followed by the IEFVPTC and classic subtypes. All HGDTC cases showed tumor necrosis, and the mean mitotic count was 3 per 2 mm2. The median of Ki-67 proliferation index was 14% (range, 5% to 45%). All of our cases had molecular investigation: BRAFV600E mutations in two cases, TERTp mutations in four cases, multiple mutations (BRAFV600E and TERTp (C228T), BRAFV600E and PIK3CA) were detected in two cases, while three cases showed no detectable mutations in BRAFV600E, NRAS, HRAS, TP53, PIK3CA, PTEN, or the TERTp. These findings reflect the aggressive nature of HGDTC, which clearly distinguishes it from DTC without high-grade features. Given its poorer prognosis and more advanced clinical behavior, accurate recognition of HGDTC is essential.
Supplementary Information
The Data Supplement is available with this article at https://doi.org/10.4132/jptm.2026.01.15.
Notes
Ethics Statement
This retrospective study has been approved by the Ethics Committee of Universitas Indonesia (No. KET-901/UN2.F1/ETIK/PPM.00.02/2024). The need for informed consent was waived due to the retrospective nature of the study.
Availability of Data and Material
The datasets generated or analyzed during the study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
Author Contributions
Conceptualization: N, ASH, CKJ. Data curation: N, ASH, MFH. Formal analysis: N, ASH, AW. Investigation: N, ASH, MFH, CKJ. Methodology: N, ASH, AW, CKJ. Project administration: N, AW. Resources: N, ASH, MFH. Supervision: MFH, CKJ. Validation: ASH, MFH, CKJ. Visualization: N, ASH. Writing—original draft: N, ASH, AW. Writing—review & editing: all authors. Approval of final manuscript: all authors.
Conflicts of Interest
C.K.J., the editor-in-chief of the Journal of Pathology and Translational Medicine, were not involved in the editorial evaluation or decision to publish this article. All remaining authors have declared no conflicts of interest.
Funding Statement
No funding to declare.