Phenotypic plasticity in plasma cell myeloma: a CD138-negative case with a rare BRAF G469R mutation
Article information
Abstract
CD138-negative plasma cell myeloma harboring a BRAF G469R mutation is described in a 76-year-old male presenting with multiple osteolytic lesions. Histologically, the lesion exhibited epithelioid to plasmacytoid morphology with prominent mitotic activity and vascular-like spaces. Immunophenotyping demonstrated strong vimentin and CD31 expression but absence of CD138 and other endothelial markers. Light-chain in situ hybridization confirmed a clonal κ-restricted plasma cell population. Bone marrow examination revealed near-complete replacement by atypical plasma cells, retaining CD138 negativity and demonstrating focal CD20 positivity, indicative of intratumoral heterogeneity. Next-generation sequencing identified a rare BRAF G469R variant. The patient exhibited poor response to bortezomib, lenalidomide, and dexamethasone therapy, necessitating a switch to carfilzomib-based treatment. This case underscores the diagnostic challenges of CD138-negative myeloma and highlights the importance of integrating morphology, immunophenotyping, and molecular profiling to inform accurate diagnosis and guide therapeutic strategies.
INTRODUCTION
Plasma cell myeloma is a neoplasm characterized by the clonal proliferation of terminally differentiated plasma cells, typically expressing syndecan-1 (CD138) as a defining surface marker. CD138 is a heparan sulfate proteoglycan involved in plasma-cell adhesion, differentiation, and secretion, and its expression is considered a hallmark of mature plasma cells [1,2].
However, rare cases of CD138-negative plasma cell myeloma have been reported, presenting significant diagnostic challenges due to their unusual morphology and immunophenotypic profiles that may mimic poorly differentiated sarcomas or hematolymphoid neoplasms of other lineages [3]. The loss or downregulation of CD138 has been associated with dedifferentiation, increased clonogenic potential, and resistance to chemotherapy, suggesting that this phenotype may represent a biologically distinct and clinically aggressive subset of myeloma [4-9].
In addition, molecular profiling using techniques such as next-generation sequencing (NGS) has been increasingly important in revealing genetic alterations that may contribute to disease heterogeneity and therapeutic response. Among these, BRAF mutations, most commonly the V600E substitution, have been identified in a small subset of myelomas, reflecting aberrant mitogen-activated protein kinase (MAPK) pathway activation [10-12]. Nevertheless, non-V600E variants such as BRAF G469R remain exceedingly rare, and their clinicopathologic significance in plasma cell neoplasms has not been well characterized.
Herein, we describe a case of CD138-negative plasma cell myeloma harboring a rare BRAF G469R mutation, which initially presented with atypical epithelioid morphology and an unusual immunophenotypic profile, posing a significant diagnostic challenge.
This case highlights the importance of combining immunohistochemistry, light-chain in situ hybridization (ISH), and molecular testing in atypical plasma cell neoplasms, and provides further insight into the phenotypic plasticity and molecular diversity of myeloma.
CASE REPORT
A 76-year-old man with a history of hypertension and diabetes presented with progressive left thigh pain for approximately six months. Radiographic examination performed at an outside clinic revealed multiple osteolytic lesions involving the ischium and the subtrochanteric region of the left femur, raising clinical suspicion for a metastatic bone lesion. The patient was referred to our institution for further evaluation and management. Laboratory studies on admission demonstrated anemia (hemoglobin 7.8 g/dL), thrombocytopenia (103 × 10³/µL), and mild leukopenia (white blood cell count 3.76 × 10³/µL). Serum biochemistry revealed elevated lactate dehydrogenase (264 U/L) and mild hypercalcemia (10.8 mg/dL). Urinalysis showed 30 mg/dL proteinuria, prompting quantitative assessment.
To obtain tissue for diagnosis and to stabilize the bone, curettage of the left femoral lesion was performed. Histologically, the specimen showed large epithelioid to plasmacytoid cells with macronucleoli, stippled chromatin, moderate amphophilic cytoplasm, and frequent mitoses in the background of multiple blood filled cystic spaces (Fig. 1A, B). Immunohistochemically, the tumor cells were negative for CD138, leukocyte common antigen, CD20, CD3, CD30, terminal deoxynucleotidyl transferase, myeloperoxidase, and cytokeratin, but demonstrated strong positivity for vimentin and CD31 (Fig. 1C, D), initially suggesting a vascular tumor. However, additional endothelial markers (CD34, ERG, and D2-40) were negative, and a definitive line of differentiation could not be established at that stage.
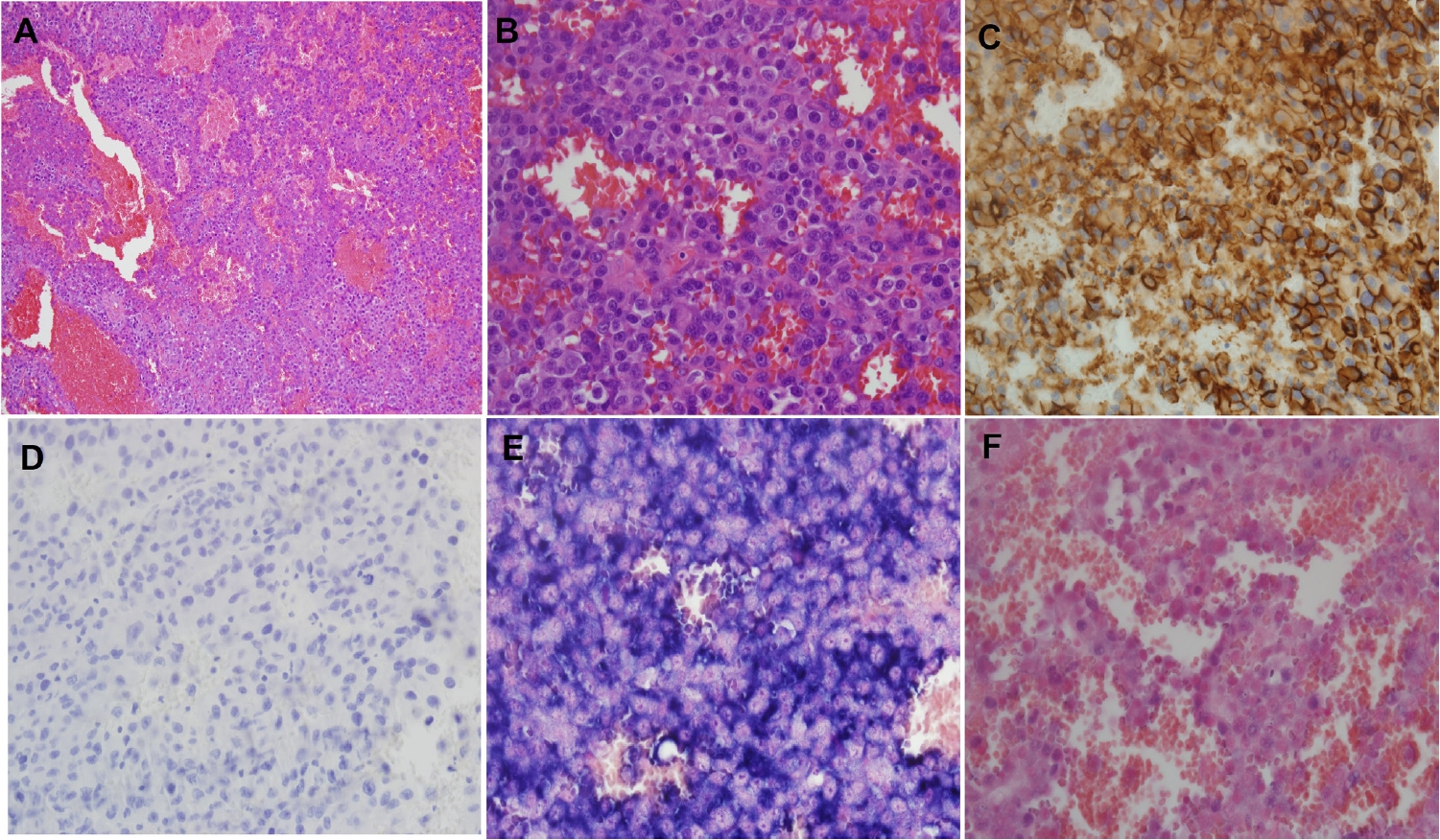
Histologic and immunohistochemical findings of the femoral lesion. (A) Low-power view showing a solid sheet of tumor cells with multiple red-blood-cell–filled cystic spaces. (B) High-power view demonstrating large epithelioid cells with macronucleoli and amphophilic cytoplasm. (C) Immunostaining for CD31 shows diffuse positivity in tumor cells. (D) CD138 is completely negative. (E) In situ hybridization for κ light chain shows diffuse and strong positivity, confirming κ-restricted clonality. (F) λ light chain is negative.
During concurrent clinical evaluation, the patient’s cytopenias and abnormal protein studies raised clinical suspicion for multiple myeloma (MM). Quantitative urine testing showed 401.5 mg/day of protein, and β₂-microglobulin was markedly elevated (11.28 mg/L). Urine immunofixation revealed a faint κ light-chain band, consistent with trace Bence-Jones proteinuria, although the overall urinary protein was predominantly non-monoclonal. In contrast, serum free light chain assay revealed markedly elevated κ light chain at 342.02 mg/L, with λ light chain 14.36 mg/L, resulting in a κ/λ ratio of 23.8, confirming systemic κ-restricted monoclonality.
Given the clinical suspicion of MM, CD138 immunostaining was repeated on multiple tissue sections, but remained negative. ISH for immunoglobulin light chains was subsequently performed, which demonstrated diffuse κ-restricted clonal plasma cell population with complete absence of λ expression (Fig. 1E, F), thus establishing the diagnosis of CD138-negative plasma cell myeloma in the bone lesion.
Subsequent bone marrow biopsy revealed near-total replacement (~99%) of marrow cellularity by atypical plasma cells, confirming advanced-stage MM. Compared with the initial bone lesion, the tumor cells in the bone marrow were morphologically more mature plasma cells (Fig. 2A, B). Immunohistochemically, the tumor cells remained negative for CD138 but demonstrated diffuse κ light-chain positivity on ISH (Fig. 2C, D). Interestingly, CD20 showed focal expression in a subset of tumor cells, indicating minor phenotypic heterogeneity within the plasma cell population, whereas paired box gene 5 (PAX5) was negative, excluding true B-cell lineage differentiation (Fig. 2E, F).
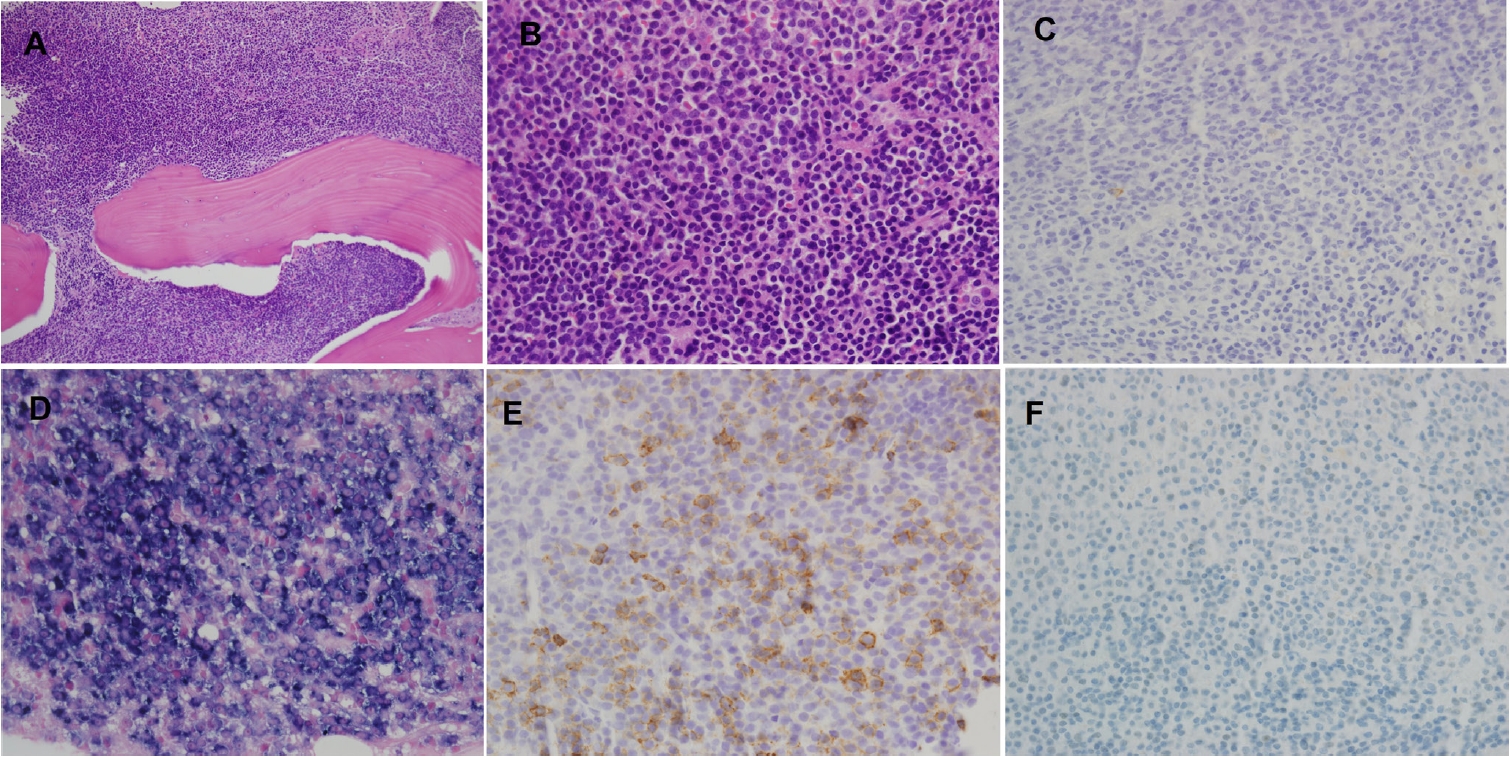
Bone marrow findings. (A) Diffuse infiltration of the bone marrow by atypical plasma cells, replacing nearly all hematopoietic elements. (B) The tumor cells show relatively smaller size and more mature morphology compared with the femoral lesion. (C) Immunohistochemistry for CD138 shows complete loss of expression. (D) In situ hybridization for κ light chain demonstrates diffuse and strong positivity, confirming κ-restricted clonality. (E) CD20 shows focal cytoplasmic and membranous positivity in a subset of tumor cells, indicating minor phenotypic heterogeneity. (F) PAX5 is completely negative, confirming absence of B-cell lineage differentiation.
These findings supported systemic involvement by κ-restricted plasma cell myeloma with partial maturation but persistent loss of CD138 expression. NGS of the bone marrow sample identified a BRAF c.1405G>A (p.Gly469Arg) mutation.
The patient was started on bortezomib, lenalidomide, and dexamethasone (VRD) chemotherapy; however, after six cycles, disease progression was observed, and therapy was transitioned to a carfilzomib and dexamethasone regimen.
DISCUSSION
CD138-negative plasma cell myeloma represents a rare and diagnostically challenging subtype of MM. In the present case, the initial biopsy exhibited an epithelioid morphology with vascular-like spaces and an unusual immunophenotypic profile, including vimentin and CD31 expression in the absence of conventional plasma cell markers, which complicated the diagnostic process. Ultimately, demonstration of κ-restricted plasma cells by ISH established the diagnosis of CD138-negative plasma cell myeloma, highlighting the importance of ancillary molecular studies in diagnostically ambiguous cases.
CD31 (PECAM-1) is widely recognized as an endothelial marker; however, its expression is not restricted to endothelial cells. Multiple studies show that CD31 is also expressed in reactive and neoplastic plasma cells, plasmablastic variants of myeloma, and other hematopoietic cells such as leukocytes, monocytes, and platelets, highlighting that CD31 positivity alone cannot be used to confirm endothelial lineage in tumors [13]. In plasma cell lesions specifically, CD31 expression has been documented in a high proportion of conventional myelomas and plasmablastic variants, indicating that CD31 expression in plasma cells is a recognized immunophenotypic feature and may confound differential diagnosis with vascular neoplasms if interpreted without additional lineage-specific markers [14].
CD138 (syndecan-1) is a transmembrane heparan-sulfate proteoglycan essential for plasma cell adhesion, differentiation, and secretion [1,2]. Loss or downregulation of CD138 occurs in a subset of myelomas and is associated with an immature, stem-like phenotype and drug resistance [4,7,8]. Experimental studies have shown that CD138-low or -negative myeloma cells exhibit increased clonogenicity, self-renewal potential, and reduced expression of terminal differentiation markers [4-6]. In our patient, the absence of CD138 expression correlated with atypical morphology and poor treatment response. After six cycles of VRD chemotherapy, the disease progressed and required transition to a carfilzomib-based regimen, consistent with the chemoresistant behavior described in CD138-low myelomas [7]. The microenvironmental interactions of CD138-negative cells may further contribute to therapeutic resistance through alterations in stromal stiffness and SDF-1/CXCR4 signaling [9].
Molecular analysis of the bone marrow specimen revealed a BRAF G469R mutation, a rare non-V600E variant within the kinase domain. BRAF mutations are functionally classified into three categories based on their mechanism of MAPK pathway activation [10,11]. Class I mutations, typified by V600E, signal as RAS-independent monomers with high kinase activity. In contrast, class II mutations, including G469 substitutions, activate downstream signaling through RAS-independent dimerization, while class III mutations exhibit impaired kinase activity and rely on upstream RAS activation. Accordingly, BRAF G469R is classified as a Class II mutation, leading to constitutive MAPK pathway activation via dimer-dependent signaling.
In MM, BRAF mutations are reported in a small subset of cases, with V600E being the most common variant. Non-V600E mutations such as G469R are exceedingly rare, and their clinicopathologic significance remains poorly defined [12]. Limited available data suggest that MAPK pathway activation through non-V600E BRAF mutations may contribute to disease progression, clonal evolution, and therapeutic resistance. In other malignancies, including non-small cell lung carcinoma, melanoma, and colorectal carcinoma, BRAF G469 mutations have been associated with limited responsiveness to BRAF inhibitor monotherapy, whereas downstream inhibition with MEK or ERK inhibitors may offer greater therapeutic potential [10,11]. Although targeted therapy was not applied in the present case, the identification of a class II BRAF mutation raises the possibility that aberrant MAPK signaling contributed to the aggressive clinical course and resistance to conventional therapy.
Interestingly, focal CD20 positivity was observed in the bone marrow lesion, whereas the initial femoral lesion was CD20 negative. This finding suggests phenotypic heterogeneity within the tumor and may reflect clonal evolution during disease progression. CD20-positive myelomas are uncommon, detected in roughly 10%–20% of cases, and have been associated with t(11;14)(q13;q32) translocation, lymphoplasmacytoid morphology, and sometimes adverse prognosis [15]. However, the clinical implications remain controversial. In our case, despite focal CD20 expression, PAX5 was negative, indicating that the CD20-positive population does not represent true B-cell lineage differentiation. Rather, this pattern may reflect partial re-expression of B-cell antigens during clonal evolution or phenotypic drift within the plasma cell clone. Such partial immunophenotypic reversion, combined with the absence of CD138, supports the concept of plasticity and dedifferentiation in certain myeloma subclones [6,7,9,15]. This dynamic immunophenotypic change might also underlie the chemoresistant and stem-like behavior described in CD138-low or -negative myelomas.
In conclusion, CD138-negative plasma cell myeloma represents a biologically distinct phenotype characterized by diagnostic ambiguity, reduced secretory activity, and relative resistance to therapy. Recognition of this variant and application of light-chain ISH and molecular testing are crucial for accurate diagnosis. Future studies correlating immunophenotypic variability (including CD20 and CD138) with genomic drivers such as BRAF mutations may clarify mechanisms of dedifferentiation and identify potential therapeutic targets.
Notes
Ethics Statement
This study was approved by the Institutional Review Board of Kosin University Gospel Hospital with a waiver of informed consent (IRB No. 2025-10-029).
Availability of Data and Material
The datasets generated or analyzed during the study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
Author Contributions
Conceptualization: SJO. Investigation: SJO, SHC. Formal analysis: SJO, SHC. Supervision: SJO. Visualization: SJO, SHC. Writing—original draft: SJO. Writing—review & editing: SJO. Approval of final manuscript: all authors.
Conflicts of Interest
The authors declare that they have no potential conflicts of interest.
Funding Statement
No funding to declare.
