Mutational status of non-invasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP): molecular analysis should be performed for NIFTPs with nuclear score 3
Article information
Abstract
Background
The classification of non-invasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP) was introduced to prevent the overtreatment of indolent tumors that were formerly diagnosed as non-invasive encapsulated follicular variant papillary thyroid carcinomas (NIEFV-PTCs). Although NIFTP was initially estimated to account for 10%–20% of papillary thyroid carcinomas in Western populations, its incidence is substantially lower in Asian cohorts. However, a multi-institutional Japanese study revealed that 31.0% of tumors previously diagnosed as follicular adenomas (FAs) were reclassified as NIFTPs. NIFTP diagnosis requires a nuclear score (NS) of 2–3, and according to the recent World Health Organization criteria, molecular analysis is recommended, but not mandatory, to exclude high-risk subtypes, namely cases with the BRAFV600E mutation, particularly for NS3 tumors.
Methods
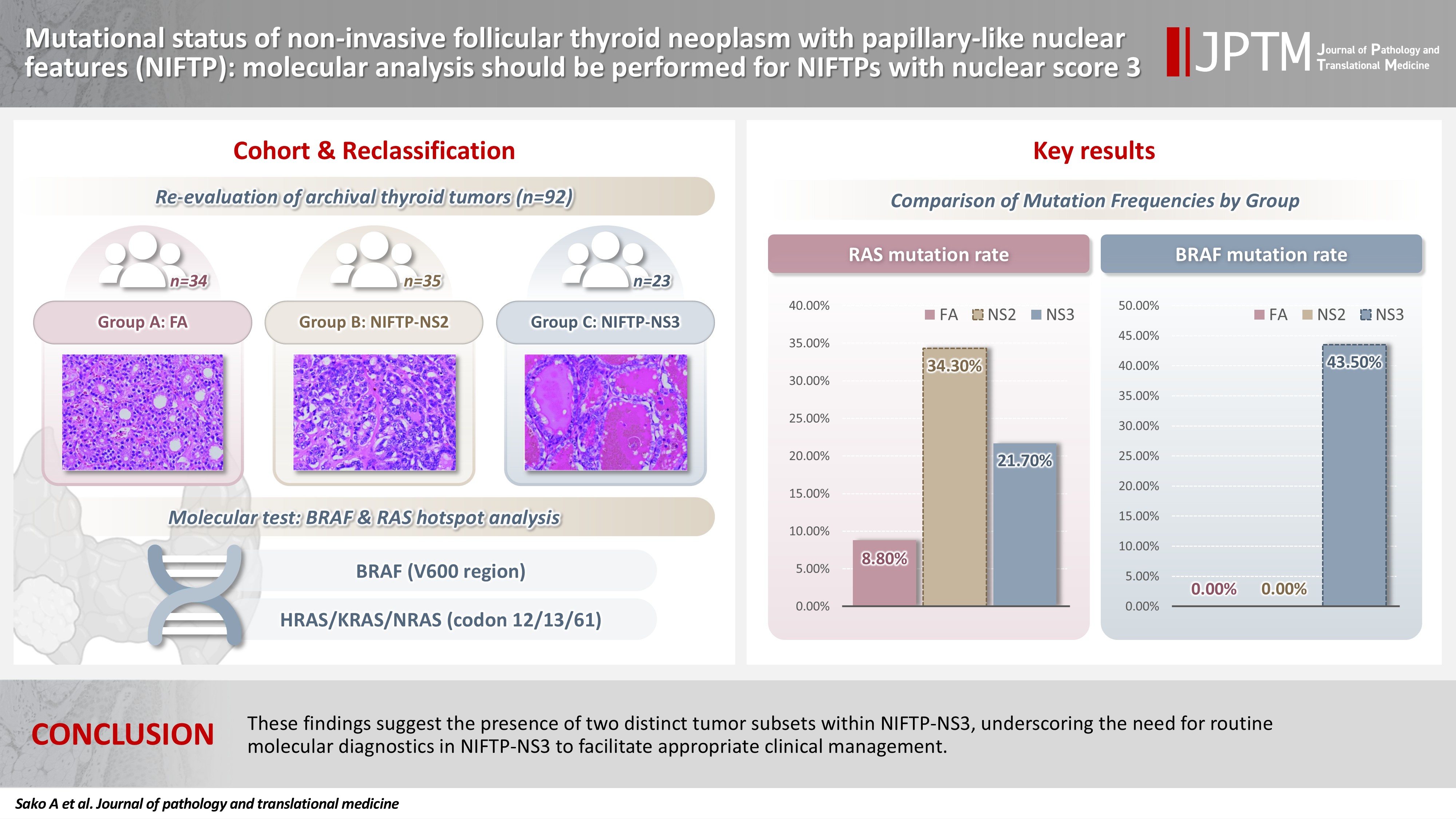
We performed genetic analysis on 92 archival thyroid tumor samples, including 69 previously diagnosed as FA, of which 34 remained as FA upon re-evaluation (group A) and 35 were reclassified as NIFTP with NS2 (group B). Additional 23 tumors previously diagnosed as NIEFV-PTC were reclassified as NIFTP with NS3 (group C).
Results
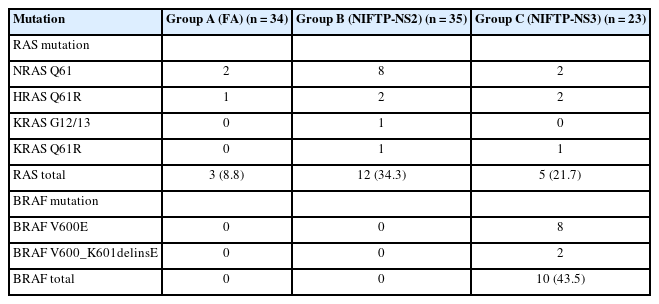
RAS mutations were detected in 8.8%, 34.3%, and 21.7% of the tumor samples in groups A, B, and C, respectively, whereas BRAF mutations were present in 43.5% of the tumor samples in group C only.
Conclusions
These findings suggest the presence of two distinct tumor subsets within NIFTP-NS3, underscoring the need for routine molecular diagnostics in NIFTP-NS3 to facilitate appropriate clinical management.
INTRODUCTION
Non-invasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP) was introduced as a new classification in 2016 [1] and categorized as a borderline tumor in the 4th edition of the World Health Organization (WHO) classification [2]. These tumors were formerly classified as non-invasive encapsulated follicular variant of papillary thyroid carcinomas (NIEFV-PTCs); however, preventing overtreatment due to their indolent biological behavior was considered necessary. Therefore, the term “neoplasm” was adopted for NIFTP. Recently, NIFTP has been categorized as a low-risk neoplasm in the 5th edition of the WHO classification [3].
The diagnostic criteria for NIFTP have undergone slight changes. The original diagnostic criteria allowed for less than 1% papillae [1]. However, broader use of the “less than 1% papillae” criterion resulted in metastasis being observed in 4%–6% of tumors diagnosed as NIFTP [4]. According to the 5th edition of the WHO classification [3], the term “<1% true papillae” is used and defined as a fibrovascular core lined by cells with score 2–3 nuclei. In addition, the new criteria introduced not only changes to the morphological criteria but also recommendation for molecular analysis, although such testing is not mandatory [3]. The detection of the BRAFV600E mutation by immunostaining or genotyping excludes the diagnosis.
The proportion of NIFTP among PTC cases has been reported to be approximately 10%–20% [5]; however, that in Asian countries has been reported to be considerably lower (0.8%) [6]. According to a multi-institutional study in Japan that assessed more than 4,000 nodules [7], 0.9% of tumors initially diagnosed as PTCs before the introduction of NIFTPs were reclassified as NIFTP. Notably, 31.0% of follicular adenomas (FAs) were recategorized as NIFTPs. This finding suggests that the introduction of NIFTP, which was originally intended to downgrade certain tumors from cancer to neoplasm, resulted in an upgrade in Japan.
The nuclear score (NS) is used to evaluate the nuclear features of PTC, ranging from 0 to 3, based on so-called papillary nuclear features such as grooves, pseudoinclusions, and clearing, and NS2–3 is required for the diagnosis of NIFTP. In the aforementioned Japanese multi-institutional study, most patients initially diagnosed with FA exhibited NS2 [7]. In other words, NIFTPs with NS2 had been diagnosed as PTC in Western countries, whereas they had been diagnosed as FA in Japan.
In the new criteria, molecular analysis or immunohistochemistry (IHC) is recommended to check for the BRAFV600E mutation or other driver mutations (RET fusions, TERT promoter, or TP53) to exclude high-risk tumors, especially for NIFTPs with NS3 [4]. Therefore, tumors with these mutations should not be diagnosed as NIFTP. This is because a small proportion of NIFTPs were found to have metastases, and many harbored the BRAFV600E mutation [4]. These findings highlight the importance of the differences between NS2 and NS3. This difference may reflect biologically distinct tumor characteristics. Comprehensive genomic and transcriptomic analyses conducted in the Cancer Genome Atlas study have revealed that PTCs can be classified into BRAF- and RAS-like tumors [8]. This distinction may also apply to tumors that fulfill the histological criteria for NIFTP. Therefore, in the present study, we conducted mutational analyses of BRAF and RAS to clarify the differences between these tumors.
MATERIALS AND METHODS
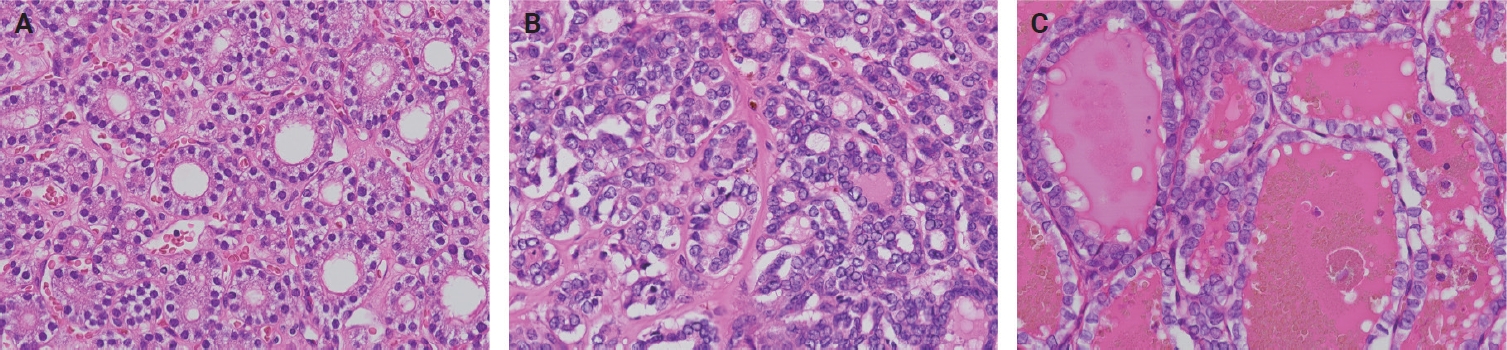
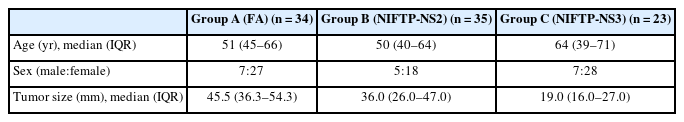
A total of 92 tumor samples that were resected at Kuma Hospital and matched the following three criteria (A–C) were used in this study. These patients were diagnosed with FA or NIEFV-PTC between 2017 and 2021 before the disease entity NIFTP was introduced into Japanese practice. (A) Thirty-four tumors with an original diagnosis of FA were still diagnosed as FA after re-evaluation. (B) Thirty-five tumors with an original diagnosis of FA were diagnosed as NIFTP after re-evaluation. (C) Twenty-three tumors with an original diagnosis of NIEFV-PTC were diagnosed as NIFTP after re-evaluation. The tumors in groups B and C had NS2 and NS3, respectively. Representative histological images are shown in Fig. 1. The re-diagnosis was made in accordance with the 5th edition of the WHO classification by a pathologist (MHiro) who specializes in thyroid pathology. Only tumors with unambiguous classifications were included. Uncertain malignant potential cases with questionable capsular invasion and/or tumors with NS between 1 and 2 were excluded. Note that we re-reviewed only slides that were preserved in the pathology department, and therefore all portions of each tumor were not examined. Patient demographics and tumor sizes are summarized in Table 1. It should be noted that these cases were intentionally selected and may therefore contain substantial selection bias.
DNA was extracted from formalin-fixed, paraffin-embedded (FFPE) sections using an AllPrep DNA/RNA FFPE kit (QIAGEN, Hilden, Germany). The mutational status of BRAF (around V600) and HRAS, KRAS, and NRAS (codons 12, 13, and 61) was analyzed by direct Sanger sequencing. First, polymerase chain reaction (PCR) was performed using KOD SYBR qPCR Mix (TOYOBO, Osaka, Japan), and the PCR product was treated with ExoSAP-IT Express (Thermo Fisher Scientific, Waltham, MA, USA). Sequencing analysis was performed using a BigDye Terminator sequencing kit v3.1 (Thermo Fisher Scientific) and an ABI3100 sequencer (Thermo Fisher Scientific). The primers used in this study are listed in Table 2.
For samples whose PCR amplification was not successful, droplet digital PCR (ddPCR) was performed. PrimePCR for ddPCR NRAS Q61 (#12001006), KRAS G12/G13 (#1863506), and KRAS Q61 (#12001626) Screening Kits (Bio-Rad, Hercules, CA, USA), ddPCR Mutation Assay KRAS p.Q61R (dHsaMDV2010135) and HRAS p.Q61R (dHsaMDV2510576) (Bio-Rad), a QX100 droplet generator (Bio-Rad), and a QX200 droplet reader (Bio-Rad) were used for the ddPCR analysis.
RESULTS
The frequencies of RAS mutations in groups A, B, and C were 8.8%, 34.3%, and 21.7%, respectively. In group A, two NRAS Q61R and one HRAS Q61R were detected. In group B (NIFTP-NS2), eight NRAS Q61R, two HRAS Q61R, one KRAS G12/13, and one KRAS Q61R were detected. In group C (NIFTP-NS3), two NRAS Q61R, two HRAS Q61R, and one KRAS Q61R were detected. The BRAFV600E mutation was found in eight cases in group C but not in group A or B. Additionally, a relatively rare BRAF mutation, c.1799_1801delTGA, resulting in p.V600_K601delinsE, was detected in two cases in group C. BRAF mutations were detected in 43.5% of the cases in group C. The summarized data are listed in Table 3. We assessed whether any morphological differences existed between BRAF- and RAS-mutated tumors in group 3, but none were identified.
DISCUSSION
The present study clearly showed that there are two pathogenetically distinct types of tumors in NIFTP-NS3. One was characterized by RAS mutations (i.e., RAS-like tumors), and the other by the BRAFV600E mutation (i.e., BRAF-like tumors). The characteristics of these tumors are entirely different. Given that the NIFTP category was originally established to downgrade the classification from cancer to neoplasm, the inability to make this distinction based solely on hematoxylin and eosin–stained slides seems to be a significant issue. The WHO recommends genetic testing, but it is not mandatory.
The frequency of RAS mutations in group A (FA) was 8.8%, which was lower than that in group B (NIFTP-NS2) (34.3%). According to previous reports, the frequencies of RAS mutations in FAs were 18% [9], 19.6% [10], and 30% [11], while those in NIFTPs were 29.6% [1], 54% [12], and 67% [13]. These findings suggest that NIFTPs exhibit a higher frequency of RAS mutations than FAs. One possible explanation is that NIFTP-NS2 is predominantly driven by RAS mutations, whereas FA may involve other driver events. Nevertheless, the presence of shared mutations also indicates a degree of molecular overlap between the two. Additionally, we selected cases with typical histological subtypes for each group, which may have contributed to the results.
No tumors in group B (NIFTP-NS2) harbored BRAF mutations, whereas 43.5% of the tumors in group C (NIFTP-NS3) did (BRAFV600E: 34.8%); such tumors should not be classified as NIFTP. Therefore, an accurate diagnosis of NIFTP-NS3 requires confirmation of the BRAF mutational status. When molecular testing is not available, at minimum, IHC for RASQ61R and/or BRAFV600E should be considered. In this context, tumors that are histologically NIFTP-NS3 but harbor the BRAF mutation may be more appropriately referred to as “encapsulated classic PTC with predominant follicular architecture.”
In this study, we detected a relatively rare BRAF mutation, p.V600_K601delinsE. Torregrossa et al. [14] reported that this mutation is the second most frequent rare mutation (except for V600E) occurring near V600, following K601E. One study reported its presence in aggressive histological subtypes [15], while the aforementioned study suggested that it is not highly aggressive. However, owing to the limited number of cases, its exact characteristics, including whether it should be classified as NIFTP, remain unclear.
Japanese pathologists have long maintained a diagnostic practice that differentiates between NIFTP-NS0–2 and NIFTP-NS3 rather than between NS0–1 and NS2–3. This perspective may have worked well in clinical practice, given that only HE-stained sections are available, and is also supported by molecular genetics. Therefore, the necessity of the NIFTP concept in Japan has been questioned in clinical practice. However, unification is essential in science for accurate communication and data comparison. If NIFTP is to be defined as a RAS-like tumor, genetic testing (or at least IHC) should be mandated for at least the NS3 cases. This also raises concerns regarding the current diagnostic categorization, which combines NIFTP-NS2 and NIFTP-NS3 under the same diagnosis of NIFTP. Attempts have been made to distinguish BRAF-like tumors from RAS-like tumors based on morphology [16,17], and such differentiation, if feasible, would greatly contribute to the clinical management of thyroid tumors. However, in our study, no morphological differences could be identified between BRAF-like and RAS-like tumors within group 3. In the future, studies should perhaps be conducted to investigate whether analyses of morphological features using, for example, artificial intelligence, can distinguish between BRAF-like and non-BRAF-like tumors.
BRAFV600E can also be detected by IHC using mutation-specific antibodies. Two meta-analyses evaluating the diagnostic accuracy of this approach reported sensitivities of 96.8% (95% confidence interval [CI], 94.1 to 98.3) and 100% (95% CI, 97 to 100), with specificities of 86.3% (95% CI, 80.7 to 90.4) and 84% (95% CI, 72 to 91), respectively [18,19]. Although the sensitivity was consistently high, the specificity was somewhat lower, and substantial inter-institutional heterogeneity was noted, suggesting that some caution is advisable in clinical applications. However, one study has reported very high sensitivity (99.3%) and 100% specificity, suggesting that optimization of the methodology may be crucial [20].
This study had several limitations. First, all the tissue samples were collected from a single institution, which introduced the possibility of sampling bias. Second, since the present study included only cases with typical histopathological features, some degree of selection bias cannot be ruled out. Third, genetic alterations other than those in RAS and BRAF hotspots were not evaluated. Fourth, not all RAS mutation variants may have been detected, particularly in samples with lower DNA quality.
In conclusion, NIFTP-NS3 contains tumors with at least two distinct characteristics, and we believe that molecular diagnostics are necessary. Otherwise, NS3 may require a different classification.
Notes
Ethics Statement
The study protocol was approved by the Institutional Review Boards of Kuma Hospital (#20231012-4, Oct 12, 2023) and Nagasaki University Graduate School of Biomedical Sciences (#20170502-14, Mar 17, 2025). The study was conducted in accordance with the ethical standards of the 1964 Declaration of Helsinki and its later amendments. Informed consent was obtained from all participants.
Availability of Data and Material
The datasets generated or analyzed during the study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
Author Contributions
Conceptualization: MH (Mitsuyoshi Hirokawa), NM. Data curation: AS, MM. Formal analysis: AS, MM. Funding acquisition: MM, NM. Investigation: AS, MM. Methodology: AS, MM, NM. Project administration: MH (Mitsuyoshi Hirokawa), NM. Resources: MH (Mitsuyoshi Hirokawa), MH (Miyoko Higuchi), AM, TA. Supervision: AK, NM. Visualization: AS. Writing—original draft: AS, NM. Writing—review & editing: MH (Mitsuyoshi Hirokawa), NM. Approval of final manuscript: all authors.
Conflicts of Interest
The authors declare that they have no potential conflicts of interest.
Funding Statement
This study was supported in part by Grants-in-Aid for Scientific Research (JSPS KAKENHI) Grant Numbers 23H02860, 20KK0217, and 22K07668, the Program of the Network-Type Joint Usage/Research Center for Radiation Disaster Medical Science, and intramurally by the Atomic Bomb Disease Institute, Nagasaki University.