Prevalence of HER2-ultralow breast cancer in South Korea: a multicenter study by reassessment of HER2-zero cases
Article information
Abstract
Background
This study aimed to determine the prevalence of human epidermal growth factor receptor 2 (HER2)–ultralow breast cancer among cases initially classified as HER2 immunohistochemistry (IHC) 0 and assess interobserver variability in interpreting low-level HER2 expression.
Methods
In this multicenter retrospective study, all invasive breast cancer cases diagnosed between January and December 2022 across 10 Korean institutions were retrieved. Institutional pathologists reexamined HER2 IHC slides originally reported as IHC 0 according to the 2018 American Society of Clinical Oncology/College of American Pathologists guidelines and reclassified them as HER2-null (0), HER2-ultralow (0+), or HER2-low (1+). Slides from 10% of HER2-null and HER2-ultralow cases were digitized for central review and independently assessed by two pathologists, with discrepancies resolved by consensus.
Results
Among 8,026 cases, 2,836 cases (35.5%) were initially reported as IHC 0. Upon re-review, 1,673 (59.0%), 1,139 (40.2%), and 24 (0.8%) cases were reclassified as HER2-null, HER2-ultralow, and HER2-low, respectively. The prevalence of HER2-ultralow breast cancer varied considerably across institutions (23.7%–78.1%). Central review of 268 digitized cases showed concordance in 193 cases (72.0%). Among the 75 discordant cases, 54 tumors (72.0%) were upgraded from HER2-null to HER2-ultralow, and 18 (24.0%) tumors were upgraded from HER2-ultralow to HER2-low. Furthermore, two tumors (2.7%) were downgraded from HER2-ultralow to HER2-null.
Conclusions
Approximately 40% of cases initially categorized as IHC 0 were reclassified as HER2-ultralow. The substantial inter-institutional variability observed in interpreting low-level HER2 expression highlights the need for standardized training and quality assurance to ensure accurate identification of patients eligible for HER2-targeted antibody–drug conjugates.
INTRODUCTION
Accurate assessment of human epidermal growth factor receptor 2 (HER2) status is essential for the management of breast cancer (BC), as it guides therapeutic decisions and provides important prognostic information. HER2 status is traditionally determined by immunohistochemistry (IHC) and in situ hybridization (ISH) assays, which classify tumors as HER2-positive (IHC 3+ or 2+ with ISH amplification) or HER2-negative (IHC 0, 1+, or 2+ without ISH amplification), directing the use of HER2-targeted therapy or conventional treatment. Therefore, diagnostic efforts have primarily focused on differentiating HER2-overexpressed/amplified tumors from non-overexpressed/non-amplified counterparts [1-4].
However, the advent of trastuzumab deruxtecan (T-DXd), a novel antibody–drug conjugate (ADC), has drawn attention to a subset of tumors termed HER2-low BCs. These tumors, previously categorized as HER2-negative, have demonstrated significant therapeutic responses to T-DXd therapy [5,6]. The HER2-low category includes tumors defined as IHC 1+ or IHC 2+ without gene amplification by ISH. The latest American Society of Clinical Oncology (ASCO)/College of American Pathologists (CAP) guidelines for HER2 testing recommend including a footnote in pathology reports highlighting the potential eligibility of patients with HER2-low expression for T-DXd therapy [7]. More recently, an additional subgroup known as HER2-ultralow has emerged. HER2-ultralow tumors are defined as IHC 0 with faint but detectable membranous staining (IHC >0 and <1+) and have also demonstrated responsiveness to T-DXd [8]. Recognition of the HER2-ultralow subgroup has prompted pathologists to further subclassify HER2 IHC 0 tumors into HER2-null (complete absence of membranous staining) and HER2-ultralow [9], even though IHC assays were not originally designed for such fine gradiations [10]. Consequently, several studies have reported low interobserver agreement in differentiating between HER2-null, HER2-ultralow, and HER2-low cases [11-14].
T-DXd initially received accelerated approval from the U.S. Food and Drug Administration (FDA) in December 2019 for treating adult patients with unresectable or metastatic HER2-positive BC after two or more prior anti-HER2 regimens. In January 2025, the indication was expanded to include patients with hormone receptor–positive, unresectable or metastatic HER2-low or HER2-ultralow BC following endocrine therapy [15,16]. In South Korea, the Ministry of Food and Drug Safety (MFDS) approved T-DXd in September 2022 for HER2-positive breast and gastric cancers and subsequently expanded its use in May 2024 to include patients with metastatic HER2-low BC [17,18]. Approval for HER2-ultralow disease is anticipated in the near future.
We previously conducted a nationwide, multicenter study involving 25 Korean institutions that evaluated the frequency of HER2-low BC, inter-institutional variability, and technical factors influencing HER2 IHC interpretation [19]. That study revealed substantial inter-institutional heterogeneity and highlighted the need for standardized diagnostic protocols. Building upon these findings, the present study aimed to specifically examine the incidence of HER2-ultralow tumors among cases initially categorized as IHC 0.
The primary objective of this study was to determine the proportion of HER2-ultralow tumors within the IHC 0 cohort, thereby establishing baseline epidemiologic data in the Korean BC patient population in the context of expanding HER2-targeted ADC therapies. In addition, we evaluated inter-pathologist concordance in interpreting minimal HER2 expression, a critical issue as indications for T-DXd are expected to include HER2-ultralow disease.
MATERIALS AND METHODS
Study population
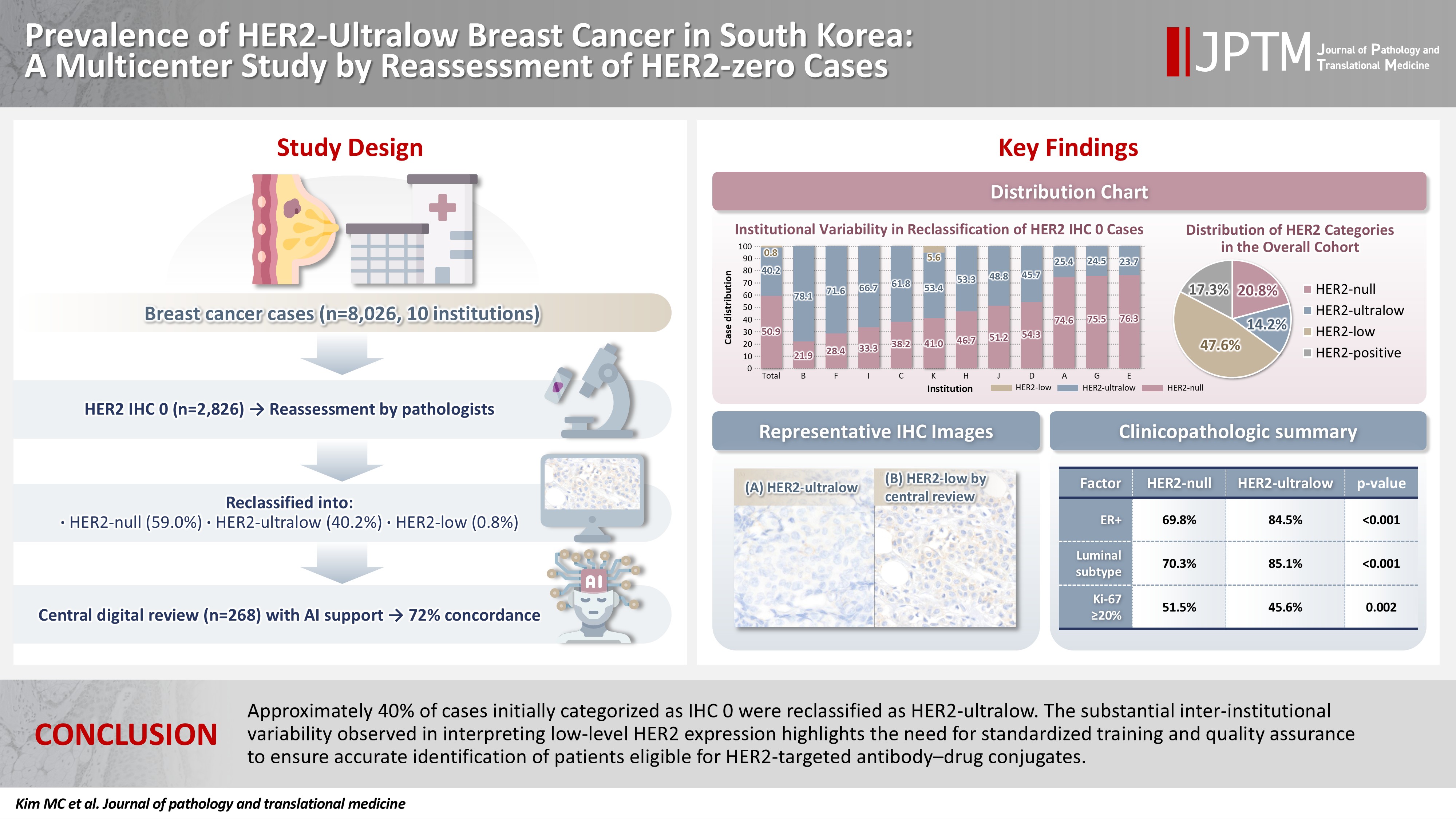
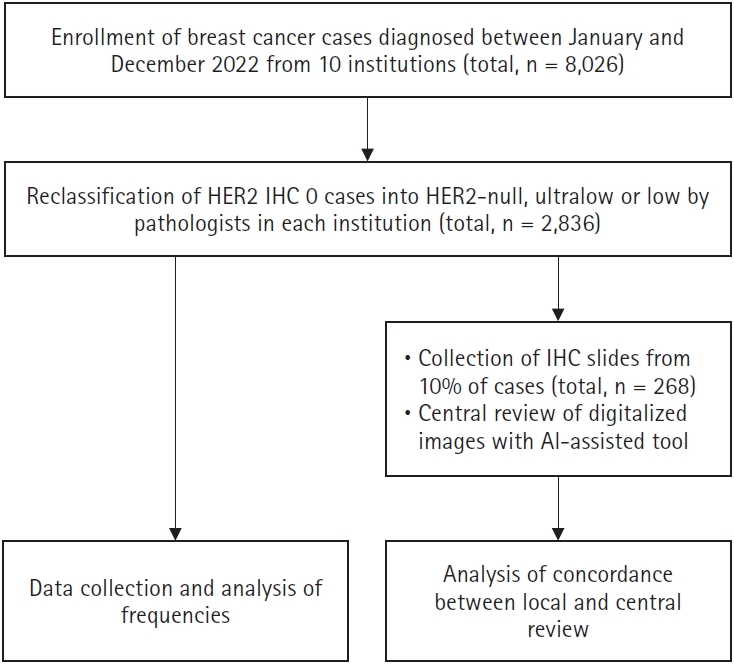
Of the 25 institutions involved in our previous nationwide study [19], 10 institutions participated in the present study. All participating centers were accredited by the Korean Society of Pathology Quality Assessment Program and had successfully completed the periodic Korean Institute of Genetic Testing Evaluation Quality Assessment Program. Ethical approval for this study was obtained from the institutional review board of each participating institution. This retrospective study included all cases of invasive BC diagnosed between January 2022 and December 2022. Cases with microinvasive carcinoma or those resected after neoadjuvant systemic therapy were excluded. For patients with multiple ipsilateral tumors, the largest lesion was selected as the representative tumor for analysis. Baseline clinicopathologic variables collected included sex, age at diagnosis, type of specimen, estrogen receptor (ER) status, progesterone receptor (PR) status, and Ki-67 labeling index (LI). ER, PR, and HER2 results were interpreted according to the 2018 ASCO/CAP guidelines. Molecular subtypes were classified based on immunohistochemical surrogate markers, using a Ki-67 cutoff of 20% to distinguish luminal A (ER+/PR+, low Ki-67) from luminal B (ER+/PR±, high Ki-67) subtypes. Among the 10 institutions, eight institutions used conventional light microscopy for HER2 assessment, and two institutions utilized digital pathology (DP) systems with visual assessment of digitalized images. A schematic overview of the study design is presented in Fig. 1.
Rescoring of HER2 IHC 0 slides
Pathologists at each institution re-evaluated HER2 IHC slides that had originally been reported as IHC 0 at the time of diagnosis. The slides were reclassified into three categories based on the latest 2025 CAP biomarker reporting template: HER2-null (complete absence of membranous staining), HER2-ultralow (faint or weak, incomplete membranous staining in <10% of tumor cells), or HER2-low (IHC score of 1+ or 2+, without HER2 gene amplification) [9]. For clarity, HER2-negative was defined as an IHC score of 0, 1+, or 2+ with a negative ISH result. Following reclassification, all data were submitted for centralized analysis.
Selective central review of IHC slides
Each participating institution submitted 10 % of its slides from the HER2 null and HER2 ultralow subgroups for central review. These slides were digitized using a whole slide scanner (SG300, Philips Healthcare, Amsterdam, Netherlands) and independently assessed by two pathologists (MCK and EYC) with support from PathoAiD-QantiIHC (Aivis, Seoul, Korea), an artificial intelligence (AI)–based analysis platform. The software was used exclusively to detect faint membranous staining patterns in large tumor sections. Nevertheless, the final HER2 classification was determined by consensus between the two pathologists and was not determined by the algorithm alone. The results of the central review were then compared with the local interpretations provided by each institution.
Statistical analysis
Statistical analyses were performed using SPSS ver. 27.0 (IBM Corp., Armonk, NY, USA). Categorical variables were compared using the chi-square test or Fisher’s exact test, whereas continuous variables were analyzed using the unpaired t-test. Concordance between institutional and central reviews was evaluated using Cohen’s κ coefficient. A κ-value ≥ 0.8 was interpreted as almost perfect agreement, while a value between 0.6 and 0.8 indicated substantial agreement [20]. A p-value < .05 was considered statistically significant.
RESULTS
Incidence of HER2-ultralow cases among tumors initially categorized as IHC 0
A total of 8,026 invasive BC cases were included from the 10 participating institutions. Of these cases, 2,836 cases had been initially classified as HER2 IHC 0. Upon re-evaluation by institutional pathologists, 1,673 (59.0%), 1,139 (40.2%), and 24 (0.8%) cases were reclassified as HER2-null, HER2-ultralow, and HER2-low, respectively. The institutional prevalence of HER2-ultralow BC among HER2 IHC 0 cases ranged from 23.7% to 78.1%. The distribution of reclassified HER2 IHC 0 cases across institutions is illustrated in Fig. 2.
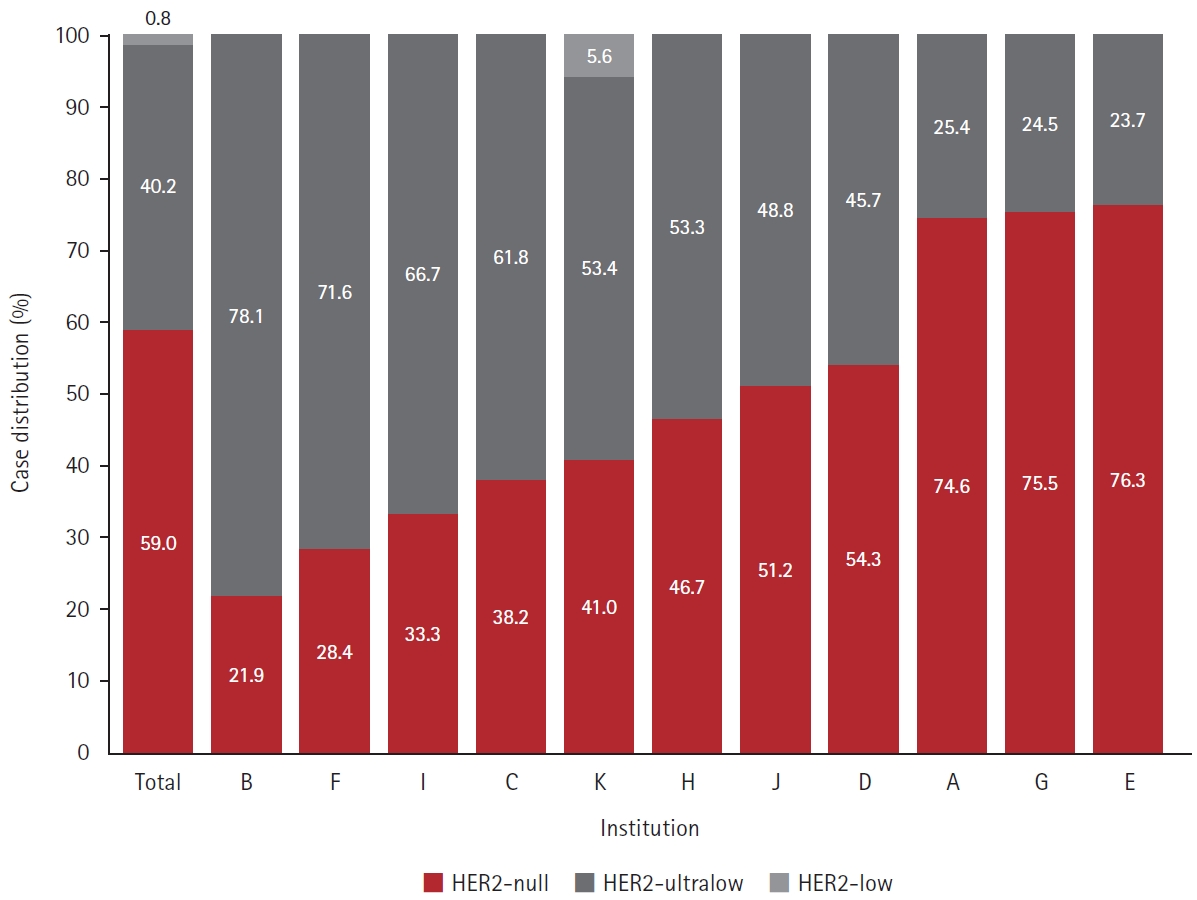
Reclassification of human epidermal growth factor receptor 2 (HER2) immunohistochemistry 0 cases by institutional pathologists.
In the overall cohort, the distribution of HER2 category was as follows: HER2-null: 1,673 (20.8%), HER2-ultralow: 1,139 (14.2%), HER2-low: 3,822 (47.6%), and HER2-positive: 1,392 (17.3%). The institutional distribution of cases according to HER2 category is summarized in Supplementary Table S1. Among the 6,634 HER2-negative cases, the proportions of HER2-null, HER2-ultralow, and HER2-low were 25.2%, 17.2%, and 57.6%, respectively.
Concordance between institutional and central reviews of HER2 IHC 0 slides
A total of 268 HER2 IHC slides, initially interpreted as HER2-null or HER2-ultralow by institutional pathologists, underwent central review by two pathologists. Representative images for discrepant cases between institutional and central reviews were provided in Supplementary Fig. S1. Overall agreement between institutional and central reviews was achieved in 193 (72.0%) cases (Table 1). When HER2 expression was dichotomized into HER2-null and HER2-ultralow/low, the Cohen’s κ coefficient was 0.59 (95% confidence interval [CI], 0.51 to 0.68), indicating moderate agreement. When excluding the 19 cases classified as HER2-low by central review and comparing only HER2-null with HER2-ultralow, the κ value was 0.57 (95% CI, 0.48 to 0.66) with 77.5% agreement.
Among the 75 discordant cases, 54 tumors (72.0%) were reclassified from HER2-null to HER2-ultralow, and one tumor (1.3%) was reclassified from HER2-null to HER2-low. Another 18 tumors (24.0%) were upgraded from HER2-ultralow to HER2-low, whereas the remaining two tumors (2.7%) were downgraded from HER2-ultralow to HER2-null. Overall, the majority of discordant cases were upgraded in HER2 category following central review.
Comparison of clinicopathologic features between HER2-null and HER2-ultralow groups
The clinicopathologic characteristics of the HER2-null and HER2-ultralow subgroups are summarized in Table 2. In comparison with HER2-null tumors, HER2-ultralow tumors were more frequently diagnosed from surgical specimens (63.0% vs. 49.8%, p < .001) and found in slightly older patients (mean age, 54.6 vs. 53.6 years; p =.015). HER2-ultralow tumors also demonstrated a higher rate of ER positivity (84.5% vs. 69.8%, p < .001), and fewer HER2-ultralow tumors had a high Ki-67 LI when using a 20% cutoff (48.5% vs. 54.4%, p = .038). In terms of histologic grade, compared with HER2-ultralow cancers, HER2-null cancers were likely to be high-grade tumors (30.5% vs. 21.9%, p < .001). Overall, HER2-ultralow tumors were predominantly ER-positive/luminal-type tumors, whereas HER2-null tumors were more commonly associated with triple-negative BC profiles.
DISCUSSION
In this multicenter study, we focused on the prevalence of HER2-ultralow BC among cases initially reported as HER2 IHC 0 and interobserver variability in distinguishing low levels of HER2 expression. Upon re-evaluation, 40.2% (range, 23.7% to 78.1%) and 0.8% of HER2 IHC 0 cases were reclassified as HER2-ultralow and HER2-low, respectively. The incidence of HER2-ultralow cases was 14.2% in the total cohort and 17.2% among HER2-negative patients. These findings are somewhat consistent with previous studies, which have reported HER2-ultralow frequencies of 10.6%–17.5% among all BCs and 23.7%–29% among HER2-negative BCs [11-13,21].
Consistent with our previous findings on HER2-low BC incidence [19], there was considerable inter-institutional variability in the frequency of HER2-ultralow tumors within the HER2 IHC 0 group. This variation may be attributed to technical and interpretive factors. As previously described [19], institutional pathology laboratories use different IHC protocols, including staining platforms, antigen retrieval solutions, primary antibody sources, incubation conditions, and detection kits. Even minor adjustments to staining protocols, such as extended antigen retrieval or prolonged antibody incubation, can enhance HER2 staining and shift classification toward the HER2-low category [22-24]. This issue is particularly relevant when evaluating borderline HER2 expression, such as HER2-ultralow. To minimize variability, adoption of standardized protocols based on those used in the DESTINY-Breast04 and DESTINY-Breast06 trials could improve consistency across institutions [25]. In addition, laboratories should pay attention to the management of pre-analytic factors, ensuring that cold ischemic time does not exceed 1 hour and formalin fixation lasts between 6 and 72 hours [26].
Interpretive challenges also contribute to inter-institutional variability in the HER2-ultralow subgroup. Historically, cases now classified as HER2-ultralow (0+) were simply reported as IHC 0, and there was no clinical impetus to distinguish them from HER2-null (0) tumors. The introduction of T-DXd has altered this paradigm. Gao et al. [27] reported an inter-pathologist concordance rate of 69.7% (κ = 0.487) when classifying cases as HER2-low, HER2-ultralow, or HER2-null, with the lowest agreement for the HER2-ultralow subgroup (κ = 0.278). Similarly, Mehta et al. [28] reported an overall concordance rate of 57% and found that the κ value was only 0.17 for the HER2-ultralow subgroup, compared with 0.21 for 1+, 0.47 for null (0), and 0.57 for 2+. These findings suggest that despite guideline adherence, diagnostic reproducibility at lower levels of HER2 expression remains a critical issue. Encouragingly, targeted training of pathologists has been shown to improve interobserver agreement in the interpretation of the HER2-low category [29-31], and this approach may benefit the assessment of HER2-ultralow tumors.
In the present study, a 72% concordance rate was observed between institutional and central reviews of 268 IHC 0 slides. Notably, 97% of the discordant cases were upgraded in HER2 category following central review, most commonly from HER2-null to HER2-ultralow. Central review utilized digitalized images with assistance from an AI-based tool, PathoAiD-QuantiIHC. The AI system facilitated the identification of borderline cases, improving efficiency and clarity. Kovacs et al. [32] reported higher HER2 scores when using digitalized slides instead of conventional microscopy due to enhanced visualization of membranous staining. Furthermore, compared with conventional assessment, an AI model identified more HER2-low and HER2-ultralow cases, thereby improving detection accuracy. Krishnamurthy et al. [33] demonstrated that the use of an AI-assisted tool increased interobserver agreement from 75.0% (digital manual review) to 83.7% and scoring accuracy from 85.3% to 88.0%. Notably, for distinguishing HER2 0 from 1+ cases, AI improved interobserver agreement (69.8% to 87.4%) and accuracy (81.9% to 88.8%). Similarly, Wu et al. [34] found that an AI algorithm significantly improved both the overall consistency and accuracy of HER2 IHC 0 and 1+ evaluation, especially in heterogeneous cases and among less-experienced pathologists. Overall, these studies suggest that AI-assisted image analysis may be useful for reducing interobserver variability and may serve as a valuable adjunct to standard pathology in the assessment of borderline HER2 expression. However, further research is required to determine whether treatment decisions based on AI-assisted classification will lead to improved clinical outcomes.
In terms of clinicopathologic characteristics, we observed that older patient age, lower histologic grade, higher ER positivity, and lower Ki-67 expression were associated with HER2-ultralow tumors compared with HER2-null tumors. These findings are consistent with those reported by Hu et al. [12], suggesting that HER2-ultralow tumors are more closely related to HER2-low than to HER2-null tumors. In contrast, Chen et al. [11] did not observe significant differences between HER2-ultralow and HER2-null tumors but reported differences between HER2-ultralow and HER2-low tumors in lymph node stage, hormone receptor status, and Ki-67 LI. Importantly, neither study found meaningful differences in disease-free survival among the HER2-null, HER2-ultralow, and HER2-low subgroups [11,12]. Taken together, these findings support the view that HER2-negative BC encompasses biologically diverse tumors [26]. However, at present, subclassification into HER2-null, HER2-ultralow, and HER2-low appears to be primarily relevant for guiding therapeutic decisions, particularly for selecting candidates for T-DXd, rather than for defining distinct biological subtypes.
This study showed that HER2-ultralow expression was more common in surgical specimens than in core biopsies. However, this finding should be interpreted with caution because the two specimen types were not matched on a case-by-case basis, and differences in tissue volume were not strictly controlled. Even so, HER2-ultralow tumors often exhibit pronounced intratumoral heterogeneity; therefore, it is reasonable to assume that larger tumor volumes may increase the likelihood of detecting HER2 ultralow expression when examined thoroughly. Several studies on HER2-low cases have supported this concept [35-37]. Accordingly, retesting HER2 on the surgical specimen, rather than relying solely on the initial biopsy result, may provide patients with an additional opportunity to receive T-DXd.
This study has several limitations. First, we did not evaluate the diagnostic experience of pathologists or the pre-analytic and analytic factors of HER2 IHC, which may have contributed to inter-institutional variability. Second, the central review was limited to a subset (10%) of the total cases. Third, inter-pathologist concordance was not assessed under standardized conditions. The central review used AI, while institutional reviews did not—so the discrepancy may partly reflect tool availability, not just interobserver variability. Despite these limitations, this work represents the first multicenter investigation in South Korea to examine the prevalence of HER2-ultralow BC and provides valuable baseline epidemiologic data for future research on HER2-targeted ADCs.
In conclusion, approximately 40% of BC cases initially categorized as IHC 0 were reclassified as HER2-ultralow with considerable inter-institutional variability (range, 23.7% to 78.1%). To enhance diagnostic accuracy and consistency, future efforts should focus on standardization of HER2 staining protocols, training of pathologists in low-level HER2 interpretation, and incorporating DP with AI-assisted image analysis. These strategies may support more reliable identification of patients who could benefit from novel HER2-targeted therapies such as T-DXd.
Supplementary Information
The Data Supplement is available with this article at https://doi.org/10.4132/jptm.2025.10.22.
Notes
Ethics Statement
This study was approved by the Institutional Review Board of each participating hospital (YUMC 2023-03-004, SMC 2023-02-077-002, PNUYH 05-2023-066, AMC 2023-0386, CNUHH-2025-028, KUH 2023-08-084, OC23RCDI0037, UUH 2023-05-037, PNUH 2303-008-124, and 2023GR0151), and the requirement for informed consent was waived.
Availability of Data and Material
The datasets generated or analyzed during the study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
Author Contributions
Conceptualization: GYG, YKB. Data curation: MCK, EYC, HJL, JSL, JYK, WSK, CYK, SYJ, HJC, SML, ARK, JYK, JYS, GYG, YKB. Formal analysis: MCK, EYC, YKB. Investigation: EYC, MCK. Methodology: MCK, EYC, GYG, YKB. Supervision: GYG, YKB. Writing—original draft: MCK, EYC. Writing— review & editing: GYG, YKB. Approval of final manuscript: all authors.
Conflicts of Interest
Y.K.B., a contributing editor of the Journal of Pathology and Translational Medicine, was not involved in the editorial evaluation or decision to publish this article. All remaining authors have declared no conflicts of interest.
Funding Statement
No funding to declare.
Acknowledgments
This study was supported by Korean Society of Pathologists Grant (KSPG2023-03).